Tuberculosis
No ideal classification scheme exists for cutaneous tuberculosis (TB), but the system listed here is logical and takes into account the mechanism of disease acquisition. Unfortunately, unlike in Hansen disease, these categories do not correlate perfectly to host immunity. The four major categories of cutaneous TB are as follows:
- 1.
Inoculation from an exogenous source (primary inoculation TB, TB verrucosa cutis)
- 2.
Endogenous cutaneous spread contiguously or by autoinoculation (scrofuloderma, TB cutis orificialis)
- 3.
Hematogenous spread to the skin (lupus vulgaris; acute miliary TB; TB ulcer, gumma, or abscess; tuberculous cellulitis) (Lupus vulgaris can also occur adjacent to lesions of scrofuloderma, suggesting that both hematogenous spread and local spread are capable of triggering this reaction pattern.)
- 4.
Tuberculids (erythema induratum [Bazin disease], papulonecrotic tuberculid, lichen scrofulosorum)
The finding of mycobacterial deoxyribonucleic acid (DNA) by polymerase chain reaction (PCR) in tuberculids suggests that tuberculids also represent hematogenous dissemination of TB, which is quickly controlled by the host, usually resulting in the absence of detectable organisms by culture and histologic methods. Miliary TB is the form with least effective host immunity. Tuberculous ulcer/abscess/cellulitis and TB cutis orificialis are conditions of poor host immunity against Mycobacterium tuberculosis. Bacilli are prominent in these forms of cutaneous TB, and histologic and microbiologic confirmation is usually straightforward. This is fortunate, because cellular-based diagnostic modalities (purified protein derivative [PPD], interferon-γ release assay [IGRA]) may be negative. TB verrucosa cutis and lupus vulgaris are conditions of high host immunity to TB, and tuberculin skin tests and IGRA for TB will usually be positive. Scrofuloderma is usually associated with a positive PPD, and identification by culture and histologic methods is positive in only 20% and 40% of cases, respectively. In its initial stage, primary inoculation TB will be multibacillary and culture positive. As host immunity develops, the skin test becomes positive, and the number of organisms on biopsy diminishes. The tuberculids also represent high host immune response manifestations of TB, and bacilli are rarely found.
Epidemiology
The increase in the numbers of cases of TB that started in the mid-1980s in the United States was associated with three phenomena: large numbers of immigrants from high-prevalence countries, the human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS) epidemic, and an increasing number of persons in congregative facilities (shelters for homeless persons, prisons). Asians, African Americans, and Hispanics have the greatest risk for developing TB in the United States. Aggressive diagnosis and treatment programs have led to a reduction in new U.S. cases of TB. The infection rate in the U.S.-born population of adults is now 3 cases per 100,000 population. However, local pockets of TB are still found in U.S. regions of otherwise very low incidence. This is partly attributable to the persistently high infection rate in the foreign-born U.S. population, which has also fallen dramatically over the years, but is still at 15 cases per 100,000.
In the developing world, TB is the number one cause of death resulting from a single infectious agent. The increasing number of infections has been driven largely by the HIV/AIDS epidemic. One third or more of HIV-infected persons in Africa are also infected with M. tuberculosis. Latent TB is 100 times more likely to reactivate in persons with HIV infection, and HIV-infected persons are much more likely to acquire new tuberculous infection. Countries such as India still account for a quarter of the total global TB burden, although great progress has been made. Hematopoietic stem cell transplantation and solid organ transplant recipients are particularly prone to TB, at a rate of 10 to 70 times the general population.
TB has increasingly become resistant to first-line treatments. Strains classified as multidrug-resistant tuberculosis (MDR-TB) are resistant to at least isoniazid and rifampin. Extensively drug-resistant tuberculosis (XDR-TB) is, in addition, resistant to any fluoroquinolone and at least one of the following: capreomycin, kanamycin, or amikacin. The emergence of these resistant strains of TB has made treatment more costly and more difficult. However, aggressive treatment protocols using multiple drugs for up to 2 years and, when indicated, surgical techniques can cure up to 60% of even XDR-TB patients.
Cutaneous TB is an uncommon complication of tuberculous infection, with less than 2% of TB patients having skin lesions, even in highly endemic areas. The types of cutaneous lesion that the patient will develop depend on the following host factors:
- 1.
Age: About 25% of scrofuloderma cases and most cases of lichen scrofulosorum occur in children.
- 2.
Gender: Women are 10 times more likely to develop erythema induratum, but men are two to three times more likely to have other forms of cutaneous TB.
- 3.
Anatomic location: Lupus vulgaris occurs on the face and extremities, whereas TB verrucosa cutis occurs predominantly on the hands.
- 4.
Nutritional status: Tuberculous abscesses and scrofuloderma are associated with malnutrition.
The pattern of cutaneous TB has been changing over the last few decades and is different in developed than in developing nations. The average age of patients with cutaneous TB has increased in developed countries, and tuberculids, especially erythema induratum, represent a larger proportion of cases. In Hong Kong 85% of cases of cutaneous TB are tuberculids. This suggests that most cutaneous TB in adults will be found in patients infected in the distant past who are reactivating their disease, not recently infected persons. Cutaneous TB is uncommon in immunosuppressed hosts; when they acquire new TB or reactivate their TB, it usually reactivates at a noncutaneous site and is diagnosed before skin disease occurs. Miliary TB is the most frequently reported form of cutaneous TB in the HIV-infected patient. In areas of high TB endemicity in the developing world, cutaneous TB is still common. More than 50% of cases will occur before age 19. The likelihood of finding associated systemic TB is higher in children than adults. Nonetheless, unlike in all other forms of extrapulmonary TB, failure to find an underlying focus of TB in patients with cutaneous TB can occur. Between 3% and 12% of patients with cutaneous TB will have an abnormal chest radiograph. Most often, TB of the lymph nodes will be found.
Tuberculin Testing
The tuberculin skin test (TST) is designed to detect a memory cell–mediated immune response to M. tuberculosis. The test becomes positive 2–10 weeks after infection and remains positive for many years, although it may wane with age. PPD preparations are currently used for testing in the United States and Canada at a dose of 5 tuberculin units (TU). The intradermal, or Mantoux, test is the standard and offers the highest degree of consistency and reliability. The test is read 48–72 hours after intradermal injection. Induration measuring 5 mm or more is considered positive in HIV-infected patients, in solid organ transplantation patients, in those with risk factors for developing TB (e.g., patients who will receive or are receiving anti–tumor necrosis factor [TNF] therapy), in those on greater than 15 mg/day of prednisone for over 1 month, in patients on current cancer chemotherapy, in recent close contacts, or in those with chest x-ray findings consistent with healed TB. Because children are at increased risk of developing active TB after exposure, a 5-mm or larger reaction in contact investigations is considered positive. If the PPD measures more than 10 mm, it is considered positive in injection (intravenous) drug users (IDUs), HIV-negative IDUs, those born in foreign countries of high TB prevalence, health care workers, residents and employees in high-risk congregate facilities, and those with medical conditions that predispose to TB. If induration is more than 15 mm, it is positive in all others; 0- to 4-mm induration is negative.
The lower the threshold for positivity for the TST, the less this represents true positivity (the higher number of false positives). This is why a TST of less than 15 mm is considered positive only in patients at higher risk for having latent TB. Conversely, as the cutoff for true positivity is raised, the number of infected persons the TST detects will decrease (the number of false negatives increases). A TST of over 6 mm will detect 89% of persons with latent TB, over 10 mm will detect 75%, and over 15 mm will detect only 47% of latently infected patients. At least 7% of patients with latent TB will have a completely negative TST. Many intermediate TST responses may represent cross-reaction with atypical mycobacteria. Bacillus (bacille) Calmette-Guérin (BCG) immunization leads to a positive tuberculin result in immunized children, but this reaction usually does not persist beyond 10 years. Repeated BCG immunization or BCG administration after age 2 years is more likely to result in a persistently positive TST on this basis. However, positive reactions in adults should not automatically be attributed to childhood BCG administration.
Reactivity to the tuberculin protein is impaired in certain conditions in which cellular immunity is impaired. Lymphoproliferative disorders, sarcoidosis, corticosteroid and immunosuppressive drugs (including TNF inhibitors), severe protein deficiency, chronic renal failure, and numerous infectious illnesses, including HIV infection, are capable of diminishing tuberculin reactivity. In overwhelming TB (miliary disease), more than 50% of patients have a negative skin test before beginning therapy. A negative or doubtful reaction to a PPD preparation does not rule out TB infection, particularly in the patient with suggestive symptoms and signs.
TST then has significant limitations, which also include negative tests in a substantial number of patients with active TB (sensitivity only 77%), repeat visits to interpret, technical competence of person applying the test, booster effect of repeat testing creating potential false-positive results, and false-positive tests in persons with prior BCG vaccination. To overcome these obstacles, antigen-specific in vitro assays have been developed. These assays measure the amount of interferon (IFN)–γ released by peripheral blood T cells (IGRAs; e.g., QuantiFERON-TB Gold, ELISpot PLUS , T-SPOT). Results are variable with respect to the sensitivity and specificity of these assays, but they are valuable in certain settings. They are considerably more specific in the BCG-vaccinated population, in whom the TST is only 60% specific, whereas IGRAs are 93% specific. In addition, in HIV-infected patients and those receiving corticosteroids, IGRAs are much more likely to be positive than a TST in those with M. tuberculosis infection. In children under age 5 the TST is preferred, as it is in those requiring serial testing. In other settings either TST or IGRAs may be used. If the TST is combined with an IGRA and the tests are concordant, false-negative results are 2% and false-positive tests only 1%. Therefore a combination of TST and IGRA may be used to clarify a TST result. In such cases the two tests should be done simultaneously, as the IGRA results may be boosted in a patient who has had a TST within the prior 3 months.
Appropriate screening before initiating anti-TNF therapy or immunosuppression in a dermatology patient would include the following:
- 1.
Screen for active TB by history and physical examination (and chest x-ray where suspicion for TB is elevated).
- 2.
Administer a TST or an IGRA.
- 3.
Interpret the test results with caution in patients already on significant iatrogenic immunosuppressive or anti-TNF treatments.
- 4.
Regularly monitor patients on anti-TNF agents for the development of TB with appropriate history, physical examination, and laboratory testing; and suspect and screen for TB if clinical symptoms may indicate infection.
- 5.
Rescreen with a TST or IGRA annually.
Bacille Calmette-Guérin Vaccination
BCG is a live attenuated strain of Mycobacterium bovis used in most parts of the world (except North America and Western Europe) to immunize infants. It enhances immunity to TB and is effective in reducing childhood TB, especially if given to neonates. Once the patient has been vaccinated, the TST becomes positive and remains so for a period of less than 10 years (unless the person is BCG immunized after age 2 or repeatedly immunized). In an adult who was vaccinated as a child in a foreign country with a high prevalence of TB and whose TST measures more than 10 mm, active TB should be assumed. The use of BCG instillation in the bladder to treat bladder cancer has been associated with disseminated disease, usually pneumonitis, hepatitis, prostatitis, and abdominal aneurysms.
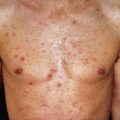
Dermatologic complications of BCG vaccination are rarely seen. Localized abscesses and regional suppurative adenitis occur at a rate of about 0.4 per 1000 vaccines. Excessive ulceration may occur if the BCG is inoculated too deeply. Scrofuloderma is rare. Disseminated infection is seen in 1–4 cases per 1 million infants vaccinated and is associated with high mortality. Disseminated BCG develops only in the setting of immunodeficiency. Lupus vulgaris can occur rarely at the vaccination site or at a distant site and will respond to appropriate antituberculous treatment. Papular and papulonecrotic tuberculids, as well as erythema induratum, can occur after BCG immunization, appearing 10 days to several months after vaccination. Treatment may not be necessary for the BCG-induced tuberculids; they frequently heal in a few months with no treatment.
Inoculation Cutaneous Tuberculosis From Exogenous Source
Primary Inoculation Tuberculosis (Primary Tuberculous Complex, Tuberculous Chancre)
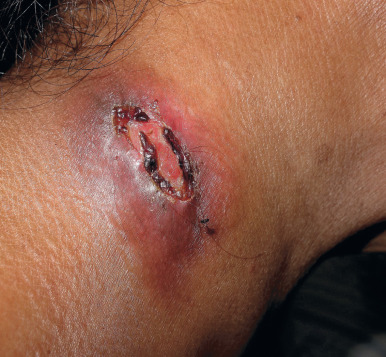
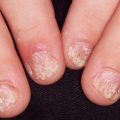
Primary inoculation TB develops at the site of inoculation of tubercle bacilli into a TB-free individual ( Fig. 16.1 ). Regional lymphadenopathy usually occurs, completing the “complex.” It occurs chiefly in children and affects the face or extremities. The inoculation can occur during tattooing, medical injections, nose piercing, or external physical trauma. The earliest lesion, appearing 2–4 weeks after inoculation, is a painless brown-red papule that develops into an indurated nodule or plaque that may ulcerate. This is the tuberculous chancre. Prominent regional lymphadenopathy appears 3–8 weeks after infection and, occasionally, suppurative and draining lesions may appear over involved lymph nodes. Primary tuberculous complex occurs on the mucous membranes in about one third of patients. Spontaneous healing usually occurs within 1 year, with the skin lesion healing first, then the lymph node, which is often persistently enlarged and calcified. Delayed suppuration of the affected lymph node, lupus vulgaris overlying the involved node, and occasionally dissemination may follow this form of cutaneous TB.
Histologically, there is a marked inflammatory response during the first 2 weeks, with many polymorphonuclear leukocyte neutrophils (PMNs) and tubercle bacilli. During the next 2 weeks, the picture changes. Lymphocytes and epithelioid cells appear and replace the PMNs. Distinct tubercles develop within 3 or 4 weeks of inoculation. Simultaneously, with the appearance of epithelioid cells, the number of tubercle bacilli decreases rapidly.
The differential diagnosis of primary inoculation TB extends over the spectrum of chancriform conditions of deep fungal or bacterial origin, such as sporotrichosis, blastomycosis, histoplasmosis, coccidioidomycosis, nocardiosis, syphilis, leishmaniasis, yaws, tularemia, and atypical mycobacterial disease. Pyogenic granuloma and cat-scratch disease must also be considered.
Paucibacillary Cutaneous Tuberculosis From Exogenous or Endogenous Source in Persons With High Immunity
Tuberculosis Verrucosa Cutis.
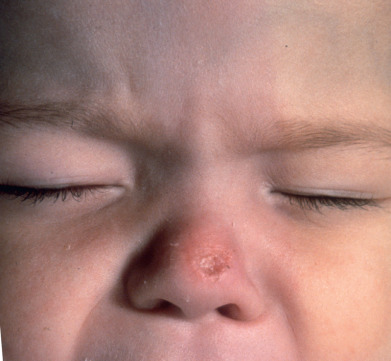
TB verrucosa cutis occurs from exogenous inoculation of bacilli into the skin of a previously sensitized person with strong immunity against M. tuberculosis. The tuberculin test is strongly positive. The prosecutor’s wart resulting from inoculation during an autopsy is the prototype of TB verrucosa cutis.
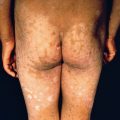
Clinically, the lesion begins as a small papule, which becomes hyperkeratotic, resembling a wart. The lesion enlarges by peripheral expansion, with or without central clearing, sometimes reaching several centimeters or more in diameter ( Fig. 16.2 ). Fissuring of the surface may occur, discharging purulent exudate. Lesions are almost always solitary, and regional adenopathy is usually present only if secondary bacterial infection occurs. Frequent locations for TB verrucosa cutis are on the dorsa of the fingers and hands in adults and the ankles and buttocks in children. The lesions are persistent but usually superficial and limited in extent. Local scarring, as seen in lupus vulgaris, can occur. Although sometimes separated by exudative or suppurative areas, the lesions seldom ulcerate and may heal spontaneously.
Histologically, there is pseudoepitheliomatous hyperplasia of the epidermis and hyperkeratosis. Suppurative and granulomatous inflammation is seen in the upper and middle dermis, sometimes perforating through the epidermis. Caseation is rare. The number of acid-fast bacilli (AFB) is usually scant, and failure to find AFB should not be used to exclude the diagnosis. Culture will be positive in slightly more than 50% of cases.
TB verrucosa cutis is differentiated only by culture from atypical mycobacteriosis caused by Mycobacterium marinum. It must also be distinguished from North American blastomycosis, chromoblastomycosis, verrucous epidermal nevus, hypertrophic lichen planus, halogenoderma, and verruca vulgaris.
Lupus Vulgaris.
Lupus vulgaris may appear at sites of inoculation, in scrofuloderma scars, or most frequently at distant sites from the initial infectious focus, probably by hematogenous dissemination. Approximately half of such cases will have evidence of TB elsewhere, so a complete evaluation is mandatory. Because lupus vulgaris is associated with moderately high immunity to TB, most patients will have a positive tuberculin test.
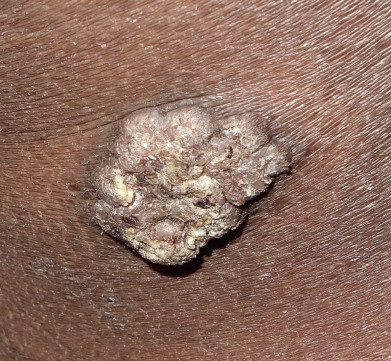
Lupus vulgaris typically is a single plaque composed of grouped red-brown papules, which, when blanched by diascopic pressure, have a pale, brownish yellow or “apple jelly” color. On dermoscopy a yellowish-orange patch may indicate the presence of dermal granulomas secondary to lupus vulgaris, sarcoidosis, a foreign body reaction, or cutaneous leishmaniasis. The papules, called lupomas, tend to heal slowly in one area and progress in another. They are minute, translucent, and embedded deeply and diffusely in the infiltrated dermis, expanding by the development of new papules at the periphery, which coalesce with the main plaque ( Figs. 16.3 and 16.4 ). The plaques are slightly elevated. The disease is destructive, frequently causes ulceration, and on involution leaves deforming scars as it slowly spreads peripherally over the years. Lupus vulgaris lesions of the head and neck can be associated with lymphangitis or lymphadenitis in some cases. If lesions involve the nose or the earlobes, these structures are shrunken and scarred, as if nibbled away. Atrophy is prominent, and ectropion and eclabion may occur. The tip of the nose may be sharply pointed and beaklike, or the whole nose may be destroyed, with only the orifices and the posterior parts of the septum and turbinates visible. The upper lip, a site of predilection, may become diffusely swollen and thickened, with fissures, adherent thin crusts, and ulcers. On the trunk and extremities, lesions may be annular or serpiginous or may form gyrate patterns. On the hands and feet and around the genitals or buttocks, lesions may cause mutilation by destruction, scar formation, warty thickenings, and elephantiasic enlargement.
An unusual form of lupus vulgaris may follow measles or another significant febrile illness. The window of immune deficiency caused by the acute illness results in dissemination of the TB hematogenously from a single focus of lupus vulgaris. Multiple erythematous papules in a generalized distribution appear a month or more after the illness. These lesions evolve to small papules and plaques, clinically and histologically resembling lupus vulgaris. The TST is negative during the immediate period after the febrile illness, then rapidly reverts to strongly positive. This is called “lupus vulgaris postexanthematicus.”
Although classically considered a scarring and atrophying process, lesions of the lips and ears may be quite hyperplastic. The lips may resemble cheilitis granulomatosis clinically and histologically. Uniform hyperplasia of the ear pinna and lobe may closely mimic “turkey ear,” as described in sarcoidosis. When the mucous membranes are involved, the lesions become papillomatous or ulcerative. They may appear as circumscribed, grayish, macerated, or granulating plaques. On the tongue, irregular, deep, painful fissures occur, sometimes associated with microglossia to the degree that nutrition is compromised.
The rate of progression of lupus vulgaris is slow, and a lesion may remain limited to a small area for several decades. The onset may be in childhood and persist throughout life. It may slowly spread, and new lesions may develop in other regions. In some patients, the lesions become papillomatous, vegetative, or thickly crusted, with a rupioid appearance. Squamous cell carcinoma may develop in long-standing lesions.
Histologically, classic tubercles are the hallmark of lupus vulgaris. Caseation within the tubercles is seen in about half the cases and is rarely marked. Sarcoidosis may be simulated. The epidermis is affected secondarily, sometimes flattened and at other times hypertrophic. AFB are found in 10% or less of cases with standard acid-fast stains. PCR may only be positive in the minority of cases of paucibacillary forms of cutaneous TB. Cultures of the skin lesions grow M. tuberculosis in about half the cases.
Colloid milia, acne vulgaris, sarcoidosis, and rosacea may simulate lupus vulgaris. Differentiation from tertiary syphilis, chronic discoid lupus erythematosus, Hansen disease, systemic mycoses, and leishmaniasis may be more difficult, and biopsy and tissue cultures may be required.
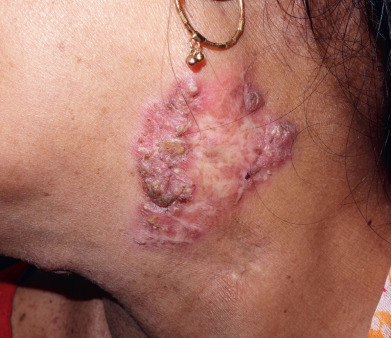
Cutaneous Tuberculosis From Endogenous Source by Direct Extension (Scrofuloderma and Periorificial Tuberculosis)
Scrofuloderma is tuberculous involvement of the skin by direct extension from an underlying focus of infection. It occurs most frequently over the cervical lymph nodes but also may occur over bone or around joints if these are involved. Clinically, the lesions begin as subcutaneous masses, which enlarge to form nodules. Suppuration occurs centrally. They may be erythematous or skin colored, and usually the skin temperature is not increased over the mass. Lesions may drain, forming sinuses, or they may ulcerate with reddish granulation at the base ( Figs. 16.5 and 16.6 ). Surgical procedures may incite lesions of scrofuloderma over joints or the abdominal cavity, apparently by releasing the loculated focus and contaminating the track along which instruments are inserted. Scrofuloderma heals with characteristic cordlike scars, frequently allowing the diagnosis to be made many years later.