Pityriasis Rosea
Clinical Features
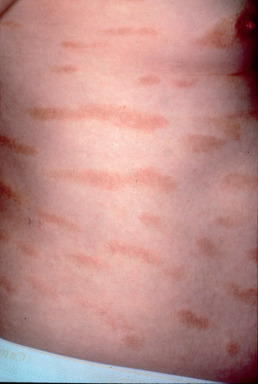
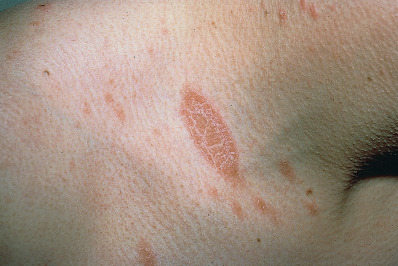
Pityriasis rosea is a mild inflammatory exanthem characterized by salmon-colored thin papules and plaques that are at first discrete but may become confluent ( Fig. 11.1 ). The individual patches are oval or circinate and covered with finely crinkled, dry epidermis, which often desquamates, leaving a collarette of scaling in the center. When stretched across the long axis, the scales tend to fold across the lines of stretch, the so-called hanging curtain sign or Christmas tree sign. The disease most frequently begins with a single herald or mother patch ( Fig. 11.2 ), usually larger than succeeding lesions, which may persist 1 week or longer before others appear. This is often confused for tinea corporis because it is an isolated patch with central colarette of scale mistaken for being annular. By the time involution of the herald patch has begun, new lesions arise rapidly ( Fig. 11.3 ). The total course is typically 3–8 weeks, and then the lesions spontaneously resolve. Relapses and recurrences are observed infrequently. The incidence is highest between ages 15 and 40, and the disease is most prevalent in the spring and autumn. Women are more frequently affected than men.
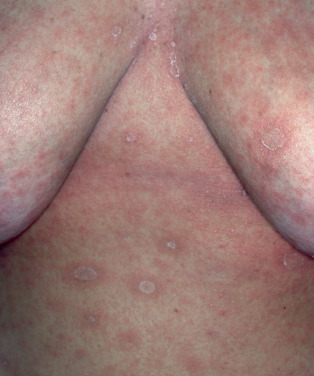
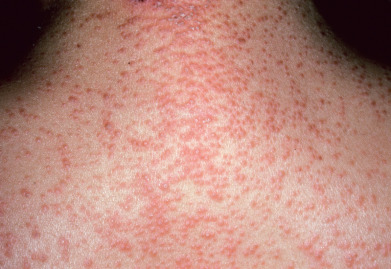
The fully developed eruption has a striking appearance because of the distribution and definite characteristics of the individual lesions. These are arranged so that the long axis of the macules runs parallel to the lines of cleavage. The eruption is usually generalized, affecting chiefly the trunk and sparing sun-exposed surfaces. Less common presentations are localized (involving neck, thighs, groins, or axillae), distribution (sparing covered areas), unilateral, papular, and segmental pattern. Children with darker skin pigmentation are particularly predisposed to the papular variant and are also more prone to facial and scalp involvement. In darker-skinned patients, the lesions often resolve with hypopigmentation. A vesicular variant has also been described, and erythema multiforme–like lesions may occur. Purpuric pityriasis rosea may manifest with petechiae and ecchymoses along Langer lines of the neck, trunk, and proximal extremities, and may rarely be a sign of an underlying leukemia. Pityriasis rosea occurring during pregnancy may be associated with premature delivery, neonatal hypotonia, and fetal loss, especially if the eruption occurs within the first 15 weeks of gestation.
When the eruption localized, it can be difficult to distinguish from unilateral laterothoracic exanthema (asymmetric periflexural exanthema of childhood). Confluent circinate patches with gyrate borders may form and may resemble tinea corporis. Rarely, the eyelids, palms and soles, scalp, or penis may be involved, and syphilis should be ruled out, especially with palm and sole involvement. Oral lesions are relatively uncommon; they are asymptomatic, erythematous macules with raised borders and clearing centers or aphthous ulcer–like lesions. They involute simultaneously with the skin lesions. Moderate pruritus may be present, particularly during the outbreak, and mild constitutional symptoms may occur before the onset.
Etiology
Pityriasis rosea is most likely a response to a virus. The most commonly implicated viruses are herpes viridae such as human herpesvirus (HHV)–6 and HHV-7. Watanabe et al. (2002) demonstrated active replication of HHV-6 or HHV-7 in mononuclear cells of lesional skin, as well as identifying the viruses in serum samples of patients including women who experienced miscarriage in association with pityriasis rosea. Although these viruses are almost universally acquired in early childhood and remain in a latent phase as mononuclear cells, the eruption is likely secondary to reactivation leading to viremia. HHV-2 and hepatitis C virus (HCV) have also been implicated in individual cases.
A pityriasis rosea–like eruption may occur as a reaction to medications such as captopril, imatinib mesylate, interferon, ketotifen, arsenicals, gold, bismuth, clonidine, methoxypromazine, tripelennamine hydrochloride, ergotamine, lisinopril, acyclovir, lithium, adalimumab, nortriptyline, lamotrigine, rituximab, imatinib, asenapine, barbiturates, or bacille Calmette-Guérin (BCG) vaccine.
Histology
The histologic features of pityriasis rosea include mild acanthosis, focal parakeratosis, and extravasation of erythrocytes into the epidermis. Spongiosis may be present in acute cases. A mild perivascular infiltrate of lymphocytes is found in the dermis. Histologic evaluation is especially helpful in excluding the conditions with which pityriasis rosea may be confused.
Differential Diagnosis
Pityriasis rosea may closely mimic seborrheic dermatitis, tinea corporis, secondary syphilis (macular syphilid), drug eruption, other viral exanthems, and psoriasis. In seborrheic dermatitis, the scalp and eyebrows are usually scaly; there is a predilection for the sternal and interscapular regions, as well as the flexor surfaces of the articulations, where the patches are covered with greasy scales. Tinea corporis is rarely so widespread. Tinea versicolor may also closely simulate pityriasis rosea but the individual lesions tend to be flatter and smaller. A positive potassium hydroxide (KOH) examination serves well to differentiate these last two. In macular syphilid, the lesions are of a uniform size and assume a brownish tint. Scaling and itching are absent or slight, and there is typically generalized adenopathy with mucous membrane lesions, palmoplantar lesions, positive nontreponemal and treponemal tests, and often the remains of a chancre. Because syphilis can so closely mimic pityriasis rosea and syphilis incidence is rising again, strong consideration for syphilis testing should be considered in patients with pityriasis rosea. Scabies and lichen planus may be confused with the papular type.
Treatment
Most patients with pityriasis rosea require no therapy because they are asymptomatic; however, the duration of the eruption may be notably reduced by several interventions. A Cochrane review cited inadequate evidence for efficacy for most published treatments; however, lack of evidence does not equate to lack of efficacy. The most data exists for the use of acyclovir with multiple trials showing decrease the eruption and itching. In pregnant patients, given the small risk of harm to the fetus early in pregnancy, acyclovir could be considered along with input from an obstetrician to help weigh the risks and benefits. Erythromicin has demonstrated benefit in small studies. The gastrointestinal side effects of erythromycin may limit its use. Use of ultraviolet B (UVB) light in erythema exposures may expedite the involution of the lesions after the acute inflammatory stage has passed. The erythema produced by ultraviolet (UV) treatment is followed by superficial exfoliation. Narrow Band UVB therapy may help improve individual lesions but may not shorten the course or alleviate pruritis. Corticosteroid lotions or creams provide some relief from itching. For dryness and irritation, simple emollients are recommended .
Bangash HK, et al: Pityriasis rosea–like drug eruption due to nortriptyline in a patient with vulvodynia. J Low Genit Tract Dis 2013; 17: 226.
Chuh A, et al: A position statement on the management of patients with pityriasis rosea. J Eur Acad Dermatol Venereol 2016; 30: 1670.
Drago F, et al: Evidence of human herpesvirus-6 and -7 reactivation in miscarrying women with pityriasis rosea. J Am Acad Dermatol 2014; 71: 198.
Drago F, et al: Pityriasis rosea. Dermatology 2016; 232: 431.
Ganguly S: A randomized, double-blind, placebo-controlled study of efficacy of oral acyclovir in the treatment of pityriasis rosea. J Clin Diagn Res 2014; 8: YC01.
Oh CW, et al: Pityriasis rosea–like rash secondary to intravesical bacillus Calmette-Guérin immunotherapy. Ann Dermatol 2012; 24: 360.
Relhan V, et al: Pityriasis rosea with erythema multiforme–like lesions. Indian J Dermatol 2013; 58: 242.
Sezer E, et al: Pityriasis rosea–like drug eruption related to rituximab treatment. J Dermatol 2013; 40: 495.
Sharma PK, Yadav TP, Gautam RK, Taneja N, Satyanarayana L. Erythromycin in pityriasis rosea: A double-blind, placebo-controlled clinical trial. Journal of the American Academy of Dermatology. 2000 Feb 1;42(2):241-4.
Pityriasis Rubra Pilaris
Clinical Features
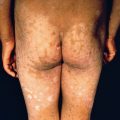
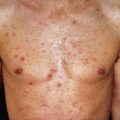
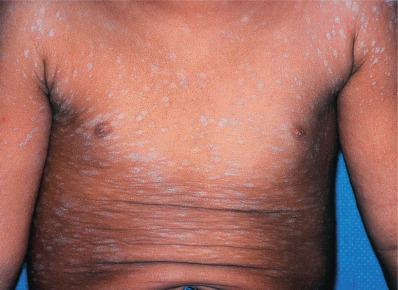
Pityriasis rubra pilaris (PRP) is a chronic skin disease characterized by small follicular papules that coalesce into salmon colored pink scaling patches, and often, solid confluent yellow-orange palmoplantar hyperkeratosis. The papules are the most important diagnostic feature, being more or less acuminate (pointy), salmon colored to reddish brown, about pinhead sized, and topped by a central horny plug ( Fig. 11.4 ). A hair, or part of one, may be embedded in the horny center. There are two peaks of during the first 5 years of life or in the fifties. Some cases of PRP are caused by genetic mutations, thus explaining their chronic recalcitrant nature.
PRP is generally symmetric and diffuse, with characteristic small islands of normal skin within the affected areas ( Fig. 11.5 ). There is a hyperkeratosis of the palms and soles ( Fig. 11.6 ), with a tendency to fissures. The classic disease generally manifests first by scaliness and erythema of the scalp. The eruption is limited in the beginning, having a predilection for the sides of the neck and trunk and the extensor surfaces of the extremities, especially the backs of the first and second phalanges. Then, as new lesions occur, extensive areas are converted into sharply marginated patches of various sizes, which look like exaggerated gooseflesh and feel like a nutmeg grater. Any part or the entire skin surface may be affected. On the soles especially, the hyperkeratosis typically extends up the sides, in the distribution of a sandal. The nails may be dull, rough, thickened, brittle, and striated, and are apt to crack and break. They are rarely, if ever, pitted. The exfoliation may become generalized and the follicular lesions less noticeable, finally disappearing and leaving a widespread dry, scaly erythroderma. The skin becomes dull red, glazed, atrophic, sensitive to slight changes in temperature, and over the bony prominences, subject to ulcerations. In the classic juvenile type, limited plaques occur on extensor surfaces, with adjacent “nutmeg-grater” papules.
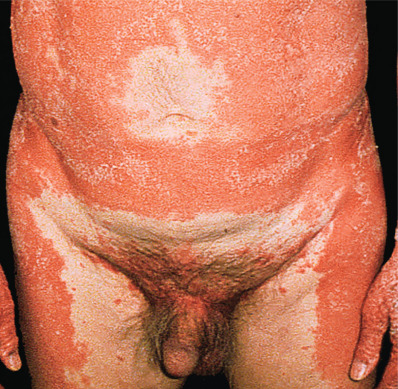
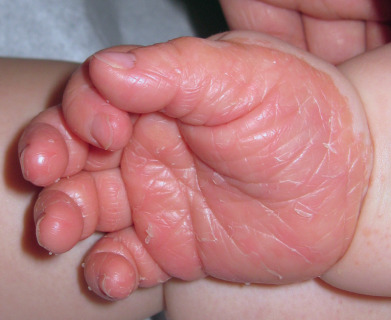
The skin may be itchy or asymptomatic. The Koebner phenomenon may be present. The general health of most patients is not affected. Occasionally arthritis may accompany the eruption and rarely a protein-losing enteropathy may occur if there is very widespread involvement. A number of cases of associated malignancy have recently been reported. It remains to be established whether these are true associations or chance findings. Both hypothyroidism and hypoparathyroidism have been reported, as has the combination of sacroiliitis and autoimmune thyroiditis.
PRP may be classified according to familial (typically autosomal dominant) or acquired types and to the onset of disease in childhood or adulthood. Griffith’s classification is useful in this regard. Type I, the classic adult type, is seen most often and carries a good prognosis, with 80% involuting over a 3-year period. Most patients with the classic juvenile type (type III) have clearing of the disease in 1 year, although it may recur, even into adulthood. The atypical adult (type II) and circumscribed juvenile (type IV) form account for a minority of cases and carry a poorer prognosis for spontaneous recovery. Type V appears in infancy or early childhood and has a more chronic course that has been associated with a CARD14 mutation, likely explaining the chronic nature. CARD14 mutations have also been demonstrated in pustular psoriasis. A type VI has been proposed that is seen in patients infected with human immunodeficiency virus (HIV) and is associated with acne conglobata, hidradenitis suppurativa, or lichen spinulosus.
Etiology
Although CARD14 has been found in chronic early-onset forms, the etiology of spontaneous PRP is unknown. Either gender may be affected, with equal frequency. Both clinically and histologically, the disease has many features that suggest it is a vitamin deficiency disorder, particularly of vitamin A. Some reports of patients with low serum levels of retinol-binding protein have appeared, but this is not a reproducible finding. Streptococcal infections have rarely been implicated in children. A similar eruption has been described secondary to imatinib, sorafenib, and telaprevir.
Histology
There is hyperkeratosis, follicular plugging, and focal parakeratosis at the follicular orifice. Parakeratosis may alternate both vertically and horizontally, producing a checkerboard pattern. Acantholysis may be present, especially within adnexal structures. The inflammatory infiltrate in the dermis is composed of mononuclear cells and is generally mild. Although making an unequivocal histologic diagnosis of PRP may be difficult, the findings of psoriasis, which is the most common clinical entity in the differential diagnosis, are not present.
Diagnosis
Once fully manifested, the diagnosis of fully developed PRP is rarely difficult because of its distinctive features, such as the peculiar orange or salmon-yellow color of the follicular papules, containing a horny center, on the backs of the fingers, sides of the neck, and extensor surfaces of the limbs; the thickened, rough, and slightly or moderately scaly, harsh skin; the sandal-like palmoplantar hyperkeratosis; and the islands of normal skin in the midst of the eruption. It is distinguished from psoriasis by the scales, which in the latter are silvery and light, and overlap like shingles, and by the papules, which extend peripherally to form patches. Phrynoderma (follicular hyperkeratosis) caused by vitamin A deficiency gives a somewhat similar appearance to the skin, as may eczematous eruptions caused by vitamin B deficiency. Rheumatologic disorders, such as subacute cutaneous lupus erythematosus and dermatomyositis, may present with similar cutaneous findings.
Treatment
The management of PRP is generally with systemic retinoids, although topical tazarotene has also been reported to be of benefit. Isotretinoin, may induce prolonged remissions or cures. It may take 6–9 months for full involution to occur, and tapering of the drug may prevent recurrence. Acitretin and methotrexate have also been shown to improve PRP. Ustekinumab has shown efficacy including in patients with familial PRP from CARD14 mutations. Resolution by way of an erythema gyratum repens–like pattern has been described during methotrexate therapy. UV light may flare some patients, but in others, psoralen plus ultraviolet A (PUVA), UVA I, or narrow-band (NB) UVB, alone or in combination with retinoids, may be effective. Phototesting before initiating light therapy is recommended. Extracorporeal photochemotherapy, cyclosporine, anti–tumor necrosis factor (TNF) agents, and azathioprine have also been reported to be effective in resistant and severe cases.
Topical applications of calcineurin inhibitors, lactic acid, or urea-containing preparations may be helpful. Responses to topical corticosteroids are not very effective as a rule. Systemic corticosteroids are beneficial only for acute, short-term management, but are not recommended for chronic use. In HIV-related disease, multiagent antiviral therapy may be useful alone or in combination with retinoids.
Adnot-Desanlis L, et al: Effectiveness of infliximab in pityriasis rubra pilaris is associated with pro-inflammatory cytokine inhibition. Dermatology 2013; 226: 41.
Eastham AB, et al: Treatment options for pityriasis rubra pilaris including biologic agents. JAMA Dermatol 2014; 150: 92.
Griffiths WA. Pityriasis rubra pilaris. Clinical and experimental dermatology. 1980 Mar;5(1):105-12.
Ko CJ, et al: Pityriasis rubra pilaris. Int J Dermatol 2011; 50: 1480.
Marchetti MA, et al: Pityriasis rubra pilaris treated with methotrexate evolving with an erythema gyratum repens–like appearance. J Am Acad Dermatol 2013; 69: e32.
Mercer JM, et al: Familial pityriasis rubra pilaris. J Cutan Med Surg 2013; 17: 226.
Möhrenschlager, M. and Abeck, D., 2002. Further clinical evidence for involvement of bacterial superantigens in juvenile pityriasis rubra pilaris (PRP): report of two new cases. Pediatric dermatology, 19(6), pp.569-569.
Napolitano M, et al: Ustekinumab treatment of pityriasis rubra pilaris. J Dermatol 2017 Oct 28; ePub ahead of print.
Pampín A, et al: Successful treatment of atypical adult pityriasis rubra pilaris with oral alitretinoin. J Am Acad Dermatol 2013; 69: e105.
Paz C, et al: Sorafenib-induced eruption resembling pityriasis rubra pilaris. J Am Acad Dermatol 2011; 65: 452.
Petrof G, et al: A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists. J Eur Acad Dermatol Venereol 2013; 27: e131.
Plana A, et al: Pityriasis rubra pilaris–like reaction induced by imatinib. Clin Exp Dermatol 2013; 38: 520.
Takeichi T, et al: Pityriasis rubra pilaris type V as an autoinflammatory disease by CARD14 mutations. JAMA Dermatol 2017; 153: 66.
Small Plaque Parapsoriasis
Small plaque parapsoriasis (SPP) is characterized by hyperpigmented or yellowish red scaling patches, round to oval in configuration, with sharply defined, regular borders. Most lesions occur on the trunk, and all are 1–5 cm in diameter. In the digitate variant, yellowish tan, elongated, fingerprint-like lesions are oriented along the cleavage lines, predominantly on the flank ( Fig. 11.7 ). These lesions may at times be longer than 5 cm. There is an absence of the induration, the large, erythematous to purplish red lesions, and poikiloderma that characterize small patches of cutaneous T-cell lymphoma in its early stages. Histology can help differentiate from pityriasis lichenoides. The eruption may be mildly itchy or asymptomatic and has a male preponderance. Typical SPP rarely progresses to mycosis fungoides, although the histologic changes can overlap, and clonality may be demonstrated in SPP. Debate continues on this issue. A hypopigmented variant may have a somewhat higher rate of progression to hypopigmented mycosis fungoides. SPP has been reported in the setting of liposarcoma, with resolution of the eruption after resection of the tumor.