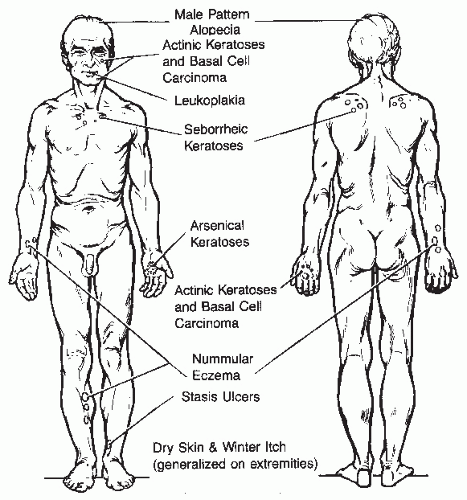
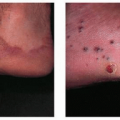
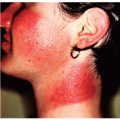
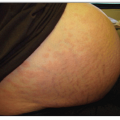
According to one study by Gip and Molin (see Suggested Readings), the 10 most prevalent dermatologic disorders in a large group of elderly Swedish patients are as follows (the numbers in parentheses refer to the total number of dermatologic disorders found in the 286 patients examined):
These investigators studied 286 patients over 60 years of age who were hospitalized in a Swedish geriatric clinic. Each patient underwent a full-body skin exam. Histopathologic, bacteriologic, or mycologic examinations were undertaken in some cases. In the 107 men, there were 231 skin diagnoses (2.2 per person), and in the 179 women, 372 skin diagnoses were made (2.1 per person). The number of skin diagnoses per person ranged from 1 to 5. No skin diagnoses were registered in 22 cases (8% of patients, 5 men and 17 women).
Based on common and concerning skin conditions in the elderly, we provide some general advice (
Table 42-1). Understanding the most common dermatologic problems that are seen in elderly patients requires an appreciation of the mechanisms of aging in the skin. Thus, the next part of this chapter addresses the clinical and histologic features and prevention and treatment of intrinsic aging and photoaging.
Intrinsic versus Extrinsic Skin Aging
Although much remains to be known, we present a brief overview of the science of aging skin since it provides a basis for the clinical changes discussed in more detail. Aging skin results from two forces: (1) intrinsic, or chronologic factors, and (2) extrinsic factors, such as sun damage.
Intrinsic Aging
Intrinsic factors cause the clinical, histologic, and physiologic changes observed in sun-protected aged skin. These changes include reductions in epidermal regeneration rate, rete ridge depth, dermal thickness, thermoregulatory ability, immune defense, wound healing, mechanical defense, sensory perception, sweat and sebum production, and vitamin D synthesis.
Clinical signs of intrinsic skin aging result mainly from thinning, fragility, and loss of elasticity. In the dermis, both intrinsic and extrinsic aging begin with fragmentation of the dermal collagen matrix. Collagen fragmentation is a normal process resulting from the actions of matrix metalloproteinases (MMPs). Fibroblasts typically keep the activity level of these collagen-degrading enzymes in balance with new collagen production. However, fibroblasts require attachment to unfragmented collagen in order to do this effectively. Once significant collagen fragmentation occurs, fibroblasts become less able to produce collagen and regulate MMPs. This leads to a self-perpetuating cycle of decreased fibroblast attachment to intact collagen, decreased collagen production, increased MMP activity, and increased collagen fragmentation.
A second underlying mechanism of intrinsic senescence involves telomere shortening. Telomere length is maintained
in stem cells by the enzyme telomerase, which re-lengthens the DNA ends after each replication cycle. When inefficient duplication of telomeres occurs in dermal fibroblasts, senescence is the end result. Because many tumor cells express telomerase, it is thought that telomerase may play a role in preventing cancer.
Stem cell changes have recently been shown to play a key role in age-associated graying of hair. Hair pigmentation requires regular replenishment of hair melanocytes by stem cells residing in the follicular bulge. Age-related changes in PAX3 and MITF change the balance between melanocyte stem cell maintenance and differentiation and lead to the gradual loss of melanocyte stem cells, resulting in gray hair.
Extrinsic Aging/Photoaging
Most of what we see as aging is in fact extrinsic aging, due mainly to solar ultraviolet (UV) radiation exposure. This so-called photoaging resembles accelerated chronologic aging,
and it occurs via at least two well-documented mechanisms. First, solar UV irradiation generates deleterious reactive oxygen species such as superoxide anions, hydrogen peroxide, and singlet oxygen through NADPH oxidase activity. UV exposure also activates proinflammatory cytokine receptors in human skin, including those for epidermal growth factor, interleukin-1β, interleukin-6, interleukin-8, intercellular adhesion molecule-1, and tumor necrosis factor-α. The increased inflammation further exacerbates oxidative stress. Second, it appears that UV irradiation decreases collagen production. It does this by upregulating multiple kinase-based signaling pathways as well as increasing expression and function of
AP-1. AP-1 is a nuclear transcription factor that shifts the balance between collagen degradation and production toward increased degradation by directly stimulating MMPs (
MMP-1, MMP-3, MMP-9) and impeding collagen gene expression by dermal fibroblasts. In studies by Fisher et al., solar UV irradiation has been shown to reversibly reduce collagen production in young skin by approximately 80% within 24 hours after a single short exposure. In aged skin, it has been shown that decreased fibroblasts and elevated levels of partially degraded collagen act to reduce collagen synthetic activity.
On a cellular level, photoaging changes the structure and function of both the dermis and the epidermis. Photoaged epidermis, for example, can become either hyperplastic or atrophic. Histologic examination of severely photodamaged skin reveals atypical keratinocytes and loss of epidermal polarity, both of which can signify premalignant or malignant changes. In photoaged dermis, degraded and/or irregularly thickened collagen and elastic fibers can present clinically as wrinkling and yellow-brown discoloration. In addition, irregular dilation and increased fragility of dermal blood vessels can show up as telangiectasias and senile purpura.
In brief, sun damage causes the majority of age-related cosmetic and clinical problems on facial skin, including skin cancers, irregular pigmentation, rhytides, and increased skin fragility and coarseness.
Histologic Changes
Histologically, the epidermis of aged skin demonstrates decreased numbers of all major cell types in the skin—keratinocytes, melanocytes, Langerhans cells, and fibroblasts. Accordingly, the function of these cell types is decreased. Keratinocytes, which form the basic barrier that confers the primary mechanical and immunologic defense to our skin, exhibit decreased proliferation, cell signaling, and response to growth factors with aging. These changes result in a reduction of the barrier function leading to increased skin fragility. The decrease in melanocytes, which results in decreased melanin synthesis for UV protection, is evident in the decreased number of nevi present as we age. Langerhans cells are antigenpresenting cells that play an important role in fighting skin infections and neoplasia. Reduced Langerhans cells result in increased rates of skin cancers and infections and decreased rates of contact dermatitis in the elderly. Reduced numbers of mast cells may also contribute to these changes.
The dermoepidermal junction of aged skin becomes flatter, which results in a lower threshold for separation and blister formation. Likewise, reductions in fibroblasts result in a thinned dermis with less collagen and mucopolysaccharide, especially hyaluronic acid. Capillary loops also shrink, resulting in decreased cutaneous blood flow. Appendages such as hair follicles, eccrine and apocrine glands, and pacinian and Meissner’s corpuscles are similarly decreased, resulting in drier, less hairy, less sensitive skin.
Prevention and Treatment
Primary photoprotection (reviewed in
Chapter 7) is the most effective way to prevent photoaging. Even though this is obvious, not enough emphasis is given to this element of skin care. Multiple studies have demonstrated that daily liberal use of a broad-spectrum sunscreen that shields both UVA and UVB radiation with a high sun protection factor (SPF) reduces the number of new premalignant actinic keratoses (AKs) and hastens reduction of preexisting AKs in people at high risk for skin cancer.
With the precautions against solar UV radiation comes a risk of vitamin D deficiency, which is highly prevalent among many elderly patients. Vitamin D, which is naturally produced in the skin after sun exposure, is necessary for bone health, and emerging data indicates that it probably plays important roles in prevention of cancer, including skin cancer, and in prevention of autoimmune disease, such as multiple sclerosis. We do not recommend suntanning as a method of acquiring adequate levels of vitamin D. Instead, we recommend oral supplementation of vitamin D, which is widely available, as well as “smart sun” precautions such as applying sunscreen everyday, wearing sun-protective clothing including hats and gloves when doing outdoor activities like gardening and fishing, and also trying to stay out of the sun between 10:00 AM and 4:00 PM. These precautions can help prevent sunburns and reduce the risk for skin cancer.
Retinoids
At this time, topical retinoids are the only longer-term treatment for photodamage. Tretinoin (all
trans-retinoic acid) has well-documented rejuvenating effects on both chronologically aged and photodamaged skin. Recent work by Fisher has shown that through complex and not fully elucidated molecular pathways, retinoid compounds can cause deposition of new, nonfragmented collagen in both photodamaged and chronologically aged human skin, thereby markedly improving skin texture and appearance. Tretinoin has also been shown to block UV induction of nuclear transcription factors
AP-1 and NF-κB as well as increases in interstitial collagenase and gelatinases in irradiated skin samples that were pretreated 48 hours prior to exposure. The restorative effects of tretinoin have also been demonstrated histologically in non-sun exposed, chronologically aged skin. Daily application of 0.025% tretinoin cream for 9 months in women aged 68 to 79 years resulted in thickened epidermal and granular cell layers with corresponding increased height in the
dermoepidermal junction through the rete pegs, increased uniformity in keratinocyte density, and decreased melanocyte vacuolization. Dermal morphology showed new microvasculature and increased numbers and sizes of fibroblasts, collagen, elastin, microfibrils, and anchoring fibrils. Such changes suggest that tretinoin cream stimulated metabolic activation of chronologically aged skin in a similar fashion to that observed in photodamaged skin.
Alpha-Hydroxy Acids
Alpha-hydroxy acids (AHAs), which include the compounds listed in
Table 42-2, are known to increase epidermal thickness and dermal glycosaminoglycans and decrease hyperkeratinization. As such, AHA-containing products are employed for disorders such as ichthyosis, dry skin, and other disorders associated with retention of the stratum corneum or hyperkeratinization. Application of AHAs in higher concentrations can be useful in treating AKs, warts, and seborrheic keratoses.
Estrogens
Estrogen deficiency, seen largely in postmenopausal women, causes wrinkling, dryness, atrophy, laxity, poor wound healing, hot flashes, and vulvar atrophy. Although data is sparse, there are reports of positive localized epidermal and dermal changes resulting from exogenous estrogen administration. These include increased skin surface lipid production and water-retention capacity, new collagen synthesis, and improved wound healing and quality of scarring. There are reports suggesting that postmenopausal HRT (hormone replacement therapy) may enhance the skin’s thickness and barrier function. In light of recent data regarding chronic use of systemic estrogen supplementation, the risk-to-benefit ratio of HRT needs to be carefully evaluated by physicians and their patients.
Antioxidants
Antioxidants are reviewed in
Chapter 7 and therefore will be discussed here only briefly. A well-balanced diet rich in fruits and vegetables is a good source for antioxidants. In regard to oral supplements, it should be noted that oral vitamin E has been shown to have deleterious effects, including reduced life expectancy.
Oral supplements containing ingredients such as L-proline, L-lysine, manganese, copper, zinc, quercetin, grape seed extract, and N-acetyl-D-glucosamine sulfate have been touted as potential wrinkle reducers. A supplement containing vitamin E, vitamin C, carotenoid, selenium, and proanthocyanidin showed the ability to decrease UV induction of MMPs.
Topical vitamin C prevents erythema following UV exposure. Studies have shown that vitamin C can upregulate collagen and tissue inhibitor of metalloproteinases (TIMP) synthesis as well as decrease wrinkles. Topical CoQ10 has been shown to reduce wrinkles through its antioxidant properties. α-Lipoic acid may have a role in the treatment of photoaging by reducing transcription factors, such as NF-κB, that affect the production of cytokines.
Interventional Treatments
More details on interventional treatments are discussed elsewhere, but a brief overview is included here.
Botulinum toxin A inhibits neuromuscular transmission by blocking acetylcholine release. Cosmetically, botulinum relaxes the underlying musculature of the face, lessening the appearance of wrinkles. Understanding underlying anatomy is important for optimal outcome.
Soft tissue augmentation, or “fillers,” can partially offset age-associated subcutaneous atrophy. Maintenance is required every 6 to 12 months, depending on location, depth, and choice of filler. Immunogenicity can present a major drawback. Acellular dermal grafts from human cadavers show less immunogenicity than bovine collagen but have fallen out of favor since the development of newer labgenerated fillers. Both hyaluronic acid and calcium hydroxyapatite derivatives are less immunogenic, and calcium hydroxyapatite is also potentially more durable.
A variety of resurfacing techniques have been used to improve the appearance of aged skin. Microdermabrasion exfoliates the superficial epidermis, and microablation uses low-frequency radiofrequency via an electrode. The goal of therapy is to injure the superficial epidermis enough to activate cytokines, MMPs, and type I procollagen mRNA and trigger a healing cascade. Effects tend to be short-lived. Other radiofrequency devices aim to improve cheek and neck laxity via heat generation. Adverse affects range from burning and erythema to subcutaneous atrophy.
Laser systems treat multiple age-associated changes such as mottled pigmentation, wrinkling, and dermal atrophy. Fractionated-delivery laser systems have fewer side effects. Care must be taken to avoid ablating the surface of a potentially malignant pigmented lesion, thus allowing a deeper component to proliferate undetected.