Acne Vulgaris
Clinical Features
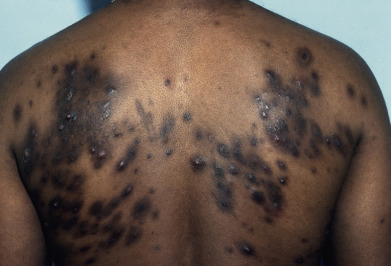
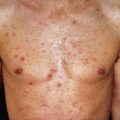
Acne vulgaris is a chronic inflammatory disease of the pilosebaceous follicles, characterized by comedones, papules, pustules, nodules, and often scars. The comedo is the primary lesion of acne. It may be seen as a flat or slightly elevated papule with a dilated central opening filled with blackened keratin (open comedo or blackhead) ( Fig. 13.1 ). Closed comedones (whiteheads) are usually 1-mm yellowish papules that may require stretching of the skin to visualize. Macrocomedones, which are uncommon, may reach 3–4 mm in size. The papules and pustules are 1–5 mm in size and are caused by inflammation, so erythema and edema occur ( Fig. 13.2 ). They may enlarge, become more nodular ( Fig. 13.3 ), and coalesce into plaques of several centimeters that are indurated or fluctuant, contain sinus tracts, and discharge serosanguineous or yellowish pus.
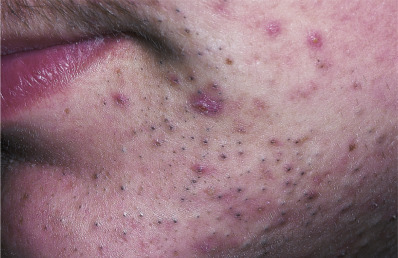
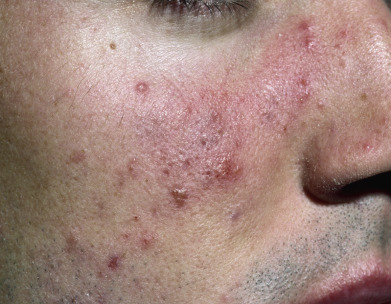
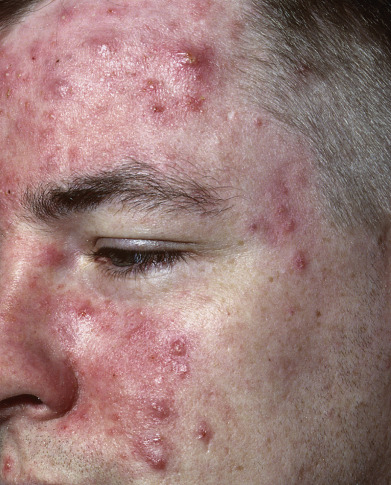
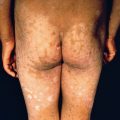
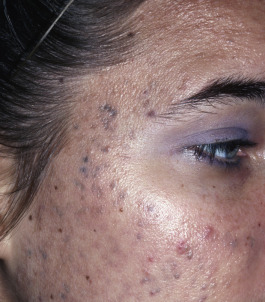
Patients typically have a variety of lesions in various states of formation and resolution. In light-skinned patients, lesions often resolve with a reddish purple macule that is short lived. In dark-skinned individuals, macular hyperpigmentation results and may last several months ( Fig. 13.4 ). Acne scars are heterogeneous in appearance. Morphologies include deep, narrow, ice pick scars seen most often on the temples and cheeks; canyon-type atrophic lesions on the face ( Fig. 13.5 ); whitish yellow papular scars on the trunk and chin; anetoderma-type scars on the trunk; and hypertrophic and keloidal elevated scars on the neck and trunk.
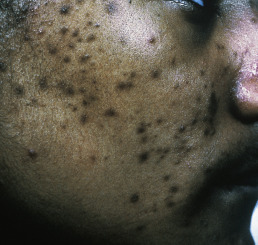
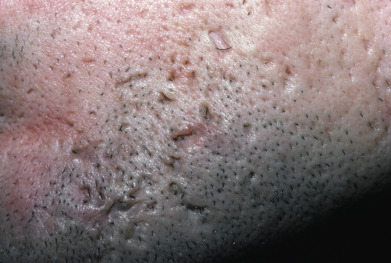
Acne affects primarily the face, neck, upper trunk ( Fig. 13.6 ), and upper arms. On the face, acne occurs most frequently on the cheeks and to a lesser degree on the nose, forehead, and chin. The ears may be involved, with large comedones in the concha, cysts in the lobes, and sometimes preauricular and retroauricular comedones and cysts. On the neck, especially in the nuchal area, large cystic lesions may predominate.
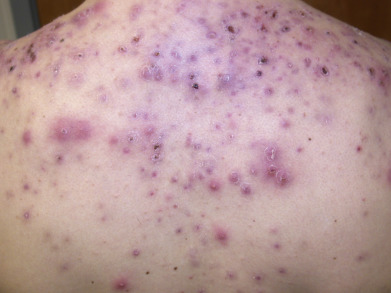
Acne typically begins at puberty and is often the first sign of increased sex hormone production. When acne begins at age 8–12 years, it is frequently comedonal in character, affecting primarily the forehead and cheeks. It may remain mild in its expression, with only an occasional inflammatory papule. However, as hormone levels rise into the middle teenage years, more severe inflammatory pustules and nodules occur, with spread to other sites. Young men tend to have an oilier complexion and more severe widespread disease than young women. Women may experience a flare of their papulopustular lesions about 1 week before menstruation. Acne may also begin in 20- to 35-year-old women who have not experienced teenage acne. This acne frequently manifests as papules, pustules, and deep, painful, persistent nodules on the jawline, chin, and upper neck.
Acne is primarily a disease of the adolescent, with 85% of all teenagers being affected to some degree. It occurs with greatest frequency between ages 15 and 18 in both genders. Generally, involution of the disease occurs before age 25; however, great variability in age at onset and of resolution occurs. About 12% of women and 3% of men will continue to have clinical acne until age 44. A few will have inflammatory papules and nodules into late adulthood.
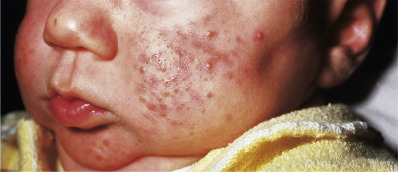
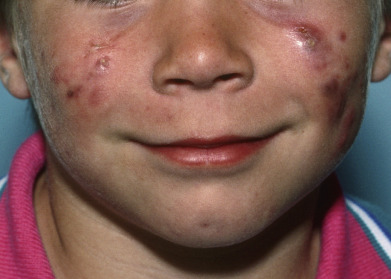
Neonatal acne is a common condition that develops a few days after birth, has male preponderance, and is characterized by transient facial papules or pustules that usually clear spontaneously in a few days or weeks ( Fig. 13.7 ). Neonatal cephalic pustulosis (infantile acne) includes cases that persist beyond the neonatal period or that have an onset after the first 6 weeks of life. Most patients’ disease remits by age 1 year, although occasionally cases extend into childhood and through puberty. In prolonged cases, topical benzoyl peroxide, erythromycin, or the retinoids may be effective. With more inflammatory disease, oral erythromycin, 125 mg twice daily, or trimethoprim, 100 mg twice daily, may be added to topical medications. Oral isotretinoin has been used in the infantile period and is effective. Midchildhood acne may evolve from persistent infantile acne or begin after age 1 year. It is uncommon and has a male predominance. Grouped comedones, papules, pustules, and nodules can occur alone or in any combination, usually limited to the face ( Fig. 13.8 ). The duration is variable, from a few weeks to several years, and occasionally extends into more severe pubertal acne. Often, there is a strong family history of moderately severe acne. A pediatric endocrinology workup is indicated for midchildhood acne and for earlier-onset patients with physical findings suggestive of a hormonal disorder, such as sexual precocity, virilization, or growth abnormality. Acne onset from age 7–12 is categorized as preadolescent acne. This is the time of adrenarche, and unless there are signs of androgen excess, no workup is needed.
Pathogenesis
Acne vulgaris is exclusively a follicular disease, with the principal abnormality being comedo formation. It is produced by the impaction and distention of the follicles with a keratinous plug in the lower infundibulum. The keratinous plug is caused by hyperproliferation and abnormal differentiation of keratinocytes. Androgens, alterations in lipid composition, and an abnormal response to local cytokines are all hypothesized to be causally important. Androgen stimulation of the sebaceous glands is critical. Acne begins after sebum secretion increases, and women with hyperandrogenic states often manifest acne, along with hirsutism and menstrual abnormalities. Treatment directed at reducing sebaceous secretion, such as isotretinoin, estrogens, or antiandrogens, is effective in clearing acne.
As the retained cells block the follicular opening, the lower portion of the follicle is dilated by entrapped sebum. Disruption of the follicular epithelium permits discharge of the follicular contents into the dermis. The combination of keratin, sebum, and microorganisms, particularly Propionibacterium acnes, leads to the release of proinflammatory mediators and the accumulation of lymphocytes, neutrophils, and foreign body giant cells. This in turn causes the formation of inflammatory papules, pustules, and nodulocystic lesions.
Additional factors may exacerbate acne or, in a predisposed patient, cause the onset of acne. Comedogenic greasy or occlusive products such as hair pomades may induce closed comedones and at times inflammatory lesions. Other types of cosmetics may initiate or worsen acne, but acne cosmetica is uncommon because most cosmetics are tested for comedogenicity.
Many types of mechanical or frictional forces can aggravate existing acne. A common problem is the overexuberant washing some patients think may help rid them of their blackheads or oiliness. A key feature of mechanical or frictional acne is an unusual distribution of the acne lesions. Provocative factors include chin straps, violins, hats, collars, surgical tape, orthopedic casts, chairs, and seats. One acne patient who had laser hair removal developed flares of inflammatory lesions localized to the acne-prone sites after each laser session; the legs and abdomen were spared. All these factors are likely to irritate the follicular epithelium and exacerbate the changes that lead to comedogenesis and follicular rupture. Prophylactic measures designed to interdict these various mechanical forces are beneficial.
The role of diet in causing or exacerbating acne is being actively investigated. A high-glycemic diet, skim milk, and whey protein may negatively influence acne. Dietary regimens used for therapeutic intervention have yet to be defined, however.
In all women or children with acne, the possibility of a hyperandrogenic state should be considered. In women, the presence of irregular menses, hirsutism, seborrhea, acanthosis nigricans, or androgenic alopecia increases the likelihood of finding clinically significant hyperandrogenism. Additionally, gynecologic endocrine evaluation may be indicated in women who have acne resistant to conventional therapy, who relapse quickly after a course of isotretinoin, or who experience sudden onset of severe acne. Screening tests to exclude a virilizing tumor include serum dehydroepiandrosterone sulfate (DHEAS) and testosterone, obtained 2 weeks before the onset of menses. DHEAS levels may be very high in adrenal tumors (>800 µg/dL) or less dramatic in congenital adrenal hyperplasia (400–800 µg/dL). Ovarian tumor is suggested by testosterone levels greater than 200 ng/dL. Many patients with late-onset 21-hydroxylase deficiency congenital adrenal hyperplasia will have normal levels of DHEAS. Although 17-hydroxyprogesterone and adrenocorticotropic hormone (ACTH) stimulation tests have been used in this setting, the baseline 17-hydroxyprogesterone may be normal and ACTH stimulation may result in overdiagnosis of the syndrome. It is not clear that screening for adult-onset 21-hydroxylase deficiency improves patient outcome. Cushing syndrome is also to be considered in the differential diagnosis.
The diagnosis of polycystic ovarian syndrome (PCOS) requires that two of three of the following criteria be present: oligoovulation or anovulation, biochemical or clinical signs of hyperandrogenism, and echographic polycystic ovaries. Thus the diagnosis of PCOS may be made clinically by the presence of anovulation (<9 periods per year or periods >40 days apart) and signs of hyperandrogenism, such as acne and hirsutism. Approximately 15% of women of childbearing age will meet the criteria for PCOS. This diagnosis has important implications for affected patients. Systemic disease such as insulin resistance (which may manifest as acanthosis nigricans), obesity, cardiovascular disease, obstructive sleep apnea, nonalcoholic steatohepatitis, endometrial cancer and infertility (with chronic anovulation), and psychiatric disease. Nearly half of PCOS patients have metabolic syndrome. In women with hirsutism of the chest and acanthosis nigricans of the axillae, PCOS is frequently found. Suggested biochemical investigation includes total testosterone, free testosterone, dehydroepiandrosterone (DHEAS), androstenedione, luteinizing hormone, and follicle-stimulating hormone. PCOS is a complex disorder and referral for investigation for ovarian cysts, interpretation of abnormal results, consideration of other testing such as thyroid-stimulating hormone and prolactin, and management of any associated disorders is recommended.
Acne neonatorum is explained by infantile production of androgens, which wanes at 6–12 months. Occasional patients have persistent acne, although acne developing after age 1 and before age 7 (with onset of adrenarche) may be a form of acne cosmetica, acne venenata, drug-induced acne or part of an endocrinologic disorder. A workup should be initiated if acne develops between ages 1 and 7 and no obvious external factor is present. In the absence of any discovered abnormalities, the qualitative or quantitative alteration of cutaneous androgen, metabolism, and increased end-organ sensitivity could be postulated as pathogenic mechanisms for preadolescent acne.
Pathology
Comedones reveal a thinned epithelium and a dilated follicular canal filled with lamellar lipid-impregnated keratinous material. In pustular cases, there are folliculocentric abscesses surrounded by a dense inflammatory exudate of lymphocytes and polymorphonuclear leukocytes. In addition to these findings, indolent nodular lesions frequently show plasma cells, foreign body giant cells, and proliferation of fibroblasts. Epithelial-lined sinus tracts may form.
Treatment
General Principles
It is important to take a complete historical record of prior therapies, including all over-the-counter (OTC) products. The dose, timing, combinations, side effects, and response to interventions should be obtained. Corticosteroids, anabolic steroids, neuroleptics, epidermal growth factor inhibitors, lithium, and cyclosporine may worsen acne. A family history of acne and, if present, its tendency to scarring should be noted. Women should be queried regularly about menstrual irregularities and hair growth in a male pattern, as well as use of cosmetics.
Treatment may fail because of drug interactions, coexisting conditions, or antibiotic resistance, but the most common and important cause is lack of adherence to the treatment plan. Utilizing medications that are well tolerated, have convenient dosing regimens, and are cosmetically acceptable will help. Thorough patient education is essential: explain how lesions form, define the expected response to and the duration and side effects of treatment, and give clear, unambiguous instructions. Patients should know the difference between active inflammatory lesions and the purplish red or hyperpigmented macules of inactive resolved lesions. Topical application should be to the entire affected area rather than to specific lesions, and oral and topical medications should be used daily as preventive treatment.
A high-glycemic diet may worsen acne, although the strength of its influence is unknown. The authors in general do not counsel patients to alter their diets unless large quantities of skim milk are being ingested or obesity is present. A trial lessening skim milk intake is worthwhile, with appropriate calcium and vitamin D supplementation given. In obese patients, dietary counseling is recommended, especially if PCOS, or other syndromes known to be associated with insulin resistance and metabolic syndrome (e.g., HAIR-AN syndrome) are present. For some patients who want a more “natural” approach to therapy and a change in diet, a low-glycemic diet may be recommended. Scrubbing of the face increases irritation and may worsen acne. Use of only prescribed medications and avoidance of potentially drying OTC products, such as astringents, harsh cleansers, and antibacterial soaps, should be emphasized. Noncomedogenic cosmetics are recommended, and pressed powders and oil-based products should be avoided.
Medical Therapy
Systemic and topical retinoids, systemic and topical antimicrobials, and systemic hormonal therapy are the main therapeutic classes of treatment available. Treatment guidelines are outlined in Box 13.1 . Pregnancy and lactation labeling is undergoing significant changes which will allow a better understanding of the potential risks. This will likely result in less stringent recommendations regarding topical therapies. Such changes are ongoing and in this section pregnancy categories will still be referenced.
Mild
- 1.
Comedonal
- •
Topical retinoid ± physical extraction (first line)
- •
Alternate retinoid, benzoyl peroxide, salicylic acid, azelaic acid (second line)
- •
- 2.
Papular/pustular
- •
Benzoyl peroxide or topical antimicrobial combination + topical retinoid, benzoyl peroxide wash if mild truncal lesions (first line)
- •
Alternate antimicrobial combination + alternate topical retinoid, azelaic acid, sodium sulfacetamide–sulfur (second line)
- •
Moderate
- 1.
Papular/pustular
- •
In men, oral antibiotic + topical retinoid + benzoyl peroxide (first line)
- •
Alternate antibiotic, alternate topical retinoid (second line); if moderately severe, isotretinoin
- •
In women, spironolactone and/or oral contraceptive + topical retinoids ± benzoyl peroxide; oral antibiotic + topical retinoid + benzoyl peroxide (alternative)
- •
Isotretinoin if relapses quickly off oral antibiotics, does not clear, or scars
- •
Severe
- 1.
Nodular/conglobate
- •
Isotretinoin
- •
Oral antibiotic + topical retinoid + benzoyl peroxide
- •
In women, spironolactone + oral contraceptive + topical retinoid ± topical or oral antibiotics and/or benzoyl peroxide
- •
Topical Treatment.
All topical treatments are preventive, and use for 8–12 weeks is required to judge their efficacy. The entire acne-affected area is treated, not just the lesions, and long-term use is the rule. In many patients, topical therapy may be effective as maintenance therapy after initial control is achieved with a combination of oral and topical treatment.
Topical Retinoids.
It has long been appreciated that topical retinoids are especially effective in promoting normal desquamation of the follicular epithelium, reducing comedones and inhibiting the development of new lesions. Additionally, they have a marked antiinflammatory effect, inhibiting the activity of leukocytes, the release of proinflammatory cytokines and other mediators, and the expression of transcription factors and toll-like receptors involved in immunomodulation. These agents also help penetration of other active agents. Thus the topical retinoids should be used in most patients with acne and are the preferred agents in maintenance therapy.
Tretinoin was the first of this group of agents to be used for acne. Popular forms of tretinoin are 0.025% and 0.05% in a cream base and the micronized gels because these are less irritating than standard gels and liquids. Its incorporation into microspheres and a polyolprepolymer also helps limit irritation and make the product more stable in the presence of light and oxidizers. Tretinoin treatment may take 8–12 weeks before improvement occurs. When patients are tolerating the medication and are slow to respond, retinoic acid gel or solution may be used. Tretinoin should be applied at night and is in pregnancy category C.
Adapalene is a well-tolerated retinoid-like compound that has efficacy equivalent to the lower concentrations of tretinoin. Because it is light stable, adapalene may be applied in either the morning or the evening. It is in pregnancy category C.
Tazarotene is comparatively strong in its action, but also relatively irritating. It should be applied once at night or every other night, and as it is in pregnancy category X, contraceptive counseling should be provided.
Initially using retinoids every other night or adding a moisturizer with their use may lessen their irritant effects. They are also particularly useful in patients of color because retinoids may lighten postinflammatory hyperpigmentation.
Benzoyl Peroxide.
Benzoyl peroxide has a potent antibacterial effect. Propionibacterium acnes resistance does not develop during use. Its concomitant use during treatment with antibiotics will limit the development of resistance, even if only given for short 2- to 7-day pulses. Although benzoyl peroxide is most effective in inflammatory acne, some studies have shown it to be comedolytic as well. The wash formulations may be used for mild truncal acne when systemic therapies are not required. These need to be in place 2 minutes to be effective.
Treatment is usually once or twice daily. Benzoyl peroxide may irritate the skin and produce peeling. Water-based formulations of lowest strength are least irritating and do not compromise efficacy. Application limited to once a day or every other day will also help. Allergic contact dermatitis will rarely develop, suggested by the complaint of itch rather than stinging or burning. Benzoyl peroxide is in pregnancy category C.
Topical Antibacterials.
Topical clindamycin and erythromycin are available in a number of formulations. In general, they are well tolerated and are effective in mild inflammatory acne. These topical products are in pregnancy category B. Use of these topical antibiotics alone, however, is not recommended because they may induce antibiotic resistance. As mentioned, concurrent therapy with benzoyl peroxide will limit this problem. Concomitant use with a topical retinoid will hasten the response and allow for more rapid discontinuance of the antibiotic.
Dapsone is available topically in a gel formulation. Hemolytic anemia may occur, and skin discoloration is possible when benzoyl peroxide is applied after topical dapsone. Additionally, concomitant oral use of trimethoprim-sulfamethoxazole will increase the systemic absorption of topical dapsone. Dapsone is in pregnancy category C. A topical minocycline product is being actively investigated.
Sulfur, Sodium Sulfacetamide, Resorcin, and Salicylic Acid.
Although benzoyl peroxide, retinoids, and topical antibiotics have largely supplanted these older medications, sulfur, resorcin, and salicylic acid preparations are still useful and moderately helpful if the newer medications are not tolerated. They are frequently found in OTC preparations. Sulfacetamide-sulfur combination products are mildly effective in both acne and rosacea, but should be avoided in patients with known hypersensitivity to sulfonamides.
Azelaic Acid.
This dicarboxylic acid is usually well tolerated and has mild efficacy in both inflammatory and comedonal acne. Azelaic acid may help to lighten postinflammatory hyperpigmentation and is in pregnancy category B.
Combination Topical Therapy.
Several products are available that combine antibiotics such as clindamycin and benzoyl peroxide or combine retinoids and either antibiotics or benzoyl peroxide. In general, these medications increase adherence because they require less frequent application, and they may also limit irritation compared with the cumulative topical application of each product separately. However, combination topical therapy limits flexibility and may cause more irritation than a single product used alone.
Oral Antibiotics.
Oral antibiotics are indicated for moderate to severe acne; in patients with inflammatory disease who do not tolerate or respond to topical combinations; for the treatment of chest, back, or shoulder acne; and in patients for whom absolute control is deemed essential, such as those who scar with each lesion or who develop inflammatory hyperpigmentation. It generally takes 8–12 weeks to judge efficacy. Starting at a high dose and stopping it after achieving control is preferred. Limitation on the use of antibiotics is best. Working to maintain control eventually with topical retinoids or retinoid–benzoyl peroxide combination therapy is ideal. A subset of patients will require longer term use if alternate therapies such as isotretinoin or hormonal agents are inappropriate. In these patients reevaluation at regular intervals to judge continued need is recommended.
There is concern that oral antibiotics may reduce the effectiveness of oral contraceptives (OCs). It is appropriate for this as-yet unproved (except with rifampin, which is not used for acne) association be discussed with patients and a second form of birth control offered.
Tetracycline.
Tetracycline’s availability and utility are limited.
Doxycycline.
The usual dose of doxycycline is 50–100 mg once or twice a day, depending on the disease severity. Photosensitivity reactions can occur with this form of tetracycline and can be dramatic. Vaginitis or perianal itching may result and occurs in about 5% of patients, with Candida albicans usually present in the involved site. The only other common side effects are gastrointestinal (GI) symptoms such as nausea. To reduce the incidence of esophagitis, tetracyclines should not be taken at bedtime. An enteric-coated formulation is available and limits the GI side effects. Staining of growing teeth occurs, precluding use of tetracyclines in pregnant women and in children under age 9 or 10. The tetracyclines should also be avoided when renal function is impaired.
Sub–antimicrobial-dose doxycycline hyclate, given as 20 mg twice daily or as a sustained-release 40-mg once-daily dose, will also serve to limit antibiotic use, as antiinflammatory activity is being utilized, but no antibiotic resistance results because of the low dose. A sustained-release 40-mg formulation of doxycycline monohydrate is also available.
Minocycline.
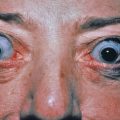
Minocycline is effective in treating acne vulgaris. The usual dose is 50–100 mg once or twice daily, depending on the severity of disease. Its absorption is less affected by milk and food than is doxycycline. Vertigo may occur, and beginning minocycline therapy with a single dose in the evening is prudent. An extended-release preparation is also available, which limits the vestibular side effects. Pigmentation in areas of inflammation, of oral tissues, in postacne osteoma or scars, in a photodistributed pattern, on the shins, or in the sclera, nail bed, ear cartilage, or teeth or in a generalized pattern may also be seen ( Fig. 13.9 ). Additionally, lupus-like syndromes, a hypersensitivity syndrome (fever, hepatitis, and eosinophilia), serum sickness, pneumonitis, and hepatitis are uncommon but potentially serious adverse effects of minocycline.
Amoxicillin.
For those who cannot take tetracyclines because of side effects, or in pregnant women requiring oral antibiotic therapy, amoxicillin may be useful. Amoxicillin and the much less effective erythromycin are in pregnancy category B. Amoxicillin can be given in doses ranging from 250 mg daily to 500 mg three times daily. Side effects are allergic reactions, which may be serious, and GI upset. Many patients of acne age have taken amoxicillin in the past and are aware of their allergy status.
Clindamycin.
Past experience has shown that clindamycin provides an excellent response in the treatment of acne. However, the potential for the development of pseudomembranous colitis and the availability of isotretinoin have limited its use. The initial dose of clindamycin is 150 mg three times daily, reduced gradually as control is achieved.
Other Antibiotics.
Sulfonamides may be effective in many cases unresponsive to other antibiotics; however, the potential for severe drug eruptions limits their use by dermatologists. Trimethoprim-sulfamethoxazole (TMP-SMX; Bactrim, Septra), in double-strength dose twice daily, is recommended initially when given to moderately to severely affected patients who have failed other oral medication. Trimethoprim alone, 300 mg twice daily, is also useful. Oral dapsone has been used in severe acne conglobata (AC) but is rarely used today. Isotretinoin is favored.
Bacterial Resistance.
Propionibacterium acnes antimicrobial resistance has been a clinically relevant problem. However, with the limited use of erythromycin, clindamycin, and tetracycline, this consideration is less problematic. Doxycycline resistance may occur, and minocycline is a suitable alternative if this problem is suspected. Although concomitant use of benzoyl peroxide will help limit cutaneous drug resistance problems, Staphylococcus aureus in the nares, streptococci in the oral cavity, and enterobacteria in the gut may also become resistant. Also, close contacts, including treating dermatologists, may harbor such drug-resistant bacteria. Strategies to prevent antibiotic resistance include limiting the duration of treatment, stressing the importance of adherence to the treatment plan, restricting the use of antibiotics to inflammatory acne, encouraging repeat treatment with the same antibiotic unless it has lost its efficacy, avoiding the use of dissimilar oral and topical antibiotics at the same time, and using isotretinoin if unable to maintain clearance without oral antibacterial therapy.
Hormonal Therapy.
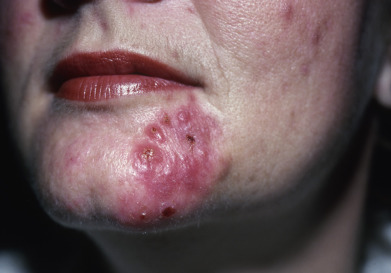
Hormonal interventions in women may be beneficial even in the absence of abnormal laboratory tests. The workup for the woman with signs of hyperandrogenism, such as acne, menstrual irregularities, hirsutism, or androgenic alopecia, is presented earlier. Women with normal laboratory values respond to hormonal therapy. Results take longer to be seen with these agents, with first evidence of improvement often not apparent for 3 months and continued improved response seen for at least 6 months. Excellent candidates for hormonal treatment include women with PCOS, late-onset adrenal hyperplasia, or another identifiable endocrinologic condition and women with late-onset moderate to severe acne, acne unresponsive to other oral and topical therapies, or acne that has relapsed quickly after isotretinoin treatment. Women with acne primarily located on the lower face and neck and with deep-seated nodules that are painful and long-lasting ( Fig. 13.10 ) are often quite responsive to hormonal intervention. As emphasis is placed on limiting antibiotic therapy, hormonal treatment may considered a first-line therapy in most women needing an oral intervention.
Oral Contraceptives.
The OCs block both adrenal and ovarian androgens. Ortho Tri-Cyclen, Estrostep Fe, Alesse, Yasmin, Beyaz, and Yaz are examples of OCs that have beneficial effects on acne. The progestins that these contain have either low androgenic activity or antiandrogenic activity. Both the physician and the patient should be familiar with the adverse reactions associated with OCs, such as nausea, vomiting, abnormal menses, melasma, weight gain, breast tenderness, and rarely thrombophlebitis, pulmonary embolism, and hypertension.
Spironolactone.
Antiandrogen treatment during pregnancy will result in feminization of a male fetus, and thus spironolactone is usually prescribed in combination with OCs. It may be effective in doses from 25–200 mg/day. Most women will tolerate a starting dose of 100 mg at night. Most also tolerate 150 mg/day (50 in the am , 100 at night), but many will have side effects at 200 mg/day (100 twice daily). Side effects include irregular menstrual periods, breast tenderness, headache, dizziness, lightheadedness, fatigue, and occasionally diuresis; the non–central nervous system (CNS) effects are dose dependent. Four large recent retrospective studies of over 700 patients confirm the majority experience significant improvement, and many cleared with combination oral and/or topical intervention. Its use during pregnancy will result in feminization of a male fetus. Combination use with OCs serves to limit this problem and improve results. Several months of treatment are usually required to see benefit.
Dexamethasone.
Dexamethasone, 0.125–0.5 mg given once at night, reduces androgen excess and may alleviate cystic acne. Although corticosteroids are effective in the treatment of adult-onset adrenal hyperplasia, antiandrogens are often used in this setting.
Prednisone.
Although corticosteroids may produce steroid acne, they are also effective antiinflammatory agents in severe and intractable acne vulgaris. In severe cystic acne and AC, corticosteroid treatment is effective; however, side effects restrict its use. Prednisone is generally only given to patients with severe inflammatory acne during the first 1 or 2 months of treatment with isotretinoin, for initial reduction of inflammation, and to reduce isotretinoin-induced flares.
Other Hormonal Agents.
Finasteride, flutamide, estrogen, gonadotropin-releasing agonists, and metformin (by decreasing testosterone levels) have all showed a beneficial effect on acne. Because of side effects, expense, and other considerations, however, these agents are not typically used.
Oral Retinoid Therapy
Isotretinoin.
Isotretinoin is approved only for severe cystic acne. However, it is useful in less severe forms of acne to prevent the need for continuous treatment and the repeated office visits required. A consensus of experts found that oral isotretinoin is warranted for severe acne, poorly responsive acne that improves by less than 50% after 3 months of therapy with combined oral and topical antibiotics, acne that relapses after oral treatment, scars, and acne that induces psychological distress. Other indications are gram-negative folliculitis, inflammatory rosacea, pyoderma faciale, acne fulminans, and AC.
This retinoid is a reliable remedy in almost all acne patients ( Fig. 13.11 ). The dose of isotretinoin is 0.5–1 mg/kg/day in one or two daily doses. For severe truncal acne in patients who tolerate higher doses, up to 2 mg/kg/day may be given. In practice, most patients are started at 20–40 mg to avoid an early flare, then increased to 40–80 mg/day to limit side effects, which generally are dose related. Doses as low as 0.1 mg/kg/day are almost as effective as the higher doses in clearing acne; the disadvantage is that lower doses are less likely to produce a prolonged remission, even after 20 weeks of treatment. To achieve potentially prolonged remission, patients should receive 120–150 mg/kg over the treatment course. An easy way to calculate the total isotretinoin dose needed is to multiply the patient’s weight in kilograms by 3. The product is the total number of 40-mg capsules needed to reach the low end of the dosage spectrum. Two groups reported treating patients with 1.5–2 mg/kg for a total dose of approximately 300 mg/kg. These patients had a low relapse rate, although side effects often limit tolerance of such dosages.
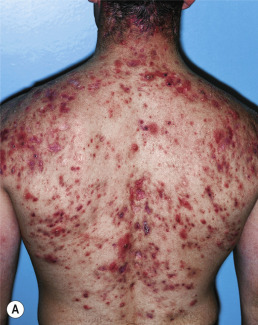
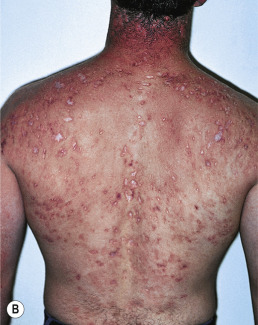
The major advantage of isotretinoin is that it is the only acne therapy that is not open ended (i.e., leads to a remission that may last many months or years). Approximately 40%–60% of patients remain acne free after a single course of isotretinoin. Approximately one half of the relapsing patients will need only topical therapy, with the others requiring oral treatments. Many patients in the latter category prefer to be retreated with isotretinoin because of its reliable efficacy and predictable side effects, which will be similar to those experienced in the first course. Many treated patients will require at least a second course of isotretinoin in 2 years.
Some subsets of patients tend to relapse more often. In patients under age 16 years, 40% need a second course of isotretinoin within 1 year and 73% within 2 years. Adult women and patients with mild acne tend to relapse more often and more quickly than severely affected 17- to 22-year-olds. Although patients’ tolerance and response to repeated courses are similar to their experience with the first course, adult women who relapse may be better managed with hormonal therapies and mild acne treated with standard therapy.
In adult acne patients, who frequently tolerate the side effects of isotretinoin less well, lower doses and intermittent therapy are possible. In 80 adult acne patients treated with 0.5 mg/kg/day for 1 week in every 4 weeks over 6 months, acne resolved in 88%, and 39% relapsed after 1 year. In nine patients age 56–75 treated with 0.25 mg/kg/day for 6 months, all cleared and all except one remained clear 36 months later.
Patient education is critical in isotretinoin therapy. Its most serious adverse effect is the risk of severe damage to the fetus if given during pregnancy. Retinoid embryopathy is a well-defined syndrome characterized by craniofacial, cardiovascular, CNS, and thymus abnormalities. It is crucial that a woman of childbearing potential follow closely the manufacturer’s recommendations. The use of consent forms, contraception education, and unequivocal documentation of the absence of pregnancy through monthly laboratory testing are important components of a U.S. Food and Drug Administration (FDA)–mandated verification program designed to prevent pregnancy during treatment. Women should not become pregnant until stopping medication for at least 1 month. Isotretinoin is not mutagenic, and there is no risk to a fetus while the male partner is taking the drug.
A second major area of educational emphasis concerns the psychological effects of the medication. Reports of depression, psychosis, suicidal ideation, suicide, and attempted suicide have prompted numerous studies of the mental health of patients taking isotretinoin. Although the usual outcome is improved mood because the disease clears, and a systematic review and meta-analysis found treatment did not appear to be associated with an increased risk of depression, a small number of patients have developed depression and have positive dechallenge and rechallenge tests. Close monitoring for depression, fully educating the patient, and enlisting the help of a roommate or family member to look for changes in mood are methods used to assess the psychological status of the patient taking isotretinoin.
Inflammatory bowel disease (IBD) is a third concern. Patients with IBD have been successfully treated with isotretinoin without flaring, but new-onset IBD in patients exposed to isotretinoin has been a concern. The age of onset of IBD overlaps with the age when acne will frequently be treated with isotretinoin and antibiotics. A meta-analysis has concluded that there was no increased risk of IBD or the subtypes. Long-term use of tetracycline medications and severe acne itself may be predisposing factors for IBD. Due to the past controversy, patients should be educated and monitored appropriately.
Other side effects of isotretinoin are dose dependent and generally not serious. Dry lips, skin, eyes, and oronasal mucosa occur in up to 90% of patients. These effects can be treated with moisturization. Dryness of the nasal mucosa leads to colonization by S. aureus in 80%–90% of treated patients. Skin abscesses, staphylococcal conjunctivitis, impetigo, facial cellulitis, and folliculitis may result. Such colonization can be avoided by the use of bacitracin ointment applied to the anterior nares twice daily during isotretinoin therapy. Arthralgias may occur but, as with other side effects, do not require interruption of therapy unless severe. Monitoring of serum lipids is done when initiating and increasing the dose because some patients will develop hypertriglyceridemia. This may be controlled by avoiding smoking and alcohol and following a low-fat diet. It should be emphasized that patients who develop this complication, as well as their family, are at risk for the development of the metabolic syndrome.
Liver function tests should be checked at regular intervals when initiating and increasing the dose. Isotretinoin should be taken with a high-fat meal to ensure excellent absorption. A formulation not requiring this type of meal is available.
Tumor Necrosis Factor Inhibitors.
Adalimumab, etanercept, and infliximab have been reported in individual patients to improve or clear severe resistant acne. Some cases have been part of an inflammatory syndrome (e.g., SAPHO, PAPA, PASS) or found in patients with IBD. Paradoxically, acne has also been reported as an adverse reaction to these medications.
Intralesional Corticosteroids.
Intralesional corticosteroids are especially effective in reducing inflammatory nodules. Triamcinolone acetonide at 10 mg/mL (Kenalog-10) is best diluted with sterile normal saline solution to 2.5 mg/mL. Injecting less than 0.1 mL directly into the center of the nodule will help safeguard against atrophy and hypopigmentation.
Physical Modalities
Local surgical treatment is helpful in quickly resolving the comedones, although many clinicians wait until after 2 or more months of topical retinoid therapy to extract the remaining comedones. The edge of the follicle is nicked with a No. 11 scalpel blade, and the contents are expressed with a comedo extractor. Scarring is not produced by this procedure. Light electrode desiccation is an alternative. In isotretinoin-treated patients, macrocomedones present at weeks 10–15 may be expressed, because they tend to persist throughout therapy.
The use of photodynamic therapy and various forms of light, laser, or radiofrequency energy is under investigation. Such interventions clearly are capable of destroying sebaceous glands and killing P. acnes, but the methods to deliver such treatment in an efficient, cost-effective, safe, relatively pain free, and practical manner are still evolving. These treatments will be a welcome addition with the potential to provide care without the concerns associated with systemic drugs. More studies of larger patient populations with appropriate controls are needed to evaluate the role of light and related energy in the spectrum of acne therapy.
Complications
Even with the excellent treatment options available, scarring may occur. This may be quite prominent and often results from the cystic type of acne, although smaller lesions may produce scarring in some individuals. Pitted scars, wide-mouthed depressions, and keloids, primarily seen along the jawline and chest, are common types of scarring. These may improve spontaneously over 1 year or longer. Many treatment options are available. Procedures reported to be effective in improving appearance include chemical peeling; ablative, nonablative, and vascular laser therapy; skin needling or rolling; dermabrasion; scar excision; subcision; punch grafts alone or followed by dermabrasion or laser smoothing; intralesional corticosteroids or fluorouracil; fractionated laser resurfacing; fat transfer; and use of filler substances.
Other complications from acne are prominent residual hyperpigmentation, especially in darker-skinned patients ( Fig. 13.12 ); pyogenic granuloma formation, which is more common in acne fulminans and in patients treated with high-dose isotretinoin; osteoma cutis, which consists of small, firm papules resulting from long-standing acne vulgaris; and solid facial edema. The latter is a persistent, firm facial swelling that is an uncommon but distressing result of acne vulgaris or acne rosacea. Both corticosteroids and isotretinoin have been reported to be effective treatments.
Abdel Hay R, et al: Interventions for acne scars. Cochrane Database Syst Rev 2016; 4: CD011946.
Asai Y, et al: Management of acne. CMAJ 2016; 188: 118.
Awan SZ, Lu J: Management of severe acne during pregnancy. Int J Womens Dermatol 2017; 13: 145.
Azoulay L, et al: Isotretinoin therapy and the incidence of acne relapse. Br J Dermatol 2007; 157: 1240.
Baldwin H: Treating acne during pregnancy and lactation. Cutis 2015; 96: 11.
Barbieri JS, et al: Approaches to limit systemic antibiotics in acne. J Am Acad Dermatol 2018; Oct 5.
Berard A, et al: Isotretinoin, pregnancies, abortions and birth defects. Br J Clin Pharmacol 2007; 63: 196.
Blasiak RC, et al: High-dose isotretinoin treatment and the rate of retrial, relapse, and adverse effects in patients with acne vulgaris. JAMA Dermatol 2013; 149: 1392.
Brown RJ, et al: Minocycline-induced drug hypersensitivity syndrome followed by multiple autoimmune sequelae. Arch Dermatol 2009; 145: 63.
Buzney E, et al: Polycystic ovary syndrome. J Am Acad Dermatol 2014; 71: 859.e1.
Çerman AA, et al: Dietary glycemic factors, insulin resistance, and adiponectin levels in acne vulgaris. J Am Acad Dermatol 2016; 75: 155.
Charney JW, et al: Spironolactone for the treatment of acne in women, a retrospective study of 110 patients. Int J Womens Dermatol 2017; 3: 111.
Chen W, et al: Acne-associated syndromes. J Eur Acad Dermatol Venereol 2011; 25: 637.
Cyrulnik AA, et al: High-dose isotretinoin in acne vulgaris. Int J Dermatol 2012; 51: 1123.
Dréno B: What is new in the pathophysiology of acne, an overview. J Eur Acad Dermatol Venereol 2017; 31 Suppl 5: 8.
Dréno B, et al: Acne in pregnant women. Acta Derm Venereol 2014; 94: 82.
Dressler C, et al: How much do we know about maintaining treatment response after successful acne therapy? Systematic review of the efficacy and safety of acne maintenance therapy. Dermatology 2016; 232: 371.
Eichenfield LF, et al: Evidence-based recommendations for the diagnosis and treatment of pediatric acne. Pediatrics 2013; 131: S163.
Fitzpatrick L, et al: Oral contraceptives for acne treatment. Cutis 2017; 99: 195.
Grandhi R, Alikhan A: Spironolactone for the treatment of acne. Dermatology 2017; 233: 141.
Hansen TJ, et al: Standardized laboratory monitoring with use of isotretinoin in acne. J Am Acad Dermatol 2016; 75: 323.
Hassoun LA, et al: The use of hormonal agents in the treatment of acne. Semin Cutan Med Surg 2016; 35: 68.
Housman E, Reynolds RV: Polycystic ovary syndrome. J Am Acad Dermatol 2014; 71: 847.e1.
Hu S, et al: Fractional resurfacing for the treatment of atopic facial acne scars in Asian skin. Dermatol Surg 2009; 35: 826.
Huang YC, Cheng YC: Isotretinoin treatment for acne and risk of depression. J Am Acad Dermatol 2017; 76: 1068.
Isvy-Joubert A, et al: Adult female acne treated with spironolactone. Eur J Dermatol 2017; 27: 393.
Jacobs A, et al: Systemic review of the rapidity of the onset of action of topical treatments in the therapy of mild-to-moderate acne vulgaris. Br J Dermatol 2014; 170: 557.
James WD: Clinical practice: acne. N Engl J Med 2005; 352: 1463.
Lee SY, et al: Does exposure to isotretinoin increase the risk for the development of inflammatory bowel disease? Eur J Gastroenterol Hepatol 2016; 28: 210.
Lee YH, et al: Laboratory monitoring during isotretinoin therapy for acne. JAMA Dermatol 2016; 152: 35.
Leyden JJ, et al: The use of isotretinoin in the treatment of acne vulgaris. J Clin Aesthet Dermatol 2014; 7: S3.
Lortscher D, et al: Hormonal contraceptives and acne. J Drugs Dermatol 2016; 15: 670.
Manolache L, et al: A case of solid facial oedema successfully treated with isotretinoin. J Eur Acad Dermatol Venereol 2009; 23: 965.
Margolis D, et al: Potential association between the oral tetracycline class of antimicrobials used to treat acne and inflammatory bowel disease. Am J Gastroenterol 2010; 105: 2610.
McCarty M: Evaluation and management of refractory acne vulgaris in adolescent and adult men. Dermatol Clin 2016; 34: 203.
Morrone A, et al: Clinical features of acne vulgaris in 444 patients with ethnic skin. J Dermatol 2011; 38: 405.
Nast A, et al: European evidence-based guidelines for the treatment of acne. J Eur Acad Dermatol Venereol 2012; 26: S1.
Rademaker M: Making sense of the effects of the cumulative dose of isotretinoin in acne vulgaris. Int J Dermatol 2016; 55: 518.
Rademaker M, et al: Isotretinoin 5 mg daily for low-grade adult acne vulgaris. J Eur Acad Dermatol Venereol 2014; 28: 747.
Rashtak S, et al: Isotretinoin exposure and risk of inflammatory bowel disease. JAMA Dermatol 2014; 150: 1322.
Schmidt TH, et al: Cutaneous findings and systemic associations in women with polycystic ovary syndrome. JAMA Dermatol 2016; 152: 391.
Spring LK, et al: Isotretinoin and timing of procedural interventions. JAMA Dermatol 2017; 153: 802.
Strahan JE, et al: Cyclosporine-induced infantile nodulocystic acne. Arch Dermatol 2009; 145: 797.
Tan J, et al: Oral isotretinoin. Dermatol Clin 2016; 34: 175.
Thiboutot DM, et al: Practical management of acne for clinicians. J Am Acad Dermatol 2018;78: S1.
Zaenglein AL, et al: Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol 2016; 74: 945.