Syphilis
Syphilis, also known as lues, is a contagious, sexually transmitted disease caused by the spirochete Treponema pallidum subspecies pallidum. The only known host is the human. The spirochete enters through the skin or mucous membranes, where the primary manifestations are seen. In congenital syphilis, the treponeme crosses the placenta and infects the fetus. The risk of acquiring infection from sexual contact with an infected partner in the previous 30 days is 16%–30%. Syphilis results in multiple patterns of skin and visceral disease and can be lifelong.
Syphilis, yaws, pinta, and endemic syphilis are closely related infectious conditions caused by “genetically monomorphic bacteria,” with less than 2% difference in the genomes of the treponemes (treponemas) that cause these infections. Historically, yaws first arose with humans in Africa and spread with human migrations to Europe and Asia. Endemic syphilis evolved from yaws and became endemic in the Middle East and the Balkans at some later date. Yaws moved with human migration to the New World and became endemic in South America. Syphilis, T. pallidum pallidum, may have originated in the New World from T. pallidum pertenue, the organism causing yaws, much as human immunodeficiency virus (HIV) evolved in Africa from simian immunodeficiency virus (SIV). A tribe in Guyana with a spirochetal infection with features of both yaws and syphilis was identified. Sequencing the genome of this spirochete suggested that it was the ancestor of T. pallidum pallidum. This lends support to the theory that syphilis originated more recently in the New World and was brought back to Europe by sailors who went to the New World with Christopher Columbus. Exactly how and when it became primarily a venereally transmitted disease is unclear, but apparently this happened toward the end of the 15th century.
Treponema pallidum is a delicate, spiral spirochete that is actively motile. The number of spirals varies from 4 to 14, and the entire length is 5–20 µm. It can be demonstrated in preparations from fresh primary or secondary lesions by darkfield microscopy or by fluorescent antibody techniques. The motility is characteristic, consisting of three movements: a projection in the direction of the long axis, a rotation on its long axis, and a bending or twisting from side to side. The precise uniformity of the spiral coils is not distorted during these movements. Microscopic characteristics of T. pallidum cannot be distinguished from commensal oral treponemes, so darkfield examination of oral lesions is unreliable. Direct fluorescent antibody testing can be used for confirmation.
The genome of T. pallidum has been sequenced and contains about one quarter of the number of genes of most bacteria. It lacks significant metabolic capacity. It is very sensitive to temperature, with some enzymes working poorly at typical body temperature (perhaps explaining why fever therapy was effective). These two factors may contribute to the inability to culture the organism in vitro. T. pallidum is an effective pathogen because it disseminates widely and rapidly after infection. It is in the bloodstream within hours of intratesticular injection and in numerous organs, including the brain, within 18 hours after inoculation. Once the organisms reach a tissue, they are able to persist for decades. In each tissue, the number of organisms is very low, perhaps below a “critical antigenic mass.” In addition, T. pallidum expresses very few antigenic targets on its surface (only about 1% as many as Escherichia coli ). The outer membrane proteins of T. pallidum also undergo rapid mutation, so that during an infection, the host accumulates numerous subpopulations of organisms with different surface antigens. This low infection load, widespread dissemination, poor surface antigen expression, and rapid evolution of antigenically distinct subpopulations may allow the infection to persist despite the development by the host of antigen-specific antibodies and immune cells.
Syphilis remains a major health problem worldwide, despite a highly effective and economical treatment for more than 50 years. The story of the U.S. and world epidemiology of syphilis illustrates a movement of infection from one population to another due to changing social conditions and behaviors. Just as the health systems respond to one epidemic, another appears. Using serologic testing, contact tracing, and penicillin treatment, U.S. health departments reduced the incidence of syphilis dramatically from the turn of the 20th century through the mid-1950s. The incidence then gradually increased through the next two decades and into the 1980s. In the early 1980s half the cases of syphilis diagnosed were in men who have sex with men (MSM). Changes in sexual behavior patterns among gay men in response to the acquired immunodeficiency syndrome (AIDS) epidemic reduced the number of these cases, but in the late 1980s syphilis again began to increase dramatically, associated with drug use, especially crack cocaine. The incidence of syphilis increased disproportionately among socioeconomically disadvantaged minority populations, especially in major cities. Throughout the 1990s the rate of syphilis fell in the United States, so that by 1999 the national rate of 2.6 cases in 100,000 population was the lowest level ever recorded. In addition, half of new cases were concentrated in 28 counties, mainly in the southeastern United States and in select urban areas. With the advent of effective antiretroviral therapy for HIV, there was a change in sexual behavior in MSM, including those with HIV infection. Epidemics of syphilis in this group have now occurred in many major North American, European, and Asian cities. This epidemic is characterized by an older average age, anonymous sex partners (often met on the Internet), use of amphetamines and sildenafil citrate (Viagra), HIV-positive status, and oral sex as the sole sexual exposure. In addition, there was a syphilis epidemic in Russia and the newly independent states starting in the late 1990s, with rates of syphilis 34 times that of Western Europe. Beginning in the mid-1990s, China had a syphilis epidemic affecting primarily unmarried men, female sex workers, and MSM, so that in 2008 one province in China (Guangdong) had more syphilis cases than the whole European Union.
Worldwide, an estimated 12 million persons are infected annually with syphilis, 2 million of whom are pregnant women. The U.S. Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) have undertaken campaigns to eradicate syphilis. The shifting epidemiology of syphilis over more than five decades suggests it will not be an easy task without an effective vaccine. Until then, reporting of all cases to public health departments for tracing and treatment of contacts should be continued, along with widespread screening of persons at risk, including all pregnant women, female sex workers, MSM, and men with HIV infection.
Serologic Tests
Serologic testing for infection with T. pallidum, as in tuberculosis, is undergoing changes that incorporate newer technologies into establishing the diagnosis. Tests are considered either “treponemal” or “nontreponemal.” Treponemal tests detect specific antitreponemal antibodies by enzyme immunoassay (EIA) or T. pallidum particle assay (TPPA). These new treponemal tests have specificity and sensitivity exceeding 95%, even in patients with primary syphilis. These generate more positive tests that require further testing than do older strategies. Nontreponemal tests are based on the fact that serum of persons with syphilis aggregates a cardiolipin-cholesterol-lecithin antigen. This aggregation can be viewed directly in tubes or on cards or slides, or it can be examined in an autoanalyzer. Because these tests use lipoidal antigens rather than T. pallidum or its components, they are called nontreponemal antigen tests. The most widely used nontreponemal tests are the rapid plasma reagin (RPR) and Venereal Disease Research Laboratories (VDRL) tests. These nontreponemal tests are the standard tests used in the United States and generally become positive within 5–6 weeks of infection, shortly before the chancre heals. Tests are usually strongly positive throughout the secondary phase, except in rare patients with AIDS, whose response is less predictable, and usually become negative during therapy, especially if therapy is begun within the first year of infection. Results may also become negative after a few decades, even without treatment. EIA tests are available that detect both immunoglobulin G (IgG)– and immunoglobulin M (IgM)–specific antibodies against T. pallidum. The IgM becomes detectable 2–3 weeks after infection, about the time the chancre appears. The IgG test becomes positive at 4–5 weeks; thus the IgM test is much more useful in diagnosing primary syphilis. The “treponemal” tests used in the United States are the microhemagglutination assay for T. pallidum (MHA-TP) or the fluorescent treponemal antibody absorption (FTA-ABS) test. These specific treponemal tests are also positive earlier than the nontreponemal tests and may be used to confirm the diagnosis of primary syphilis in a patient with a negative RPR/VDRL. The EIA, TPPA, FTA-ABS, and MHA-TP remain positive for life in the majority of patients, although in 13%–24% of patients these tests will become negative with treatment, regardless of stage and HIV status. The IgM EIA test, however, becomes negative after treatment in early syphilis, so that at 1 year 92% of these patients are negative on the IgM EIA.
All these tests can have false-positive results, so all positive results are confirmed by another test. In most U.S. cities, this involves screening the patient with a nontreponemal test, usually an RPR, and confirming all positives with a specific treponemal test, usually an MHA-TP. If a treponemal test, such as the TPPA or EIA, is used for initial screening, either a nontreponemal test or the other, specific treponemal test should be used to confirm the first test. A nontreponemal RPR/VDRL is also performed on all positives to determine the titer and monitor treatment success. If the initial screening treponemal-specific test is positive, but the nontreponemal test is negative, a history of prior syphilis and treatment should be sought. If prior syphilis and adequate treatment can be documented, and if examination shows no evidence of either primary or late syphilis, the patient is followed and considered serofast after treatment. If the nontreponemal test is negative, but a second treponemal test is positive, and if no prior history of syphilis and its treatment can be found, the patient is considered to have late latent syphilis (less likely, recent infection) and is treated appropriately. This patient is considered noninfectious. If the two treponemal tests are discordant, one positive and the other negative, a third treponemal-specific test can be ordered, or the case can be referred to a public health department for expert consultation. Because the nontreponemal tests are falsely negative in 25% or more of patients with primary syphilis and in up to 40% with late syphilis, in these patients a specific treponemal test should also be performed as a screening test.
In resource-poor countries, serologic testing for syphilis is largely unavailable because reagents require refrigeration or the tests require electrical equipment for processing. In Bangladesh and in some countries in sub-Saharan Africa and South America, more than 75% of women receive prenatal care, but only about 40% receive prenatal syphilis screening. Syphilis is endemic in these regions, with infection rates in pregnant women exceeding 1%, and thus millions of pregnant women with syphilis go undiagnosed. More than half a million babies die of congenital syphilis in sub-Saharan Africa every year. New, rapid treponemal-specific tests that can be used in these resource-poor countries have been developed. They cost only $0.31–$0.41 (U.S.) per test and await available funding to be put into use.
Nontreponemal tests are very valuable in following the efficacy of treatment in syphilis. By diluting the serum serially, the strength of the reaction can be stated in dilutions; the number given is the highest dilution giving a positive test result. In primary infection, the titer may be only 1 : 2; in secondary syphilis, it is regularly high, 1 : 32 to 1 : 256, or higher; in late syphilis, generally much lower, perhaps 1 : 4 or 1 : 8. The rise of titer in early infection is of great potential diagnostic value, as is the fall after proper treatment or the rise again if there is reinfection or relapse. Patients with very high antibody titers, as occur in secondary syphilis, may have a false-negative result when undiluted serum is tested. This “prozone” phenomenon will be overcome by diluting the serum.
Biologic False-Positive Test Results
“Biologic false-positive” (BFP) test result is used to denote a positive serologic test for syphilis in persons with no history or clinical evidence of syphilis. The term BFP is usually applied to the situation of a positive nontreponemal test and a negative treponemal test. About 90% of BFP test results are of low titer (<1 : 8). “Acute” BFP reactions are defined as those that revert to negative in less than 6 months; those that persist for more than 6 months are categorized as “chronic.” Acute BFP reactions may result from vaccinations, infections (infectious mononucleosis, hepatitis, measles, typhoid, varicella, influenza, lymphogranuloma venereum, malaria), and pregnancy. Chronic BFP reactions are seen in connective tissue diseases, especially systemic lupus erythematosus (SLE) (44%), chronic liver disease, multiple blood transfusions/intravenous drug use, and advancing age.
False-positive results to specific treponemal tests are less common but have been reported to occur in lupus erythematosus, drug-induced lupus, scleroderma, rheumatoid arthritis, smallpox vaccination, pregnancy, other related treponemal infections (see next), and genital herpes simplex infections. A pattern of beaded fluorescence associated with FTA-ABS testing may be found in the sera of patients without treponemal disease who have SLE. The beading phenomenon, however, is not specific for SLE or even for connective tissue diseases.
Cutaneous Syphilis
Chancre (Primary Stage)
The chancre is usually the first cutaneous lesion, appearing 18–21 days after infection. The typical incipient chancre is a small red papule or a crusted superficial erosion. In a few days to weeks, it becomes a round or oval, indurated, slightly elevated papule, with an eroded but not ulcerated surface that exudes a serous fluid ( Fig. 18.1 ). On palpation, it has a cartilage-like consistency. The lesion is usually, but not invariably, painless. This is the uncomplicated or classic hunterian chancre. The regional lymph nodes on one or both sides are usually enlarged, firm, and nontender and do not suppurate. Adenopathy begins 1 or 2 weeks after the chancre appears. The hunterian chancre leaves no scar when it heals.
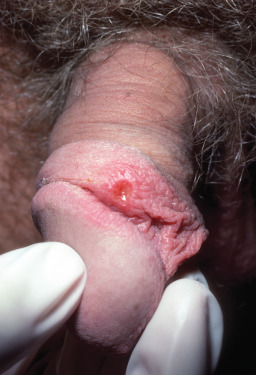
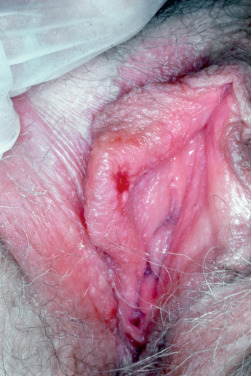
Chancres generally occur singly, although they may be multiple, especially on the penis in MSM who are infected through oral intercourse. The lesions vary in diameter from a few millimeters to several centimeters. The genital chancre is less often observed in women because of its location within the vagina or on the cervix ( Fig. 18.2 ). Extensive edema of the labia or cervix may occur. In men, the chancre is commonly located in the coronal sulcus or on either side of the frenum. A chancre in the prepuce, being too hard to bend, will flip over all at once when the prepuce is drawn back, a phenomenon called a “dory flop” (resembling a broad-beamed skiff or dory being turned upside down). Untreated, the chancre tends to heal spontaneously in 1–4 months. About the time it disappears, or slightly before, constitutional symptoms and objective signs of generalized (secondary) syphilis occur ( Fig. 18.3 ).
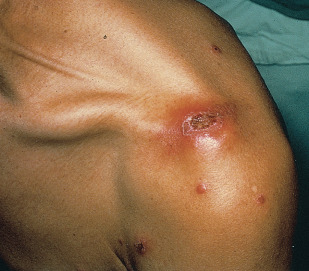
Extragenital chancres may be larger than those on the genitalia. They affect the lips, tongue, tonsil, female breast, index finger, and, especially in MSM, the anus. Oral chancres form firm, eroded papules on the lip, tongue, uvula, or tonsillar pillar and are associated with a history of oral sex. Unilateral cervical adenopathy can be present. The presenting complaints of an anal chancre include an anal sore or fissure and irritation or bleeding on defecation. Anal chancre must be ruled out in any anal fissure not at the 6 or 12 o’clock positions. When there is a secondary eruption, no visible chancre, and the glands below Poupart ligament are greatly enlarged, anal chancre should be suspected.
Atypical chancres are common. Simultaneous infection by a spirochete and another microbial agent may produce an atypical chancre. The mixed chancre caused by infection with Haemophilus ducreyi and Treponema pallidum will produce a lesion that runs a course different from either chancroid or primary syphilis alone. Such a sore begins a few days after exposure, because the incubation period for chancroid is short, and later the sore may transform into an indurated syphilitic lesion. A phagedenic chancre results from the combination of a syphilitic chancre and contaminating bacteria that may cause severe tissue destruction and result in scarring. Edema indurativum, or penile venereal edema, is marked solid edema of the labia or the prepuce and glans penis accompanying a chancre. Chancre redux is relapse of a chancre with insufficient treatment, accompanied by enlarged lymph nodes. Pseudochancre redux is a gumma occurring at the site of a previous chancre; it is distinguished from relapsing chancre by the absence of lymphadenopathy and a negative darkfield examination. Syphilitic balanitis of Follmann may occur in the absence of a chancre. The lesions may be exudative, circinate, or erosive.
Histologic evaluation of a syphilitic chancre reveals an ulcer covered by neutrophils and fibrin. Subjacent, there is a dense infiltrate of lymphocytes and plasma cells. Blood vessels are prominent with plump endothelial cells. Spirochetes are numerous in untreated chancres and can be demonstrated with an appropriate silver stain, such as the Warthin-Starry, Levaditi, or Steiner methods, or by immunoperoxidase staining. Spirochetes are best found in the overlying epithelium or adjacent or overlying blood vessels in the upper dermis.
In a patient who presents with an acute genital ulceration, darkfield examination should be performed if available. The finding of typical T. pallidum in a sore on the cutaneous surface establishes a diagnosis of syphilis. Treponema pertenue, which causes yaws, and Treponema carateum, which causes pinta, are both indistinguishable morphologically from T. pallidum, but the diseases that they produce are usually easy to recognize. Commensal spirochetes of the oral mucosa are indistinguishable from T. pallidum, making oral darkfield examinations unreliable. If the darkfield examination results are negative, the examination should be repeated daily for several days, especially if the patient has been applying a topical antibacterial agent.
The lesion selected for examination is cleansed with water and dried. It is grasped firmly between the thumb and index finger and abraded sufficiently to cause clear or faintly blood-stained plasma to exude when squeezed. In the case of an eroded chancre, a few vigorous rubs with dry gauze are usually sufficient. If the lesion is made to bleed, it is necessary to wait until free bleeding has stopped to obtain satisfactory plasma. The surface of a clean coverslip is touched to the surface of the lesion so that plasma adheres. Then it is dropped on a slide and pressed down so that the plasma spreads out in as thin a film as possible. Immersion oil forms the interface between the condenser and slide and between the coverslip and objective. The specimen must be examined quickly, before the thin film of plasma dries.
An alternative to darkfield microscopy is the direct fluorescent antibody test (DFAT-TP) for the identification of T. pallidum in lesions. Serous exudate from a suspected lesion is collected as just described, placed on a slide, and allowed to dry. Many health departments will examine such specimens with fluorescent antibodies specific to T. pallidum. The method, unlike the darkfield examination, can be used for diagnosing oral lesions. Multiplex polymerase chain reaction (PCR) is also an accurate and reproducible method for diagnosing genital ulcerations, with the advantage of being able to diagnose multiple infectious agents simultaneously. In genital ulcer disease outbreaks, PCR should be made available.
The results of serologic tests for syphilis are positive in 75% (nontreponemal tests) to 90% (treponemal tests) of patients with primary syphilis; both these tests should be performed in every patient with suspected primary syphilis. The likelihood of positivity depends on the duration of infection. If the chancre has been present for several weeks, test results are usually positive.
A syphilitic chancre must be differentiated from chancroid. The chancre has an incubation period of 3 weeks; is usually a painless erosion, not an ulcer; has no surrounding inflammatory zone; and is round or oval. The edge is not undermined, and the surface is smooth and at the level of the skin. It has a dark, velvety red, lacquered appearance; it has no overlying membrane; and it is cartilage hard on palpation. Lymphadenopathy may be bilateral and is nontender and nonsuppurative. Chancroid, on the other hand, has a short incubation period of 4–7 days; the ulcer is acutely inflamed, is extremely painful, and has a surrounding inflammatory zone. The ulcer edge is undermined and extends into the dermis. It is covered by a membrane and feels soft. Lymphadenopathy is usually unilateral and tender and may suppurate. Chancroid lesions are usually multiple and extend into each other. Cultures for chancroid on special media confirm the diagnosis. However, because a combination of a syphilitic chancre and chancroid (mixed sores) is indistinguishable from chancroid alone, appropriate direct and serologic testing should be performed to investigate the presence of syphilis. Again, multiplex PCR allows for the simultaneous diagnosis of many infectious agents in genital ulcer diseases.
The primary lesion of granuloma inguinale begins as an indurated nodule that erodes to produce hypertrophic, vegetative granulation tissue. It is soft and beefy-red and bleeds readily. A smear of clean granulation tissue from the lesion stained with Wright or Giemsa reveals Donovan bodies in the cytoplasm of macrophages.
The primary lesion of lymphogranuloma venereum (LGV) is usually a small, painless, transient papule or a superficial nonindurated ulcer. It most often occurs on the coronal sulcus, prepuce, or glans in men or on the fourchette, vagina, or cervix in women. A primary genital lesion is noticed by about 30% of infected heterosexual men, but less frequently in women. Primary lesions are followed in 7–30 days by adenopathy of the regional lymph nodes. LGV is confirmed by serologic tests.
Herpes simplex begins with grouped vesicles, often accompanied or preceded by burning pain. After rupture of the vesicles, irregular, scalloped, tender, soft erosions form.
Secondary Syphilis
Cutaneous Lesions.
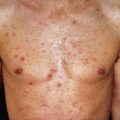
The skin manifestations of secondary syphilis occur in 80% or more of patients with secondary syphilis. The early eruptions are symmetric, more or less generalized, superficial, nondestructive, exanthematous, transient, and macular; later they are maculopapular or papular eruptions, which are usually polymorphous, and less often, scaly, pustular, or pigmented. The early manifestations tend to be distributed over the face, shoulders, flanks, palms and soles, and anal or genital regions. The severity varies widely. The presence of lesions on the palms and soles is strongly suggestive. However, a generalized syphilid can spare the palms and soles. The individual lesions are generally less than 1 cm in diameter, except in the later secondary or relapsing secondary eruptions.
Macular Eruptions.
The earliest form of macular secondary syphilis begins with the appearance of an exanthematous erythema 6–8 weeks after the development of the chancre, which may still be present. The syphilitic exanthem extends rapidly, so that it is usually pronounced a few days after onset. It may be evanescent, lasting only a few hours or days, or it may last several months, or partially recur after having disappeared. This macular eruption appears first on the sides of the trunk, about the navel, and on the inner surfaces of the extremities.
Individual lesions of macular secondary syphilis consist of round, indistinct macules that are nonconfluent and rarely may be slightly elevated or urticarial. The color varies from a light pink or rose to brownish red. The macular eruption may not be noticed on black skin and may be so faint that it is also not recognized on other skin colors. Pain, burning, and itching are usually absent, although pruritus may be present in 10%–40% of patients. Simultaneous with the onset of the eruption, there is a generalized shotty adenopathy, most readily palpable in the posterior cervical, axillary, and epitrochlear areas. Rarely, secondary syphilis may cause livedo reticularis. The macular eruption may disappear spontaneously after a few days or weeks without residua or may result in postinflammatory hyperpigmentation. After a varying interval, macular syphilis may be followed by other eruptions.
Papular Eruptions.
Papular eruptions usually arise slightly later than the macular eruption. The fully developed lesions are round and of a raw-ham or coppery shade ( Fig. 18.4 ). Although papules most frequently are 2–5 mm in diameter, nodules coalescing to large plaques can occur. Lesions often are only slightly raised, but a deep, firm infiltration is palpable. The surface is smooth, sometimes shiny, and at other times covered with a thick, adherent scale. When this desquamates, it leaves a characteristic collarette of scales overhanging the border of the papule.
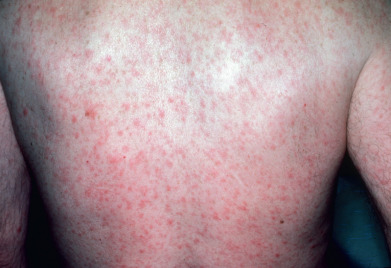
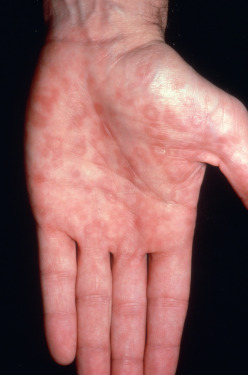
Papules are frequently distributed on the face and flexures of the arms and lower legs and are often distributed all over the trunk ( Fig. 18.5 ). Palmar and plantar involvement characteristically appears as indurated, yellowish red spots ( Fig. 18.6 ). Ollendorf (Buschke-Ollendorff) sign is present; the papule is exquisitely tender to the touch of a blunt probe. Healing lesions frequently leave hyperpigmented spots that, especially on the palms and soles, may persist for weeks or months. Split papules are hypertrophic, fissured papules that form in the creases of the alae nasi and at the oral commissures. These may persist for a long period. The papulosquamous syphilids, in which the adherent scales covering the lesions more or less dominate the picture, may produce a psoriasiform eruption. Follicular or lichenoid syphilids appear as minute, scale-capped papules. If they are at the ostia of hair follicles, syphilids are likely to be conical; elsewhere on the skin, they are domed. Often, syphilids are grouped to form scaling plaques in which the tiny, coalescing papules are still discernible.
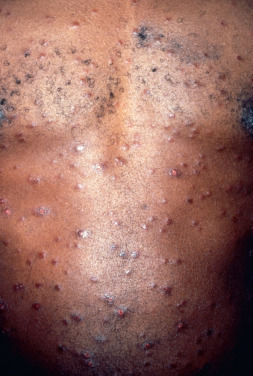
As with the other syphilids, papular eruptions tend to be disseminated but may also be localized, asymmetric, configurate, hypertrophic, or confluent. The arrangement may be corymbose or in patches, rings, or serpiginous patterns.
The annular syphilid, as with sarcoidosis, which it may mimic, is more common in blacks ( Fig. 18.7 ). It is often located on the cheeks, especially close to the angle of the mouth, where it may form annular, arcuate, or gyrate patterns of delicate, slightly raised, infiltrated, finely scaling ridges. These ridges are made up of minute, flat-topped papules, and the boundaries between ridges may be difficult to discern. An old term for annular syphilids was “nickels and dimes.”
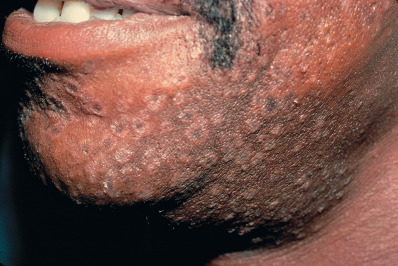
The corymbose syphilid is another infrequent variant, usually occurring late in the secondary stage, in which a large central papule is surrounded by a group of tiny satellite papules. The pustular syphilids are among the rarer manifestations of secondary syphilis. They occur widely scattered over the trunk and extremities, but they usually involve the face, especially the forehead. The pustule usually arises on a red, infiltrated base. Involution is usually slow, resulting in a small, rather persistent, crust-covered, superficial ulceration. Lesions in which the ulceration is deep are called ecthymatous. Closely related is the rupial syphilid, a lesion in which a relatively superficial ulceration is covered with a pile of terraced crusts resembling an oyster shell. Lues maligna is a rare form of secondary syphilis with severe ulcerations, pustules, or rupioid lesions, accompanied by severe constitutional symptoms. This form of secondary syphilis appears to be more common in HIV-infected men.
Involvement of the palms and soles is a characteristic feature of secondary syphilis. In some cases, instead of discrete lesions, the whole area of the palms and soles can be symmetrically involved, resembling keratoderma blennorrhagicum, hyperkeratotic hand eczema, or even an acquired keratoderma, such as Howel-Evans syndrome. Similarly, cutaneous lesions can be very psoriasiform, and if they develop in a person with known psoriasis, lesions can be mistaken for a flare of that disease. Anetoderma may occur after treatment of secondary syphilis.
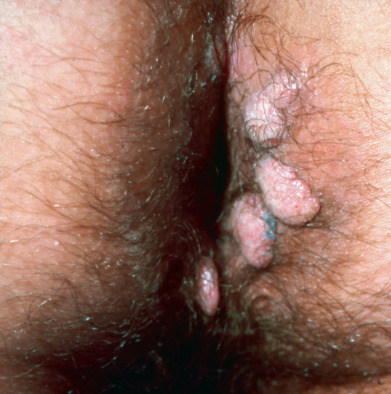
Condylomata lata are papular lesions, relatively broad and flat, located on folds of moist skin, especially around the genitalia and anus, but also at the angles of the mouth, nasolabial fold, and toe webs. They may become hypertrophic and, instead of infiltrating deeply, protrude above the surface, forming a soft, red, often mushroom-like mass 1–3 cm in diameter, usually with a smooth, moist, weeping, gray surface ( Fig. 18.8 ). Condyloma lata may be lobulated but are not covered by the digitate elevations characteristic of venereal warts (condylomata acuminata). Secondary syphilis may initially present with perianal erosions and plaques that may mimic cutaneous Crohn disease.
Syphilitic alopecia is irregularly distributed so that the scalp has a moth-eaten appearance. It is unusual, occurring in about 5% of patients with secondary syphilis. Smooth, circular areas of alopecia mimicking alopecia areata may occur in syphilis, and an ophiasis pattern may rarely be seen.
Mucous membrane lesions are present in one third of patients with secondary syphilis and may be the only manifestation of the infection. The most common mucosal lesion in the early phase is the syphilitic sore throat, a diffuse pharyngitis that may be associated with tonsillitis or laryngitis. Hoarseness and sometimes complete aphonia may be present. On the tongue, smooth, small or large, well-defined patches devoid of papillae may be seen, most frequently on the dorsum near the median raphe ( Fig. 18.9 ). Ulcerations may occur on the tongue and lips during the late secondary period, at times resembling aphthae or major aphthae. A rare variant of syphilis is one presenting with oral and cutaneous erosions that histologically show the features of pemphigus vulgaris, with a suprabasilar acantholytic blister, as well as positive direct and indirect immunofluorescence findings of pemphigus.
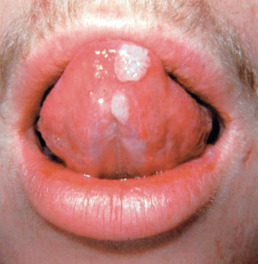
Mucous patches are the most characteristic mucous membrane lesions of secondary syphilis. They are macerated, flat, grayish, rounded erosions covered by a delicate, soggy membrane. These highly infectious lesions are about 5 mm in diameter and teem with treponemas. They occur on the tonsils, tongue, pharynx, gums, lips, and buccal areas or on the genitalia, chiefly in women, in whom the lesions are most common on the labia minora, vaginal mucosa, and cervix. Such mucous erosions are transitory and change from week to week, or even from day to day. Lesions of the oral mucosa frequently contain plasma cells, so the oral pathologist may not consider syphilis. Because the oral lesions of secondary syphilis are usually teeming with spirochetes, a T. pallidum –specific immunoperoxidase stain is useful in confirming the diagnosis.
Relapsing Secondary Syphilis.
The early lesions of syphilis undergo involution either spontaneously or with treatment. Relapses occur in about 25% of untreated patients, 90% within the first year. Such relapses may take place at the site of previous lesions, on the skin or in the viscera. Recurrent eruptions tend to be more configurate or annular, larger, and asymmetric.
Systemic Involvement.
The lymphatic system in secondary syphilis is characteristically involved. The lymph nodes most frequently affected are the inguinal, posterior cervical, postauricular, and epitrochlear. The nodes are shotty, firm, slightly enlarged, nontender, and discrete.
Acute glomerulonephritis, gastritis or gastric ulceration, proctitis, hepatitis, acute meningitis, unilateral sensorineural hearing loss, iritis, anterior uveitis, optic neuritis, Bell palsy, multiple pulmonary nodular infiltrates, periostitis, osteomyelitis, polyarthritis, and tenosynovitis may all be seen in secondary syphilis.
Histopathology
Lesions of secondary syphilis may demonstrate neutrophils within the stratum corneum, elongation of rete ridges, interface dermatitis with a lymphocyte in every vacuole, an interstitial infiltrate (busy dermis), perivascular plasma cells, lymphocytes with visible amphophilic cytoplasm, and endothelial swelling (plump endothelial cells with absence of a lumen). Plasma cells are absent about 30% of cases, and many cases lack other characteristic features as well. Examples of syphilis with fewer histologic features are more likely to demonstrate effacement of the rete pattern rather than elongation of the rete ridges. A high index of suspicion is required, and any combination of interface dermatitis, interstitial inflammation, endothelial swelling, lymphocytes with visible cytoplasm, or plasma cells should raise the possibility of secondary syphilis. As lesions age, macrophages become more numerous, so that in late secondary lues, granulomatous foci are often present, mimicking sarcoidosis, or less often, a granuloma annulare–like pattern. Condylomata lata show spongiform pustules within areas of papillated epithelial hyperplasia, and spirochetes are numerous.
In early-syphilis skin lesions, spirochetes are most numerous within the epidermis and around superficial vessels. Silver stains are technically difficult, but because the number of organisms is high in early syphilis, the tests are usually positive. PCR and immunoperoxidase assay may identify T. pallidum infection when silver stains are negative. However, immunoperoxidase stains may be negative and silver stains positive; therefore if suspicion of early syphilis is high, both silver stains and immunoperoxidase assays may need to be performed.
Diagnosis and Differential Diagnosis
Syphilis has long been known as the “great imitator,” because the various cutaneous manifestations may simulate almost any cutaneous or systemic disease. Pityriasis rosea may be mistaken for secondary syphilis, especially because both begin on the trunk. The herald patch, the oval patches with a fine scale at the edge, patterned in the lines of skin cleavage, the absence of lymphadenopathy, and infrequent mucous membrane lesions help to distinguish pityriasis rosea from secondary syphilis. Drug eruptions may produce a similar picture to secondary syphilis but tend to be morbilliform and also pruritic, whereas secondary syphilis is not. Drug eruptions in pityriasis rosea are often pruritic, whereas those in secondary syphilis usually are not.
Lichen planus may resemble papular syphilid. The characteristic papule of lichen planus is flat topped and polygonal, has Wickham striae, and exhibits Koebner phenomenon. Pruritus is severe in lichen planus but is less common and less severe in syphilis. Psoriasis may be distinguished from papulosquamous secondary syphilis by the presence of adenopathy, mucous patches, and alopecia in the latter. Sarcoidosis may produce lesions morphologically identical to secondary syphilis. Histologically, multisystem involvement, adenopathy, and granulomatous inflammation are common to both diseases. Serologic testing and biopsy specimens will distinguish sarcoidosis from syphilis.
The differential diagnosis of mucous membrane lesions of secondary syphilis is of importance. Infectious mononucleosis may cause a biologic false-positive test for syphilis but is diagnosed by serology. Geographic tongue may be confused with the desquamative patches of syphilis or with mucous patches. Lingua geographica occurs principally near the edges of the tongue in relatively large areas, which are often fused and have lobulated contours. It continues for several months or years and changes in extent and degree of involvement from day to day. Recurrent aphthous ulceration produces one or several painful ulcers, 1–3 mm in diameter, surrounded by hyperemic edges, with a grayish covering membrane, on nonkeratinized mucosal epithelium, especially in the gingival sulcus. A prolonged, recurrent history is characteristic. Syphilis of the lateral tongue may resemble oral hairy leukoplakia.
Latent Syphilis
After the lesions of secondary syphilis have involuted, a latent period occurs. This may last for a few months or continue for the remainder of the infected person’s life. Between 60% and 70% of untreated infected patients remain latently asymptomatic for life. During this latent period, there are no clinical signs of syphilis, but the serologic tests for syphilis are reactive. During the early latent period, infectivity persists; for at least 2 years, a woman with early latent syphilis may infect her unborn child. For treatment purposes, it is important to distinguish early latency (<1-year duration) from late latency (>1 year or unknown duration).
Late Syphilis
For treatment purposes, late syphilis is defined by the CDC as infection of more than 1 year in duration and by the WHO as more than 2 years in duration. Only about one third of patients with late syphilis will develop complications of their infection.
Tertiary Cutaneous Syphilis
Tertiary syphilids most often occur 3–5 years after infection. About 16% of untreated patients will develop tertiary lesions of the skin, mucous membrane, bone, or joints. Skin lesions tend to be localized, to occur in groups, to be destructive, and to heal with scarring. Treponemas are usually not found by silver stains or darkfield examination but may be demonstrated by immunoperoxidase techniques.
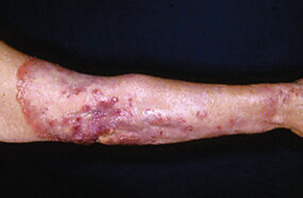
Two main types of cutaneous tertiary syphilis are recognized, the nodular syphilid and the gumma, although the distinction is sometimes difficult to make. The nodular, noduloulcerative, or tubercular type consists of firm, reddish brown or copper-colored papules or nodules, 2 mm in diameter or larger. The individual lesions are usually covered with adherent scales or crusts ( Fig. 18.10 ). The lesions tend to form rings and to undergo involution as new lesions develop just beyond them, producing characteristic circular or serpiginous patterns. A distinctive type is the kidney-shaped lesion, which typically occurs on the extensor surfaces of the arms and on the back. Individual lesions are composed of nodules in different stages of development, so that scars and pigmentation often are found together with fresh as well as ulcerated lesions. On the face, the nodular eruption closely resembles lupus vulgaris. When the disease is untreated, the process may last for years, slowly marching across large areas of skin. The nodules may enlarge and eventually break down to form painless, rounded, smooth-bottomed ulcers a few millimeters deep. These punched-out ulcers arise side by side and form serpiginous syphilitic ulcers, palm sized in aggregate, enduring for many years ( Fig. 18.11 ).