Skin is composed of three layers: the epidermis, dermis, and subcutaneous fat (panniculus) ( Fig. 1.1 ). The outermost layer, the epidermis, is composed of viable keratinocytes covered by a layer of keratin, the stratum corneum. The principal component of the dermis is the fibrillar structural protein collagen. The dermis lies on the panniculus, which is composed of lobules of lipocytes separated by collagenous septa that contain the neurovascular bundles.
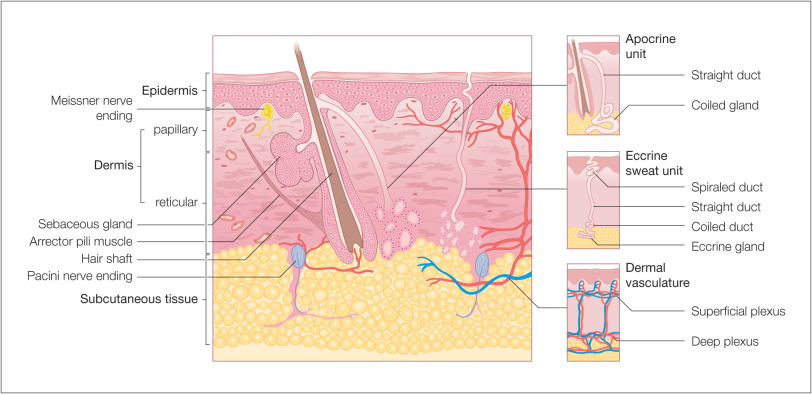
There is considerable regional variation in the relative thickness of these layers. The epidermis is thickest on the palms and soles, measuring approximately 1.5 mm. It is very thin on the eyelid, where it measures less than 0.1 mm. The dermis is thickest on the back, where it is 30–40 times as thick as the overlying epidermis. The amount of subcutaneous fat is generous on the abdomen and buttocks compared with the nose and sternum, where it is meager.
Epidermis and Adnexa
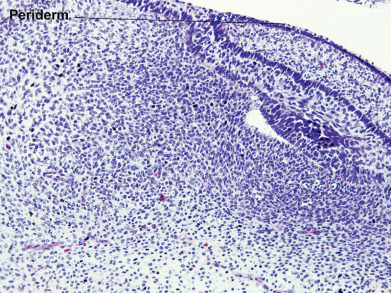
During the first weeks of life, the fetus is covered by a layer of nonkeratinizing cuboidal cells called the periderm ( Fig. 1.2 ). Later, the periderm is replaced by a multilayered epidermis. Adnexal structures, particularly follicles and eccrine sweat units, originate during the third month of fetal life as downgrowths from the developing epidermis. Later, apocrine sweat units develop from the upper portion of the follicular epithelium and sebaceous glands from the midregion of the follicle. Adnexal structures appear first in the cephalic portion of the fetus and later in the caudal portions.
The adult epidermis is composed of three basic cell types: keratinocytes, melanocytes, and Langerhans cells. An additional cell, the Merkel cell, can be found in the basal layer of the palms and soles, oral and genital mucosa, nail bed, and follicular infundibula. Located directly above the basement membrane zone, Merkel cells contain intracytoplasmic dense-core neurosecretory-like granules and, through their association with neurites, act as slow-adapting touch receptors. They have direct connections with adjacent keratinocytes by desmosomes and contain a paranuclear whorl of intermediate keratin filaments. Both polyclonal keratin immunostains and monoclonal immunostaining for keratin 20 stain this whorl of keratin filaments in a characteristic paranuclear dot pattern. Merkel cells also label for neuroendocrine markers such as chromogranin and synaptophysin.
Keratinocytes
Keratinocytes are of ectodermal origin and have the specialized function of producing keratin, a complex filamentous protein that not only forms the surface coat (stratum corneum) of the epidermis but also is the structural protein of hair and nails. Multiple distinct keratin genes have been identified and consist of two subfamilies, acidic and basic. The product of one basic and one acidic keratin gene combines to form the multiple keratins that occur in many tissues. Mutations in the genes for keratins 5 and 14 are associated with epidermolysis bullosa simplex. Keratin 1 and 10 mutations are associated with epidermolytic hyperkeratosis. Mild forms of this disorder may represent localized or widespread expressions of mosaicism for these gene mutations.
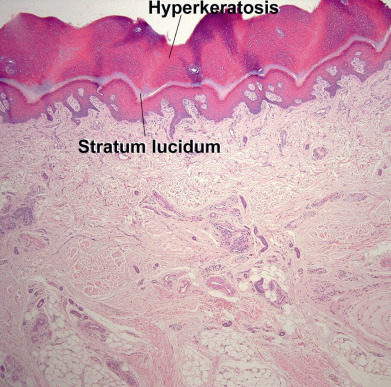
The epidermis can be divided into the innermost basal layer (stratum germinativum), the malpighian or prickle layer (stratum spinosum), the granular layer (stratum granulosum), and the horny layer (stratum corneum). On the palms and soles, a pale clear to pink layer, the stratum lucidum, is noted just above the granular layer ( Fig. 1.3 ). When the skin in other sites is scratched or rubbed, the malpighian and granular layers thicken, a stratum lucidum forms, and the stratum corneum becomes thick and compact. Histones appear to regulate epidermal differentiation, and histone deacetylation suppresses expression of profilaggrin. Slow-cycling stem cells provide a reservoir for regeneration of the epidermis. Sites rich in stem cells include the deepest portions of the rete, especially on palmoplantar skin, as well as the hair bulge. Stem cells divide infrequently in normal skin, but in cell culture they form active, growing colonies. They can be identified by their high expression of β1-integrins and lack of terminal differentiation markers. Stem cells can also be identified by their low levels of desmosomal proteins, such as desmoglein 3. The basal cells divide, and as their progeny move upward, they flatten and their nucleus disappears. Abnormal keratinization can manifest as parakeratosis (retained nuclei), as corps ronds (round, clear to pink, abnormally keratinized cells), or as grains (elongated, basophilic, abnormally keratinized cells).
During keratinization, the keratinocyte first passes through a synthetic and then a degradative phase on its way to becoming a horn cell. In the synthetic phase, within its cytoplasm the keratinocyte accumulates intermediate filaments composed of a fibrous protein, keratin, arranged in an α-helical coiled pattern. These tonofilaments are fashioned into bundles, which converge on and terminate at the plasma membrane, where they end in specialized attachment plates called desmosomes. The degradative phase of keratinization is characterized by the disappearance of cell organelles and the consolidation of all contents into a mixture of filaments and amorphous cell envelopes. This programmed process of maturation resulting in death of the cell is called terminal differentiation. Terminal differentiation is also seen in the involuting stage of keratoacanthomas, where the initial phase of proliferation gives way to terminal keratinization and involution. Degradation of the mitochondrial network within keratinocytes occurs with aging. Oxidation injury to keratinocytes occurs with environmental exposure and thermal burns, and can be partially prevented by vitamin C in the form of L-ascorbic acid.
Premature programmed cell death, or apoptosis, appears in hematoxylin and eosin (H&E)–stained sections as scattered bright-red cells, some of which may contain small, black pyknotic nuclei. These cells are present at various levels of the epidermis, because this form of cell death does not represent part of the normal process of maturation. Widespread apoptosis is noted in the verrucous phase of incontinentia pigmenti. It is also a prominent finding in catagen hairs, where apoptosis results in the involution of the inferior segment of the hair follicle.
In normal skin, the plasma membranes of adjacent cells are separated by an intercellular space. Electron microscopic histochemical studies have shown that this interspace contains glycoproteins and lipids. Lamellar granules (Odland bodies or membrane-coating granules) appear in this space, primarily at the interface between the granular and cornified cell layers. Lamellar granules contribute to skin cohesion and impermeability. Conditions such as lamellar ichthyosis and Flegel hyperkeratosis demonstrate abnormal lamellar granules. Glycolipids such as ceramides contribute a water-barrier function to skin and are typically found in topical products meant to restore the epidermal barrier. Lamellar bodies form abnormally in the absence of critical ceramides such as glucosylceramide, or there is disproportion of critical lipids. Desmosomal adhesion depends on cadherins, including the calcium-dependent desmogleins and desmocollins. Antibodies to these molecules result in immunobullous diseases, but desmogleins function not only in adhesion but also in differentiation. The binding of the desmoglein 1 cytoplasmic tail to the scaffolding-protein Erbin downregulates the Ras-Raf pathway to promote stratification and differentiation of keratinocytes in the epidermis.
Keratinocytes of the granular zone contain, in addition to the keratin filament system, keratohyaline granules, composed of amorphous particulate material of high sulfur-protein content. This material, profilaggrin, is a precursor to filaggrin, so named because it is thought to be responsible for keratin filament aggregation. Conversion to filaggrin takes place in the granular layer, and this forms the electron-dense interfilamentous protein matrix of mature epidermal keratin. Kallikrein-related peptidase 5, a serine protease secreted from lamellar granules, appears to function in profilaggrin cleavage.
Keratohyalin is hygroscopic, and repeated cycles of hydration and dehydration contribute to normal desquamation of the stratum corneum. Ichthyosis vulgaris is characterized by a diminished or absent granular layer, contributing to the retention hyperkeratosis noted in this disorder. Keratohyalin results in the formation of soft, flexible keratin. Keratin that forms in the absence of keratohyaline granules is typically hard and rigid. Hair fibers and nails are composed of hard keratin.
Keratinocytes play an active role in the immune function of the skin. In conditions such as allergic contact dermatitis, these cells participate in the induction of the immune response, rather than acting as passive casualties. Keratinocytes secrete a wide array of cytokines and inflammatory mediators, including tumor necrosis factor–alpha (TNF-α). They also can express molecules on their surface, such as intercellular adhesion molecule 1 (ICAM-1) and major histocompatibility complex (MHC) class II molecules, suggesting that keratinocytes actively respond to immune effector signals.
During wound healing, epithelial cell migration occurs before dermal remodeling. Tight junction proteins claudin-1 and occludin are critical for effective migration. Downregulation of claudin-1 expression results in delayed migration and reduced epithelial proliferation. For occludin, downregulation impairs wound healing when cells are also subjected to mechanical stress. Wound healing occurs best in a moist environment but can be impaired by excessive maceration.
Melanocytes
Melanocytes are derived from the neural crest and by the eighth week of development can be found within the fetal epidermis. In normal, sun-protected trunk epidermis, melanocytes reside in the basal layer at a frequency of about 1 in every 10 basal keratinocytes. Areas such as the face, shins, and genitalia have a greater density of melanocytes, and in heavily sun-damaged facial skin, Mart-1 immunostaining can demonstrate ratios of melanocytes to basal keratinocytes that approach 1 : 1. Recognition of the variation in melanocyte/keratinocyte ratio is critical in the interpretation of biopsies of suspected lentigo maligna (malignant melanoma in situ) on sun-damaged skin.
Racial differences in skin color are not caused by differences in the number of melanocytes. It is the number, size, and distribution of the melanosomes or pigment granules within keratinocytes that determine differences in skin color. Pale skin has fewer melanosomes, and these are smaller and packaged within membrane-bound complexes. Dark skin has more melanosomes, and these tend to be larger and singly dispersed. Chronic sun exposure can stimulate melanocytes to produce larger melanosomes, thereby making the distribution of melanosomes within keratinocytes resemble the pattern seen in dark-skinned individuals.
In histologic sections of skin routinely stained by H&E, the melanocyte appears as a cell with ample amphophilic cytoplasm or as a clear cell in the basal layer of the epidermis. The apparent halo is an artifact formed during fixation of the specimen. This occurs because the melanocyte, lacking tonofilaments, cannot form desmosomal attachments with keratinocytes. Keratinocytes also frequently demonstrate clear spaces but can be differentiated from melanocytes because they demonstrate cell-cell junctions and a layer of cytoplasm peripheral to the clear space.
The melanocyte is a dendritic cell. Its dendrites extend for long distances within the epidermis, and any one melanocyte is therefore in contact with a great number of keratinocytes; together they form the so-called epidermal melanin unit. Keratinocytes actively ingest the tips of the melanocytic dendrites, thus imbibing the melanosomes.
Melanosomes are synthesized in the Golgi zone of the cell and pass through a series of stages in which the enzyme tyrosinase acts on melanin precursors to produce the densely pigmented granules. Melanocytes in red-haired individuals tend to be rounder and to produce more pheomelanin. The melanocortin 1 receptor (MC1R) is important in the regulation of melanin production. Loss-of-function mutations in the MC1R gene bring about a change from eumelanin to pheomelanin production, whereas activating gene mutations can enhance eumelanin synthesis. Most redheads are compound heterozygotes or homozygotes for a variety of loss-of-function mutations in this gene.
Antimicrobial peptides, including cathelicidin and β-defensins, are key components of the innate immune system. They protect against infection, are implicated in the pathogenesis of atopic dermatitis, and play a role in control of pigmentation. The β-defensins encompass a class of small, cationic proteins important to both the innate and the adaptive immune system. β-Defensin 3 also functions as a melanocortin receptor ligand.
Eumelanin production is optimal at pH 6.8, and changes in cellular pH also result in alterations of melanin production and the eumelanin/pheomelanin ratio. Within keratinocytes, melanin typically forms a cap over the nucleus, where it presumably functions principally in a photoprotective role. Evidence of keratinocyte photodamage in the form of pyrimidine dimer formation can be assessed using gas chromatography–mass spectrometry or enzyme-linked immunosorbent assays. Pigment within melanocytes also serves to protect the melanocytes themselves against photodamage, such as ultraviolet A (UVA)–induced membrane damage.
Areas of leukoderma, or whitening of skin, can be caused by very different phenomena. In vitiligo, the affected skin becomes white because of destruction of melanocytes. In albinism, the number of melanocytes is normal, but they are unable to synthesize fully pigmented melanosomes because of defects in the enzymatic formation of melanin. Local areas of increased pigmentation can result from a variety of causes. The typical freckle results from a localized increase in production of pigment by a near-normal number of melanocytes. Black “sunburn” or “ink spot” lentigines demonstrate basilar hyperpigmentation and prominent melanin within the stratum corneum. Nevi are benign proliferations of melanocytes. Melanomas are their malignant counterpart. Melanocytes and keratinocytes express neurotrophins (ectodermal nerve growth factors). Melanocytes release neurotrophin 4, but the release is downregulated by ultraviolet B (UVB) irradiation, suggesting neurotrophins as possible targets for therapy of disorders of pigmentation. Melanocytes express toll-like receptors (TLRs) and stimulation by bacterial lipopolysaccharides increases pigmentation. Melatonin and its metabolites protect melanocytes from UVB damage.
Langerhans Cells
Langerhans cells are normally found scattered among keratinocytes of the stratum spinosum. They constitute 3%–5% of the cells in this layer. As with melanocytes, Langerhans cells are not connected to adjacent keratinocytes by the desmosomes. The highest density of Langerhans cells in the oral mucosa occurs in the vestibular region, and the lowest density is in the sublingual region, suggesting the latter is a relatively immunologically “privileged” site.
At the light microscopic level, Langerhans cells are difficult to detect in routinely stained sections. However, they appear as dendritic cells in sections impregnated with gold chloride, a stain specific for Langerhans cells. They can also be stained with CD1α or S-100 immunostains. Ultrastructurally, they are characterized by a folded nucleus and distinct intracytoplasmic organelles called Birbeck granules. In their fully developed form, the organelles are rod shaped with a vacuole at one end, resembling a tennis racquet. The vacuole is an artifact of processing.
Functionally, Langerhans cells are of the monocyte-macrophage lineage and originate in bone marrow. They function primarily in the afferent limb of the immune response by providing for the recognition, uptake, processing, and presentation of antigens to sensitized T lymphocytes and are important in the induction of delayed-type sensitivity as well as humoral immunity. Once an antigen is presented, Langerhans cells migrate to the lymph nodes. Hyaluronan (hyaluronic acid) plays a critical role in Langerhans cell maturation and migration. Langerhans cells express langerin, membrane adenosine triphosphatase (ATPase, CD39), and CCR6, whereas CD1α+ dermal dendritic cells express macrophage mannose receptor, CD36, factor XIIIa, and chemokine receptor 5, suggesting different functions for these two CD1α+ populations. If skin is depleted of Langerhans cells by exposure to UV radiation, it loses the ability to be sensitized until its population of Langerhans cell is replenished. Macrophages that present antigen in Langerhans cell–depleted skin can induce immune tolerance. In contrast to Langerhans cells, which make interleukin-12 (IL-12), the macrophages found in the epidermis 72 hours after UVB irradiation produce IL-10, resulting in downregulation of the immune response. At least in mice, viral immunity appears to require priming by CD8α+ dendritic cells, rather than Langerhans cells, suggesting a complex pattern of antigen presentation in cutaneous immunity.
Vaccine studies suggest the importance of various cutaneous dendritic cells. Microneedle delivery of vaccine into skin can provoke CD8+ T-cell expansion mediated by CD11c(+) CD11b(+) langerin-negative dendritic cells. Intradermal immunization is dependent on Langerhans cells to stimulate follicular T helper cells and germinal center formation.
Chen Y, et al: Biomaterials as novel penetration enhancers for transdermal and dermal drug delivery systems. Drug Deliv 2013; 20: 199.
Homberg M, et al: Beyond expectations: novel insights into epidermal keratin function and regulation. Int Rev Cell Mol Biol 2014; 311: 265.
Janjetovic Z, et al: Melatonin and its metabolites protect human melanocytes against UVB-induced damage. Sci Rep 2017; 7: 1274.
Levin C, et al: Critical role for skin-derived migratory DCs and Langerhans cells in T(FH) and GC responses after intradermal immunization. J Invest Dermatol 2017; 137: 1905.
Mellem D, et al: Fragmentation of the mitochondrial network in skin in vivo. PLoS One 2017; 12: e0174469.
Pielesz A, et al: The role of topically applied l-ascorbic acid in ex-vivo examination of burn-injured human skin. Spectrochim Acta A Mol Biomol Spectrosc 2017; 185: 279.
Roberts N, et al: Developing stratified epithelia. Wiley Interdiscip Rev Dev Biol 2014; 3: 389.
Volksdorf T, et al: Tight junction proteins claudin-1 and occludin are important for cutaneous wound healing. Am J Pathol 2017; 187: 1301.
Whitehead F, et al: Identifying, managing and preventing skin maceration. J Wound Care 2017; 26: 159.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree