Epidemiology
The World Health Organization (WHO) has committed itself to eliminating Hansen disease as a public health problem, but the disease remains endemic in certain regions, with 95% of cases reported from 16 countries. Brazil, India, and Indonesia account for 80% of all cases worldwide. Although 90% of diagnosed U.S. cases are imported, Hansen disease is endemic in the coastal southeastern United States and in Hawaii. In the southeastern states, cases may be related to exposure to armadillos, a natural host for the infectious agent.
It is believed that more than 90% of persons exposed to Mycobacterium leprae are able to resist infection. In endemic areas, between 1.7% and 31% of the population is seropositive for antibodies to leprosy-specific antigens, suggesting widespread exposure to the bacillus. About 17% of household contacts of multibacillary patients have M. leprae, which is detectable by polymerase chain reaction (PCR) on skin swabs, with 4% detectable in nasal swabs. This clears after the multibacillary patient has been treated with multidrug therapy (MDT) for 2 months. Thus although many persons can be transiently infected, they apparently are able to resist overt clinical infection.
There appears to be a genetic basis for susceptibility to acquire Hansen disease. Monozygotic twins have concordant disease in 60%–85% of cases, and dizygotic twins in only 15%–25%. Numerous genes have been identified as possibly conferring susceptibility to infection with M. leprae. Different genes have been identified in different populations, suggesting that multiple genetic causes of susceptibility to infection are possible with M. leprae. Tight genetic linkage with the PARK2/PACRG regulatory region, HLA-DRB1, and lymphotoxin A (LTA+80) has been detected. PARK2 is a gene involved in the development of Parkinson disease, and LTA+80 is a low-production lymphotoxin A allele associated with malaria parasitemia. Interleukin (IL)–17F single nucleotide polymorphism (SNP) is associated with an increased susceptibility to Hansen disease, and type 1 reactions in paucibacillary patients. In Chinese patients, susceptibility was linked to multiple genes, including CCDC12, C13orf31, and a series of genes known to be associated with susceptibility to other mycobacterial diseases, including NOD2, IL12B, RIP2K, and TNFSF15.
In adult cases, men outnumber women 1.5:1. Although Hansen disease occurs at all ages, most cases appearing or acquired in endemic areas present before age 35. Patients exposed to armadillos present on average at age 50. The latency period between exposure and overt signs of disease is usually 5 years for paucibacillary cases and an average of 10 years in multibacillary cases. Infected women are likely to present during or immediately after pregnancy.
The mode of transmission remains controversial. Except for cases associated with armadillo exposure, other patients with Hansen disease are thought to be the only possible source of infection. Rarely, tattooing or other penetrating injury to the skin can be the route of infection. Multibacillary cases are much more infectious than paucibacillary cases, so the nature of the source case is the most important factor in transmission. Contact is associated with acquiring infection. Household contacts represent 28% of new Hansen disease patients; there is an 8–10 times greater risk of acquiring disease if the household contact has lepromatous disease, versus only 2–4 times if the contact has tuberculoid leprosy. In 80% of all new cases of Hansen disease, there is a clear history of social contact with an untreated patient with Hansen disease. PCR can detect M. leprae on the intact skin by saline washings in up to 90% of multibacillary patients with a high bacterial load (bacterial index [BI] >3). Up to 70% of nasal swabs are similarly positive. Whereas the swabs from the patients remain positive after 3 months of MDT, the swabs of household contacts become negative, suggesting that the bacilli seen in patients are nonviable, and that the risk of transmission is substantially reduced after the index patient is treated. Unfortunately, persons may be infectious from their skin or nasal secretions, with no clinical evidence of Hansen disease (multibacillary patients who are not yet symptomatic and without identifiable skin lesions). This may make strategies relying on treatment of contacts of known Hansen disease patients ineffective in eradicating the disease. In nonendemic areas, transmission to contacts is rare, a reassuring fact for the families of patients diagnosed in areas where Hansen disease is uncommon. The last case of secondary transmission of Hansen disease in the United Kingdom was in 1923.
The Infectious Agent
Until recently, it had been thought that all cases of human and animal leprosy are caused by the same organism, Mycobacterium leprae. This is a weakly acid-fast organism that has not been successfully cultured in vitro. M. leprae grows best at temperatures (30°C) below the core body temperature of humans. This explains the localization of Hansen disease lesions to cooler areas of the body and the sparing of the midline and scalp. The organism may be cultivated in mouse footpads and most effectively in armadillos, whose lower body temperature is more optimal for growth of M. leprae. Phenolic glycolipid 1 (PGL-1) is a surface glycolipid unique to the leprosy bacillus. In infected tissues, the leprosy bacillus favors intracellular locations, within macrophages and nerves. The genome of the leprosy bacillus has been sequenced and compared with its close relative, the tuberculous bacillus. The genome of M. leprae contains only 50% functional genes, apparently the result of significant reductive evolution. As with other intracellular parasites, and in the absence of the ability to share DNA with other bacteria, M. leprae has lost many nonessential genes, including those involved in energy metabolism, making it dependent on the intracellular environment for essential nutrients. This may explain the extremely long generation time, 12–14 days, and the inability to culture M. leprae in vitro.
A second organism, Mycobacterium lepromatosis, has been isolated from Hansen disease patients in Mexico and reported as the major cause of leprosy in some regions. Some patients are infected with both M. leprae and M. lepromatosis. This second mycobacterium is specifically associated with the diffuse type of lepromatous leprosy (DLL), also known as “Lucio leprosy.” These are the patients who develop Lucio phenomenon. Invasion of the endothelial cells characterizes infection with the new organism. Although not all researchers accept M. lepromatosis as a second, separate species capable of causing Hansen disease, it does suggest more than one causative organism for leprosy.
Diagnosis
A diagnosis of Hansen disease must be considered in any patient with neurologic and cutaneous lesions. The diagnosis is frequently delayed in the developed world; clinicians do not readily think of Hansen disease because they may never have seen it. In the United States this diagnostic delay averages ![]() –2 years. In the United Kingdom, in more than 80% of patients with Hansen disease, the correct diagnosis was not suspected during the initial medical evaluation.
–2 years. In the United Kingdom, in more than 80% of patients with Hansen disease, the correct diagnosis was not suspected during the initial medical evaluation.
Hansen disease is diagnosed, as with other infectious diseases, by identifying the infectious organism in affected tissue. Because the organism cannot be cultured, this may be very difficult. Biopsies from skin or nerve lesions, stained for the bacillus with Fite-Faraco stain, are usually performed in the developed world. In some Hansen disease clinics, and in the developing world where the disease is endemic, organisms are identified in slit smears of the skin. Smears are very specific, but 70% of all patients with Hansen disease have negative smears. Smears are taken from lesions and cooler areas of the skin, such as the earlobes, elbows, and knees. If organisms are found on skin smears, the patient is said to be multibacillary. If the results of skin smears are negative (and there are five or fewer lesions), the patient is called paucibacillary.
Nerve involvement is detected by enlargement of peripheral nerves and lesional loss of sensation. Enlarged nerves are found in more than 90% of patients with multibacillary Hansen disease and in 75%–85% of patients with paucibacillary disease. About 70% of lesions have reduced sensation, but lesional dysesthesia is not detected in patients with multibacillary Hansen disease, the most infectious form.
Serologic tests to detect antibodies against M. leprae –unique antigens have limitations in the diagnosis of leprosy and reactional states. They are universally positive in patients with multibacillary disease, in whom the diagnosis is not difficult. In paucibacillary patients, these tests are often negative, and in endemic areas there is a high background rate of positivity of serologic tests. In pure neural Hansen disease, however, about 50% of patients are seropositive, and serologic testing might be of use. Based on the technology of the T-cell interferon-γ (IFN-γ) production–based assays for M. tuberculosis infection, researchers have identified unique peptides of M. leprae and developed a research IFN-γ release assay (IGRA). This was able to detect all paucibacillary cases in a Hansen disease cohort. In addition, 13 of 14 household contacts of Hansen disease patients were positive with IGRA. Ideally, in endemic areas, both serologic and cell-based assays can be used to detect all patients with Hansen disease.
In endemic areas, active surveillance of contacts is recommended. In Brazil, about 2% of both in-domicile and extradomiciliary contacts are found to have Hansen disease. Most cases are associated with patients who have multibacillary Hansen disease.
Classification
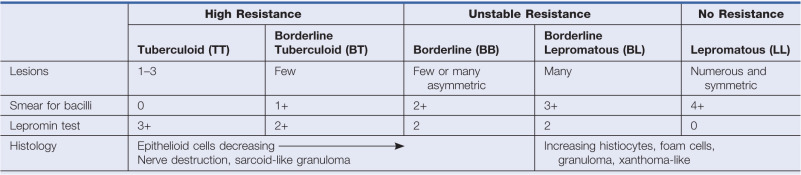
Hansen disease may present with a broad spectrum of clinical diseases. The Ridley and Jopling scale classifies cases based on clinical, bacteriologic, immunologic, and histopathologic features ( Table 17.1 ). In many exposed patients, the infection apparently clears spontaneously, and no clinical lesions develop. Patients who do develop clinical disease are broadly classified into two groups for the purposes of treatment and for trials that compare treatment strategies. Paucibacillary patients have few or no organisms in their lesions and usually have three to five lesions or fewer (for treatment purposes, the finding of acid-fast bacilli by stains or smears classifies a patient as having multibacillary Hansen disease). Multibacillary patients have multiple, symmetric lesions and organisms detectable by biopsy or smears. The individual’s cell-mediated immune response to the organism determines the form that Hansen disease will take in the individual. If the cell-mediated immune response against M. leprae is strong, the number of organisms will be low (paucibacillary), and conversely, if this response is inadequate, the number of organisms will be high (multibacillary).
The most common outcome after exposure is probably spontaneous cure. If skin disease does appear, the initial clinical lesion may be a single, hypopigmented patch, perhaps with slight anesthesia. This is called indeterminate disease, because the course of the disease cannot be predicted at this stage. The lesion may clear spontaneously or may progress to any other form of Hansen disease.
The spectrum of Hansen disease has two stable poles, the tuberculoid and lepromatous forms (see Table 17.1 ). These so-called polar forms do not change; the patient remains in one or the other form throughout the course of the disease. The polar tuberculoid form (called TT), the type with high cell-mediated immunity, is characterized by less than five lesions (often only one) and very few organisms (paucibacillary disease). The patient has strong cell-mediated immunity against the organism. The natural history of many TT leprosy patients is for spontaneous cure over several years. The polar lepromatous form (LL) has very limited cell-mediated immunity against the organism; lesions are numerous and contain many organisms (multibacillary). Between these two poles is every possible degree of infection, forming the borderline spectrum. Cases near the tuberculoid pole are called borderline tuberculoid (BT), those near the lepromatous pole are called borderline lepromatous (BL), and those in the middle are called borderline borderline (BB). Borderline Hansen disease is characteristically unstable, and with time, cases move from the TT to the LL pole, a process called downgrading.
Hansen disease may involve only the nerves. This pure neural disease may be indeterminate, tuberculoid, or lepromatous (paucibacillary or multibacillary) and is so classified. In Nepal and India, pure neural Hansen disease may represent as much as 5% of all new cases.
Early and Indeterminate Hansen Disease
Usually, the onset of Hansen disease is insidious. Prodromal symptoms are generally so slight that the disease is not recognized until the appearance of a cutaneous eruption. Actually, the first clinical manifestation in 90% of patients is numbness, and years may elapse before skin lesions or other signs are identified. The earliest sensory changes are loss of the senses of temperature and light touch, most often in the feet or hands. The inability to discriminate hot from cold may be lost before pinprick sensibility. Such dissociation of sensibility is especially suspicious. The distribution of these neural signs and their intensity will depend on the type of disease that is evolving.
Often, the first lesion noted is a solitary, poorly defined, hypopigmented macule that merges into the surrounding normal skin. Less often, erythematous macules may be present. Such lesions are most likely to occur on the cheeks, upper arms, thighs, and buttocks. Examination reveals that sensory functions are either normal or minimally altered. Peripheral nerves are not enlarged, and plaques and nodules do not occur. Histologically, a variable lymphocytic infiltrate (without granulomas) is seen, sometimes with involvement of the cutaneous nerves. Usually, no bacilli, or only a few, are seen on biopsy of this indeterminate form. It is the classification, not the diagnosis, that is indeterminate. Few cases remain in this state; they evolve into lepromatous, tuberculoid, or borderline types, or (if cell-mediated immunity is good) often spontaneously resolve and never develop other signs or symptoms of Hansen disease.
Tuberculoid Leprosy
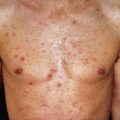
Tuberculoid lesions are solitary or few in number (five or less) and asymmetrically distributed. Lesions may be hypopigmented or erythematous and are usually dry, scaly, and hairless ( Fig. 17.1 ). The typical lesion of tuberculoid leprosy is the large, erythematous plaque with a sharply defined and elevated border that slopes down to a flattened atrophic center. This has been described as having the appearance of “a saucer right side up.” Lesions may also be macular and hypopigmented or erythematous, resembling clinically indeterminate lesions. The presence of palpable induration and neurologic findings distinguish tuberculoid lesions from indeterminate lesions clinically. The most common locations are the face, limbs, or trunk; the scalp, axillae, groin, and perineum are not involved.
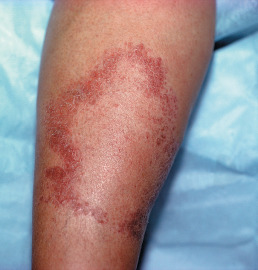
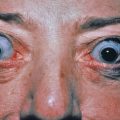
A tuberculoid lesion is anesthetic or hypesthetic and anhidrotic, and superficial peripheral nerves serving or proximal to the lesion are enlarged, tender, or both. The greater auricular nerve and the superficial peroneal nerve may be visibly enlarged. Nerve involvement is early and prominent in tuberculoid leprosy, leading to characteristic changes in the muscle groups served. There may be atrophy of the interosseous muscles of the hand, with wasting of the thenar and hypothenar eminences, contracture of the fingers, paralysis of the facial muscles, and footdrop. Facial nerve damage dramatically increases the risk for ocular involvement and vision loss.
The evolution of the lesions is generally slow. There is often spontaneous remission of the lesions in about 3 years, or remission may result sooner with treatment. Spontaneous involution may leave pigmentary disturbances.
Borderline Tuberculoid (BT) Leprosy
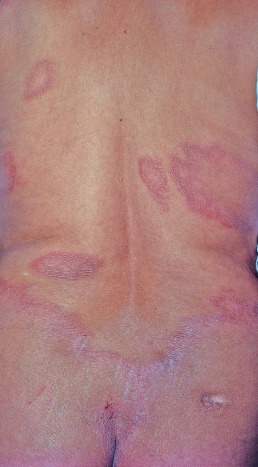
BT lesions are similar to tuberculoid lesions, except that they are smaller and more numerous ( Fig. 17.2 ). Satellite lesions around large macules or plaques are characteristic.
Borderline Borderline (BB) Leprosy
In BB leprosy, the skin lesions are numerous (but countable) and consist of red, irregularly shaped plaques. Small satellite lesions may surround larger plaques. Lesions are generalized but asymmetric. The edges of lesions are not as well defined as the ones seen at the tuberculoid pole. Nerves may be thickened and tender, but anesthesia is only moderate in the lesions.
Borderline Lepromatous (BL) Leprosy
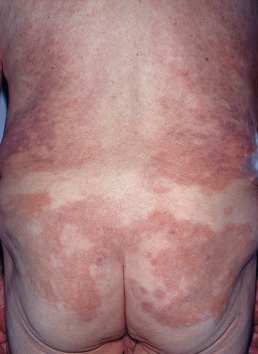
In BL leprosy, the lesions are symmetric and numerous (too many to count) and may include macules, papules, plaques, and nodules ( Fig. 17.3 ). The number of small, lepromatous lesions outnumbers the larger, borderline-type lesions. Nerve involvement appears later; nerves are enlarged, tender, or both, and it is important to note that involvement is symmetric. Sensation and sweating over individual lesions are normal. Patients usually do not show the features of full-blown lepromatous leprosy, such as madarosis (loss of the eyebrows), keratitis, nasal ulceration, and leonine facies.
Lepromatous Leprosy
Lepromatous leprosy may begin as such or develop after indeterminate leprosy or from downgrading of borderline leprosy. The cutaneous lesions of lepromatous leprosy consist mainly of pale macules ( Fig. 17.4 ) or diffuse infiltration of the skin. There is a tendency for the disease to become progressively worse without treatment. Lepromatous leprosy may be divided into a polar form (LLp) and a subpolar form (LLs); these forms may behave differently.
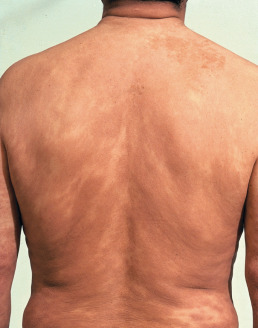
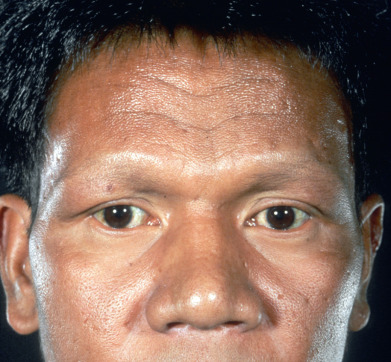
Macular lepromatous lesions are diffusely and symmetrically distributed over the body. Tuberculoid macules are large and few in number, whereas lepromatous macules are small and numerous. Lepromatous macules are poorly defined, show no change in skin texture, and blend imperceptibly into the surrounding skin. There is minimal or no loss of sensation over the lesions, no nerve thickening, and no change in sweating. A slow, progressive loss of hair takes place from the outer third of the eyebrows, then the eyelashes, and finally the body; however, the scalp hair usually remains unchanged.
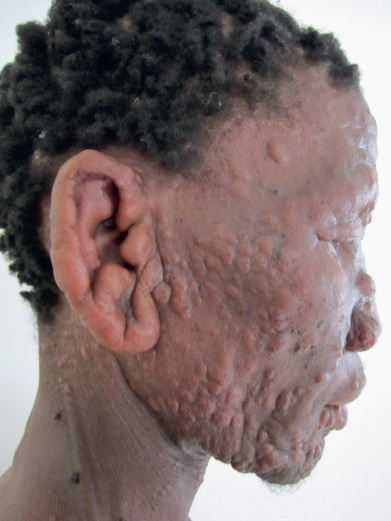
Lepromatous infiltrations may be divided into the diffuse, plaque, and nodular types. The diffuse type is characterized by the development of a diffuse infiltration of the face, especially the forehead, madarosis, and a waxy, shiny appearance of the skin, sometimes described as “varnished.” Diffuse leprosy of Lucio (DLL) is a striking form, uncommon except in western Mexico and certain other Latin American areas, where almost one third of lepromatous cases may be of this type. This form of lepromatous leprosy is characterized by diffuse lepromatous infiltration of the skin ( Fig. 17.5 ); localized lepromas do not form. A unique complication of this subtype is the reactional state referred to as Lucio phenomenon (erythema necroticans).
The infiltrations may be manifested by the development of nodules called lepromas ( Fig. 17.6 ). The early nodules are poorly defined and occur most often in acral parts: ears, brows, nose, chin, elbows, hands, buttocks, or knees.
Nerve involvement invariably occurs in lepromatous leprosy but develops very slowly. As with the skin lesions, nerve disease is bilaterally symmetric, usually in a stocking-glove pattern. This is frequently misdiagnosed as diabetic neuropathy in the United States if it is the presenting manifestation.
Histoid Leprosy
Histoid leprosy is an uncommon form of multibacillary Hansen disease in which skin lesions appear as large, yellow-red, shiny papules and nodules in the dermis or subcutaneous tissue. Lesions appear on a background of normal skin. They vary in size from 1–15 mm in diameter, and may appear anywhere on the body but favor the buttocks, lower back, face, and bony prominences. They may closely resemble molluscum contagiosum. This pattern may appear de novo but has mostly been described in patients with resistance to dapsone.
Nerve Involvement
Nerve involvement is characteristic and unique to Hansen disease. This neural predilection or neurotropism is a histopathologic hallmark of Hansen disease. Nerve involvement is responsible for the clinical findings of anesthesia within lesions (paucibacillary and borderline leprosy) and of a progressive stocking-glove peripheral neuropathy (lepromatous leprosy). The neuropathy is termed primary impairments (WHO grade 1). Secondary (or visible) impairments (WHO grade 2) are a consequence of the neuropathy and include skin fissures, wounds, clawing of digits, contractures, shortening of digits, and blindness. Neural damage leads to deformities and in endemic regions results in Hansen disease being a major cause of “limitations of activity” (formerly called disability) and “restrictions in social participation” (formerly called handicap). Neuropathy is present in 1.3%–3.5% of paucibacillary patients and 7.5%–24% of multibacillary patients undergoing MDT. Secondary impairments occur in 33%–56% of multibacillary patients. Neuropathy may progress, even after effective MDT, and secondary impairments may continue to appear for years as a consequence of the neuropathy. This requires patients with neuropathy to be constantly monitored, even though they are “cured” of their infection.
Nerve enlargement is rare in other skin diseases, so the finding of skin lesions with enlarged nerves should suggest Hansen disease. Nerve involvement tends to occur with skin lesions, and the pattern of nerve involvement parallels the skin disease. Tuberculoid leprosy is characterized by asymmetric nerve involvement localized to the skin lesions. Lepromatous nerve involvement is symmetric and not associated with skin lesions. Nerve involvement without skin lesions, called pure neural leprosy, can occur and may be either tuberculoid (paucibacillary) or lepromatous (multibacillary). Nerve disease can be symptomatic or asymptomatic.
Leprosy bacilli may be delivered to the nerves through the perineural and endoneural blood vessels. Once the bacilli transgress the endothelial basal lamina and are in the endoneurium, they enter resident macrophages or selectively enter Schwann cells. Damage to the nerves could then occur by the following mechanisms:
- 1.
Obstruction of neural vessels
- 2.
Vasculitis of neural vessels
- 3.
Interference with metabolism of the Schwann cell, making it unable to support the neuron
- 4.
Immunologic attack on endothelium or nerves
- 5.
Infiltration and proliferation of M. leprae in the closed and relatively nonexpandable endoneural and perineural spaces
Different and multiple mechanisms may occur in different forms of Hansen disease and in the same patient over time. The selective ability of M. leprae to enter Schwann cells is unique among bacteria. M. leprae –unique PGL-1, expressed abundantly on the surface of leprosy bacilli, binds selectively to the α2 G module of laminin 2. This α2 chain is tissue restricted and specifically expressed on peripheral nerve Schwann cells. Other accessory binding molecules may facilitate the binding and endocytosis. The nerves become immunologic targets when they present M. leprae antigens on their surface in the context of major histocompatibility complex (MHC) class II molecules. Schwann cells and thus nerves are usually protected from immunologic attack mediated by the adaptive immune system because they rarely present MHC class II antigens on their surface. In Hansen disease, expression of these immunologic molecules occurs on the surface of Schwann cells, making them potential targets for CD4+ cytotoxic T cells. This mechanism may be important in the nerve damage that occurs in type 1 (reversal) reactions.
Schwann cells have been infected with M. leprae in vitro. Infected Schwann cells with high bacterial load are reprogrammed into mesenchymal stem cell–like cells. In association with Schwann cells, these dedifferentiated cells attract histiocytes and form granulomas. The attracted histiocytes are infected by the mycobacteria-containing Schwann cells and are released from the granulomas. If this process also occurs in vivo, it may be the mechanism by which multibacillary disease is spread throughout the body from a reservoir of infected nerves.
The neural signs in Hansen disease are dysesthesia, nerve enlargement, muscular weakness and wasting, and trophic changes. The lesions of the vasomotor nerves accompany the sensory disturbances or may precede them. Dysesthesia develops in a progressive manner. The first symptom is usually an inability to distinguish hot and cold. Subsequently, the perception of light touch is lost, then that of pain, and last the sense of deep touch. At times, the sensory changes in large Hansen disease lesions are not uniform because of the variation in the involvement of the individual neural filaments supplying the area. Therefore the areas of dysesthesia may not conform to the distribution of any particular nerve and, except in lepromatous cases, are not symmetric.
Nerve involvement mainly affects (and is most easily observed in) the more superficial nerve trunks, such as the ulnar, median, radial, peroneal, posterior tibial, fifth and seventh cranial, and especially the great auricular nerve. Beaded enlargements, nodules, or spindle-shaped swellings may be found, which at first may be tender. Neural abscesses may form. The ulnar nerve near the internal condyle of the humerus may be as thick as the little finger, round, and stiff and is often easily felt several centimeters above the elbow.
Because the presentation of neural involvement in Hansen disease is variable, the diagnosis is often not suspected, especially in nonendemic areas. Even in endemic areas, the diagnosis may be delayed. Between one half and one third of patients with pure neural Hansen disease, a biopsy of hypesthetic skin can show specific leprotic skin changes, and if nonspecific inflammation is considered confirmatory, the positivity of such biopsies is greater than 50%. Therefore skin biopsy of a hypesthetic skin site should be considered before nerve biopsy when pure neural Hansen disease is a possibility.
As a result of the nerve damage, areas of anesthesia, paralysis, and trophic disorders in the peripheral parts of the extremities gradually develop. Muscular paralysis and atrophy generally affect the small muscles of the hands and feet or some of the facial muscles, producing weakness and progressive atrophy. Deeper motor nerves are only rarely involved. The fingers develop contractures, with the formation of a clawhand ( Fig. 17.7 ). Also, as the result of resorption of phalangeal bones, fingers and toes become shorter. Ptosis, ectropion, and a masklike appearance occur from damage to the fifth and seventh cranial nerves.