Skin Disease in Transplant Patients
E.B. Olasz MD, PhD
M. Neuburg MD
Introduction
Since the first successful cadaver kidney transplant in 1962, the field of solid organ transplantation has undergone a remarkable journey, resulting in an ever-growing number of transplantations and increased transplant and recipient survival. The success of transplant survival is built on the advances in our understanding of physiology, immunology, improved surgical techniques, more efficient immunosuppressive treatments, and the multidisciplinary care of solid organ transplant recipients (OTRs). Longer recipient survival has led to an increased understanding of the consequences of long-term immunosuppression. Potent immunosuppressive drugs allow for long-term organ and patient survival. However, these drugs have numerous side effects, many of which manifest on the skin. These include direct effects from the individual drugs, immune-mediated effects from the grafted organ, and indirect effects of acute and chronic immunosuppression such as opportunistic infections and an increased incidence of skin cancers. The recognition of the accelerated and accentuated cutaneous carcinogenesis and the increased risk for cutaneous infections in OTRs opened the field of transplant dermatology. In 2001 the International Transplant Skin-Cancer Collaborative (ITSCC) was formed, joined by its European counterpart Skin Cancer in Organ Transplant Patients, Europe (SCOPE), in an effort to educate and care for the growing number of OTRs. The following sections outline the broad scope of skin diseases observed in OTRs.
SAUER’S NOTES
1. Transplant patients will continue to increase in the foreseeable future and dermatologists need to help meet the challenge of their complex care.
2. Infections of the skin tend to be associated with less common organisms, to be more aggressive, and to be more recalcitrant to therapy.
3. Malignancies of the skin tend to be more common, more aggressive, and more apt to recur.
4. The skin is the window to other organ systems of the body. In this challenging group of patients, “this window” needs to be used to detect systemic infections and malignancies.
Epidemiology
According to the United Network for Organ Sharing, over 28,000 solid organ transplants are performed in the United States each year and 74,000 worldwide. Approximately 250,000 solid OTRs are alive in the United States today, while over 90,000 people await transplantation. The overall five-year survival rate has steadily increased in the past 10 years, reaching 80% to 90% in kidney, 72% to 86% in liver, 70% in cardiac, and 42% in lung transplant recipients.
Cutaneous Effects of Immunosuppressive Medications
The cutaneous adverse effects of commonly used immunosuppressive agents are detailed in the following text and summarized in Table 44-1.
TABLE 44-1 ▪ Cutaneous Adverse Effects of Commonly Used Immunosuppressive Agents | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
Corticosteroids
Cushing Syndrome
Cushing syndrome is caused by the administration of excess glucocorticoids as part of an OTR patient’s immunosuppressive regimen. The patient may have any of the following typical physical stigmata: moon facies, facial plethora, supraclavicular fat pads, buffalo hump, truncal obesity, and purple striae. Individuals often complain of proximal muscle weakness, easy bruising, weight gain, hirsutism, and impaired wound healing. Reduction or discontinuation of glucocorticoid therapy should be managed by, or in conjunction with, the transplant physician due to the risks of (1) steroid withdrawal syndrome, (2) organ rejection, and (3) possible suppression of HPA axis with secondary adrenal insufficiency.
Steroid Acne
Steroid acne presents with monomorphous, erythematous papules and pustules that appear relatively abruptly on the upper trunk, often sparing the face. The monomorphous nature and abrupt appearance distinguish steroid acne from acne vulgaris, which typically has a slower onset and is composed of acneiform lesions in different stages of development, usually involving the face. Cysts and comedones are common in acne vulgaris.
Treatment. In addition to the traditional treatments of acne vulgaris, including benzoyl peroxide wash or gel, topical or oral antibiotics, and topical retinoids, reduction of offending oral steroids, if possible, may have good results in treating steroid acne. Commonly, pityrosporum folliculitis is a cofactor in the development of steroid acne; therefore, treatment with appropriate topical antifungals should also be considered.
Striae
Striae rubra distensae are linear bands of atrophic, cigarette paper-like skin, which are originally red and indurated, later becoming hypopigmented and atrophic. These may be widely distributed, especially over the abdomen, lower back, buttocks, and thighs.
Treatment. In the early erythematous stage, pulsed-dye laser has been shown to be somewhat helpful. After striae reach the late atrophic stage, treatment is very difficult. Topical retinoids, cryotherapy, and ablative laser resurfacing have been used with inconsistent results.
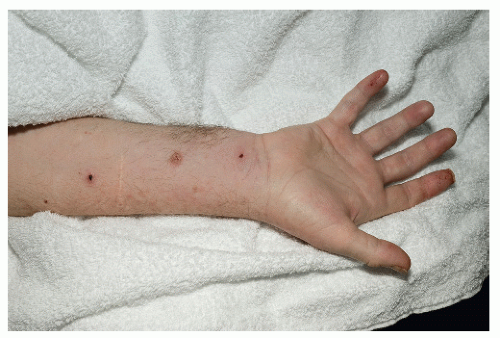
Cutaneous Fragility and Ecchymosis
The skin may become thin and fragile. Spontaneous tearing may occur with trivial trauma. Purpura and ecchymoses are most commonly located on the dorsal forearms and dorsal hands. Avoidance of shearing trauma on the dorsal hands and forearms may help prevent the ecchymoses. Adhesive bandages should be removed with extreme care as skin may tear.
Cyclosporine
Hypertrichosis
Hypertrichosis is a cosmetically undesirable dose-dependent side effect of cyclosporine therapy, characterized by excessive hair growth not localized to the androgen-dependent areas of the body. It appears months after initiation of cyclosporine therapy in about 75% of patients. The cessation of cyclosporine therapy results in a progressive resolution of hypertrichosis. Laser-assisted hair removal or switching to tacrolimus may improve the hypertrichosis.
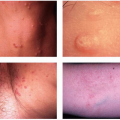
Sebaceous Hyperplasia
Sebaceous hyperplasia is a well-known side effect of cyclosporine therapy, presenting within several years after starting treatment. About 10% to 15% of patients taking cyclosporine develop multiple small, yellowish umbilicated papules measuring 2 to 6 mm located usually on the central face. However, ectopic sites such as the oral mucosa may be affected as well. These lesions are benign but can be cosmetically bothersome to patients. Electrosurgery, laser treatment, shave excision, and photodynamic therapy (PDT) have been employed to treat sebaceous hyperplasia.
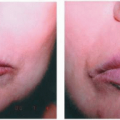
Gingival Hyperplasia
Gingival overgrowth affects 30% to 50% of patients. It is first observed approximately 3 months following the initiation of cyclosporine therapy. Topical or systemic azithromycin may induce marked improvement. In some cases, periodontal surgery may be necessary. In addition, changing cyclosporine to tacrolimus has been reported to improve this condition.
Tacrolimus
Alopecia
Tacrolimus-induced hair loss presents in about 29% of patients as widespread hair thinning, occurring at a mean of 30 to 422 days after initiating tacrolimus. Rapid reversal of alopecia has been reported with the use of minoxidil. Reducing the dose of tacrolimus or switching to cyclosporine is helpful.
Azathioprine
Azathioprine (AZA) has been reported to cause cutaneous hypersensitivity reactions including urticarial, maculopapular, and vasculitic eruptions. Less commonly reported side effects are mucositis, photosensitivity, and increased susceptibility to verrucae, herpes zoster, and Norwegian scabies. In addition, AZA has been shown in laboratory models to have direct carcinogenic effects.
Mycophenolate Mofetil
Nonspecific cutaneous eruptions have been reported in 8% to 22% of patients on mycophenolate mofetil (MMF). Acne and peripheral edema, as well as exacerbation of dyshidrotic eczema, have been linked to MMF therapy.
Sirolimus
Infections and Pilosebaceous Eruptions
Infections and acne-like eruptions are common cutaneous side effects of sirolimus. More specifically, 34% of patients developed viral infections, 4% developed bacterial, and 16% of patients developed fungal infections. With predominance in men, inflammatory eruptions resembling acne were noted in 46% of renal transplant patients on sirolimus. Scalp folliculitis and hidradenitis suppurativa, as well as other skin eruptions resembling seborrheic dermatitis involving almost every body part, have also been reported. Topical and systemic antibiotics, benzoyl peroxide, and topical or systemic retinoids are recommended.
Edema
Chronic edema, mainly affecting the lower legs (98%) was noted in more than half of OTRs (65%) treated with sirolimus. In some patients, upper extremity edema or angioedema involving the face and oral cavity was observed. The exact mechanism of sirolimus-induced edema is unknown, but it is thought to be due to vasculitis, lymphatic obstruction, or capillary obstruction. Sirolimus-induced edema is often resistant to diuretics; therefore, dietary restrictions, blood pressure control, and compression therapy are advised. Discontinuation of the inciting drug is often necessary. Depending on the severity of angioedema, airway support, antihistamines, glucocorticoids, and epinephrine may be required.
Impaired Wound Healing
Sirolimus has been found to cause delayed wound healing, wound dehiscence, and superficial and deep wound fluid collections. The mechanism of impaired wound healing has been shown to be due to the inhibitory effect of sirolimus on fibroblast and endothelial cells through the blockade of growth factors, leading to anti-angiogenesis. Appropriate medical or surgical wound care, drainage of seromas, and in severe cases discontinuation of the drug are necessary. Temporary use of alternate immunosuppressive agents in anticipation of elective surgery is advised.
Cutaneous Effects of the Transplanted Organ
Graft Versus Host Disease (GVHD)
Although GVHD is most frequently associated with bone marrow or stem cell transplantation, it is also a rare but severe complication of solid organ transplantation. Transplanted solid organs contain a variable amount of lymphoid tissue, enabling the allografts to function as a mini bone marrow transplant and initiate an immunologically mediated and injurious set of reactions by cells genetically disparate to their host. In OTRs, GVHD is seen most frequently after liver transplantation, but the incidence of GVHD is highest after small bowel transplantation (about 5%), possibly owing to a large number of donor lymphocytes present in the gut. Risk factors for developing GVHD in liver transplant recipients include age greater than 65, closely matched HLA recipients, and a donor more than 40 years younger than the recipient.
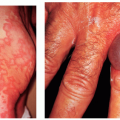
The diagnosis of acute GVHD is established by clinical judgment, imaging studies, laboratory workup, and biopsy results. Skin involvement often precedes hepatic (except in liver transplant patients), hematologic, or gastrointestinal symptoms and presents from 2 days to 6 weeks post-transplant with an erythematous maculopapular eruption. The eruption has a predilection for the palms and soles and in severe cases can progress to generalized erythema with bullae and desquamation, often making it difficult to distinguish it from a severe drug eruption. Skin biopsy and the presence of other systemic symptoms including diarrhea, pancytopenia, and fever aid in confirming the diagnosis. When evaluating a maculopapular eruption in an OTR, in addition to GVHD, drug eruption, chemotherapy toxicity, and viral eruption should be considered in the differential diagnosis. The mortality rate of GVHD in OTRs is high (75% to 90%), with death usually resulting from overwhelming sepsis, gastrointestinal bleeding, pneumonia, or renal failure. Management of acute GVHD after solid organ transplantation requires a multidisciplinary approach. Current therapies include antilymphocyte regimens and various approaches resulting in either increased or decreased immunosuppression. New biologics including basiliximab, a chimeric mouse-human monoclonal antibody to the IL-2Rα receptor (CD25) of T cells, have been successfully used in some cases.
Infectious Diseases of the Skin in OTR Patients
Due to their suppressed host immune defense mechanism, OTRs are more susceptible to bacterial, fungal, and viral infections. Most infections during the first month are related to surgical complications. Opportunistic infections typically occur from the second to the sixth month post-transplant. During the late post-transplant period (beyond 6 months), OTRs suffer from the same infections seen in the general community. Opportunistic bacterial infections seen in transplant recipients include those caused by Legionella spp., Nocardia spp., Salmonella spp., and Listeria monocytogenes. Cytomegalovirus (CMV) is the most common cause of viral infections. Herpes simplex virus (HSV), varicella zoster virus (VZV), Epstein-Barr virus (EBV), human papillomavirus (HPV), and others are also significant pathogens. Fungal infections caused by both yeasts and mycelial fungi are associated with the highest mortality rates. Mycobacteria, pneumocystis, and parasitic diseases may also occur. Due to the heightened risk of infection, the clinician should maintain an increased index of suspicion. More aggressive and earlier diagnostic approaches including skin biopsy with special stains, superficial and tissue culture, and diagnostic imaging modalities may be warranted. Many systemic antimicrobials
require dose adjustments in the setting of renal insufficiency. Use of these drugs in renal transplant recipients should include comanagement by transplant nephrologists.
require dose adjustments in the setting of renal insufficiency. Use of these drugs in renal transplant recipients should include comanagement by transplant nephrologists.
Bacterial Infections
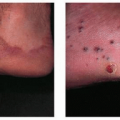
Staphylococcal Infections
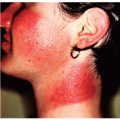
Staphylococcus aureus is a common pathogen and causes the majority of pyodermas and soft-tissue infections seen in solid OTRs. It often colonizes the anterior nares as well as superficial skin breaks and skin disruptions in these patients. Treatment of S. aureus colonization with topical mupirocin ointment has been shown to decrease the occurrence of S. aureus pyodermas including folliculitis, furuncles, carbuncles, impetigo, bullous impetigo, and ecthyma in certain nonimmunosuppressed patients. However, the same results in OTRs have not been proven. Topical treatment with clindamycin, mupirocin, retapamulin, or oral antibiotics according to sensitivities is recommended. Methicillin-resistant S. aureus is an increasing concern in OTRs (Fig. 44-1). Use of Vancomycin or treatment with Linezolid 600 mg b.i.d. may be employed if sensitivities are determined and infection is of significant concern.
Streptococcus Infections
Group A beta-hemolytic streptococci can cause superficial pyodermas presenting as impetiginized skin and soft-tissue infections. Necrotizing fasciitis is a severe form of soft-tissue infection extending into the subcutaneous fat and deep fascia caused by beta-hemolytic streptococcus or a combination of non-group A streptococci and anaerobe bacilli. Pain out of proportion to clinical signs should raise the clinical suspicion of necrotizing fasciitis. Treatment includes clindamycin and gamma globulin in addition to extensive, emergency surgical debridement and intensive care monitoring.
Gram-negative bacteria, such as Klebsiella pneumoniae, Escherichia coli, Pseudomonas aeruginosa, Proteus mirabilis, and Enterobacter, may cause cellulitis in OTR patients, presenting with varying morphology including bullae, ulcers, or cutaneous necrosis with vascular involvement. Initial empiric coverage with gentamicin is helpful until tissue and blood cultures return.
Nocardiosis
Nocardial infections have been reported primarily in renal and heart transplant patients, fewer than 4% of whom develop this type of infection. Nocardial infection is most commonly caused by Nocardia asteroides, a gram-positive, weakly acid fast bacterium. It can be directly introduced to the skin or more commonly spread by the hematogenous route from the lung. Primary cutaneous infection can present as abscesses, ulcers, granulomas, soft-tissue infection, mycetoma, or lymphocutaneous infection. Sulfonamides, either alone or in combination with trimethroprim, are the treatment of choice for nocardiosis. Antimicrobial therapy should be continued for a prolonged period after cure because there is a tendency to relapse and the optimal duration of therapy is not known.
Bartonella Infection
Infection by Bartonella henselae, a gram-negative bacillus, has been reported in kidney, liver, and heart transplant recipients as early as 11 months and as late as 14 years after transplantation. The clinical manifestation of Bartonella infection in OTRs can vary greatly. Patients may present with the typical features of cat-scratch disease with regional lymphadenopathy and fever, but this will usually progress to a more severe, systemic illness if not treated promptly. Transplant recipients have also been reported to develop bacillary angiomatosis, the form of Bartonella infection caused by B. henselae and B. quintana, commonly seen in individuals with HIV infection. Patients with bacillary angiomatosis present with red-to-violaceous, dome-shaped friable papules and nodules, ranging in size from a few millimeters up to 2 to 3 cm in diameter. Culture of the organism from the skin or a lymph node may be difficult to obtain and requires an incubation period of as long as 30 days. For this reason, polymerase chain reaction of the tissue specimen may be preferable. The antibiotics of choice are erythromycin 250 to 500 mg PO four times daily or doxycyline 100 mg two times daily, continued for 3 months.
Vibrio vulnificus
Vibrio vulnificus, a gram-negative rod, may contaminate shellfish and oysters. Infection occurs either through ingestion or direct inoculation through open wounds from seawater mainly along the Atlantic seacoast. Skin lesions begin within 24 to 48 hours after exposure as erythematous plaques and rapidly progress to hemorrhagic bullae, subsequently becoming necrotic ulcers. Aggressive early surgical debridement is mandatory. Mortality is not insignificant. It is important to remember that infection with V. vulnificus can lead to sepsis in immunocompromised hosts and these patients should be cautioned about eating uncooked shellfish. Treatment with Doxycyline 100 mg twice daily is recommended.
Fungal Skin Infections
Fungal skin infections in OTR patients include classical dermatomycosis, opportunistic fungal infections, and infections with rare fungal pathogens. Fungal infections in solid OTRs continue to be a significant cause of morbidity and mortality. Candida spp. and Aspergillus spp. account for most invasive fungal infections. The incidence of fungal infection varies with the type of solid organ transplant. Liver transplant recipients have the highest reported incidence of Candida infections while lung transplant recipients have the highest rate of Aspergillus infections.
The classic superficial fungal infections, such as tinea cruris, corporis, and pedis, are seen in up to 50% of OTR patients and are most commonly caused by Trichophyton rubrum. Onychomycosis is also a frequent finding. Onychomycosis more commonly involves multiple toe- or fingernails in OTRs, and more importantly it is frequently caused by molds, such as Scopulariopsis species. In an immunosuppressed host this fungus may cause subcutaneous infection or fatal systemic infection. Proximal white subungual onychomycosis is a pathognomic sign of immunosuppression, involves the nail plate adjacent to the proximal nail fold, and is produced by T. rubrum and T. megnini. In addition to topical antifungal medications, treatment with terbinafine for extensive superficial fungal infections is a good option because the potential for drug interactions with immunosuppressants is minimal.
Opportunistic Infections
Opportunistic fungal infections in OTRs may be caused by fungi normally occurring in our environment that do not typically cause infection in a normal host, such as Candida, Aspergillus, Cryptococcus, Zygomycetes, and Scedosporium spp. In a severely immunocompromised host these saprophytes can cause serious infections. Candida




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree