Eczema
The word eczema seems to have originated in 543 ad and is derived from the Greek word ekzein, meaning to “to boil forth” or “to effervesce.” The term encompasses such disorders as dyshidrotic eczema and nummular eczema (NE), but at times is used synonymously for atopic dermatitis (atopic eczema). The acute stage generally presents as a red edematous plaque that may have grossly visible, small, grouped vesicles. Subacute lesions present as erythematous plaques with scale or crusting. Later, lesions may be covered by a drier scale or may become lichenified. In most eczematous reactions, severe pruritus is a prominent symptom. The degree of irritation at which itching begins (the itch threshold) is lowered by stress. Itching is often prominent at bedtime and usually results in insomnia. Heat and sweating may also provoke episodes of itching.
Histologically, the hallmark of all eczematous eruptions is a serous exudate between cells of the epidermis (spongiosis), with an underlying dermal perivascular lymphoid infiltrate and exocytosis (lymphocytes present in overlying epidermis singly or in groups). Spongiosis is generally out of proportion to the lymphoid cells in the epidermis. This is in contrast to mycosis fungoides, which demonstrates minimal spongiosis confined to the area immediately surrounding the lymphocytes.
In most eczematous processes, spongiosis is very prominent in the acute stage, where it is accompanied by minimal acanthosis or hyperkeratosis. Subacute spongiotic dermatitis demonstrates epidermal spongiosis with acanthosis and hyperkeratosis. Chronic lesions may have minimal accompanying spongiosis, but acute and chronic stages may overlap because episodes of eczematous dermatitis follow one another. Scale corresponds to foci of parakeratosis produced by the inflamed epidermis. A crust is composed of serous exudate, acute inflammatory cells, and keratin. Eczema, regardless of cause, will manifest similar histologic changes if allowed to persist chronically. These features are related to chronic rubbing or scratching and correspond clinically to lichen simplex chronicus or prurigo nodularis. Histologic features at this stage include compact hyperkeratosis, irregular acanthosis, and thickening of the collagen bundles in the papillary portion of the dermis. The dermal infiltrate at all stages is predominantly lymphoid, but an admixture of eosinophils may be noted. Neutrophils generally appear in secondarily infected lesions. Spongiosis with many intraepidermal eosinophils may be seen in the early spongiotic phase of pemphigoid, pemphigus, and incontinentia pigmenti, as well as some cases of allergic contact dermatitis.
Atopic Dermatitis (Atopic Eczema)
Atopic dermatitis (AD) is a chronic, inflammatory skin disease characterized by pruritus and a chronic course of exacerbations and remissions. It is associated with other atopic conditions, including food allergies, asthma, allergic rhinoconjunctivitis, eosinophilic esophagitis, and eosinophilic gastroenteritis. Because AD usually precedes the appearance of these other atopic conditions, it has been proposed that AD is the first step in an “atopic march” whereby sensitization to allergens through the skin may lead to allergic responses in the airways or digestive tract. Although this sequence of atopic conditions does occur in many children, whether the AD is causal in the development of the other manifestations of atopy is unproved but plausible. For this reason, early and effective treatment of AD is encouraged in an effort to prevent other atopic conditions. The genetic defect(s) predisposing at-risk individuals to the development of AD is the same for asthma and allergic rhinoconjunctivitis, and thus it has been difficult to prove that AD is causal in the development of other atopic conditions.
Epidemiology
The prevalence of AD, asthma, and allergic rhinoconjunctivitis increased dramatically in the last half of the 20th century, becoming a major health problem in many countries. The increase began first in the most developed nations, and as the standard of living has increased worldwide, so has the prevalence of AD. Rates of AD are about 30% in the most developed nations and exceed 10% in many countries, resulting in a worldwide cumulative prevalence of 20%. In the most developed nations, the rates of AD plateaued in the 1990s, whereas developing nations have rates that continue to increase. Factors associated with high rates of AD are high latitude (perhaps associated with low levels of annual sun exposure) and lower mean annual temperature. A role for exposure to allergens thought to “trigger” AD is not supported by epidemiologic studies. Iceland has a very high rate of AD (27%) yet has no dust mites, few trees, and low pet ownership. However, children in Iceland often have positive skin prick tests to environmental allergens (24%). This questions the value of such tests in predicting causal environmental allergens in AD. Girls are slightly more likely to develop AD. In the United States an increased risk of AD during the first 6 months of life is noted in infants with African and Asian race/ethnicity, male gender, greater gestational age at birth, and a family history of atopy, particularly a maternal history of eczema. Other factors that increase the risk for the development of AD early in childhood include consumption of a Western diet, birth order (first children at greater risk), and delivery by cesarean section, all of which alter the intestinal microbiome. Exposure to antibiotics prenatally during the first or second and third trimesters also increases risk of AD. Therefore biodiversity in the gut microbiome seems to be protective. Gut colonization with Clostridium cluster I is associated with development of AD. Dog ownership before age 1 year decreases the risk of developing AD by age 4, but cat ownership has no effect. The hygiene hypothesis suggests that being raised on a farm lowers the risk of AD whereas living in modernized, cleaner indoor environments leads to a higher risk of AD.
About 50% of cases of AD appear in the first year of life, the vast majority within the first 5 years of life, and the remaining cases of “adult” AD usually before age 30. Atopy is now so common in the population that most individuals have a family history of atopy. Elevated immunoglobulin E (IgE) levels are not diagnostic of atopic disease in the adult. Therefore elevated IgE and a family history of “atopy” in an adult with new-onset dermatitis should not be used to confirm the diagnosis of adult AD. Adult AD should only be considered when the dermatitis has a characteristic distribution and when other significant diagnoses, such as allergic contact dermatitis, photodermatitis, and cutaneous T-cell lymphoma, have been excluded. Rather, a dermatologist should infrequently make the diagnosis of adult “atopic dermatitis” for a dermatitis appearing for the first time after age 30.
Genetic Basis and Pathogenesis
Eighty percent of identical twins show concordance for AD. A child is at increased risk of developing AD if either parent is affected. More than one quarter of offspring of atopic mothers develop AD in the first 3 months of life. If one parent is atopic, more than half the children will develop allergic symptoms by age 2. This rate rises to 79% if both parents are atopic. All of these findings strongly suggested a genetic cause for AD. Filaggrin is a protein encoded by the gene FLG, that resides in the epidermal differentiation complex (EDC) on chromosome 1q21. Filaggrin is processed by caspase 14 during terminal keratinocyte differentiation into highly hydroscopic pyrrolidone carboxylic acid and urocanic acid, collectively known as the “natural moisturizing factor” (NMF). Null mutations in FLG lead to reduction in NMF, which probably contributes to the xerosis that is almost universal in AD. Transepidermal water loss (TEWL) is increased. This may be caused by subclinical dermatitis, but also by abnormal delivery of lamellar body epidermal lipids (especially ceramide) to the interstices of the terminally differentiated keratinocytes. The resulting defective lipid bilayers retain water poorly, leading to increased TEWL and clinical xerosis. Ichthyosis vulgaris is caused by mutations in the FLG gene and is frequently associated with AD. Four FLG mutations have an estimated combined allelic frequency of 7%–10% in individuals of European descent. Different FLG gene mutations are associated with AD in other ethnicities although the rates do not necessarily match AD prevalence, demonstrating that the disease is multifactorial. Filaggrin 2 (FLG2), also in the EDC and with similar function to FLG, is associated with persistent AD in African Americans. Inheriting one null FLG mutation slightly increases one’s risk of developing AD, and inheriting two mutations, either as a homozygote or a compound heterozygote, dramatically increases one’s risk. Between 42% and 79% of persons with one or more FLG null mutations will develop AD. However, 40% of carriers with FLG null mutations never have AD. FLG mutations are associated with AD that presents early in life, tends to persist into childhood and adulthood, and is associated with wheezing in infancy and with asthma. FLG mutations are also associated with allergic rhinitis and keratosis pilaris, independent of AD. Hyperlinear palms are strongly associated with FLG mutations, with a 71% positive predictive value (PPV) for marked palmar hyperlinearity. LAMA3 gene mutations, encoding the alpha chain of laminin 5, may also predispose to AD. In murine models, decreased Claudin 3 expression can lead to leakage of sweat in the superficial dermis contributing to impaired sweating in atopic dermatitis.
Not all cases of AD are associated with FLG mutations. AD patients often demonstrate immunologic features consistent with a T-helper 2 (Th2) phenotype, with elevated IgE, eosinophils on skin biopsy, and positive skin tests and radioallergosorbent test (RAST). The cytokines, especially interleukin-4 (IL-4) and interleukin-13 (IL-13), that are released due to the Th2 immune response play an important role in AD by increasing inflammatory cell infiltration and stimulating the inflammatory feedback loop. Basophils and innate type 2 lymphoid cells can also secrete IL-4 and IL-13. Thymic stromal lymphopoietin (TSLP) is an important interleukin-7 (IL-7)–like cytokine that, through its interaction with Th2 cells, basophils, mast cells, and dendritic cells, promotes the secretion and production of Th2 cytokines and the development of inflammatory Th2 CD4+ T cells (through production of OL40L). TSLP is produced by keratinocytes and is found in high levels in AD skin lesions. In addition, interleukin-31 (IL-31) is produced by Th2 and Th22 cells. IL-31 binds directly to nerves leading to itching and also downregulates expression of filaggrin. The JAK-STAT pathway is also critical in causing overactivation of the TH2 response and itch. Interleukin-17 (IL-17) and interleukin-22 (IL-22), which are released by Th17 cells, are also elevated in AD. Thus AD appears to represent a disorder characterized by a barrier defect that activates a specific Th2 response leading to a cycle of inflammation mediated by various inflammatory signals. The cytokines produced then worsen the already defective barrier. This leads to a vicious cycle of barrier failure and progressive inflammation, producing a chronic, relapsing, pruritic disorder, and explains why moisturization alone is not enough once the inflammatory cascade has started.
Cotter DG, et al: Emerging therapies for atopic dermatitis: JAK inhibitors. Journal of the American Academy of Dermatology 2018; 78(3): S53-62.
Yamaga K, et al: Claudin-3 Loss Causes Leakage of Sweat from the Sweat Gland to Contribute to the Pathogenesis of Atopic Dermatitis. Journal of Investigative Dermatology 2018; 138(6): 1279-87.
Prevention in High-Risk Children
Extensive studies have been undertaken to determine whether it is possible to prevent the development of AD in children at high risk—those with parents or siblings with atopy. The most promising studies now repeated multiple times show that early moisturization, starting before 3 weeks of life, with thick emollient, may prevent AD in children at high risk of developing AD. Twice daily moisturization with various emollients such as petrolatum, sunflower seed oil, and others leads to an approximately 50% reduction in the expected rate of AD development in the first 6 months of life. This supports the idea that breaks in the skin barrier are an essential step in developing AD.
Switching to soy formula does not appear to reduce the risk of developing AD. Prolonged exclusive breastfeeding beyond 3–4 months of age is not protective for the development of AD. Extensively hydrolyzed casein formulas may be used as a supplement or substitute for breast milk during the first 4 months of life. Maternal allergen avoidance during pregnancy does not reduce the risk of AD in the offspring. Probiotic administration during and after pregnancy has been shown to decrease AD incidence by 14% based on data from a meta-analysis. However, the type of probiotic to use, exactly when to start, and the safety during pregnancy are not fully elucidated. House dust mite (HDM) avoidance does not reduce AD, even in sensitized individuals, and high levels of HDMs in the environment in early life reduce AD risk.
Food Allergy
The role of food allergy in AD is complicated, and the purported role of foods in AD has changed in recent years. Approximately 35% of children with moderate to severe AD have food allergies. However, 85% of children with AD will have elevated IgE to food or inhalant allergens, making a diagnosis of food allergy with serum or prick tests alone challenging due to the high false-positive rate of testing. Before food allergy testing is undertaken, treatment of the AD should be optimized. Parents are often seeking a “cause” for the child’s AD, when in fact it could be controlled with appropriate topical measures. Food restriction diets can be difficult and could put the child at risk for malnourishment and possibly development of allergy and should never be pursued without the oversight of an allergist and nutritionist to ensure proper nutrition. Food allergy should be pursued only in younger children with moderate to severe AD in whom standard treatments have failed. These children should also have a history of possible triggering of AD by specific food exposures. Testing, if performed, should be targeted foods to which the child is likely to be exposed, but generally wheat, egg, soy, cow’s milk, and peanut testing has been the most relevant to the AD. Double-blind placebo-controlled food challenges are the “gold standard” for diagnosing food allergy. Skin prick tests have a high negative predictive value (>95%) but a PPV of only 30%–65%. For example, more than 8% of the U.S. population has a positive prick test to peanut, but only 0.4% are actually clinically allergic. Possible food allergy detected by testing should be confirmed by clinical history. A positive RAST or skin prick test for a food that the child rarely or never ingests is probably not causally relevant to the child’s AD. Higher serum IgE levels and larger wheal sizes (>8–10 mm) are associated with greater likelihood of reacting to these foods when challenged. About 90% of food allergy is caused by a limited number of foods, as follows:
- •
Infants: cow’s milk, egg, soybean, wheat
- •
Children (2–10 years): cow’s milk, egg, peanut, tree nuts, fish, crustacean shellfish, sesame, kiwi fruit
- •
Older children: peanut, tree nuts, fish, shellfish, sesame, pollen-associated foods
There has been a rapid rise in peanut allergy in the United States. Recommendations to limit peanut exposure in childhood has been revised. A large randomized placebo control trial showed that early exposure in children at high risk for peanut allergy (due to strong family history of atopy) decreased the rate of development of peanut allergy by 86%. Therefore the revised guidelines that were developed in conjunction with dermatologists and allergists for peanut exposure are based on AD in the child. Of course a whole peanut should never be given to an infant due to choking hazard; therefore this was done with smooth peanut butter or the peanut snack Bamba (trademark).
- 1.
Children with severe AD or egg allergy should be considered for peanut testing before exposure to peanuts around 4–6 months of age.
- 2.
Mild to moderate AD: Introduce peanuts around 6 months.
- 3.
No AD or food allergy: Introduce peanuts in accordance with family and cultural preferences.
Aeroallergens
It is debated how much of a role aeroallergens play in the pathophysiology of AD. Although early exposure to allergens may lessen the incidence of AD, exposure to dust mites, animal allergens, mold, pollen, tree allergens, and other airborne allergens in those who are allergic may exacerbate AD. Testing for IgE responses to aeroallergens may not be predictive of their effect on patients with AD. In patients who note worsening around specific allergens, they should avoid these. Common allergens to avoid if possible include tobacco smoke, pollen, house dust mites, cats, and dogs (if allergic) although early exposure to dogs may prevent AD. Reduction of dust mite exposure can be achieved through vacuuming with a filtered vacuum, using dust mite covers on mattresses, avoidance of stuffed animals, and wall-to-wall carpeting if feasible.
Clinical Manifestations
AD can be divided into three stages: infantile AD, occurring from 2 months to 2 years of age; childhood AD, from 2–10 years; and adolescent/adult AD. In all stages, pruritus is the hallmark. Itching often precedes the appearance of lesions, thus the concept that AD is “the itch that rashes.” Useful diagnostic criteria include those of Hannifin and Rajka, the UK Working Party, and the American Academy of Dermatology’s Consensus Conference on Pediatric Atopic Dermatitis ( Boxes 5.1 and 5.2 ). These criteria have specificity at or above 90% but have much lower sensitivities (40%–100%). Therefore these criteria are useful for enrolling patients in studies and ensuring that they have AD, but less practical in diagnosing a specific patient with AD.
Eichenfield LF, et al: Consensus conference on pediatric atopic dermatitis. J Am Acad Dermatol 2003; 49: 1088-95
Hanifin JM, et al: Diagnostic features of atopic dermatitis. Acta Derm Venereol (Stockh) 1980; (Suppl. 92): 44-7
Major Criteria
Must have three of the following:
- 1.
Pruritus
- 2.
Typical morphology and distribution
- •
Flexural lichenification in adults
- •
Facial and extensor involvement in infancy
- •
- 3.
Chronic or chronically relapsing dermatitis
- 4.
Personal or family history of atopic disease (e.g., asthma, allergic rhinitis, atopic dermatitis)
- 1.
Minor Criteria
Must also have three of the following:
- 1.
Xerosis
- 2.
Ichthyosis/hyperlinear palms/keratosis pilaris
- 3.
IgE reactivity (immediate skin test reactivity, RAST test positive)
- 4.
Elevated serum IgE
- 5.
Early age of onset
- 6.
Tendency for cutaneous infections (especially Staphylococcus aureus and HSV)
- 7.
Tendency to nonspecific hand/foot dermatitis
- 8.
Nipple eczema
- 9.
Cheilitis
- 10.
Recurrent conjunctivitis
- 11.
Dennie-Morgan infraorbital fold
- 12.
Keratoconus
- 13.
Anterior subcapsular cataracts
- 14.
Orbital darkening
- 15.
Facial pallor/facial erythema
- 16.
Pityriasis alba
- 17.
Itch when sweating
- 18.
Intolerance to wool and lipid solvents
- 19.
Perifollicular accentuation
- 20.
Food hypersensitivity
- 21.
Course influenced by environmental and/or emotional factors
- 22.
White dermatographism or delayed blanch to cholinergic agents
- 1.
HSV, Herpes simplex virus; IgE, immunoglobulin E; RAST, radioallergosorbent assay.
Essential Features
- 1.
Pruritus
- 2.
Eczema
- •
Typical morphology and age-specific pattern
- •
Chronic or relapsing history
- •
Important Features
- 1.
Early age at onset
- 2.
Atopy
- 3.
Personal and/or family history
- 4.
IgE reactivity
- 5.
Xerosis
Associated Features
- 1.
Atypical vascular responses (e.g., facial pallor, white dermatographism)
- 2.
Keratosis pilaris/ichthyosis/hyperlinear palms
- 3.
Orbital/periorbital changes
- 4.
Other regional findings (e.g., perioral changes/periauricular lesions)
- 5.
Perifollicular accentuation/lichenification/prurigo lesions
Infantile Atopic Dermatitis
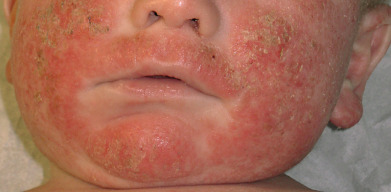
Fifty percent or more of AD cases present in the first year of life, but usually not until after 2 months. Widespread dermatitis in children under 2 months may be from an irritant, ichthyosis, or a hallmark of severe immunodeficiency. AD in infancy usually begins as erythema and scaling of the cheeks ( Fig. 5.1 ). The eruption may extend to the scalp, neck, forehead, wrists, extensor extremities, and buttocks. There can be overlap with seborrheic dermatitis on the scalp and in the folds, but papular or nodular involvement in the axillae and inguinal folds is more typical of scabies infestation. Children with AD who have FLG gene mutations specifically have more cheek and extensor arm/hand involvement. There may be significant exudate; secondary effects from scratching, rubbing, and infection include crusts, infiltration, and pustules, respectively. Occlusion of saliva due to teething, extensive breastfeeding, and drooling may cause the cheeks and upper chest to flare and thick emollients can help prevent this. Breastfeeding should not be curtailed. The infiltrated plaques eventually take on a characteristic lichenified appearance. The infantile pattern of AD usually disappears by the end of the second year of life.
Worsening of AD is often observed in infants after immunizations and viral infections likely due to activation of the immune system. Partial remission may occur during the summer, with relapse in winter. This may relate to the therapeutic effects of ultraviolet (UV) B light and humidity in many atopic patients, as well as the aggravation by wool and dry air in the winter. Extensive airborne environmental allergies may lead to worsening in the warmer months.
Childhood Atopic Dermatitis
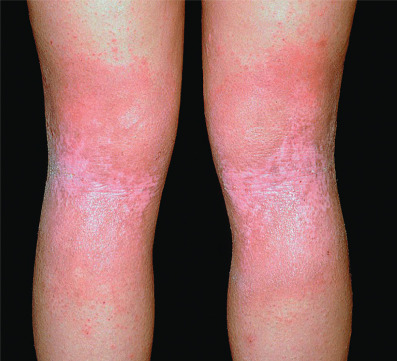
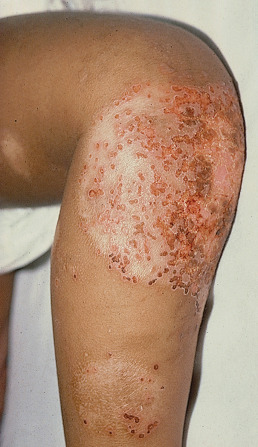
During childhood, lesions tend to be less exudative. The classic locations are the antecubital and popliteal fossae ( Fig. 5.2 ), flexor wrists, ankles, eyelids, face, and around the neck. Lesions are often lichenified, indurated plaques. These are intermingled with isolated, excoriated, 2–4 mm papules that are scattered more widely over the uncovered parts. Nummular morphology and involvement of the feet are more common in childhood AD.
Pruritus is a constant feature, and most of the cutaneous changes are secondary to it. Itching is paroxysmal. Scratching induces lichenification and may lead to secondary infection. A vicious cycle may be established, the itch-scratch cycle, as pruritus leads to scratching, and scratching causes secondary changes that in them cause itching. Instead of scratching causing pain, in the atopic patient the “pain” induced by scratching is perceived as itch and induces more scratching. The scratching impulse is beyond the control of the patient. Severe bouts of scratching occur during sleep, leading to poor rest and chronic tiredness in atopic children. This can affect school performance. Parents often scold children who are scratching, and this leads to more anxiety and thus more scratching.
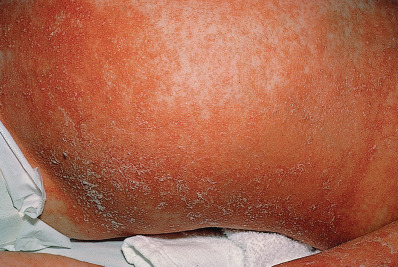
Severe AD involving a large percentage of the body surface area (BSA) can be associated with growth retardation ( Fig. 5.3 ). Restriction diets and steroid use may exacerbate growth impairment. Aggressive management of such children with phototherapy or systemic immunosuppressive agents may allow for rebound growth. Children with severe AD may also have substantial psychological disturbances. Parents should be questioned with regard to school performance and socialization. Although using light therapy and systemic immunosuppressive or immunomodulatory therapy in children can be daunting for patients and practitioners, the benefits to quality of life can dramatically outweigh the risks.
Atopic Dermatitis in Adolescents and Adults
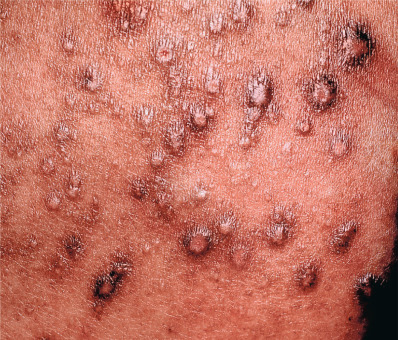
Most adolescents and adults with AD will give a history of childhood disease. AD will begin after age 18 years in only 6%–14% of patients diagnosed with AD. One exception is the patient who moves from a humid, tropical region to a more temperate area of higher latitude. This climatic change is often associated with the appearance of AD. In older patients, AD may occur as localized erythematous, scaly, papular, exudative, or lichenified plaques. In adolescents, the eruption often involves the classic antecubital and popliteal fossae, front and sides of the neck, forehead, and area around the eyes. In older adults, the distribution is generally less characteristic, and localized dermatitis may be the predominant feature, especially hand, nipple, or eyelid eczema. At times, the eruption may generalize, with accentuation in the flexures. The skin generally is dry and somewhat erythematous. Lichenification and prurigo-like papules are common ( Fig. 5.4 ). Papular lesions tend to be dry, slightly elevated, and flat topped. They are almost always excoriated and often coalesce to form plaques. Staphylococcal colonization is common. In darker-skinned patients, the lesions are often hyperpigmented, frequently with focal hypopigmented areas related to healed excoriations.
Itching usually occurs in crises or paroxysms. Adults frequently complain that flares of AD are triggered by acute emotional upsets. Stress, anxiety, and depression reduce the threshold at which itch is perceived and result in damage to the epidermal permeability barrier, further exacerbating AD. Atopic persons may sweat poorly and may complain of severe pruritus related to heat or exercise. Physical conditioning and liberal use of emollients improve this component, and atopic patients can participate in competitive sports.
Even in patients with AD in adolescence or early adulthood, improvement usually occurs over time, and dermatitis is uncommon after middle life. In general, these patients retain mild stigmata of the disease, such as dry skin, easy skin irritation, and itching in response to heat and perspiration. They remain susceptible to a flare of their disease when exposed to a specific allergen or environmental situation. Photosensitivity develops in approximately 3% of AD patients and may manifest as either a polymorphous light eruption–type reaction or simply exacerbation of the AD by UV exposure. The average age for photosensitive AD is the middle to late thirties. Human immunodeficiency virus (HIV) infection can also serve as a trigger, and new-onset AD in an at-risk adult should lead to counseling and testing for HIV if warranted.
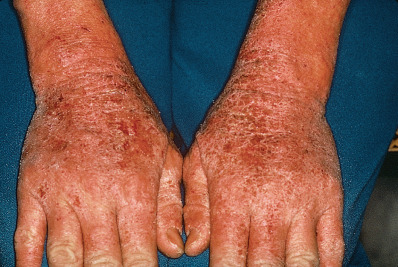
The hands, including the wrists, are frequently involved in adults, and hand dermatitis is a common problem for adults with a history of AD. It is common for irritant or atopic hand dermatitis to appear in young women after the birth of a child, when increased exposure to soaps and water triggers their disease. There is a new trend in children who can buy kits or mix their own various household liquids such as glue, borax, contact solution, baking soda and others to make “slime”. This can lead to irritant dermatitis or worsening of hand atopic dermatitis. Wet work is a major factor in hand eczema in general, including those patients with AD. Atopic hand dermatitis can affect both the dorsal and the palmar surface ( Fig. 5.5 ). Keratosis punctata of the creases, a disorder seen almost exclusively in black persons, is also more common in atopic patients. Patients with AD have frequent exposure to preservatives and other potential allergens in the creams and lotions that are continually applied to their skin. Contact allergy may manifest as chronic hand eczema. Patch testing can help differentiate an allergic contact dermatitis.
Eyelids are often involved ( Fig. 5.6 ). In general, the involvement is bilateral and the condition flares with cold weather. As in hand dermatitis, irritants and allergic contact allergens must be excluded by a careful history and patch testing.
Gittler JK, et al: “Slime” May Not be so Benign: A Cause of Hand Dermatitis. The Journal of pediatrics. 2018.

Associated Features and Complications
Cutaneous Stigmata
A linear transverse fold just below the edge of the lower eyelids, known as the Dennie-Morgan fold, is indicative of the atopic diathesis, although it may be seen with any chronic dermatitis of the lower lids. In atopic patients with eyelid dermatitis, increased folds and darkening under the eyes is common. When there is extensive facial involvement, the nose is still typically spared. This is called the “headlight sign.” The axillary vault and inguinal folds are also typically spared likely due to high humidity in these areas. A prominent nasal crease may also be noted due to chronic upward wiping of the nose when there is rhinitis secondary to seasonal allergies. This is called the “nasal salute.” When taken together with other clinical findings, these remain helpful clinical signs.
The less involved skin of atopic patients is frequently dry and slightly erythematous and may be scaly. Histologically, the apparently normal skin of atopic patients is frequently inflamed subclinically. The dry, scaling skin of AD may represent low-grade dermatitis. Pityriasis alba is a form of subclinical dermatitis, frequently atopic in origin. It presents as poorly marginated, hypopigmented, slightly scaly patches on the cheeks, upper arms, and trunk, typically in children and young adults with types III to V skin. It usually responds to emollients and mild topical steroids, preferably in an ointment base.
Keratosis pilaris (KP) consists of horny follicular lesions of the outer aspects of the upper arms, legs, cheeks, and buttocks and is often associated with AD, AD and occurs in patients with filagrin mutations. The keratotic papules on the face may be on a red background, a variant of KP called keratosis pilaris rubra faceii. KP is often refractory to treatment. Moisturizers alone are only partially beneficial. Some patients will respond to topical lactic acid, urea, or retinoids but they can easily irritate the skin of atopic patients and should be avoided in young children. If older patients desire, treatment should begin with applications only once or twice a week. KP must be differentiated from follicular eczema which tends to affect the trunk and is often more prominent in patients with skin types III-VI.
Thinning of the lateral eyebrows (Hertoghe sign) is sometimes present. This apparently occurs from chronic rubbing caused by pruritus and subclinical dermatitis. Hyperkeratosis and hyperpigmentation, which produce a “dirty neck” appearance, are also common in AD patients.
Vascular Stigmata
White dermatographism is blanching of the skin at the site of stroking or scratching. Chronic exposure to the vasoconstrictive effects of topical and oral steroids may lead to erythroderma due to vasodilation when steroids are tapered. This can lead to dysesthesias and belies the importance of using maintenance therapies that limit steroid exposure or lessen strength.
Atopic patients are at increased risk of developing various forms of urticaria, including contact urticaria. Episodes of contact urticaria may be followed by typical eczematous lesions at the affected site because skin that is scratched may flare with AD.
Ophthalmologic Abnormalities
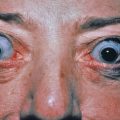
Up to 10% of patients with AD develop cataracts, either anterior or posterior subcapsular. Posterior subcapsular cataracts in atopic individuals are indistinguishable from corticosteroid-induced cataracts. Development of cataracts is more common in patients with severe dermatitis. Keratoconus is an uncommon finding, occurring in approximately 1% of atopic patients. Contact lenses, keratoplasty, and intraocular lenses may be required to treat this condition. Environmental allergies may also lead to allergic rhinoconjunctivitis.
Susceptibility to Infection
Chronic eczematous lesions are often colonized with Staphylococcus aureus. Staphylococcus epidermitis may actually be protective as it is predominates in patients with fewer flares. In addition, the apparently normal nonlesional skin of atopic patients may also be colonized by S. aureus. The finding of increasing numbers of pathogenic staphylococci on the skin of a patient with AD is frequently associated with weeping and crusting of skin lesions, retroauricular and infraauricular and perinasal fissures, folliculitis, and adenopathy. In a flaring atopic patient, the possibility of secondary infection must be considered. IgE antibodies directed against Staphylococcus and its toxins have been documented in some atopic individuals. Staphylococcal production of superantigens is another possible mechanism for staphylococcal flares of disease. Treatment of lesions of AD with topical steroids is associated with reduced numbers of pathogenic bacteria on the surface, even if antibiotics are not used. Despite the frequent observation that the presence of staphylococcal infection of lesions of AD is associated with worsening of disease, it has not been proven that oral antibiotics make a long term difference in the course of AD. Nonetheless, treatment of the “infected” AD patient with oral antibiotics is a community standard of dermatologists worldwide.
With the widespread presence of antibiotic-resistant S. aureus, dermatologists have shifted from the chronic use of oral antibiotics in managing patients with frequent flares of AD associated with staphylococcal infection. Rather, dilute sodium hypochlorite (bleach) baths and reduction of nasal carriage have become the basis for preventing infection-triggered AD. The typical formula is one quarter of a US cup (approximately 60 milliliters) of plain, unscented, NOT concentrated, NOT splashproof bleach per 20 gallon bathtub of water. In patients with AD and frequent infections, chronic suppressive oral antibiotic therapy may stabilize the disease. Options include cephalosporins, trimethoprim-sulfamethoxazole (TMP-SMX), clindamycin, and (in older patients) doxycycline. Identifying and treating S. aureus carriers in the family and pets may also be of benefit. An unusual complication of S. aureus infection in patients with AD is subungual infection, with osteomyelitis of the distal phalanx. In atopic patients with fever who appear very toxic, the possibility of streptococcal infection must be considered. These children may require hospital admission and intravenous antibiotics. Group A streptococcal superinfection of AD typically presents with a more vesiculopustular look that may mimic herpes simplex virus (HSV). Culture of the skin can differentiate and guide therapy typically with a penicillin- or cephalosporin-based antibiotic.
Patients with AD have increased susceptibility to viral superinfection, especially from HSV, varicella-zoster virus (VZV), enteroviruses (Coxsackie), vaccinia, and molluscum. Kaposi varicelliform infection is a term used to describe an acute vesicular eruption on top of AD and is usually caused by HSV, VZV, or enterovirus superinfection. Eczema herpeticum is seen most frequently in young children and is usually associated with HSV type 1 transmitted from a caretaker or sibling. Once infected, there can be frequent flares of eczema herpeticum. Eczema herpeticum presents as the sudden appearance of vesicular, pustular, crusted, or eroded lesions concentrated in the areas of dermatitis ( Fig. 5.7 ). The patients may have a fever and appear toxic. The lesions may continue to spread, and most of the skin surface may become involved. Secondary staphylococcal or streptococcal infection is common, and local edema and regional adenopathy frequently occur. If lesions of eczema herpeticum occur on or around the eyelids, ophthalmologic evaluation is recommended. The severity of eczema herpeticum is quite variable, but most cases require systemic antiviral therapy and an antistaphylococcal antibiotic. Delayed administration of acyclovir in hospitalized patients is associated with prolonged hospital stay. Genetic variants in TSLP and interferon regulatory factor 2 (IFR-2) in addition to DOCK 8 mutations are associated with AD and eczema herpeticum.
Recently a more virulent form of “hand foot and mouth” caused by enterovirus A6 instead of the typical A16 (also termed Coxackie A16 ) has been recognized and labeled “eczema coxsackium.” Superinfection with the A6 strain often leads to very widespread papules and vesicles with low-grade fever. There will typically be at least a few lesions on the palms and soles and in the mouth but there is not the typical predominance in these areas compared with areas of AD. Therapy is supportive.
Vaccination against smallpox is contraindicated in persons with AD, even when the dermatitis is in remission. Widespread and even fatal vaccinia can occur in patients with an atopic diathesis.
Atopic individuals may also develop extensive flat warts or molluscum contagiosum (MC). If the infection is extremely exuberant, an immunodeficiency such as DOCK-8 mutation should be ruled out. A common presentation of MC in children is a new patch of AD in an unusual area such as an axilla due to MC causing an immune response leading to a flare of AD. Chemical therapies such as salicylic acid and cantharidin may cause severe irritation within lesions of AD. Destruction with curettage (for molluscum), cryosurgery, or electrosurgery may be required to clear the lesions.
Differential Diagnosis
Typical AD in infancy and childhood is straightforward because of its characteristic morphology; predilection for symmetric involvement of the face, neck, and antecubital and popliteal fossae; and association with food allergy, asthma, and allergic rhinoconjunctivitis. Dermatoses that may resemble AD include seborrheic dermatitis (especially in infants), irritant or allergic contact dermatitis, nummular dermatitis, photodermatitis, scabies, and cases of psoriasis with an eczematous morphology. Certain immunodeficiency syndromes (see later discussion) may exhibit a dermatitis remarkably similar or identical to AD. In older patients with new onset dermatitis, cutaneous T cell lymphoma and allergic contact dermatitis should be considered.
Histopathology
The histology of AD varies with the stage of the lesion, with many of the changes induced by scratching. Hyperkeratosis, acanthosis, and excoriation are common. Staphylococcal colonization may be noted histologically. Although eosinophils may not be seen in the dermal infiltrate, staining for eosinophil major basic protein (MBP) reveals deposition in many cases.
General Management
Education and Support
Parental and patient education is of critical importance in the management of AD. In the busy clinic setting, dermatologists frequently have insufficient time to educate patients adequately regarding the multiple factors that are important in managing AD. Educational formats that have proved effective have been immediate nursing education on the correct use of medications, weekly evening educational sessions, and multidisciplinary day treatment venues. There are also now multiple online resources from major academic centers, professional societies, and associations that can be helpful. In all cases, “written action plans” outlining a “stepwise approach” have been important for parent/patient education. In addition, patients with chronic disease often become disenchanted with medical therapies or simply “burn out” from having to spend significant amounts of time managing their skin disease. The psychological support that can be incorporated into educational sessions can help motivate parents/patients and keep them engaged in the treatment plan. Having a child with AD is extremely stressful and generates significant stress within the family. Sleep is lost by both the patient and the parents. Supportive educational techniques and sometimes professional behavioral health consultation can help the family cope with this burden. In addition, the dermatologist must consider the complexity and time commitment of any prescribed regimen and ensure that the parents/patient understand and are committed to undertaking the treatments proposed.
Barrier Repair
AD patients have an impaired epidermal barrier. The cornerstone of treatment and prevention of AD lies in addressing this problem. Patients should moisturize daily, especially immediately after bathing, ideally while the skin is still damp within a few minutes of getting out of the water. This may be with petrolatum or a petrolatum-based product, an oil-based product, a dimethicone-based product, vegetable shortening, or a “barrier repair” moisturizer that contains the essential lipids of the epidermal barrier. These special barrier repair moisturizers have similar benefits in AD to low-potency topical steroids. They are easier to apply and, if available to the patient, may enhance compliance. Petrolatum and petrolatum-based moisturizers are most often recommended and are often the least expensive. However, men with significant body hair, AD patients triggered by heat, and the very rare patient with true allergic contact dermatitis to petrolatum may be unable to tolerate petrolatum-based agents. Moisturizers should ideally be fragrance and formaldehyde free because these are two of the most common allergens in patients with AD. The ratio and content of ceramides in the epidermis of patients with AD is abnormal. Moisturizers with ceramides added have been shown to provide some benefit beyond the actual emollient. Patients should be instructed on the barrier-damaging properties of soaps, hot water, and scrubbing. The pH of the skin that promotes normal epidermal function is slightly acidic (the acid mantle) so synthetic detergents that have a more acidic pH are preferred to more basic soaps that have a high concentration of surfactant. Detergent use should be restricted to the axilla, groin, face, soles, and scalp. Oil-based cleansers can be used to “wash” the skin without water. For flares of AD, the soak and smear technique (soak in tub, then seal in water with a heavy moisturizer or medicated ointments) or wet dressings (wet wraps) with topical steroids can be very effective. In dry climates, AD patients may note some benefit with humidifiers. α-Hydroxy acid–containing products (lactic acid, glycolic acid) can be irritating and can exacerbate inflamed AD. These products should only be used for the xerosis of AD when there is absolutely no inflammation or pruritus.
Antimicrobial Therapy
When the AD patient has evidence of infection, treatment with topical or systemic antibiotics may be appropriate. Rather than treating once an infection occurs, it appears that the key in AD is to reduce nasal staphylococcal carriage preemptively and to keep the skin decolonized from Staphylococcus. Dilute sodium hypochlorite (bleach) baths have rapidly become a mainstay in AD patients. Twice-weekly bathing in a tepid bath with one quarter of a US cup (approximately 60 ml) of standard household bleach (5–6%) diluted in 20 gallons of water dramatically improves AD on the trunk and extremities, but less so on the face. For a smaller baby bathtub this is typically 2.5 mL = ![]() teaspoon per gallon of water. Many bleach manufacturers make splashless or double-concentrated bleach, which should be avoided. This treatment combines decolonization of the skin with hydration, addressing two of the major factors in worsening of AD. Dilute bleach baths have been shown to decrease Nuclear Factor (NF) kappa-beta and thus may serve a role as a primary antiinflammatory therapy as well. Adequate moisturizing after bathing is critical. Intranasal application of mupirocin is beneficial in reducing nasal carriage although resistance is emerging. In 80% of families, at least one parent is carrying the same staphylococcal strain as a colonized AD child. If the AD patient has recurrent infections, other carriers in the family and their pets are sought and treated aggressively. Recurrent infections, especially furunculosis, are a cardinal feature of children and adults with AD who have systemic immunologic abnormalities, especially hyper-IgE syndrome.
teaspoon per gallon of water. Many bleach manufacturers make splashless or double-concentrated bleach, which should be avoided. This treatment combines decolonization of the skin with hydration, addressing two of the major factors in worsening of AD. Dilute bleach baths have been shown to decrease Nuclear Factor (NF) kappa-beta and thus may serve a role as a primary antiinflammatory therapy as well. Adequate moisturizing after bathing is critical. Intranasal application of mupirocin is beneficial in reducing nasal carriage although resistance is emerging. In 80% of families, at least one parent is carrying the same staphylococcal strain as a colonized AD child. If the AD patient has recurrent infections, other carriers in the family and their pets are sought and treated aggressively. Recurrent infections, especially furunculosis, are a cardinal feature of children and adults with AD who have systemic immunologic abnormalities, especially hyper-IgE syndrome.
Environmental Factors
Stress, heat, sweating, and external irritants may precipitate an attack of itching and flare in the AD patient. Wool garments should be avoided. Addressing these triggers may improve the AD. Itch nerves are more active at higher temperatures, so overheating should be avoided. Irritants and allergens in the numerous products that AD patients may use can lead to flares of AD. Patients should avoid products that contain common allergens and should be evaluated for allergic contact dermatitis if a topical agent is associated with worsening of their AD.
Antipruritics
The primary treatment for the pruritus of AD is to reduce the severity of the AD. Antihistamines are frequently used for the pruritus of AD but are mainly beneficial for their sedative properties. If the patient has environmental allergies that lead to itch, the scratched skin can flare with AD, so in these patients long-acting histamine-2 blockers such as cetirizine, loratadine, and fexofenadine may help by decreasing the symptoms of the environmental allergies. Sedating antihistamines can lose some of their sedative effect if used consistently, so ideally they are only used when needed. Diphenhydramine, hydroxyzine, and doxepin can all be efficacious. Nonsedating antihistamines do not appear to benefit the pruritus of AD in standard doses. In some patients, gabapentin, selective serotonin reuptake inhibitors (SSRIs), mirtazapine, and even opiate antagonists may reduce pruritus. Applying ice during intense bouts of itch may help to “break” an itch paroxysm. Moisturizing lotions containing menthol, phenol, or pramoxine can be used between steroid applications to moisturize and reduce local areas of severe itch. More widespread use of topical doxepin (Sinequan) is limited by systemic absorption and sedation.
Specific Treatment Modalities
Topical Corticosteroid Therapy
Topical corticosteroids are the most common class of medications, along with moisturizers, used for the treatment of AD. They are effective and economical. Ointments are preferred because they can serve a double purpose as an emollient, they do not burn when applied, and they typically have fewer ingredients leading to lower of a chance of allergic contact dermatitis.
In infants, low-potency steroid ointments, are preferred. Regular application of emollients must be emphasized. Once corticosteroid receptors are saturated, additional applications of a steroid preparation contribute nothing more than an emollient effect. In most body sites, once-daily application of a corticosteroid is almost as effective as more frequent applications, at lower cost and with less systemic absorption and likely increased compliance. In some areas, twice-daily applications may be beneficial, but more frequent applications are almost never of benefit. Steroid phobia is common in parents and patients with AD. Less frequent applications of lower-concentration agents or intermittent use of steroids 2–5 times weekly, with emphasis on moisturizing, address these concerns. It can also be useful to point out that the human body must produce endogenous steroids daily to function. Application of topical corticosteroids under wet wraps or vinyl suit occlusion especially after soaking in a tub of water (soak and smear) can increase efficiency. For refractory areas, a stronger corticosteroid may be used. A more potent molecule is more appropriate than escalating concentrations of a weaker molecule because the effect of the latter plateaus rapidly as receptors become saturated, and the potent medication should be stopped when the flare subsides. Because AD is an inflammatory cascade that must be halted, it is important not to undertreat. Undertreatment leads to loss of faith on the part of the patient/parents and prolongs the suffering of the patient. For severe disease, using more potent topical steroids in short bursts of a few days to a week can help gain control of the disease. In refractory and relapsing AD, twice-weekly steroid application to the areas that commonly flare may reduce flares. Another maintenance method is to mix 1 part of hydrocortisone 2.5% ointment in with 1 to 4 parts of the patient’s preferred emollient 2–5 days per week, lessening the amount this is used as tolerated.
In older children and adults, medium-potency steroids are often used, except on the face, where milder steroids or calcineurin inhibitors are preferred. For thick plaques and lichen simplex chronicus–like lesions, extremely potent steroids may be necessary. Ointments are more effective because of their moisturizing properties and require no preservatives, reducing the likelihood of allergic contact dermatitis. If an atopic patient worsens or fails to improve after the use of topical steroids and moisturizers, the possibility of allergic contact dermatitis to a preservative or the corticosteroids must be considered. Contact allergy to the corticosteroid itself can occur. Corticosteroid allergy seldom manifests as acute worsening of the eczema. Instead, it manifests as a flare of eczema whenever the corticosteroid is discontinued, even for a day. This may be difficult to differentiate from stubborn AD.
Although the potential for local and even systemic toxicity from corticosteroids is real, the steroid must be strong enough to control the pruritus and remove the inflammation. Even in small children, strong topical steroids may be necessary in weekly pulses to control severe flares. Monitoring of growth parameters should be carried out in infants and young children.
Ideally prevention of AD flares is with just emollients, but patients with more severe disease often need some consistent antiinflammatory medicines (similar to the paradigm used for asthma). Maintenance therapy with topical steroids as discussed earlier can be with twice-weekly application, mixing a low-potency steroid in with an emollient or with nonsteroidal antiinflammatory medications.
Topical Calcineurin Inhibitors
Topical calcineurin inhibitors (TCIs) such as tacrolimus or pimecrolimus offer an alternative to topical steroids. Systemic absorption is generally not significant with either of these agents. Although a 0.03% tacrolimus ointment is marketed for use in children, it is unclear whether it really offers any safety advantage over the 0.1% formulation. Patients may experience a warm or stinging feeling in the skin when the medicines are first started. This tends to resolve and tolerability is improved if the ointment is applied to “bone-dry” skin. Patients experience less burning if eczematous patches are treated initially with a corticosteroid, with transition to a TCI after partial clearing. Improvement tends to be steady, with progressively smaller areas requiring treatment. TCIs are particularly useful on the eyelids and face, in areas prone to steroid atrophy, when steroid allergy is a consideration, or when systemic steroid absorption is a concern. Tacrolimus is more effective than pimecrolimus, with tacrolimus 0.1% ointment equivalent to triamcinolone acetonide 0.1%, and pimecrolimus equivalent to a class V or VI topical corticosteroid. There is a black box warning on calcineurin inhibitors in the United States, but a recent study that followed patients after marketing found no increase in malignancy in over 26,000 patient-years studied.
Topical Phosphodiesterase Inhibitors
Crisaborole is an antiinflammatory topical PDE4 inhibitor introduced in 2017 for AD in children over 2 years. It can be used on the face and skinfolds without concern for striae but may sting when applied. It may be most useful as a maintenance medication or for mild to moderate disease.
Tar
Crude coal tar 1%–5% in white petrolatum or hydrophilic ointment USP, or liquor carbonis detergens (LCDs) 5%–20% in hydrophilic ointment USP, is sometimes helpful for an area of refractory AD. Tar preparations are especially beneficial when used for intensive treatment for adults in an inpatient or day care setting, especially in combination with UV phototherapy. Goeckerman therapy with tar and UVB in a day treatment setting will lead to improvement in more than 90% of patients with refractory AD, and prolonged remission can be induced.
Phototherapy
If topical modalities fail to control AD, phototherapy is another step on the therapeutic ladder. Narrow-band (NB) UVB is highly effective and has replaced broadband UV for treating AD. When acutely inflamed, AD patients may tolerate UV poorly. Initial treatment with soak and smear topical steroids or a systemic immunosuppressive may cool off the skin enough to institute UV treatments. Patients with significant erythema must be introduced to UV at very low doses to avoid nonspecific irritancy and flaring of the AD. Often, the initial dose is much lower and the dose escalation much slower than in patients with psoriasis. In acute flares of AD, UVA I can be used. For patients unresponsive to NB UVB, photochemotherapy with psoralen plus UVA (PUVA) can be effective but the benefits must outweigh the risks. It requires less frequent treatments, and can be given either topically (soak/bath PUVA) or systemically (oral PUVA).
Systemic Therapy
Dupilumab is an IL-4 receptor inhibitor that thus blocks the function of IL-4 and IL-13. It is an injection given every 2 weeks after a loading dose and decreases pruritus and Eczema Area and Severity Index (EASI) scores. It is the first targeted systemic therapy for AD. The most common significant side effect is keratoconjunctivitis.
Systemic Corticosteroids.
In general, systemic corticosteroids should be avoided due to concerns for reexacerbation when the steroids are stopped. Some will use systemic steroids to control acute exacerbations. In patients requiring systemic steroid therapy, short courses (≤3 weeks) are preferred. If repeated or prolonged courses of systemic corticosteroids are required to control the AD, phototherapy or a steroid-sparing systemic therapy should be considered. Chronic corticosteroid therapy for AD frequently results in significant steroid-induced side effects such as striae, infection, boney changes, and growth delay. Osteoporosis in women requires special consideration and should be addressed with a bisphosphonate early in the course of therapy when bone loss is greatest. Preventive strategies, such as calcium supplements, vitamin D supplementation, bisphosphonates, regular exercise, and smoking cessation, should be strongly encouraged. Dual-energy x-ray absorptiometry (DEXA) scans are recommended.
Cyclosporine
Cyclosporine (cyclosporin A) is highly effective in the treatment of severe AD, but the response is rarely sustained after the drug is discontinued. It is very useful to gain rapid control of severe AD. Cyclosporine has been shown to be safe and effective in both children and adults, although probably tolerated better in children. Potential long-term side effects, especially renal disease and hypertension, require careful monitoring, with attempts to transition the patient to a potentially less toxic agent if possible. The dose range is 3–5 mg/kg in children and 150–300 mg in adults, with a better and more rapid response at the higher end of the dose range. Cyclosporin should not be used long term due to risks of renal and other toxicity. Rebound flaring of AD is possible and can be significant after stopping cyclosporin A, and a plan should be in place for this eventuality.
Methotrexate
Methotrexate has a long history of safe use in children for other diseases. Recently studies have been published demonstrating that its efficacy is similar to cyclosporine, although the onset to action is significantly slower and it is not FDA approved for this indication. Doses are similar to what is used in psoriasis (0.3–0.6 mg/kg given once weekly (up to 25 mg) in children and 10–25 mg weekly in adults. Patients on methotrexate should take folic acid as well. The time to relapse with methotrexate may be longer than with cyclosporine. Switching from oral to subcutaneous administration may increase the benefit.
Other Immunosuppressive Agents
Several other immunosuppressive agents have demonstrated efficacy in patients with AD. They include axathioprine and mycophenolate mofetil. These agents do not appear to be as quick to work as cyclosporine. Patients with FLG mutations respond to azathioprine and methotrexate, but the response is slower than in those without FLG mutations. However, over time they may have a better safety profile, so patients requiring long-term immunosuppression may benefit from one of these agents. The dosing of azathioprine is guided by the serum thiopurine methyltransferase level. Mycophenolate mofetil (MMF) is generally well tolerated and, as with azathioprine, takes about 6 weeks to begin to reduce the AD. Unfortunately, the response of AD patients to MMF is variable, with 20%–40% not responding. If cyclosporin A is not to be used, the choice of steroid-sparing agent is personalized to the patient’s risk factors and tolerance of the medication. Hydroxyurea, as used for psoriasis in the past, can be effective in AD if other steroid-sparing agents fail, as well as in the patient with liver disease.
Intravenous immune globulin (IVIG) has had some limited success in managing AD, but its high cost precludes it use, except when other reasonable therapeutic options have been exhausted. There are also reports of a dyshidrotic flare of dermatitis on the hands in patients on IVIG. Interferon-γ (IFN-γ) given by daily injection has demonstrated efficacy in both children and adults with severe AD but is very rarely used. IFN-γ is well tolerated but can cause flulike symptoms. Omalizumab can be considered in refractory cases, but only 20% of patients achieve a 50% or greater reduction of their AD. Infliximab has not been beneficial in AD. Ustekinumab has been effective in a few reports, because the inflammatory cascade triggering and maintaining AD does involve Th17 cells.
Traditional Chinese herb mixtures have shown efficacy in children and in animal models for AD. The active herbs appear to be ophiopogon tuber and Schisandra fruit. Chinese herbs are usually delivered as a brewed tea to be drunk daily. Their bitter taste makes them difficult to palate, and there is a risk of allergy from herbal products.
Management of Acute Flare
Initially, the precipitating cause of the flare should be sought. Recent stressful events may be associated with flares. Secondary infection with S. aureus is one of the most common causes for flares and will manifest with pustules and honey-colored erosions. Less frequently, HSV or coxsackievirus may be involved. Pityriasis rosea may also cause AD to flare. The development of contact sensitivity to an applied medication or photosensitivity must be considered.
In the patient with an acute flare, treating triggers may lead to improvement (see earlier discussion). Soaking and smearing (wet wrapping) is recommended as first-line treatment. Often, 3–4 days of such intensive home therapy will break a severe flare. A short course of systemic corticosteroids may be of benefit as a last resort.
Agusti-Mejias AM, et al: Severe refractory atopic dermatitis in an adolescent patient successfully treated with ustekinumab. Ann Dermatol 2013; 25: 3.
Andreae DA, Wang J: Immunologic effects of omalizumab in children with severe refractory atopic dermatitis. Pediatrics 2014; 134: S160.
Annesi-Maesano I, et al: Time trends in prevalence and severity of childhood asthma and allergies from 1995 to 2002 in France. Allergy 2009; 64: 798.
Benninger MS, et al: Prevalence of atopic disease in patients with eosinophilic esophagitis. Int Forum Allergy Rhinol 2017; 7: 757.
Byrd AL, et al: Staphylococcus aureus and Staphylococcus epidermidis strain diversity underlying pediatric atopic dermatitis. Sci Transl Med 2017; 9.
Cao L, et al: Long-term effect of early-life supplementation with probiotics on preventing atopic dermatitis. J Dermatolog Treat 2015; 26: 537.
Damm JA, et al: The influence of probiotics for preterm neonates on the incidence of atopic dermatitis. Arch Dermatol Res 2017; 309: 259.
Du Toit G, et al: Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med 2015; 372: 803.
Dvorakova V, et al: Methotrexate for severe childhood atopic dermatitis. Pediatr Dermatol 2017; 34: 528.
Eichenfeld LF, et al: Guidelines of care for the management of atopic dermatitis. Section 1. J Am Acad Dermatol 2014; 70: 2.
Eichenfeld LF, et al: Guidelines of care for the management of atopic dermatitis. Section 2. J Am Acad Dermatol 2014; 71:116.
Epstein TG, et al: Opposing effects of cat and dog ownership and allergic sensitization on eczema in an atopic birth cohort. J Pediatr 2011; 158: 2.
Fleischer DM, et al: Primary prevention of allergic disease through nutritional interventions. J Allergy Clin Immunol 2013; 1: 29.
Flohr C, Mann J: New approaches to the prevention of childhood atopic dermatitis. Allergy 2014; 69: 56.
Flohr C, Mann J: New insights into the epidemiology of childhood atopic dermatitis. Allergy 2014; 69: 3.
Gao PS, et al: Genetic variants in interferon regulatory factor 2 (IRF2) are associated with atopic dermatitis and eczema herpeticum. J Invest Dermatol 2012; 132: 650.
Gao PS, et al: Genetic variants in thymic stromal lymphopoietin are associated with atopic dermatitis and eczema herpeticum. J Allergy Clin Immunol 2010; 125: 6.
Gittlem JK, et al: Progressive activation Th2/Th22 cytokines and selective epidermal proteins characterizes acute and chronic atopic dermatitis. J Allergy Clin Immunol 2012; 130: 6.
Goujon C, et al: Methotrexate versus cyclosporine in adults with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol Pract 2017 Sep 26. Epub ahead of print.
Haeck IM, et al: Enteric-coated mycophenolate sodium versus cyclosporine as a long-term treatment in adult patients with severe atopic dermatitis. J Am Acad Dermatol 2011; 64: 6.
Hotze M, et al: Increased efficacy of omalizumab in atopic dermatitis patients with wild-type filaggrin status and higher serum levels of phosphatidylcholines. Allergy 2014; 69: 132.
Huang JT, et al: Treatment of Staphylococcus aureus colonization in atopic dermatitis decreases disease severity. Pediatrics 2009; 123: e808.
Luca NJ, et al: Eczema herpeticum in children: clinical features and factors predictive of hospitalization. J Pediatr 2012; 161: 4.
Margolis DJ, et al: Filaggrin-2 variation is associated with more persistent atopic dermatitis in African American subjects. J Allergy Clin Immunol 2014; 133: 784.
Margolis DJ, et al: Thymic stromal lymphopoietin variation, filaggrin loss of function, and the persistence of atopic dermatitis. JAMA Dermatol 2014; 150: 254.
Otsuka A, et al: The interplay between genetic and environmental factors in the pathogenesis of atopic dermatitis. Immunol Rev 2017; 278: 246.
Ozkaya E: Adult-onset atopic dermatitis. J Am Acad Dermatol 2005; 52: 579.
Penders J, et al: Establishment of the intestinal microbiota and its role for atopic dermatitis in early childhood. J Allergy Clin Immunol 2013; 132: 601.
Polcari I, et al: Filaggrin gene mutations in African Americans with both ichthyosis vulgaris and atopic dermatitis. Pediatr Dermatol 2014; 31: 489.
Rahman SI, et al: The methotrexate polyglutamate assay supports the efficacy of methotrexate for severe inflammatory skin disease in children. J Am Acad Dermatol 2013; 70: 252.
Ricci G, et al: Three years of Italian experience of an educational program for parents of young children affected by atopic dermatitis. Pediatr Dermatol 2009; 26: 1.
Ring J, et al: Guidelines for treatment of atopic eczema (atopic dermatitis). J Eur Acad Dermatol Venereol 2012; 26: 1176.
Rupnik H, et al: Filaggrin loss-of-function mutations are not associated with atopic dermatitis that develops in late childhood or adulthood. Br J Dermatol 2015; 172: 455.
Salimi M, et al: A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med 2013; 210: 13.
Sidbury R, et al: Guidelines of care for the management of atopic dermatitis: section 3. J Am Acad Dermatol 2014; 71: 327.
Sidbury R, et al: Guidelines of care for the management of atopic dermatitis: section 4. J Am Acad Dermatol 2014; 71: 1218.
Simpson EL, et al: Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol 2014; 134: 818.
Stemmler S, et al: Association of variation in the LAMA3 gene, encoding the alpha-chain of laminin 5, with atopic dermatitis in a German case-control cohort. BMC Dermatol 2014; 14: 17.
Stott B, et al: Human IL-31 is induced by IL-4 and promotes Th2-driven inflammation. J Allergy Clin Immunol 2013; 132: 2.
Thomsen SF, et al: Outcome of treatment with azathioprine in severe atopic dermatitis. Br J Dermatol 2015; 172: 1122.
Van Drongelen V, et al: Reduced filaggrin expression is accompanied by increased Staphylococcus aureus colonization of epidermal skin models. Clin Exp Allergy 2014; 44: 1515.
Wang WL, et al: Thymic stromal lymphopoietin. Int Arch Allergy Immunol 2013; 160: 18.
Regional Eczemas
Some eczema is different clinically based on location. Localized eczema can be from AD, contact dermatitis (both allergic and irritant), sebopsoriasis, and others as reviewed here.
Ear Eczema
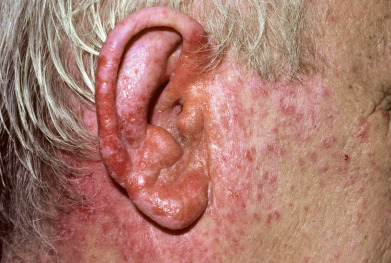
Eczema of the ears or otitis externa may involve the helix, postauricular fold, and external auditory canal. By far the most frequently affected site is the external canal, where eczema is often a manifestation of seborrheic dermatitis or allergic contact dermatitis caused by topical medications, especially neomycin ( Fig. 5.8 ), benzocaine, and preservatives or earphones. Secretions of the ear canal derive from the specialized apocrine and sebaceous glands, which form cerumen. Rubbing, wiping, scratching, and picking exacerbate the condition. Secondary bacterial colonization or infection is common. Infection is usually caused by staphylococci, streptococci, or Pseudomonas. Pseudomonas aeruginosa can result in malignant external otitis with ulceration and sepsis. Earlobe dermatitis in people with pierced ears is virtually pathognomonic for metal contact dermatitis (especially nickel).