Superficial and Deep Mycoses
Fungal infections, including dermatophytes and candida, are the most common type of infection worldwide. Cutaneous infections are divided into superficial (those that affect mostly the skin) and deep (those that are typically more invasive) mycoses. In addition immunosuppressed patients are at high risk for opportunistic fungal infections, including from molds and other saprophytic fungi that are not typically pathogens in normal hosts.
Most mycotic infections are superficial and are limited to the stratum corneum, hair, and nails. In contrast, most deep mycoses are evidence of disseminated infection, typically with a primary pulmonary focus. Although blastomycosis, histoplasmosis, and coccidioidomycosis generally present as skin lesions, they are almost always evidence of a systemic infection. A few deep mycoses result from direct inoculation into the skin by a thorn or other foreign body, including cutaneous lymphangitic sporotrichosis, primary cutaneous phaeohyphomycosis, and chromomycosis. Phaeohyphomycosis may begin as a skin infection, but similar to all nondermatophyte fungi, immunosuppressed patients are at great risk of dissemination and death. Even cutaneous sporotrichosis may occasionally disseminate. Cutaneous aspergillosis can occur due to cutaneous embolization from a systemic (often a pulmonary) focus, but in immunosuppressed populations, direct inoculation of any saprophytic infection can lead to severe infection and then disseminate. In burn victims, Aspergillus can colonize the burn eschar. This colonization may often be treated with debridement alone. Deep incisional biopsies are required to determine whether viable tissue has been invaded beneath the eschar. Evidence of viable tissue invasion suggests a likelihood of systemic dissemination and is usually an indication for systemic antifungal therapy.
The major fungi that cause only stratum corneum, hair, and nail infection are the dermatophytes. They are classified in three genera: Microsporum, Trichophyton, and Epidermophyton. The identity of the pathogen may be important for determining a zoonotic reservoir of infection (e.g., cats or dogs for Microsporum canis infections, cattle for Trichophyton verrucosum, and rats for granular zoophilic Trichophyton mentagrophytes ).
Susceptibility and Prevalence
Local immunosuppression from a potent topical corticosteroid or calcineurin inhibitor may promote larger lesions of cutaneous spread of dermatophyte infection. A defective cutaneous barrier, as in patients with ichthyosis, can also predispose to widespread tinea infection. Close contact with other infected individuals can lead to local breakouts of tinea especially tinea capitis. Maceration of the feet due to sweating, athletics, and swimming can increase the risk of tinea pedis.
Dermatophytes almost exclusively infect the skin. Patients with blood type A are somewhat more prone to chronic disease, and those with autosomal recessive CARD9 deficiency are susceptible to invasive dermatophyte fungal infection with dissemination to lymph nodes and the central nervous system (CNS). Many individuals will carry Trichophyton rubrum asymptomatically, which may be an autosomal dominant inherited tendency. When they are exposed to a hot, humid climate or occlusive footwear, these patients often become symptomatic. Reported prevalence rates are therefore greatly affected by climate, footwear, and lifestyle.
Antifungal Therapy
Specific therapeutic recommendations will be made in each section; the following is an overview of available options. Topical agents provide safe, cost-effective therapy for limited superficial fungal infections. When considering the use of an oral antifungal agent, factors include the type of infection, organism, spectrum, pharmacokinetic profile, safety, compliance, age, and cost. Laboratory monitoring recommendations for systemic antifungals change frequently so the reader is advised to read the latest guidelines before prescribing systemic therapy. Griseofulvin has a long safety record but requires longer courses of therapy than newer agents. Topical antifungals remain very cost-effective for limited cutaneous disease. Dermatophyte infection involving hair-bearing areas (scalp, beard, thick hair on arms or legs) often requires oral therapy due to infection of the hair follicle. Infants may respond to topical therapy even in hair-bearing areas due to immature hair follicles. Lesions accidentally pretreated with topical steroids or calcineurin inhibitors may also require systemic therapy.
Various classes of antifungals are in use. The imidazoles include clotrimazole, miconazole, econazole, sulconazole, oxiconazole, voriconazole, efinaconazole, fenticonazole, and ketoconazole. They work by inhibition of cytochrome P450 14α-demethylase, an essential enzyme in ergosterol synthesis. Nystatin is a polyene that works by irreversibly binding to ergosterol, an essential component of fungal cell membranes but is used for Candida infections and is not effective for treating dermatophytes. Naftifine, terbinafine, and butenafine are allylamines, and their mode of action is inhibition of squalene epoxydation. The triazoles include itraconazole and fluconazole, which affect the CYP450 system.
For both itraconazole and griseofulvin, fatty food increases absorption. For itraconazole and ketoconazole, antacids, H2 antagonists, and proton pump inhibitors lower absorption. Terbinafine is less active against Candida and Microsporum species (spp.) in vitro. In vivo, adequate doses can be effective against these organisms. Terbinafine has limited efficacy in the oral treatment of tinea versicolor but is effective topically. Although few drug interactions have been reported with terbinafine, and the bioavailability is unchanged in food, hepatotoxicity, leukopenia, toxic epidermal necrolysis, and taste disturbances occur infrequently. Ketoconazole has a wide spectrum of action against dermatophytes, yeasts, and some systemic mycoses, but has the potential for hepatotoxicity. The U.S. Food and Drug Administration (FDA) has advised that systemic ketoconazole should not be used for skin and nail infections and should only be used for systemic fungal infections when no other medication is available ( http://www.fda.gov/Drugs/DrugSafety/ucm500597.htm ).
Fluconazole is mainly used to treat Candida infections, but has shown efficacy in the treatment of dermatophytoses both in daily and in weekly doses. It also has benefit in infants and young children because fluconazole has approval for candidal infections in infants and therefore is a reasonable off-label option for dermatophyte infections when needed in this population. Griseofulvin is FDA approved for tinea capitis in children over 2 and terbinafine only over age 4 years, but fluconazole has approval in infancy for various fungal infections. Although pulse therapy with fluconazole has been shown effective, patients may have trouble remembering intermittent dosing schedules. Both terbinafine and itraconazole have been shown to be effective and well tolerated in several studies of the treatment of tinea capitis and onychomycosis in children. However, itraconazole has been associated with reports of heart failure.
Voriconazole has exceptional activity against a wide variety of yeasts, as well as many other fungal pathogens, but has been associated with photosensitivity, premature photoaging, actinic keratoses, squamous cell carcinoma, melanoma, and porphyria. Posaconazole has significant in vitro activity against Candida spp., although some resistance has been reported to this drug.
The echinocandins inhibit β-(1,3)-glucan synthesis, thus damaging fungal cell walls. These drugs are active against most Candida spp. and fungistatic against Aspergillus spp. The echinocandins have limited activity against zygomycetes, Cryptococcus neoformans, or Fusarium spp. Caspofungin was the first drug in this class to be marketed in the United States for refractory invasive aspergillosis. Micafungin also belongs to this antifungal class. Adverse events are uncommon but include phlebitis, fever, elevated liver enzymes, and mild hemolysis. The drugs must be given intravenously. Metabolism is mainly hepatic. In the setting of Candida sepsis, results are similar to those achieved with amphotericin B, with substantially lower toxicity. The echinocandins may be used together with other antifungal agents in the treatment of life-threatening systemic fungal infections, such as disseminated aspergillosis refractory to other regimens.
The Superficial Mycoses
Tinea Capitis
Tinea capitis, known colloquially as scalp “ringworm,” can be caused by all of the pathogenic dermatophytes. In the United States most cases are caused by Trichophyton tonsurans. Pet exposure (especially cat) is associated with tinea capitis caused by Microsporum canis. As people travel and emigrate from other countries, the diversity of fungi causing tinea capitis in a region can rise.
Tinea capitis occurs mainly in children, although it may be seen at all ages. Boys have tinea capitis more frequently than girls; however, in epidemics caused by T. tonsurans, both genders are often affected equally. African American children have a higher incidence of T. tonsurans infections than other groups. The infection is also common among Latin American children.
Trichophyton tonsurans can produce black dot tinea ( Fig. 15.1 ), scaling alopecic plaques, subtle seborrheic-like scaling, and/or inflammatory kerion. Black dot tinea occurs due to the fungi invading the follicle (endothrix) and may also be caused by Trichophyton violaceum, an organism rarely seen in the United States. Both of these organisms produce chains of large spores within the hair shaft (large-spore endothrix). They do not fluoresce with Wood’s light.
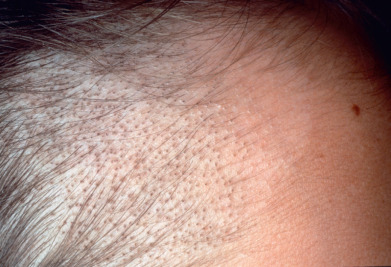
The Microsporum canis complex includes a group of organisms that produce small spores visible on the outside of the hair shaft (small-spore ectothrix). These fungi fluoresce under Wood’s light examination. The M. canis complex includes M. canis, M. canis distortum, Microsporum ferrugineum, and M. audouinii. M. canis infections begin as scaly, erythematous, papular eruptions with loose and broken-off hairs. The lesions typically become highly inflammatory, although M. audouinii produces less inflammatory lesions. Deep, tender, boggy plaques exuding pus are known as kerion celsii ( Fig. 15.2 ). Kerion may be followed by scarring and permanent alopecia in the areas of inflammation and suppuration, although with early treatment hair regrowth is common. Prompt administration of oral antifungals is most important but systemic corticosteroids for a short period, along with appropriate antifungal therapy, can greatly diminish the inflammatory response and reduce the risk of scarring, and this therapy should be considered in the patient with highly inflammatory lesions.
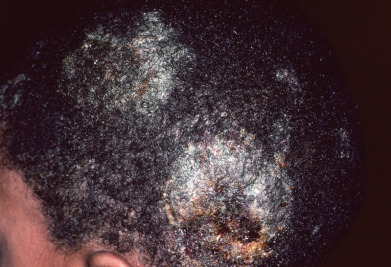
Many children are asymptomatic carriers of T. tonsurans and represent a source of infection for classmates and siblings. Numerous studies have shown that 5%–15% of urban children in Western countries have positive scalp dermatophyte cultures. In one study, 60% of African American children with a positive scalp culture were asymptomatic.
The prevalence of dermatophytes varies throughout the world. Where animal herding is an important part of the economy, zoonotic fungi account for a significant proportion of cases of tinea. In south-central Asia, T. violaceum is the most common dermatophytic species isolated, with M. audouinii a close second. Other common organisms include Trichophyton schoenleinii, T. tonsurans, Microsporum gypseum, T. verrucosum, and T. mentagrophytes. In East Asia, T. violaceum and M. ferrugineum are important pathogens. In Europe, African and Caribbean immigrants account for a large proportion of new patients with tinea capitis. Important pathogens include T. tonsurans, M. audouinii var. langeronii, Trichophyton soudanense, and T. violaceum. Trichophyton megninii is a rare cause of tinea capitis largely restricted to southwestern Europe. In Africa, large-scale epidemics are associated with T. soudanense, T. violaceum, T. schoenleinii, and Microsporum spp. In Australia, the predominantly Caucasian population experiences infections, mostly with M. canis, but T. tonsurans is now equal in prevalence in some areas of the continent. Recent immigrants have a high incidence of tinea capitis with organisms common in their regions of origin. Among African and Arab immigrants, T. soudanense, T. violaceum, and M. audouinii are particularly common.
Favus
Favus, which is extremely rare in the United States, appears chiefly on the scalp but may affect the glabrous skin and nails. On the scalp, concave sulfur-yellow crusts form around loose, wiry hairs. Atrophic scarring ensues, leaving a smooth, glossy, thin, paper-white patch. On the glabrous skin, the lesions are pinhead to 2 cm in diameter with cup-shaped crusts called scutulae, usually pierced by a hair as on the scalp. The scutulae have a distinctive mousy odor. When the nails are affected, they become brittle, irregularly thickened, and crusted under the free margins. Other types of dermatophytes such as T. rubrum may appear similar clinically.
Favus is prevalent in the Middle East, southeastern Europe, South Africa and the countries bordering the Mediterranean Sea.
Pathogenesis and Natural History
The incubation period of anthropophilic (affecting humans) tinea capitis is 2–4 days, although the period is highly variable, and asymptomatic carriers are common. The hyphae grow downward into the follicle, on the hair’s surface, and the intrafollicular hyphae break up into chains of spores. There is a period of spread (4 days to 4 months) during which the lesions enlarge and new lesions appear. At about 3 weeks, hairs break off a few millimeters above the skin surface. Within the hair, hyphae descend to the upper limit of the keratogenous zone and here form Adamson “fringe” on about the 12th day. During a refractory period (4 months to several years) fewer new lesions develop. The clinical appearance is constant, with the host and parasite at equilibrium. This may be followed by a period of involution in which the formation of spores gradually diminishes. Zoonotic (from animals) fungal infections often are more highly inflammatory but undergo similar phases of evolution.
Diagnosis
Wood’s Light
Ultraviolet (UV) light of 365-nm wavelength commonly known as the Wood’s light, is used to demonstrate fungal fluorescence. Infections caused by M. audouinii, M. canis, M. ferrugineum, M. distortum, and T. schoenleinii will fluoresce . In a darkroom, the skin under this light fluoresces faintly blue, and dandruff usually is bright blue-white. Infected hair fluoresces bright green or yellow-green due to the presence of pteridine. Large-spore endothrix organisms (e.g., T. tonsurans, T. violaceum ) and T. verrucosum (a cause of large-spore ectothrix) do not fluoresce making woods light less helpful because T. tonsurans is the most common type of tinea capitis.
Laboratory Examination
Tinea capitis can be a clinical diagnosis if the classic scaling, alopecic plaques with nuchal or posterior cervical lymphadenopathy are seen in a child. In one study 97% percent of children with tinea capitis demonstrated posterior cervical or nuchal lymphadenopathy, making this a very useful negative predictor if absent. For more diffuse “seborrheic” patterns or without significant alopecia or if another diagnosis is being considered, a culture can prove the diagnosis. Cultures from a kerion (inflammatory tinea capitis) may be false negative due to the exuberant inflammatory response so treatment is typically empiric. For demonstration of the fungus in a highly inflammatory plaque, two or three loose hairs are carefully removed with epilating forceps from the suspected areas. If fluorescence occurs, it is important to choose these hairs. Bear in mind that hairs infected with T. tonsurans do not fluoresce. In black dot ringworm or in patients with seborrheic scale, small broken fragments of infected hair or scaling can be scraped or brushed off with a sterile toothbrush. The hairs are placed on a slide and covered with a drop of a 10%–20% potassium hydroxide (KOH) solution. A coverslip is then applied, and the specimen is warmed until the hairs are macerated. Dimethyl sulfoxide (DMSO) can be added to the KOH solution in concentrations of up to 40%. This additive allows for rapid clearing of keratin without heating. Once the keratin in the hairs has dissolved somewhat, they can be examined first with a low-power objective and then with a high-power objective for detail. The mycelia may be seen under low-power microscopy, but better observation of both hyphae and spores is obtained by the 10× objective with the condenser lowered down or the light aperture closed by two thirds. A staining method using 100 mg of chlorazol black E dye in 10 mL of DMSO and adding it to a 5% aqueous solution of KOH can be helpful. Toluidine blue 0.1% can also be used on thin specimens, but contains no clearing agent to dissolve keratin. The patterns of endothrix and ectothrix involvement described earlier, together with local prevalence data, allow for identification of the organism.
Exact identification of the causative fungus is generally determined by culture, although molecular sequencing offers a more rapid alternative. For culture, several infected hairs are plated on Sabouraud dextrose agar, Sabouraud agar with chloramphenicol, Mycosel agar, or dermatophyte test medium (DTM). Laboratories differ in how they prefer the sample to be collected, but using a toothbrush to gently remove scale and loose hairs and transfer to the laboratory in a sterile container or a bacterial culturette moistened with sterile saline and then rubbed on the affected areas are the most common methods. On the first three media, a distinctive growth appears within 1–2 weeks. Most frequently, the diagnosis is made by the gross appearance of the culture growth, together with the microscopic appearance. With Trichophyton spp., growth on different nutrient agars is often required to identify the organisms beyond genus. DTM not only contains antibiotics to reduce growth of contaminants, but also contains a colored pH indicator to denote the alkali-producing dermatophytes although laboratory regulations have limited this use in academic centers. A few nonpathogenic saprophytes will also produce alkalinization, and in the occasional case of onychomycosis of toenails caused by airborne molds, a culture medium containing an antibiotic may inhibit growth of the true pathogen.
Differential Diagnosis
Tinea capitis must be differentiated clinically from chronic staphylococcal folliculitis, pediculosis capitis (head lice), psoriasis, seborrheic dermatitis, secondary syphilis, trichotillomania, alopecia areata, lupus erythematosus (LE), lichen planus, lichen simplex chronicus, and various inflammatory follicular conditions. The distinctive clinical features of tinea capitis are broken-off stumps of hairs, often in rounded patches in which there are crusts or pustules and few hairs. The broken-off hairs are loose. Diffuse seborrheic scaling with hair loss is a common presentation of T. tonsurans infections. Dermoscopy can reveal comma-shaped and corkscrew hairs. Horizontal bands and translucent hairs in M. canis infections have been demonstrated on high-power dermoscopy.
In alopecia areata, the affected patches are bald, the skin is smooth and shiny without signs of inflammation or scaling, and exclamation point hairs can be seen on dermoscopy. Stumps of broken-off hairs are infrequently found, and no fungi are demonstrable. In seborrheic dermatitis, the involved areas are covered by fine, dry, or greasy scales. Hair may be lost, but the hairs are not broken and lymphadenopathy is generally absent. Atopic dermatitis can affect the scalp but is rarely only localized to the scalp. In psoriasis, well-demarcated, sometimes diffuse, areas of erythema and white or silver scaling are noted. Although in children psoriasis may first manifest in the scalp, there is usually some evidence of psoriasis elsewhere. Lichen simplex chronicus is frequently localized to the inferior margin of the occipital scalp. In trichotillomania, as in alopecia areata, inflammation and scaling are absent and the patterns of hair loss are unusual clinically. Serologic testing, scalp biopsies, and immunofluorescent studies may be indicated if the alopecia of secondary syphilis or LE is a serious consideration. It should be noted that adult patients with LE are susceptible to tinea capitis, which may be photosensitive and difficult to distinguish from LE plaques without biopsy and KOH examinations.
Treatment
Numerous clinical trials have demonstrated the effectiveness of itraconazole, terbinafine, and fluconazole. Griseofulvin remains the most frequently used antifungal agent in children, but because there are data that terbinafine may be more effective in T. tonsurans infections and the course of therapy is shorter, terbinafine is gaining favor, and both are considered first line. Griseofulvin has a long safety record. A meta-analysis of published studies shows mean efficacy for griseofulvin treatment of about 68% for Trichophyton spp. and 88% for Microsporum. For the ultramicronized form, doses start at 10 mg/kg/day. Griseofulvin must be given with a fatty food to ensure absorption. The ultramicrosized form absorbs more easily so doses are lower. The dose of micronized is 20 mg/ kg/day (not to exceed 500 mg twice a day), although some advocate up to 25 mg/kg/day in recalcitrant cases. Treatment should continue for 6–8 weeks, or for at least 2 weeks after clinical or laboratory determined cure. For Trichophyton infections, terbinafine is usually effective in doses of 3–6 mg/kg/day for 3–6 weeks. In the United States it is FDA approved over 4 years of age, but some authors advocate for its use in younger children. Published dosing guidelines are based on the terbinafine granules, which can be difficult to obtain. Similar dosing that is used for onychomycosis in children based on the tablets is reasonable: 250-mg tablet for patients over 40 kg, 125 mg ( ![]() tablet) for those 20–40 kg, and 62.5 mg (
tablet) for those 20–40 kg, and 62.5 mg ( ![]() tablet) for those under 20 kg. Terbinafine should not be mixed with acidic foods such as apple sauce because they may limit absorption. Microsporum infections require higher doses and longer courses of therapy with terbinafine, so griseofulvin is preferred in these infections.
tablet) for those under 20 kg. Terbinafine should not be mixed with acidic foods such as apple sauce because they may limit absorption. Microsporum infections require higher doses and longer courses of therapy with terbinafine, so griseofulvin is preferred in these infections.
Second-line agents include fluconazole (not to exceed adult dose) at doses of 3–6 mg/kg/day for 3–6 weeks lower doses lead to lower cure rates. Fluconazole 6mg/kg/week for 8–12 weeks has also been reported. Itraconazole has been shown to be effective in doses of 5 mg/kg/day (not to exceed adult dose) for 2–3 weeks, but reports of heart failure with itraconazole have limited its use. In neonates and children under 2 topical therapy is occasionally effective, likely due to the less mature hair follicles, but oral antifungals are recommended by a recent review.
Selenium sulfide shampoo or ketoconazole shampoo left on the scalp for 5 minutes three times a week can be used as adjunctive therapy to oral antifungal agents to reduce the shedding of fungal spores. Combs, brushes, and hats should be cleaned carefully, and natural bristle brushes must be discarded.
Prognosis
Recurrence is uncommon when adequate amounts of griseofulvin, fluconazole, or terbinafine have been taken, although exposure to infected persons, asymptomatic carriers, or contaminated fomites will increase the relapse rate. Without medication, there may be spontaneous clearing at about age 15 years, except with T. tonsurans, which often persists into adult life.
Dermatophytids
In cases of inflammatory tinea capitis, widespread “id” eruptions may appear concomitantly on the trunk and extremities. These are vesicular, lichenoid, papulosquamous, or pustular and represent a systemic reaction to fungal antigens either during the infection or therapy for the infection. Although the eruptions are usually refractory to topical corticosteroids, they typically clear rapidly after treatment of the fungal infection.
An id reaction can be seen on the hands and sides of the fingers when there is an acute fungus infection of the feet. These lesions are mostly vesicular and are extremely pruritic and even tender. Secondary bacterial infection may occur; however, fungus is not demonstrable in the hand lesions. The onset can be accompanied by fever, anorexia, generalized adenopathy, spleen enlargement, and leukocytosis. Dermatophytid reactions from inflammatory tinea capitis may occasionally present as widespread eruption, usually follicular, lichenoid, or papulosquamous. An id reaction characterized by fine papules on the trunk, face, ears and extremities can occur during systemic therapy especially with griseofulvin for tinea capitis. Rarely, the eruption may be morbilliform or scarlatiniform. The erysipelas-like dermatophytid has been reported on the shin but must be differentiated from cellulitis that occurs as a result of skin breaks from tinea pedis.
The histologic picture of an id is characterized by spongiotic vesicles and a superficial, perivascular, predominantly lymphohistiocytic infiltrate. Eosinophils may be present. Diagnosis of a dermatophytid reaction depends on the demonstration of a fungus at some site remote from the suspect lesions of the dermatophytid, the absence of fungus in the id lesion, and involution of the lesion as the fungal infection subsides.
Tinea Barbae
Ringworm of the beard, also known as tinea sycosis and barber’s itch, is more common among those in agricultural pursuits, especially those in contact with farm animals, but may also be seen in wrestlers and through incidental contact. The involvement is mostly one sided on the neck or face. Typical scaling alopecic patches can occur in addition to a more inflammatory superficial, crusted, partially bald patches with folliculitis ( Fig. 15.3 ) or deep, nodular, suppurative lesions possibly indicating a zoonotic infection.
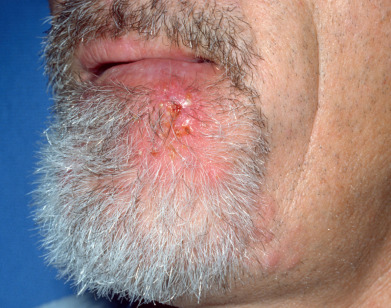
The deep type develops slowly and produces nodular thickenings and kerion-like swellings, usually caused by T. mentagrophytes or T. verrucosum. As a rule, the swellings are confluent and form diffuse, boggy infiltrations with abscesses. The overlying skin is inflamed, the hairs are loose or absent, and pus may be expressed through the remaining follicular openings. Generally, the lesions are limited to one part of the face or neck in men. The superficial crusted type is characterized by a less inflammatory pustular folliculitis and may be associated with T. violaceum or T. rubrum. The affected hairs can sometimes be easily extracted. Rarely, Epidermophyton floccosum may cause widespread verrucous lesions known as verrucous epidermophytosis.
Diagnosis
The clinical diagnosis of tinea barbae is confirmed by the microscopic mounts of extracted hairs or a biopsy and culture of the tissue specimen.
Differential Diagnosis
The differential diagnosis includes staphylococcal folliculitis (sycosis vulgaris) and herpetic infections. Tinea barbae differs from sycosis vulgaris by usually sparing the upper lip and by often being unilateral. In sycosis vulgaris, the lesions are pustules and papules, pierced in the center by a hair, which is loose and easily extracted after suppuration has occurred. Herpetic infections usually demonstrate umbilicated vesicles. Tzanck preparations have a low diagnostic yield, but viral culture or direct fluorescent antibody is virtually always positive.
Treatment
As in tinea capitis, oral antifungal agents are required to cure tinea barbae. Topical agents are only helpful as adjunctive therapy. Oral agents are used in the same doses and for the same durations as in tinea capitis.
Tinea Faciei
Fungal infection of the face is frequently misdiagnosed ( Fig. 15.4 ). Typical annular rings are usually lacking, and the lesions are exquisitely photosensitive. Frequently, a misdiagnosis of LE is made. Biopsies for direct immunofluorescence often demonstrate some reactants on sun-exposed skin, adding to the possible diagnostic confusion. Erythematous, slightly scaling, indistinct borders may be present at the periphery of the lesions and are the best location for KOH examination. If topical corticosteroids have been used, the lesions will expand and fungal folliculitis is a frequent finding. A biopsy may be required to establish the diagnosis. A high index of suspicion is required, because fungal hyphae may be few in number or confined to hair follicles. The inflammatory pattern may be psoriasiform spongiotic or vacuolar interface. The latter pattern has the potential to perpetuate confusion with LE.
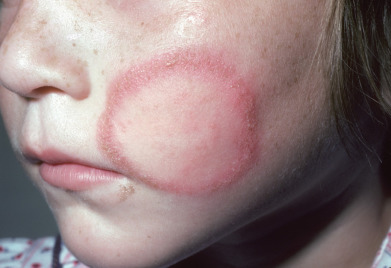
Usually, the infection is caused by T. rubrum, T. mentagrophytes, or M. canis. Tinea faciei caused by Microsporum nanum has been described in hog farmers. If hair follicles are infected such as long the eyebrow or beard area in older children or the infection is widespread, oral medication is required. Otherwise, the infection generally responds well to topical therapy.
Tinea Corporis (Tinea Circinata)
Tinea corporis includes all superficial dermatophyte infections of the skin other than those involving the scalp, beard, face, hands, feet, and groin. This form of ringworm is characterized by one or more circular, sharply circumscribed, slightly erythematous, dry, scaly, sometimes hypopigmented patches. An advancing scaling edge is usually prominent ( Fig. 15.5 ). Progressive central clearing produces annular outlines that give them the name “ringworm.” Lesions may widen to form rings many centimeters in diameter. In some cases, concentric circles or polycyclic lesions form, making intricate patterns. Although more common on the feet, lesions of tinea corporis can also form vesicles and bullae (bullous tinea). Widespread tinea corporis may be the presenting sign of acquired immunodeficiency syndrome (AIDS) or may be related to the use of a topical corticosteroid or calcineurin inhibitor.
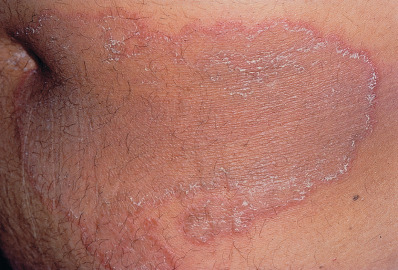
In the United States T. rubrum, M. canis, and T. mentagrophytes are common causes, although infection can be caused by any of the dermatophytes. Multiple small lesions are usually caused by exposure to a pet with M. canis. Other zoonotic fungi, such as granular zoophilic T. mentagrophytes related to Southeast Asian bamboo rats, can cause widespread epidemics of highly inflammatory tinea corporis.
Tinea gladiatorum is a common problem for wrestlers. Opponents, equipment, and mats represent potential sources of infection.
Diagnosis
The diagnosis of tinea corporis is by finding the fungus by KOH examination of skin scrapings under the microscope as described earlier. When evaluating tinea corporis, the lines of juncture of normal keratinocytes dissolve into a branching network that may easily be mistaken for fungus structures (“mosaic false hyphae”). This is the most common artifact misinterpreted as a positive KOH examination. Cotton and synthetic fibers from socks may also mimic hyphae. In addition, skin scrapings can be cultured on a suitable medium as described earlier for tinea capitis. Growth of the fungus on culture medium is apparent within 1 week or 2 weeks at most, and in most cases is identifiable to the genus level by the gross and microscopic appearance of the culture. (Biopsy of a chronic refractory dermatosis often reveals tinea incognito.)
Other diseases that may closely resemble tinea corporis are pityriasis rosea, impetigo, nummular dermatitis, secondary and tertiary syphilids, seborrheic dermatitis, and psoriasis. These are distinguished by KOH examination and culture.
Treatment
Localized disease without fungal folliculitis may be treated with topical therapy. Sulconazole (Exelderm), oxiconazole (Oxistat), miconazole (Monistat cream or lotion, or Micatin cream), clotrimazole (Lotrimin or Mycelex cream), econazole (Spectazole), naftifine (Naftin), ketoconazole (Nizoral), ciclopirox olamine (Loprox), terbinafine (Lamisil), efinaconazole, and butenafine (Mentax) are currently available and effective and therapy should be guided based on cost effectiveness. Most treatment times are between 2 and 4 weeks with twice-daily use. Econazole, ketoconazole, oxiconazole, and terbinafine may be used once a day. With terbinafine, the course can be shortened to 1 week. Combination products with a potent corticosteroid such as clotrimazole/betamethasone frequently produce widespread tinea and fungal folliculitis. Their use should be avoided.
Extensive disease or infection of hair follicles requires systemic antifungal treatment. When tinea corporis is caused by T. tonsurans , T. mentagrophytes , or T. rubrum , griseofulvin, terbinafine, itraconazole, and fluconazole are all effective.
Trichophyton species generally respond to shorter courses of terbinafine and Microsporum species respond better to griseofulvin.
Other Forms of Tinea Corporis
Fungal Folliculitis Tinea Incognita and Majocchi Granuloma
Tinea incognita is a term applied to lesions of tinea that have an atypical appearance due to therapy with topical steroids or calcineurin inhibitors. The lesions are often widespread and may lack an advancing, raised, scaly border. Alternatively multiple edges may be present and there may be a lack of central clearing, rather the center may be eczematous ( Fig. 15.6 ). The diagnosis may be established by KOH examination or biopsy.
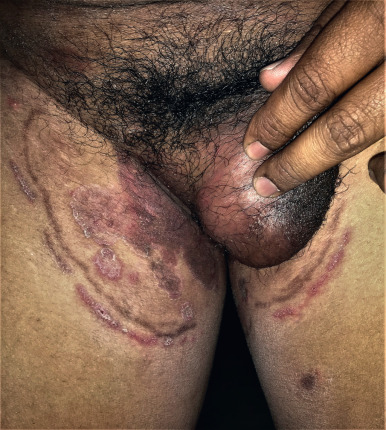
Treatment of tinea with a topical steroid or calcineurin inhibitor can lead to deeper, more inflammatory infections of the hair follicles. It presents as a circumscribed, annular, raised, crusty, and boggy granuloma in which the follicles are distended with viscid purulent material. These occur most frequently on the shins or wrists. The lesions are often seen in areas of occlusion or shaving, or when a topical corticosteroid has been used. In immunosuppressed patients, the lesions may be deep and nodular. Often, patients have been treated with both topical corticosteroids and antifungal agents. If a topical antifungal has been used recently, KOH examination and culture may be negative. A biopsy may be required to establish the diagnosis. Oral therapy is necessary to cure the lesions. Occasionally, a deep, pustular type of tinea circinata resembling a carbuncle or kerion is observed on the glabrous skin ( Fig. 15.7 ). This type of lesion is a fungal folliculitis caused most often by T. rubrum or T. mentagrophytes infecting hairs at the site of involvement.
Tinea Imbricata (Tokelau)
Tinea imbricata is a superficial fungal infection limited to southwest Polynesia, Melanesia, Southeast Asia, India, and Central America. It is characterized by concentric rings of scales forming extensive patches with polycyclic borders. Erythema is typically minimal. The eruption begins with one or several small, rounded macules on the trunk and arms. The small macular patch splits in the center and forms large, flaky scales attached at the periphery. As the resultant ring spreads peripherally, another brownish macule appears in the center and undergoes the process of splitting and peripheral extension. This cycle is repeated over and over again. When fully developed, the eruption is characterized by concentrically arranged rings or parallel, undulating lines of scales overlapping each other like shingles on a roof ( imbrex means “shingle”).
The causative fungus is Trichophyton concentricum, although a similar pattern may be produced by T. mentagrophytes and T. tonsurans. Microscopically, the scrapings show interlacing, septate, mycelial filaments that branch dichotomously. Polyhedral spores are also present. Terbinafine has been shown to be very effective while the recurrence rate may be high with griseofulvin.
Tinea Cruris
Tinea cruris, also known as “jock itch” and “crotch itch,” occurs more commonly in men on the upper and inner surfaces of the thighs, especially during the summer when the humidity is high. It begins as a small, erythematous, and scaling or vesicular and crusted patch that spreads peripherally and partly clears in the center, so that the patch is characterized chiefly by its curved, well-defined border, particularly on its lower edge. The border may have vesicles, pustules, or papules. It may extend downward on the thighs and backward on the perineum or about the anus. The scrotum is rarely involved.
Etiology and Differential Diagnosis.
Dermatophyte infection of the groin is usually caused by T. rubrum, T. mentagrophytes, or E. floccosum. Infection with Candida albicans may closely mimic tinea cruris, but is usually moister, more inflammatory, and associated with satellite macules and can involve the scrotum. Candida often produces collarette scales and satellite pustules.
The crural region is also a common site for erythrasma, seborrheic dermatitis, pemphigus vegetans, and intertriginous psoriasis. Erythrasma often has a copper color and is diagnosed by Wood’s light examination, which produces coral-red fluorescence. Seborrheic dermatitis generally involves the central chest and axillae in addition to the groin. Pemphigus vegetans produces macerated and eroded lesions. Diagnosis of tinea cruris is established by KOH examination, fungal culture, biopsy or (if caused by a fluorescent fungus) immunofluorescence. Inverse psoriasis may be associated with collarette scales or with serpiginous arrays of pustules at the border of inflammatory lesions. When more typical lesions of psoriasis are lacking, a biopsy may be required to establish the diagnosis.
Treatment.
The reduction of perspiration and enhancement of evaporation from the crural area are important prophylactic measures. The area should be kept as dry as possible by the wearing of loose underclothing and trousers. Plain talcum powder or antifungal powders are helpful. Specific topical and oral treatment for tinea cruris is the same as that described earlier for tinea corporis.
Tinea of Hands and Feet
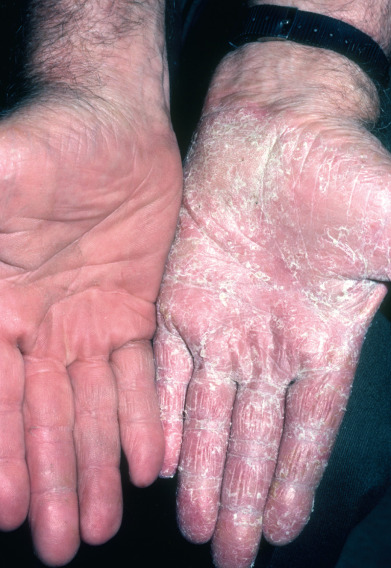
Dermatophytosis of the feet, “athlete’s foot,” is the most common fungal disease. T. rubrum causes the majority of infections although it can be caused by many other dermatophytes, including T. tonsurans. There may be an autosomal dominant predisposition to this form of infection or it may just be being spread within families. T. rubrum typically produces a relatively noninflammatory type of dermatophytosis characterized by a dull erythema and pronounced silvery scaling that may involve the entire sole and sides of the foot, giving a moccasin or sandal appearance. The eruption may also be limited to a small patch adjacent to a fungus-infected toenail or to a patch between or under the toes. In some patients, an extensive patchy, scaly eruption covers most of the trunk, buttocks, and extremities. Rarely, there is a patchy hyperkeratosis.
Generally, dermatophyte infection of the hands is of the dry, scaly, and erythematous type suggestive of T. rubrum infection. Other areas are frequently affected at the same time, especially the combination of both feet and one hand ( Fig. 15.8 ). Tinea pedis caused by anthropophilic T. mentagrophytes (interdigitale) presents with three distinct appearances: (1) multilocular bullae involving the thin skin of the plantar arch and along the sides of the feet and heel, (2) erythema and desquamation between the toes, and (3) white superficial onychomycosis. In the human immunodeficiency virus (HIV)–positive population, this latter syndrome is usually caused by T. rubrum. Interdigital tinea must be distinguished from simple maceration caused by a closed web space, which does not respond to antifungal therapy. Interdigital tinea must also be distinguished from gram-negative toe web infection and candidal infection. Diabetic patients develop interdigital fungal infections at a younger age than patients without diabetes.
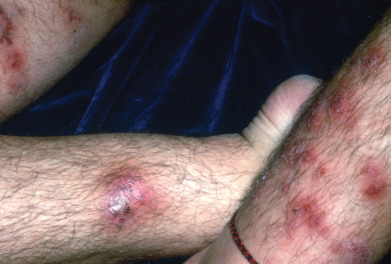
T. mentagrophytes often produces acutely inflammatory multilocular bullae ( Fig. 15.9 ). The burning and itching that accompany the formation of the vesicles may cause great discomfort, which is relieved by opening the tense vesicles. They contain a tenacious, clear, straw-colored fluid. Extensive or acute eruptions on the soles may be incapacitating. The fissures between the toes, as well as the vesicles, may become secondarily infected with pyogenic cocci, which may lead to recurrent attacks of lymphangitis and inguinal adenitis. Gram-negative toe web infections may also supervene. Hyperhidrosis is frequently present in this type of dermatophytosis. The sweat between the toes and on the soles has a high pH, and damp keratin is a good culture medium for the fungi.
Dermatophytid of the hands may be associated with inflammatory tinea of the feet and begins with the appearance of groups of minute, clear vesicles on the palms and fingers and sometimes the sides and tops of the feet. The itching may be intense. Typically, both hands are involved, and the eruption tends to be symmetric.
Diagnosis
Demonstration of the fungus by KOH microscopic examination ( Fig. 15.10 ) as described for tinea capitis and corporis earlier establishes the diagnosis. For bullous tinea, bullae should be unroofed, and either the entire roof is mounted intact or scrapings are made from the underside of the roof.
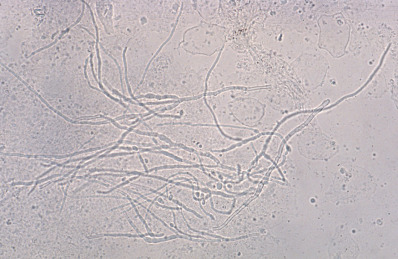
Material may also be placed on Sabouraud dextrose agar, Sabouraud agar with chloramphenicol, Mycosel agar, or DTM. The last three agars inhibit growth of bacterial or saprophytic contaminants. The last two may inhibit some pathogenic nondermatophytes. The alkaline metabolites produced by growth of dermatophytes change the color of the pH indicator in DTM medium from yellow to red.
Prophylaxis
Hyperhidrosis is a predisposing factor for tinea infections. Because the disease often starts on the feet, the patient should be advised to dry the toes thoroughly after bathing. Cold water laundering does not inactivate fungal elements in socks, which may serve as a source of recolonization. Dryness is essential to reduce the incidence of symptomatic reinfection.
The use of a antiseptic powder on the feet after bathing, particularly between the toes, is strongly advised for susceptible persons. Tolnaftate powder (Tinactin) or Zeasorb medicated powder is an excellent dusting powder for the feet. Plain talc, cornstarch, or rice powder may be dusted into socks and shoes to keep the feet dry. Periodic use of a topical antifungal agent may be required, especially when hot occlusive footwear is worn.
Treatment
Clotrimazole, miconazole, sulconazole, oxiconazole, ciclopirox, econazole, ketoconazole, naftifine, terbinafine, flutrimazole, bifonazole, efinaconazole, and butenafine are effective topical antifungal agents but especially in tinea manuum or exuberant tinea pedis, an oral agent may be necessary. When there is significant maceration between the toes, the toes may be separated by foam or cotton inserts in the evening. Aluminum chloride 10% solution or aluminum acetate, 1 part to 20 parts of water, can be beneficial. Interdigital tinea can also be treated with antifungal-impregnated socks. Topical antibiotic ointments, such as gentamicin (Garamycin), that are effective against gram-negative organisms are helpful additions in some moist interdigital lesions. In the ulcerative type of gram-negative toe web infections, systemic antibiotic therapy is necessary (see Chapter 14 ). Keratolytic agents containing salicylic acid, resorcinol, lactic acid, and urea may be useful in some cases, although all may lead to maceration if occluded.
Treatment of fungal infection of the skin of the feet and hands with griseofulvin, 500–1000 mg/day, can be effective. Dosage for children is 10–20 mg/kg/day. The period of therapy depends on the response of the lesions. Repeated KOH scrapings and cultures should be negative. Shorter courses are possible with other antifungal agents. Recommended adult dosing for terbinafine is 250 mg/day for 1–2 weeks; for itraconazole, 200 mg twice daily for 1 week; and for fluconazole, 150 mg once weekly for 4 weeks.
Tinea Nigra
Hortaea werneckii (formerly Phaeoannellomyces werneckii ) is a black, yeastlike hyphomycete that is widely distributed in hot, humid environments. The organism is common in the tropics. In the United States the infection is seen along the Gulf Coast. New taxonomic analysis has led some to classify Cladosporium castellanii as the etiologic agent of tinea nigra in humans and confirmed that this fungus is the same as Stenella araguata.
Tinea nigra presents as one or several brown or black spots on the palms or soles. The lesions may be mistaken for nevi, melanoma or talon noir hemorrhage under the skin. The pigment is confined to the stratum corneum and scrapes off easily. Dermoscopy has also been used to differentiate the lesions from melanocytic tumors although a case of tinea nigra with a parallel ridge pattern simulating melanoma was reported. The fungus can easily be demonstrated by means of KOH or culture. In KOH preparations, the hyphae appear brown or golden in color. The pigment produced by the fungal hyphae is melanin. Culture will identify the organism, and polymerase chain reaction (PCR) can be useful for rapid identification of H. werneckii.
Topical imidazoles and allylamines, such as clotrimazole, miconazole, ketoconazole, sulconazole, econazole, and terbinafine, have been reported as effective. Griseofulvin is not effective. Simply shaving away the superficial epidermis with a blade is frequently both diagnostic and curative of tinea nigra.
Onychomycosis (Tinea Unguium)
Onychomycosis is defined as the infection of the nail plate by fungus and represents up to 30% of diagnosed superficial fungal infections. T. rubrum accounts for most cases, but many fungi may be causative. Other etiologic agents include E. floccosum and various species of Microsporum and Trichophyton fungi. It may also be caused by yeasts and nondermatophytic molds.
The four classic types of onychomycosis are as follows:
- 1.
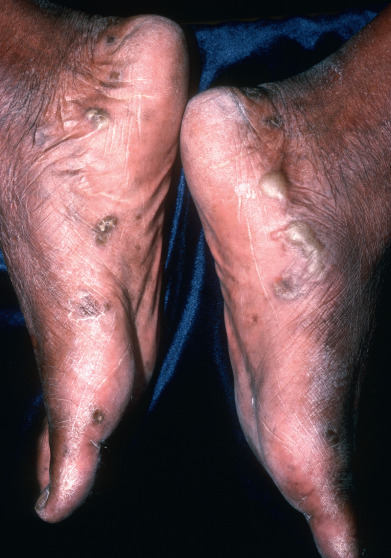
Distal subungual onychomycosis primarily involves the distal nail bed and the hyponychium, with secondary involvement of the underside of the nail plate of fingernails and toenails ( Fig. 15.11 ). It is usually caused by T. rubrum although T. tonsurans is growing in prevalence in children .
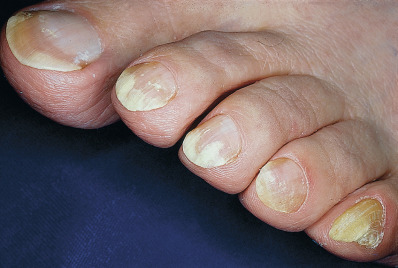
Fig. 15.11
Distal subungual onychomycosis.
- 2.
White superficial onychomycosis (leukonychia trichophytica) is an invasion of the toenail plate on the surface of the nail. It is produced by T. mentagrophytes, Cephalosporium, Aspergillus, and Fusarium oxysporum fungi. In the HIV-positive population, it is typically caused by T. rubrum.
- 3.
Proximal subungual onychomycosis involves the nail plate mainly from the proximal nailfold, producing a specific clinical picture ( Fig. 15.12 ). It is produced by T. rubrum and Trichophyton megninii and may be an indication of HIV infection.
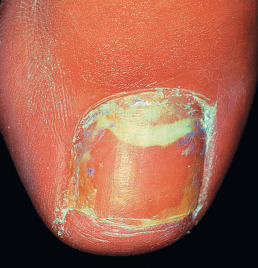
Fig. 15.12
White proximal subungual onychomycosis.
- 4.
Candida onychomycosis produces destruction of the nail and massive nail bed hyperkeratosis. It is caused by C. albicans and is seen in patients with chronic mucocutaneous candidiasis and other forms of immunodeficiency.
Onychomycosis caused by T. rubrum usually starts at the distal corner of the nail and involves the junction of the nail and its bed. A yellowish discoloration occurs, which spreads proximally as a streak in the nail. Later, subungual hyperkeratosis becomes prominent and spreads until the entire nail is affected. Gradually, the entire nail becomes brittle and separated from its bed as a result of the piling up of subungual keratin. Fingernails and toenails present a similar appearance, and the skin of the soles is likely to be involved, with characteristic branny scaling and erythema.
Onychomycosis caused by T. mentagrophytes is usually superficial, and there is no paronychial inflammation. The infection generally begins with scaling of the nail under the overhanging cuticle and remains localized to a portion of the nail. In time, however, the entire nail plate may be involved. White superficial onychomycosis is the name given to one type of superficial nail infection caused by this fungus in which small, chalky white spots appear on or in the nail plate. These are so superficial that they may be easily shaved off. T. violaceum, T. schoenleinii, and T. tonsurans occasionally invade the nails, as does Trichosporon beigelii.
Scopulariopsis brevicaulis and other nondermatophyte molds are infrequently isolated from onychomycosis. Infection usually begins at the lateral edge of the nail, burrows beneath the plate, and produces large quantities of cheesy debris. Nattrassia mangiferae (Hendersonula toruloidea) and Scytalidium hyalinum have been reported to cause onychomycosis, as well as a moccasin-type tinea pedis. Other common causes of nondermatophyte molds are Aspergillus species, Fusarium, and Acremonium. In addition to the more common features of onychomycosis, such as nail plate thickening, opacification, and onycholysis, features of infection with these fungi include lateral nail invasion alone, paronychia, and transverse fracture of the proximal nail plate. When these agents are suspected, culture must be done with a medium that does not contain cycloheximide (found in Mycosel agar). Oral griseofulvin is not effective in the treatment of these organisms.
The pathogen is heavily influenced by heredity, geography, and footwear. In the United States most tinea pedis and onychomycosis are caused by T. rubrum.
Diagnosis
The demonstration of fungus is made by microscopic examination or by culture. The submitted clippings or curettings must include dystrophic subungual debris. Samples obtained by a drilling technique may have a higher yield than those obtained by curettage. Immediate examination may be made if very thin shavings or curettings are taken from the diseased nail bed and examined with KOH solution with or without an added stain. Histologic examination, PCR, and calcofluor white microscopy and culture have also been used.
Histopathologic examination with periodic acid–Schiff (PAS) stain has been found to be 41%–93% sensitive in various studies. It has proved more sensitive than either KOH or culture in several studies. Immunofluorescent microscopy without calcofluor white is comparable to PAS staining, but high background eosin fluorescence can make the sections difficult to read. Studies have shown the sensitivity of culture to be 30%–70%. Combining KOH and culture has yielded sensitivities in the range of 80%–85%.
DTM can also be used with approximately 50% sensitivity but laboratory regulations have limited its use in the United States. PCR is emerging as an alternative method of detection.
Because no single method offers 100% sensitivity, a variety of methods are still in use. KOH has the advantage of being performed rapidly in the office. Histologic examination usually provides results within 24 hours, whereas culture can take days to weeks. Identification of genus and species is only possible with culture or PCR.
Differential Diagnosis
Dystrophic nails can be caused by psoriasis, lichen planus, eczema, and contact dermatitis and may be clinically indistinguishable from fungal nails. Confirmatory tests to identify the fungus are mandatory to establish a diagnosis. Psoriasis may involve other nails with pitting, onycholysis, oil spots, and salmon patches or by heaped-up subungual keratinization. Typical features of psoriasis may be present on other areas of skin. Lichen planus may produce rough nails or pterygium formation and may involve the oral mucosa or skin. Eczema and contact dermatitis affect the adjacent nailfold. Langerhans cell histiocytosis can also lead to subungual debris and hemorrhage and may be a marker for systemic disease. Hyperkeratotic (“Norwegian”) scabies can also produce dystrophic nails, but this is associated with generalized hyperkeratosis.
Onychomycosis among psoriasis patients is reported with varying prevalence but occurs in about 22%, compared with 13% for patients with other skin diseases. Onychomycosis occurs more frequently in men than in women with psoriasis.
Treatment
Many patients with onychomycosis are not symptomatic and may not seek treatment. Patients with diabetes or peripheral neuropathy may be at higher risk for bacterial cellulitis related to onychomycosis, and the benefits of treatment may be greater in this population. These factors, as well as cost, risk of recurrence, and spread to other family members should be considered as part of the decision to treat onychomycosis.
The topical management of onychomycosis has improved with the introduction of ciclopirox, eficonazole, and amorolfine nail lacquers. These agents are modestly effective at moderate cost. Eficonazole and ciclopirox must be used for 48 weeks. Topical therapy with ciclopirox achieved a 71% effective cure rate and 77% mycologic cure rate in children, possibly due to thinner nail plates or increased compliance. Eficonazole has a complete cure rate of 15%–17% but mycologic cure rate of 55%. No topical agent achieves the cure rates possible with oral therapy, but topical agents have lower risk of side effects because they are not systemic. Neodymium:yttrium-aluminum-garnet (Nd:YAG) laser therapy was recently reported to have a 50%–60% cure rate after multiple therapies.
For adults with disease involving fingernails, terbinafine is given in doses of 250 mg/day for 6 weeks; the dose for children is 250 mg daily for patients over 40 kg, 125 mg ( ![]() tablet) for those 20–40 kg, and 62.5 mg (
tablet) for those 20–40 kg, and 62.5 mg ( ![]() tablet) for those under 20 kg. For toenails, the course of treatment is generally 12 weeks. In adults, Itraconazole is generally given as pulsed dosing, 200 mg twice daily for 1 week of each month, for 2 months when treating fingernails and for 3–4 months when treating toenails. Fluconazole, 150–300 mg once weekly for 6–12 months, appears to be effective. About 20% of patients will not respond to treatment. The presence of a dermatophytoma within the nail may be associated with a higher risk of failure. Dermatophytomas present as yellow streaks within the nail and may respond to unroofing and curettage. Recurrence rates may be lower with itraconazole than with terbinafine monotherapy, and combined therapy does not result in a lower rate of recurrence. See monitoring recommendations earlier in this chapter.
tablet) for those under 20 kg. For toenails, the course of treatment is generally 12 weeks. In adults, Itraconazole is generally given as pulsed dosing, 200 mg twice daily for 1 week of each month, for 2 months when treating fingernails and for 3–4 months when treating toenails. Fluconazole, 150–300 mg once weekly for 6–12 months, appears to be effective. About 20% of patients will not respond to treatment. The presence of a dermatophytoma within the nail may be associated with a higher risk of failure. Dermatophytomas present as yellow streaks within the nail and may respond to unroofing and curettage. Recurrence rates may be lower with itraconazole than with terbinafine monotherapy, and combined therapy does not result in a lower rate of recurrence. See monitoring recommendations earlier in this chapter.
Several studies suggest that continuous therapy with terbinafine for 4 months is cost-effective compared with other possible agents and regimens. Most clinical trials have been industry sponsored, however, and little independent research is available for review. Terbinafine, itraconazole, and fluconazole have all been shown to be effective. Dosage depends on body weight, as previously indicated. Duration of treatment is the same as for adults.
Although data are limited, treatment with systemic antifungals is generally effective in onychomycosis caused by Aspergillus spp. Scopulariopsis brevicaulis and Fusarium spp. Infection may be difficult to eradicate, and treatment with both systemic antifungals and topical nail lacquers may be appropriate. Nail avulsion represents another option but is more painful and recurrence can still occur.
Candida onychomycosis is a sign of immunosuppression but can be seen infants as well. Systemic treatment with itraconazole or fluconazole is usually effective, but relapses are the rule. When treating Candida infections, combinations of topical and systemic treatment can be used for synergistic effect. The combination of topical amorolfine and oral itraconazole, which interferes with different steps of ergosterol synthesis, has been shown to exhibit substantial synergy in this setting. Combination treatment with topical amorolfine and two pulses of itraconazole may be as effective as three pulses of itraconazole, with lower cost.
Itraconazole pulsed treatment has been shown to have a low incidence of liver function abnormalities (alanine transaminase [ALT], aspartate transaminase [AST], alkaline phosphatase, total bilirubin). Product labeling recommends liver function tests (LFTs) for patients receiving continuous itraconazole for periods exceeding 1 month. Monitoring is required for the pulsed regimen if the patient has a history of hepatic disease, has abnormal baseline LFTs, or development of signs or symptoms suggestive of liver dysfunction.
Molds are sensitive to ozone gas, UV light, and visible light. T. rubrum in culture has been shown to be susceptible to UVC radiation, photodynamic therapy (PDT), psoralen with UVA (PUVA), and various forms of laser light. However, the mechanism of action and degree of effectiveness of these therapies require further study. For PDT with broad-band white light, the phthalocyanines and Photofrin displayed a fungistatic effect, whereas porphyrins caused photodynamic killing of the dermatophyte. 5,10,15- Tris (4-methylpyridinium)-20-phenyl-(21H,23H)-porphine trichloride and deuteroporphyrin monomethylester showed superior results in vitro. Further study of various methods of phototherapy is warranted.
Barot BS, et al: Drug delivery to the nail: therapeutic options and challenges for onychomycosis. Crit Rev Ther Drug Carrier Syst 2014; 31: 459.
Becker C, et al: Lasers and photodynamic therapy in the treatment of onychomycosis. Dermatol Online J 2013; 19:19611.
Bonifaz A, et al: Dermatophyte isolation in the socks of patients with tinea pedis and onychomycosis. J Dermatol 2013; 40: 504.
Boyd AS: Tinea capitis caused by Trichophyton rubrum mimicking favus. Cutis 2016; 98: 389.
Bristow IR: The effectiveness of lasers in the treatment of onychomycosis. J Foot Ankle Res 2014; 7: 34.
Carney C, et al: Treatment of onychomycosis using a submillisecond 1064-nm neodymium:yttrium-aluminum-garnet laser. J Am Acad Dermatol 2013; 69: 578.
Eichenfield LF, Friedlander SF: Pediatric onychomycosis. J Drugs Dermatol 2016; 16: 105.
Elewski BE, et al: Onychomycosis. J Drugs Dermatol 2013; 12: s96.
El-Gohary M, et al: Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev 2014; 8: CD009992.
Friedlander SF, et al: Onychomycosis does not always require systemic treatment for cure. Pediatr Dermatol 2013; 30: 316.
Fuller LC, et al: British Association of Dermatologists’ guidelines for the management of tinea capitis 2014. Br J Dermatol 2014; 171: 454.
Ghannoum MA, et al: Molecular analysis of dermatophytes suggests spread of infection among household members. Cutis 2013; 91: 237.
Gupta AK, et al: Medical devices for the treatment of onychomycosis. Dermatol Ther 2012; 25: 574.
Gupta AK, et al: Recurrences of dermatophyte toenail onychomycosis during long-term follow-up after successful treatments with mono- and combined therapy of terbinafine and itraconazole. J Cutan Med Surg 2013; 17: 201.
Ilkit M, et al: Tinea pedis. Crit Rev Microbiol 2015; 41: 374.
Kumar V, et al: Extensive nail changes in a toddler with multisystemic Langerhans cell histiocytosis. Pediatr Dermatol 2017; 34: 732.
Lacarrubba F, et al: Newly described features resulting from high-magnification dermoscopy of tinea capitis. JAMA Dermatol 2015; 151: 308.
Lanternier F, et al: Deep dermatophytosis and inherited CARD9 deficiency. N Engl J Med 2013; 369: 1704.
Miyajima Y, et al: Rapid real-time diagnostic PCR for Trichophyton rubrum and Trichophyton mentagrophytes in patients with tinea unguium and tinea pedis using specific fluorescent probes. J Dermatol Sci 2013; 69: 229.
Motamedi M, et al: Growing incidence of non-dermatophyte onychomycosis in Tehran, Iran. Jundishapur J Microbiol. 2016; 9: e40543.
Neri I, et al: Corkscrew hair. JAMA Dermatol 2013; 149: 990.
Noguchi H, et al: Tinea nigra showing a parallel ridge pattern on dermoscopy. J Dermatol 2015; 42: 518.
Shemer A: Update: medical treatment of onychomycosis. Dermatol Ther 2012; 25: 582.
Shemer A, et al: Increased risk of tinea pedis and onychomycosis among swimming pool employees in Netanya area, Israel. Mycopathologia 2016; 181: 851.
Shemer A, et al: Treatment of tinea capitis—griseofulvin versus fluconazole—a comparative study. J Dtsch Dermatol Ges 2013; 11: 737.
Solís-Arias MP, Garcia-Romero MT: Onychomycosis in children. Int J Dermatol 2017; 56: 123.
Tietz HJ, et al: Efficacy of 4 weeks topical bifonazole treatment for onychomycosis after nail ablation with 40% urea. Mycoses 2013; 56: 414.
Verma SB: Steroid modified tinea. BMJ 2017; 356: j973.
Wanitphakdeedecha R, et al: Efficacy and safety of 1064-nm Nd:YAG laser in treatment of onychomycosis. J Dermatol Treat 2016; 27: 75.
Yuen CW, et al: Treatment of interdigital-type tinea pedis with a 2-week regimen of wearing hygienic socks loaded with antifungal microcapsules. J Am Acad Dermatol 2013; 69: 495.
Zampella JG, et al: Tinea in tots. J Pediatr 2017; 183: 12.
Candidiasis
Candidiasis is also known as candidosis or moniliasis. Candida albicans is a common inhabitant of the human gastrointestinal (GI) tract, genitourinary tract, and skin. Under the right conditions, C. albicans becomes a pathogen, and can infect the skin, nails, and mucous membranes. Candida albicans is an opportunistic organism, acting as a pathogen in the presence of impaired immune response, or where local conditions favor growth. Warmth and moisture favor candidal growth, as can reductions in competing flora during antibiotic therapy. The intertriginous areas, including perianal and inguinal folds, abdominal creases, inframammary creases, interdigital areas, nailfolds, and axillae, are at especially high risk. Higher skin pH can occur due to occlusion from diapers and underwear liners also favor candidal growth.
Diagnosis
Candida is often a clinical diagnosis based on red maceration in skin folds with pustules and peeling at the edges of the red patch (satellite pustules). A KOH preparation will show spores and pseudohyphae. On Gram stain the yeast forms dense, gram-positive, ovoid bodies, 2–5 µm in diameter. A combination of Gomori methenamine silver (GMS) and Congo red staining can be helpful in the differential diagnosis of fungal infections. Blastomyces and Pityrosporum are positive for both, whereas Candida and Histoplasma are GMS positive and Congo red negative.
Candida proliferates in both budding and mycelial forms in the stratum corneum or superficial mucosa. Budding yeast and pseudohyphae are easier to detect in histologic section with a PAS stain. Whereas dermatophyte hyphae tend to run parallel to the skin surface, Candida pseudohyphae are more prone to vertical orientation.
Topical Anticandidal Agents
Most of the topical agents marketed for tinea are also effective for candidiasis. These include clotrimazole, econazole, ketoconazole, miconazole, oxiconazole, sulconazole, naftifine, terconazole, ciclopirox olamine, butenafine, terbinafine, nystatin, and topical amphotericin B lotion. Older agents, such as gentian violet, Castellani paint, and boric acid, are still sometimes used. Mupirocin has been show in a large study to have excellent candida coverage. Oral nystatin is as effective as intravenous (IV) fluconazole at preventing invasive Candida infections in preterm neonates. The oral preparation is more easily administered and is lower in cost.
Other Agents
Fluconazole can be used for acute infections or more chronically for prevention in Candida related to genodermatoses. Posaconazole, itraconazole, voriconazole, echinocandins, anidulafungin, and amphotericin B are also used in various settings. Various flavonoid compounds, including apigenin and kaempferol, alkaloid ibogaine (an indole), and the protoberberine alkaloid berberine, have been studied for their inhibitory effects. Topical application of each of these agents accelerated elimination from cutaneous sites of inoculation.
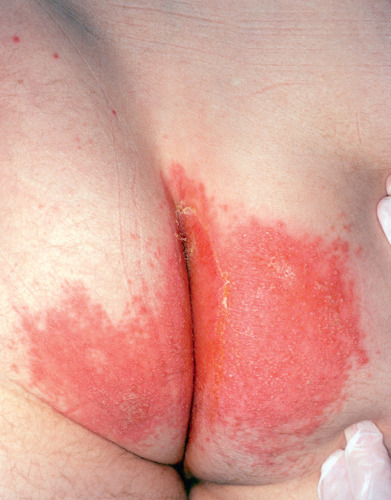
Candidal Intertrigo
The pruritic intertriginous eruptions caused by C. albicans may arise between the folds of the genitals; in groins or armpits; between the buttocks ( Fig. 15.13 ); under large, pendulous breasts; under overhanging abdominal folds; or in the umbilicus. The pink to red, intertriginous moist patches are surrounded by a thin, overhanging fringe of somewhat macerated epidermis (“collarette” scale). Some eruptions in the inguinal region may resemble tinea cruris, but usually there is less scaliness and a greater tendency to fissuring. Persistent excoriation and subsequent lichenification and drying may modify the original appearance over time. Often, tiny, superficial, white pustules are observed closely adjacent to the patches. When present, Candida can cause flares of inverse psoriasis, although prevalence of Candida is not increased in the intertriginous areas of patients with either psoriasis or atopic dermatitis.