Melanocytes originate in the embryonal neural crest and migrate to the epidermis, dermis, leptomeninges, retina, mucous membrane epithelium, inner ear, cochlea, and vestibular system. Nevus cells are a form of melanocyte with a tendency to aggregate into clusters of cells. Nevus cells lack dendritic processes but are otherwise similar to other melanocytes.
Epidermal Melanocytic Lesions
The melanocytes occurring at the dermoepidermal junction (DEJ) are dendritic cells that supply melanin to the skin. These cells contain pigment granules (melanosomes). Melanocytes stain with the dopa reaction and silver stains because they contain melanin. Immunohistochemical stains, such as S-100, HMB-45, MelanA/Mart-1, MITF, and SOX-10, do not depend on the presence of melanin. These stains have largely replaced silver stains for the identification of melanocytes in biopsy specimens. Melanocytes of the epidermis transfer melanosomes through their thin, dendritic processes, where they are actively taken up by keratinocytes. Melanocyte numbers vary by anatomic site and are increased in sun-damaged skin, but they vary little among racial groups. The type, number, size, dispersion, and degree of melanization of the melanosomes determine the pigmentation of the skin and hair.
Treatment of epidermal pigmented lesions can be directed at pigmented keratinocytes, melanocytes, or melanosomes. Q-switched (QS) lasers target the melanosome. Lasers with a longer pulse duration lasting milliseconds (ms) result in melanocyte destruction. Laser treatment produces consistent lightening of ephelides, but the response is variable for café au lait macules, Becker nevus, and nevus spilus.
Ephelis
The common freckle occurs in light-skinned individuals in response to sun exposure. Histologically, freckles demonstrate pigmented basilar keratinocytes, and a mild increase in the number of melanocytes.
Nevus Spilus
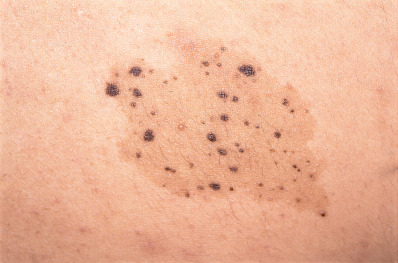
Nevus spilus (speckled lentiginous nevus) presents as a light-brown or tan macule, speckled with smaller, darker macules or papules ( Fig. 30.1 ). It frequently occurs on the trunk and lower extremities, tends to follow Blaschko lines, and is noted in approximately 2% of the population. The nevus spilus may be small, measuring less than 1 cm in diameter, or may be quite large and follow a segmental distribution, referred to as a “zosteriform” lentigo. Multiple sites may be involved in the same individual and may be widely separated by normal skin. Happle has suggested dividing the entity into two forms, a macular type and a papular type. The dark speckles in the macular type are more evenly distributed and represent junctional lentiginous nevi; malignant melanoma has been reported more frequently in this type. Nevus spilus maculosus is consistently found in phakomatosis spilorosea, whereas nevus spilus papulosus demonstrates compound or intradermal nevi and is seen in phakomatosis pigmentokeratotica.
Nevus spilus in combination with a nevus flammeus is called phakomatosis pigmentovascularis (see Chapter 28 ). Phakomatosis pigmentokeratotica includes a speckled lentiginous nevus, organoid nevus, hemiatrophy, and neurologic findings such as muscular weakness. Generalized nevus spilus has been associated with nevus anemicus and primary lymphedema. Nevus spilus has also been reported in association with nevus depigmentosus and with bilateral nevus of Ito.
Histologically, the flat, tan background may show only basilar hyperpigmentation, such as is present in a café au lait spot, or lentiginous proliferation of the epidermis with bulbous rete ridges. The darker speckles usually contain nevus cells and may occasionally demonstrate blue nevi or Spitz nevi.
Melanoma may rarely occur in nevus spilus and some have given rise to multiple melanomas, suggesting a first-hit postzygotic mutation predisposing to melanoma in some cases. A changing lesion should be biopsied. Removal by QS ruby laser or QS alexandrite laser has been reported as effective but may require many sessions for acceptable results.
Boot-Bloemen MCT, et al: Melanoma in segmental naevus spilus. Acta Derm Venereol 2017; 97: 749.
Brito MH, et al: Synchronous melanomas arising within nevus spilus. An Bras Dermatol 2017; 92: 107.
Kar H, et al: Treatment of nevus spilus with Q switched Nd:YAG laser. Indian J Dermatol Venereol Leprol 2013; 79: 243.
Tavoloni Braga JC, et al: Early detection of melanoma arising within nevus spilus. J Am Acad Dermatol 2014; 70: e31.
Lentigo
Lentigo Simplex
These lesions occur as sharply defined, round to oval, brown or black macules. Lentigines usually arise in childhood but may appear at any age. There is no predilection for areas of sun exposure. Multiple lentigines may appear after clearing of plaques of psoriasis, including during biologic therapy. Histologically, lentigo simplex shows hyperpigmentation of basilar keratinocytes and an increase in the number of melanocytes in the basal layer. Melanophages are usually present in the upper dermis.
Solar Lentigo (Lentigo Senilis)
Solar lentigines are commonly known as “liver spots.” They are persistent, benign, discrete, hyperpigmented, round to oval macules occurring on sun-damaged skin. The backs of the hands, cheeks, and forehead are favorite sites in the typical older patient. Red-haired, light-skinned individuals, especially those with high solar exposure, may develop many of these on the shoulders and central upper chest, even at an early age. Solar lentigines may be accompanied by depigmented macules, actinic purpura, and other chronic actinic degenerative changes in the skin. They may evolve into benign lichenoid keratoses and reticulated seborrheic keratoses.
Histologically, the rete ridges appear club shaped or show narrow, budlike extensions. There is a marked increase in pigmentation in the basal cell layer, especially at the tips of the bulbous rete. The number of melanocytes is slightly increased, and the upper dermis often contains melanophages.
Application of liquid nitrogen with a cotton-tipped applicator or cryospray unit is often an effective destructive modality. Argon, QS neodymium-yttrium-aluminum-garnet (Nd:YAG), frequency-doubled Nd:YAG, QS and long-pulse alexandrite, QS ruby, and Er:YAG lasers have been reported as effective. Intense pulsed light has also been used. Postinflammatory pigment alteration is the major complication seen with destructive modalities. Sun protection will reduce the number of new lesions. Bleaching creams containing 4% or 5% hydroquinone, used over several months, will induce temporary lightening. Hydroquinone-cyclodextrin (2%), 4-hydroxyanisole (4-HA), chemical peels, local dermabrasion, topical tretinoin, and adapalene are other treatment options. The combination of 2% 4-HA and 0.01% tretinoin is superior to either active component alone, and a commercial preparation containing these two ingredients plus 2% mequinol has been shown to lighten lesions.
Early lesions of lentigo maligna (melanoma in situ) may be light to medium brown and may mimic solar lentigines. Dermoscopy and confocal microscopy can improve diagnostic accuracy, but when in doubt, a biopsy is appropriate. Lentigo maligna, benign solar lentigo, and pigmented actinic keratosis all occur on sun-damaged skin, and collision lesions are common. If a lesion is not homogeneous clinically, representative biopsies should be taken from each color or shade of brown within the lesion.
PUVA Lentigines
Individuals receiving oral methoxsalen photochemotherapy (psoralen plus ultraviolet A, PUVA), may develop persistent pigmented macules with possible melanocytic atypia. These lesions may occur on sites that are normally protected from sunlight. High-dose single exposures to radiation may result in similar radiation lentigines in exposed skin.
Ink Spot Lentigo (Sunburn Lentigo)
Sunburn lentigines typically occur on the shoulders as small, extremely irregular, reticulated, dark-gray to black macules resembling spots of ink on the skin. Histologically, there is a mild increase in the number of melanocytes and increased melanin in both the basilar keratinocytes and the stratum corneum.
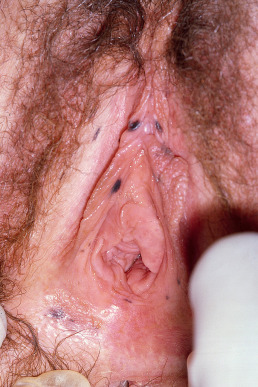
Labial, Penile, and Vulvar Melanosis (Melanotic Macules, Mucosal Lentigines)
Melanotic macules are usually light brown on the oral labial mucosa but may be strikingly irregular and darkly pigmented in the genitalia ( Fig. 30.2 ). In females, the labia minora are most often affected, and in males, the glans and prepuce. Histologically, these lesions demonstrate broad, “boxcar” rete ridges with prominent basilar hyperpigmentation and a normal to slightly increased number of melanocytes. The melanocytes are usually morphologically normal.
Multiple Lentigines Syndrome
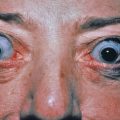
The lesions appear shortly after birth and develop a distinctive speckled appearance that has given rise to the designation LEOPARD syndrome. LEOPARD is Gorlin’s mnemonic acronym for lentigines, electrocardiographic abnormalities, ocular hypertelorism, pulmonary stenosis, abnormalities of genitalia, retardation of growth, and deafness. Inheritance is autosomal dominant. Multiple lentigines occur mainly on the trunk, but other areas may also be involved, such as the palms and soles, buccal mucosa, genitalia, and scalp. PTPN11 gene mutations are seen in both LEOPARD syndrome and Noonan syndrome. Café noir spots noted in these patients are larger and darker than café au lait spots. Histologically, some are melanocytic nevi, whereas others demonstrate histologic features of lentigo simplex.
Moynahan Syndrome
Moynahan syndrome consists of multiple lentigines, congenital mitral stenosis, dwarfism, genital hypoplasia, and mental deficiency.
Generalized Lentiginosis
An occasional patient will have generalized lentiginosis without associated abnormalities.
Centrofacial Lentiginosis
Centrofacial lentiginosis is characterized by lentigines on the nose and adjacent cheeks, variously associated with status dysraphicus, multiple skeletal anomalies, and central nervous system (CNS) disorders. Mucous membranes are spared. Onset is in the first years of life. Lentigines of the central face are also typical of Carney complex.
Carney Complex
Carney complex is also known as NAME syndrome and LAMB syndrome. This designation comprises cardiocutaneous myxomas, lentigines, blue nevi, and endocrine abnormalities. It is discussed in more detail with myxomas in Chapter 28 .
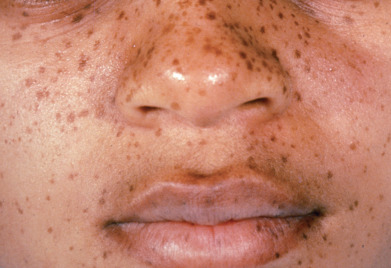
Inherited Patterned Lentiginosis
O’Neill and James reported 10 light-complexioned black patients with autosomal dominant lentigines beginning in infancy or early childhood, but no internal abnormalities ( Fig. 30.3 ). The lentigines are distributed over the central face and lips, with variable involvement of the dorsal hands and feet, elbows, and buttocks. The mucous membranes are spared.
Partial Unilateral Lentiginosis
Partial unilateral lentiginosis is a rare disorder of cutaneous pigmentation characterized by the presence of multiple simple lentigines, wholly or partially involving half the body. Conjunctival involvement has been reported. Agminated lentiginosis appears to be a similar if not identical entity.
Peutz-Jeghers Syndrome
Peutz-Jeghers syndrome is an autosomal dominant syndrome consisting of pigmented macules on the lips, oral mucosa, and perioral and acral areas. Gastrointestinal polyps, especially prominent in the jejunum, are frequently associated. It is discussed further in Chapter 36 .
Imhof L, et al: A prospective trial comparing Q-switched ruby laser and a triple combination skin-lightening cream in the treatment of solar lentigines. Dermatol Surg 2016; 42: 853.
Lv XP: Gastrointestinal tract cancers. Oncol Lett 2017; 13: 1499.
Raziee M, et al: Efficacy and safety of cryotherapy vs. trichloroacetic acid in the treatment of solar lentigo. J Eur Acad Dermatol Venereol 2008; 22: 316.
Vachiramon V, et al: Comparison of Q-switched Nd:YAG laser and fractional carbon dioxide laser for the treatment of solar lentigines in Asians. Lasers Surg Med 2016; 48: 354.
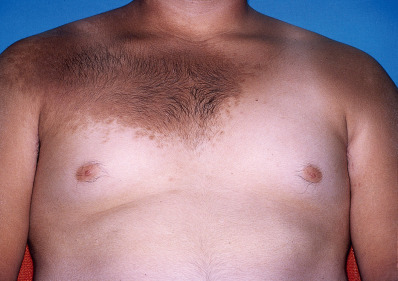
Becker Nevus
Becker nevus presents as a hyperpigmented, hypertrichotic patch on the upper trunk ( Fig. 30.4 ) or proximal upper extremity. The lesion usually begins before puberty, and almost all patients are males. The lesion is typically associated with a smooth muscle hamartoma histologically. Usually, the lesion is asymptomatic and of little consequence, but some lesions have also been associated with connective tissue nevus, inflammatory linear verrucous epidermal nevus, basal cell carcinoma (BCC), phakomatosis pigmentovascularis, ipsilateral breast hypoplasia or abnormalities of underlying bone or of vascular, neural, or other soft tissue structures (Becker nevus syndrome). The pathogenesis may be related to postzygotic mutations in beta-actin and increased expression of androgen receptors within lesional skin. Treatment may not be necessary, but some patients desire removal of pigment or terminal hair associated with the lesion. Ablative 10,600-nm fractional laser and hair removal laser therapy have been used with partial success. Topical flutamide has also been used.
Cai ED, et al: Postzygotic mutations in beta-actin are associated with Becker’s nevus and Becker’s nevus syndrome. J Invest Dermatol 2017; 137: 1795.
Hernandez-Quiceno S, et al: Becker’s nevus syndrome in a pediatric female patient. Case Rep Pediatr 2016; 2016: 3856518.
Momen S, et al: The use of lasers in Becker’s naevus. J Cosmet Laser Ther 2016; 18: 188.
Patrizi A, et al: Clinical characteristics of Becker’s nevus in children. Pediatr Dermatol 2012; 29: 571.
Taheri A, et al: Treatment of Becker nevus with topical flutamide. J Am Acad Dermatol 2013; 69: e147.
Melanoacanthoma
Cutaneous melanoacanthoma is an uncommon lesion first described by Bloch. Clinically, it resembles a pigmented seborrheic keratosis or pigmented BCC and tends to occur in older white men. Histologically, it is a benign epidermal neoplasm composed of keratinocytes and dendritic melanocytes. It is best considered a form of seborrheic keratosis. The starburst dermatoscopic appearance can be confused with that of Spitz nevus. Grouped and ulcerated lesions rarely occur.
Oral melanoacanthoma is also a proliferation of two cell types, melanocytes and epithelial cells, but appears to be a reactive lesion. It occurs as a macular or slightly raised pigmented area on the buccal mucosa, predominantly in young adult black women ( Fig. 30.5 ). Rapid onset and spontaneous resolution are typical.
Cantudo-Sanagustín E, et al: Pathogenesis and clinicohistopathological characteristics of melanoacanthoma. J Clin Exp Dent 2016; 8: e327.
Chung E, et al: Clinical and dermoscopic features of cutaneous melanoacanthoma. JAMA Dermatol 2015; 151: 1129.
Benign Melanocytic Nevi
Common moles, also known as nevocytic nevi or banal nevi, tend to increase in number during the first three decades of life. They are less common in doubly covered areas, such as the buttocks. They typically begin as sharply defined macular lesions, become papular, then gradually become soft and lose their pigment.
Sun exposure increases the number of moles in the exposed skin. Australians have more moles than Europeans. White persons have more than black persons, and individuals with a light complexion have more nevi than those with a dark complexion. Women have more total nevi and more nevi on the legs. Men have more on the trunk. Black persons have more nevi on the palms, soles, conjunctivae, and nail beds. A study of young British women showed an association of holidays overseas with an increased nevus count. The association was greatest in anatomic sites intermittently exposed to sunlight.
Eruptive nevi may occur in association with bullous diseases, severe sunburn, immunosuppression, or sulfur mustard gas exposure. The cheetah phenotype refers to patients with more than 100 uniform, dark-brown to black pigmented macules 4 mm or smaller. The evaluation of these patients can be challenging, because similar-appearing lesions range from junctional nevi to melanoma histologically.
Melanocytic lesions with a junctional component are more often removed during the summer months, whereas excision of intradermal nevi is relatively constant during the year. This suggests that some change in these lesions draws more attention during the summer months. Nevi may darken during pregnancy, but other changes should prompt consideration of a biopsy.
Clinical and Histologic Features
Features of benign nevi include a diameter of 6 mm or less, perfectly uniform pigmentation, flaccid epidermis, smooth, uniform border, and an unchanging size and color. Benign nevi tend to be round to oval and undergo a predictable course of maturation.
Junctional nevi are sharply circumscribed brown macules, varying in diameter from 1–6 mm. They usually appear between 3 and 18 years of age. During adolescence and adulthood, some become compound or intradermal. Small, well-nested junctional melanocytic proliferations are almost invariably benign. Benign junctional nevi associated with bulbous hyperplasia of the rete ridges are referred to as junctional lentiginous nevi. Lentigo maligna can appear well nested with an appearance similar to that of junctional lentiginous nevi. Any broad junctional melanocytic lesion on sun-damaged skin should be viewed with suspicion.
Compound nevi demonstrate both junctional and intradermal melanocytes. Benign compound nevi are well nested at the junction, with dispersion of individual melanocytes at the base of the lesion. They demonstrate bilateral symmetry but are not symmetric from top to bottom. Instead, with descent into the dermis, the melanocytes become smaller and spindled in appearance. Nests at the junction tend to be round to oval and are about equidistant from one another. Dermal nests are generally smaller than the junctional nests and become progressively smaller deeper in the dermis. Individual cells rather than nests are present at the base. Pigment is most prominent at the junction and becomes progressively less prominent deeper in the dermis. Intradermal nevi look similar to compound nevi, without the junctional nests.
In most benign nevi, there are no melanocytes above the DEJ. Individual melanocytes in a “buckshot” scatter throughout the epidermis are typical of superficial spreading melanoma. Sunburned benign nevi may also demonstrate buckshot intraepidermal scatter of melanocytes, and buckshot scatter may be seen in the central portion of acral nevi and Spitz nevi. Genital nevi often demonstrate large, poorly cohesive nests. They may also resemble dysplastic nevi histologically. A histologic resemblance to dysplastic nevi is also common in nevi from the scalp, ears, dorsal foot, and breast, even in patients with no other evidence of the dysplastic nevus syndrome.
On the palms and soles, the rete pattern follows the dermatoglyphs ( Fig. 30.6 ). Nests in these locations tend to run along the rete ridges. If a benign palmar nevus is bisected across the dermatoglyphs, the nests will appear round to oval. If the same lesion is sectioned parallel to the dermatoglyphs, the nests will appear elongated and may mimic those of melanoma as an artifact of sectioning. Careful communication with the pathologist is essential when submitting an acral melanocytic lesion to the laboratory.
Malignant Degeneration
Almost half of melanomas occur in preexisting nevi, and an increased number of nevi represents a risk factor for melanoma. The signs of malignant transformation in pigmented nevi are recent enlargement, an irregular or scalloped border, asymmetry, changes or variegation in color (especially red, white, or blue), surface changes (scaling, erosion, oozing, crusting, ulceration, or bleeding), development of a palpable thickening, signs of inflammation, or the appearance of satellite pigmentation. Symptoms may include development of pain, itch, or tenderness. The “ugly duckling” sign refers to nevi in an individual generally tending to share a similar appearance. Any mole that does not share the same characteristics should be considered for biopsy. Moles with dark areas that do not lie entirely within the lesion, but produce an extension beyond the border, may represent melanoma arising in association with a preexisting nevus. The clinician should alert the pathologist to the presence of these areas and the pathologist should section through the appropriate area. Perifollicular hypopigmentation is a common finding in benign nevi. When it occurs at the edge of the nevus, it may give the lesion a notched appearance. Dermatoscopic examination may be of value in this setting. Lesions with changing clinical or dermatoscopic features should be biopsied.
Nevi frequently darken with pregnancy or with oral contraceptive use. Nevi from normal persons have no estrogen or progesterone receptors, but there may be positive estrogen receptor binding in nevi from pregnant women, as is also found in malignant melanoma. The development of what appears to be a new pigmented nevus in a patient over age 35 should alert the physician to possible melanoma, because patients without the dysplastic nevus syndrome usually do not develop new nevi at this age.
Treatment
Acquired nevi should be removed if they show signs of malignant transformation. Nevi of the neckline, beltline, or other areas that are irritated may be removed to relieve the patient of the irritation. Nevi may also be removed if they are in a location where it is impractical to observe them. If a solitary, darkly pigmented lesion is present on the oral or genital mucous membrane, a biopsy should be performed, because nevi are uncommon in these locations. Nail matrix nevi and lentigines produce a pigmented nail band. The proximal matrix gives rise to the dorsal nail plate, and the distal matrix gives rise to the ventral nail plate. When the nail is observed end-on, the level of the pigment may be evident and indicates the location of the pigmented lesion in the matrix. A widening band indicates a matrix lesion increasing in diameter. Biopsy of a solitary, expanding, acquired longitudinal pigmented band in an adult is typically necessary to ascertain the cause. Hutchinson’s sign (pigmentation of the nailfold) is an indicator of melanoma. Nail matrix melanoma in children is exceptional.
Conjunctival nevi occur, and most can be followed serially if the lesion has been present since childhood or has shown no evidence of growth. Changing pigmented lesions and those acquired after childhood are best evaluated in conjunction with an ophthalmologist. Most conjunctival nevi occur on the bulbar conjunctiva and often about the nasal or temporal corneoscleral limbus. Suspicion of melanoma should arise if a pigmented lesion occurs in the palpebral or forniceal conjunctiva, if lesions are not hinged at the limbus and are immovable, if they extend into the cornea, if there is canalicular obstruction that leads to tearing, or if adjacent dilated vessels are noted.
Combined melanocytic nevi are common. They consist of a banal nevus together with a blue nevus, Spitz nevus, or deep penetrating nevus. Two or more distinct populations of melanocytes are evident.
Melanocytic nevi may occur in lymph nodes and are present in about 10% of sentinel node biopsies. Nodal nevi typically occur in the capsule, in contrast to melanoma metastases, which are typically subcapsular. Nodal nevi are frequently associated with cutaneous nevi in the draining basin, especially nevi with congenital features.
Pseudomelanoma (Recurrent Nevus)
Melanotic lesions clinically resembling a superficial spreading melanoma may occur at the site of a recent shave removal of a melanocytic nevus. Melanocytic nevi occurring in areas of lichen sclerosus or bullous disease often have similar features. On dermatoscopic examination, a regular network and the presence of streaks suggest reactive pigmentation. Any truly suspicious lesion should be removed. Histologically, the junctional component often demonstrates a predominance of nonnested melanocytes, confluence of nests, and nests that vary in size and shape. The presence of a superficial dermal scar with remnants of the original nevus beneath this zone of fibrosis is an important clue to the correct diagnosis. Although atypical in appearance, the junctional proliferation remains entirely confined to the area overlying the scar.
Recurrent Spitz nevus is a particular problem because many of the histologic features of benign Spitz nevi overlap with those of melanoma. In benign recurrent Spitz nevi, the dermal component typically retains cytologic maturation, dispersion at the base of the lesion, and an immunostaining pattern typical for benign nevi. Recurrent blue nevi also present special difficulties. High cellularity, cellular pleomorphism, mitotic figures, and a lymphoid host response may be present. In the absence of marked cytologic atypia, frequent mitotic figures or necrosis en masse, the lesions are likely to be benign. Because of the special problems posed by recurrent Spitz and blue nevi, the initial biopsy of these lesions should be a complete excisional biopsy whenever possible. Congenital nevi have a higher rate of recurrence when surgery is done at a younger age.
Balloon Cell Nevus
Clinically, balloon cell nevi are indistinguishable from ordinary nevi. Histologically, they are composed of large, pale, polyhedral balloon cells. Generally, foci of ordinary nevus cells are also evident. Rarely, the lesions are composed entirely of balloon cells. Balloon cell change has been reported in cellular blue nevus as well. Balloon cell melanoma does exist, but the nuclei are large and pleomorphic, and the architecture of the lesion is that of melanoma. Balloon cell nodal nevi may be seen in sentinel node specimens.
Halo Nevus
Halo nevus is also known as Sutton nevus, perinevoid vitiligo, and leukoderma acquisitum centrifugum. The lesions are characterized by a pigmented nevus with a surrounding depigmented zone ( Fig. 30.7 ). Halo nevi tend to be multiple and occur most frequently on the trunk, mostly in teenagers. The central nevus gradually loses its pigmentation, turns pink, and then disappears, leaving a round to oval area of depigmentation. Over time, the area repigments. Darkening of the central nevus rather than lightening has also been reported in association with the halo phenomenon. Halo nevi have been reported during infliximab therapy. Target-like pigmented nevi present with the appearance of an inverse halo nevus phenomenon.
The infiltrate contains many cytotoxic T cells and may represent immunologically induced rejection. The peripheral blood has been shown to contain activated adhesive lymphocytes that disappear when the lesion is excised. Patients also demonstrate antibodies to melanocytes and cell-mediated immunity to melanoma cells. There may be associated vitiligo.
Regressing melanoma may also have associated leukoderma, but the pattern is usually haphazard and confined to the pigmented lesion. Other lesions that may also have a surrounding zone of leukoderma include blue nevi and neurofibromas.
Histologically, halo nevi demonstrate a band of lymphocytes that extends throughout the lesion, intimately mingling with the melanocytes. In contrast, the lymphoid infiltrate associated with melanoma tends to aggregate at the periphery of the lesion. In early halo nevi, amelanotic melanocytes may be found in the leukodermic halo. Later, melanocytes are absent until repigmentation occurs. A granulomatous infiltrate may rarely be present. The term Myerson’s nevus has been applied to eczematous change associated with a nevus. Hypopigmentation may be present.
A full mucocutaneous examination at diagnosis is indicated to exclude a concurrent melanoma, but this is rarely found. The decision to remove the nevus at the center of the halo is based on its morphologic features, as with any other nevus.
Congenital Melanocytic Nevus
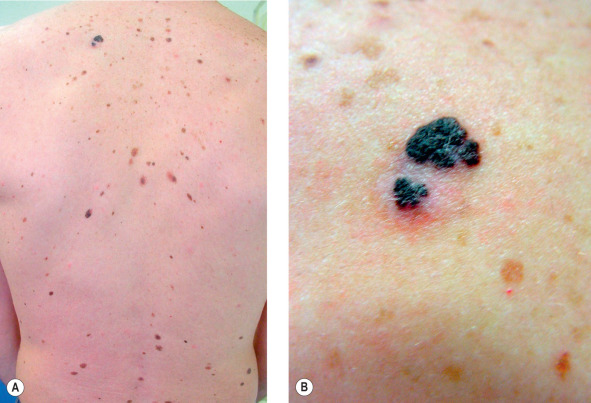
Giant Pigmented Nevus (Giant Hairy Nevus, Bathing Trunk Nevus)
Giant pigmented nevi appear as large, darkly pigmented hairy patches in which smaller, darker patches may be interspersed or present as small satellite lesions. The skin may be thickened and verrucous. The trunk is a favored site, especially the upper or lower parts of the back ( Fig. 30.8 ). Giant hairy nevi are present at birth and grow proportionally with the body. Widespread congenital dermal nevus with large nodules may affect the entire body, including the palms, soles, and oral mucous membrane. Some congenital melanocytic nevi have associated placental infiltration by benign melanocytes.
The incidence of melanomas developing in giant congenital pigmented nevi is between 2% and 15%. Approximately 60% of these melanomas appear within the first decade of life, and the majority arise in the dermis or subjacent tissue, rather than at the DEJ. About 40% of the malignant melanomas seen in children occur in large congenital nevi. The risk is greatest for axial lesions and those larger than 40 cm. In one study, 94% of large congenital nevi that gave rise to melanoma had satellite nevi.
Large axial lesions may be associated with neurocutaneous melanocytosis. The risk is greatest for large axial lesions with many satellite lesions, and almost half of patients with symptomatic neurocutaneous melanosis develop leptomeningeal melanoma. Neurocutaneous melanosis can be detected by magnetic resonance imaging (MRI).
Histologically, giant congenital nevi extend into the deep dermis and may involve the subcutis, fascia, muscle, and other underlying structures. Nevus cells are found in a patchy perivascular distribution and often extend in a patchy, single-file fashion between collagen bundles. Nests are often seen in association with adnexal structures or nerves. Extensive desmoplasia has been described. Estrogen and progesterone binding has been noted in congenital nevi. These receptors are generally absent from common acquired nevi.
Benign “proliferative” nodules within giant congenital nevi may be confused histologically with malignant change. Features useful in distinguishing the two include lack of high-grade atypia, lack of necrosis, rarity of mitoses, a lack of Ki-67 expression, evidence of transition between the cells of the nodule and those of the adjacent nevus, and lack of compressive expansile growth. Comparative genomic hybridization has demonstrated chromosomal aberrations in atypical nodular proliferations in congenital nevi, but many of these are numerical aberrations of whole chromosomes, suggesting a mitotic spindle defect. These differ from the chromosomal aberrations seen in melanoma.
Treatment decisions must be individualized. Half of all melanomas in giant congenital nevi occur in deep structures. Extensive surgery to remove the upper portions of the lesion reduces, but does not eliminate, the risk of melanoma. In patients with leptomeningeal melanosis, the risk of melanoma remains high. Satellite lesions and extremity lesions have a lower incidence of neoplastic conversion than large axial lesions, and the risk-benefit ratio of extensive surgery on these lesions differs accordingly. Some lesions are not amenable to excision because they involve functionally critical areas.
Serial staged excision is the method of choice whenever possible. Tissue expansion, cultured autologous cultured skin substitutes, and flap closure are especially useful in the head and neck region. Alternative approaches to treatment, such as dermabrasion, curettage, carbon dioxide (CO 2 ) laser ablation or treatment with QS Nd:YAG, ruby, and alexandrite lasers can lead to improvement in appearance. Therapy may also eliminate some nevus cells, with theoretic lowering of the melanoma risk. It is important to emphasize that most melanomas in giant congenital nevi occur in the dermal component rather than at the DEJ. Any treatment that alters the surface may alter detection of deep melanoma. Malignant transformation has been reported 20 years after dermabrasion. Regardless of the method of choice, lifelong periodic cutaneous examination and general medical evaluation are indicated.
Small and Medium-Sized Congenital Nevocytic Nevus
Small, congenital nevocytic nevi are generally defined as less than 2 cm in greatest diameter, and medium-sized lesions measure more than 2 cm but less than 20 cm. They are found in about 1% of newborns. About half eventually become hairy. Histologically, they share many features with giant congenital nevi but usually do not extend into the subcutaneous tissue. Many of the histologic features associated with congenital nevi also occur in acquired nevi. The risk of melanoma in small to medium-sized congenital nevi is extremely low; it may be no greater or only slightly greater than the risk of melanoma arising in ordinary acquired nevi. One important difference is that malignant degeneration may occur in the deep dermal component of small congenital nevi, rather than at the DEJ. Most of the melanomas that do occur do so after puberty. Excision is recommended for changing lesions and may be considered for those of cosmetic concern and in areas difficult to observe.
Green MC, et al: Management considerations for giant congenital melanocytic nevi in adults. Mil Med 2014; 179: e463.
Ma T, et al: Tissue expansion in the treatment of giant congenital melanocytic nevi of the upper extremity. Medicine (Baltimore) 2017; 96: e6358.
Mutti LA, et al: Giant congenital melanocytic nevi. An Bras Dermatol 2017; 92: 256. Vourc’h-Jourdain M, et al: Large congenital melanocytic nevi. J Am Acad Dermatol 2013; 68: 493.
Viana ACL, et al: A prospective study of patients with large congenital melanocytic nevi and the risk of melanoma. An Bras Dermatol 2017; 92: 200.
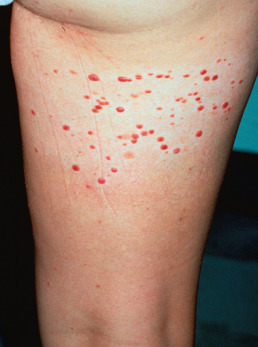
Spindle and Epithelioid Cell Nevus (Spitz Nevus)
Spitz nevi typically appear as pink, smooth-surfaced, raised, round, firm papules ( Fig. 30.9 ). Most frequently, Spitz nevi occur during the first two decades of life, although they occur in adulthood in about one third of cases. Infrequently, multiple lesions present as agminate (clustered) ( Fig. 30.10 ) or disseminated lesions in children and adults. Although they usually contain no visible pigment, some lesions are pigmented. Occasionally, Spitz nevi can be blue-black in color. A starburst pattern is characteristic on dermatoscopic examination. Although dermoscopy and confocal microscopy are being used in this setting, both false-positive and false-negative studies occur, and histologic examination remains the “gold standard” for evaluation of suspicious lesions.
As with other nevi, Spitz nevi may be junctional, compound, or intradermal. Compound nevi are most common and are characterized by compact hyperkeratosis, hypergranulosis, and pseudoepitheliomatous hyperplasia. The cells are large, with round to spindled nuclei. Epithelioid cells have large vesicular nuclei with prominent nucleoli and ample pink cytoplasm. Adjacent to the nucleus, the cytoplasm typically has a more amphophilic hue, giving it a characteristic two-tone appearance, similar to the cytoplasm of the cells in reticulohistiocytic granuloma. The nests tend to be oval and oriented in a vertical direction, as are the nuclei within the nests, so that they appear to be “raining down” the adjacent rete ridges. Clefts are typically present adjacent to some of the nests, and superficial vascular ectasia is characteristic. Dull-pink globules (Kamino bodies) are seen within the epidermis. These represent trapped basement membrane zone material and stain similar to collagen with a trichrome stain as well as with immunostains for type IV collagen. Buckshot scatter of melanocytes may be noted within the epidermis overlying the center of the lesion, but the lesion is sharply circumscribed, and cells disperse as individual units between collagen bundles at the base of the lesion. Rosette-like structures may occur. In a review of 349 Spitz nevi, the presence of epithelioid and spindled cells was the only feature present in 100% of cases. Other findings, in descending order, included maturation (72%), inflammatory infiltrate (70%), epidermal hyperplasia (66%), melanin (50%), telangiectasias (40%), Kamino bodies (34%), desmoplastic stroma (26%), mitosis (23%), pagetoid extension (13%), and hyalinization of the stroma (8%).
Melanomas may have many of the previous features, but generally lack Kamino bodies, and often demonstrate broad lateral extension, deep mitoses, and large nests at the base of the lesion. Cytologic features that favor a diagnosis of melanoma over Spitz nevus include pleomorphism, mitotic figures, notching of the nuclear envelope, and a peppered moth chromatin pattern. In questionable cases, adjunctive studies may be of value. S-100A6 shows strong and diffuse expression in Spitz nevi. Other melanocytic nevi often express S-100A6 weakly or not at all. Melanomas may express S-100A6, but the expression tends to be weak and patchy in the dermal component and is often negative in the junctional component. HMB-45 typically stains Spitz nevi in a top-heavy fashion, whereas melanomas stain uniformly top to bottom. MIB-1 (Ki-67), a proliferation marker, may also be helpful as an adjunct to the histopathologic diagnosis of Spitz nevi. MIB-1–positive nuclei are rare in the deep portion of a Spitz nevus, whereas they are often numerous in melanoma. Comparative genomic hybridization demonstrates chromosomal aberrations in the majority of melanomas, but most Spitz nevi show no aberrations. A minority of Spitz nevi show an isolated gain of chromosome 11p, the site of HRAS, but this aberration is not observed in melanoma. Specific gains or losses can be demonstrated with fluorescent in situ hybridization (FISH) probes. Studies of the mitogen-activated protein kinase (MAPK) pathway and genomic gains and losses may also prove helpful in this setting.
Junctional Spitz nevi usually show some degree of buckshot scatter of melanocytes and share many histologic features with melanoma. Lesions that lack sharp lateral circumscription are more likely to represent melanoma. Intradermal Spitz nevi lack overlying hyperkeratosis, hypergranulosis, or pseudoepitheliomatous hyperplasia, but the cells disperse as individual units at the deep margin. Dermal spitzoid lesions that remain nested at the deep margin are likely to represent melanoma. Desmoplastic Spitz nevi may be compound or intradermal, and are characterized by a dense, hypocellular collagenous stroma.
Pigmented spindle cell nevus is regarded by many as a variant of Spitz nevus. The lesions tend to be pigmented macules on the legs of young women. The cells are smaller and uniformly spindled, but other histologic features are similar to those of Spitz nevi. In contrast to Spitz nevi, they stain poorly with S-100A6. Desmoplastic Spitz nevi are moderately to strongly positive for p16, whereas most desmoplastic melanomas are negative, but studies have produced conflicting results regarding the usefulness of this antibody. Neuropilin-2 looks promising as an adjunctive study. Both Spitz nevi and spitzoid melanoma have a lower incidence of BRAF and NRAS mutations than common acquired nevi and conventional melanomas. HRAS mutations are typical of Spitz nevi but are rare to absent in spitzoid melanoma. HRAS -duplicated Spitz nevi are large, with large nuclei. BAP1- mutated nevi (Wiesner nevi) are composed of dermal nests of large epithelioid melanocytes with pleomorphism and lack of maturation, simulating melanoma. Immunoperoxidase staining for BAP1 is negative, whereas staining for VE1 (V600E) BRAF mutation is positive. BAP1 mutation is associated with the familial uveal melanoma syndrome, which includes large numbers of Wiesner nevi, mesothelioma, and renal cell carcinoma. BAP1 mutation can also be seen in melanoma (especial blue nevus–like melanoma), so lack of BAP1 staining alone is not sufficient to prove that the lesion is benign.
Although new molecular techniques may allow better differentiation, because of the histologic overlap with melanoma, the biopsy technique for suspected Spitz nevi should be complete excision whenever possible. Critical differentiating histologic features include sharp lateral circumscription and dispersion at the base of the lesion. An incomplete excision will fail to demonstrate either the lateral or the deep aspect of the tumor, and these diagnostic features will not be evident. When a lesion is incompletely excised, most authorities recommend reexcision of the site to ensure complete removal. At times, however, the dogma that all Spitz nevi should be completely excised must be tempered in the patient’s best interest. An otherwise typical Spitz nevus that extends to the deep margin on a young child’s nose may be difficult to excise without disfigurement. The risks of anesthesia must also be weighed against the likelihood that the lesion is anything but a benign Spitz nevus. Data suggest that children and teenagers with atypical spitzoid neoplasms and positive sentinel nodes have a less aggressive clinical course than those with unambiguous melanoma, but this may merely reflect mixed outcomes of benign and malignant lesions that were classified together.
Abboud J, et al: The diagnosis and management of the Spitz nevus in the pediatric population. Syst Rev 2017; 6: 81.
Busam KJ, et al: Multiple epithelioid Spitz nevi or tumors with loss of BAP1 expression. JAMA Dermatol 2013; 149: 335.
Dika E, et al: Spitz nevi and other spitzoid neoplasms in children. Pediatr Dermatol 2017; 34: 25.
Gerami P, et al: Risk assessment for atypical spitzoid melanocytic neoplasms using FISH to identify chromosomal copy number aberrations. Am J Surg Pathol 2013; 37: 676.
Lott JP, et al: Clinical characteristics associated with Spitz nevi and spitzoid malignant melanomas. J Am Acad Dermatol 2014; 71: 1077.
Luo S, et al: Spitz nevi and other spitzoid lesions. Part I. J Am Acad Dermatol 2011; 65: 1073.
Luo S, et al: Spitz nevi and other spitzoid lesions. Part II. J Am Acad Dermatol 2011; 65: 1087.
Massi D, et al: Atypical Spitz tumors in patients younger than 18 years. J Am Acad Dermatol 2015; 72: 37.
Menezes FD, et al: Spitz tumors of the skin. Surg Pathol Clin 2017; 10: 281.
Urso C: On the nature of atypical Spitz tumors. Arch Pathol Lab Med 2016; 140: 1316.
Valdebran M, et al: Nuclear and cytoplasmic features in the diagnosis of banal nevi, Spitz nevi, and melanoma. J Am Acad Dermatol 2016; 75: 1032.
Atypical Nevus (Dysplastic Nevus, Clark Nevus)
In 1978 Clark et al. described families with unusual nevi and multiple inherited melanomas, a condition they referred to as the “B-K mole syndrome” (after Family B and Family K). About the same time, Lynch et al. recognized similar findings in other families and designated this the “familial atypical multiple mole–melanoma” (FAMM) syndrome. The most widely accepted term for the marker lesions is dysplastic nevus, with the patient’s condition called the dysplastic nevus syndrome (DNS). The lesions may also be referred to as atypical nevi, Clark nevi, or nevi with architectural disorder. Patients with dysplastic nevi who have at least two blood relatives with dysplastic nevi and melanoma have the worst prognosis for development of melanoma. These individuals may have a 100% lifetime risk of melanoma. An associated increased risk of developing pancreatic carcinoma is present in some families. Some studies have indicated that ocular melanomas may occur in these patients.
The genetic basis for familial melanoma is being elucidated. One quarter to one third of patients have germline mutations on chromosome 9p in the CDKN2A tumor-suppressor gene (also known as p16, MTS1, and p16INK4A ). It encodes for an inhibitor of a cyclin-dependent kinase 4 (CDK4), which functions to suppress proliferation. Patients with mutations that impair the function of the p16 suppressor protein, referred to as the p16M alleles, have a concomitant predisposition to pancreatic cancer. In other families in whom this is not present and who have 16W alleles, the predisposition to melanoma does not correlate with an elevated risk of pancreatic cancer. Mutations in the CDK4 gene have also been found to be responsible for a lesser number of cases of familial melanomas. The products of this gene interact with the same cell growth cycle process as p16.
Moles with a histology similar to dysplastic nevi also occur frequently in patients without a personal or family history of melanoma, with 5%–20% of patients having at least one clinically dysplastic nevus, depending on the criteria used. During the growth phase, many nevi demonstrate junctional extension beyond the dermal component. This “shouldering” phenomenon is also one of the criteria for dysplastic nevi, and many growing nevi will have some histologic features of dysplastic nevi. The same is true for many congenital nevi, genital nevi, and those on the breast, dorsal foot, and scalp, none of which appears to be a marker for DNS.
Dysplastic nevi differ from common acquired nevi in several respects. Clinically, dysplastic nevi are characterized by a variegated tan, brown, and pink coloration, with the pink hues seen mainly in the macular portion of the nevus. A macular component is always present and may comprise the entire lesion but frequently surrounds a papular center. The nevi are larger than common nevi, usually 5–12 mm in diameter (common nevi usually measure 6 mm or less). The shape of dysplastic nevi is often irregular, with indistinct borders. Atypical nevi are most often seen on the back ( Fig. 30.11 ), and exposure to sun promotes the development of these lesions in individuals with DNS.