46 Implant-Based Reconstruction and Radiation
Summary
This chapter provides an overview of the effects of radiation therapy on tissues as it pertains to implant-based reconstruction. It covers the impact of radiation on decision making prior to, during, and after mastectomy as well as how to manage complications arising within irradiated tissues. It also provides comparisons and outcome studies of radiation effects on implant- and autologous-based reconstruction. The timing of radiation therapy in relation to reconstruction is also discussed.
Key Teaching Points
Radiation affects the outcome of all forms of reconstruction.
Prepectoral implant-based reconstruction tolerates radiation better than submuscular techniques.
Radiation therapy generally causes shrinkage and increased fibrosis in autologous reconstructions.
Single-stage direct-to implant reconstructions perform better than two-stage expander–implant reconstructions when exposed to radiation.
Implant-based and expander–implant-based reconstructions are a reasonable starting point for reconstruction in patients facing potential radiation therapy.
Increasing numbers of patients are being treated with postoperative radiation, including early-stage breast cancers.
Autologous reconstruction should probably be reserved as a salvage option rather than subjected to the negative effects of radiation therapy in immediate reconstruction.
46.1 Introduction
Radiation therapy has been an integral part of breast cancer management for almost 70 years. Its use spread rapidly, peaking in the late 1970s, then began to decline as complications and a failure to demonstrate improved breast cancer survival became apparent. Concomitantly, Veronesi and colleagues demonstrated that breast conservation incorporating radiation therapy yielded survival rates equivalent to mastectomy alone, stage for stage, ushering in a new era of indications for breast radiation. In 1997 two key papers published in the New England Journal of Medicine documented a significant improvement in survival for premenopausal patients with node-positive breast cancers who received radiation therapy, suggesting that this modality should be used more widely in breast cancer management as a whole. The impact of these studies for oncologists as well as plastic surgeons has been profound.
In this chapter we will discuss these issues and propose an algorithm for management.
46.2 Current Concepts in Radiation Management of Breast Cancer
Radiation therapy used in conjunction with breast conservation therapy had been steadily gaining ground since Veronesi’s published data demonstrated equivalent survival to mastectomy without radiation stage for stage. However, radiation therapy as an adjunctive treatment for breast cancer had been on the decline during the 1980s because of a complete lack of data to support its benefit in improving patient survival, particularly in early breast cancers. The meta-analysis data of 15,000 patients from the Early Breast Cancer Trialist’s Collaborative Group revealed that postmastectomy radiation therapy reduced locoregional recurrence rates in node-positive women from 27 to 9%. Although this appeared significant, the 20-year survival rates for women after a mastectomy, whether treated with radiation or not, was unchanged at 36 to 37%. The disparity was from increased mortality in the radiated group from non–cancer-related causes, including cardiac deaths arising from atherosclerosis, leading to myocardial infarction and cardiac failure. Pulmonary fibrosis contributed to complication rates, as did chest wall necrosis. Therapy tended to be confined to the chest wall and axilla.
In 1997, the landscape changed entirely. Two papers published in the New England Journal of Medicine, one from Overgaard and colleagues in Denmark and the other from Ragaz and colleagues in British Columbia, documented a statistically significant improvement in patient survival with node-positive disease. Both studies invoked the use of treatment to the breast, internal mammary node chain, axilla, and the supraclavicular nodal basin. This represented a significant addition of treatment fields not used routinely in earlier protocols and provided greater cardiopulmonary protection with better planning. The Danish patients had stage II and III disease, whereas the Canadian cases were drawn from patients with stage I and II disease. In these studies, the improved survival at 15 years was approximately 8%. These studies set the stage for a dramatic increase in the use of postmastectomy radiation in patients with four or more positive lymph nodes.
Indications for Radiation Therapy
Oncologists in the United States unequivocally support the use of radiation therapy in the following groups of patients:
Patients with advanced tumors (stage III).
Patients with four or more positive lymph nodes.
More data are emerging about the role of radiation therapy in patients with the following:
Stage I and II disease.
Patients with one to three positive lymph nodes.
Given the Danish and Canadian data, why is there debate concerning these patient groups? Part of the answer lies in the fact that the addition of radiation therapy to the internal mammary chain as well as to the supraclavicular axillary axis is labor intensive and is not performed routinely in many centers. Its use may increase the complications of brachial plexopathy and lymphedema if administered rigorously. Some institutions are already routinely administering radiation therapy to women with any node-positive disease, but not all are rigorously applying Overgaard’s and Ragaz’s recommended treatment fields. More data are being sought in the interim to clarify these issues.
46.3 Radiation: Mode of Action and Tissue Response
A basic understanding of the mode of action of radiation and its effects on tissues and wound healing is essential to reconstructive surgeons working with irradiated tissues.
Ionizing radiation acts directly on the cell nucleus, the DNA of actively replicating cells, chemically damaging and changing it, and thereby preventing its replication.
Radiation therapy for cancer is based on the premise that cancer cells replicate more actively than normal cells, resulting in a higher probability that the DNA will be damaged by this therapy.
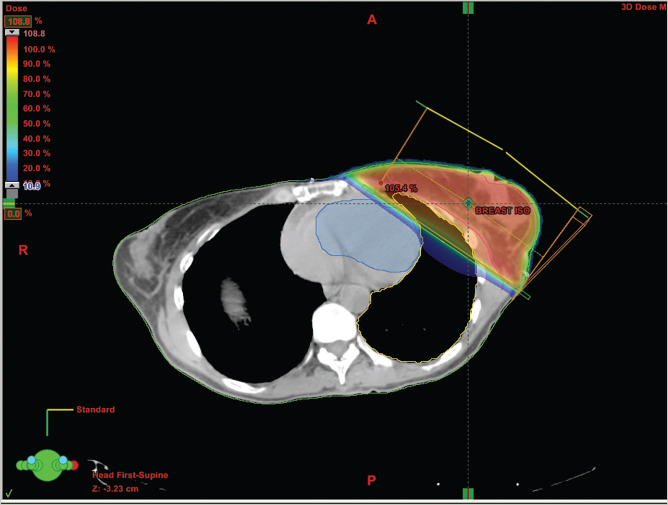
Radiation ionizes the bonds in a cell’s water molecules, resulting in the release of hydroxyl radicals, superoxide radicals, and toxic oxygen species that damage DNA in malignant cells. This not only limits a cell’s ability to replicate but also appears to compromise tissue integrity by altering the cytokine-mediated flow of information between cells. Chronic stimulation of transforming growth factor-β (TGF-β) has been suggested as one of the causes of late fibrosis in irradiated tissue. Massive amplification of the cytokine cascade and the presence of quantities of cellular debris appear to result in cellular destruction. The tumor dosage, port locations and inclination, and fractionation, as shown, are determined to maximize delivery of radiation to the tumor while sparing normal tissue (▶Fig. 46.1).
46.4 Effects of Radiation Therapy
Despite the documented therapeutic benefits of radiation therapy, it is also a source of potential problems. External beam fractionated therapy, intraoperative radiation therapy, and brachytherapy have all contributed tremendously to the management of breast cancer patient survival. Reconstructive surgeons have been faced with a variety of radiation-associated complications since the first application of adjunctive radiation therapy to prevent local recurrence of breast cancer. Radiation therapy may be associated with fibrosis, atrophy, breast necrosis, chest wall radionecrosis, accelerated coronary atherosclerosis, brachial plexus pain and paresis, lymphedema of the arm, and neoplasia. The incidence of these sequelae is decreasing as technology and planning improve and as the breast management team has gained more experience in working with irradiated tissue and with partial mastectomy deformities. Tangential planes of treatment with careful field planning have gone a long way toward reducing the impact of radiation therapy on the heart and lungs, as well as decreasing the incidence of osteoradionecrosis. Brachial plexopathy is also being seen less, although the increased use of axillary to supraclavicular fields of treatment may resurrect this problem.
The complications associated with mastectomy versus breast conservation therapy are as follows:
Complications
Mastectomy:
Fibrosis.
Atrophy.
Breast necrosis.
Osteoradionecrosis.
Lymphedema.
Atherosclerosis.
Breast Conservation Therapy:
Angiosarcomatous change.
Brachial plexopathy.
Fibrosis.
Atrophy.
Radiation of normal tissues is inevitable during radiation therapy; unfortunately, radiation therapy causes damage to these normal tissues as well as the compromised ones, although the damage is not as profound. The earliest and most noticeable effects of irradiation are evident in the skin’s basal cell layer, which is the most mitotically active.
Immediate local effects of irradiation include erythema, desquamation, and rarely, vesicle formation and ulceration. Immediately after radiation therapy the breast is edematous and erythematous; the erythema gradually resolves over a few months, and the area may eventually have a tanned appearance. Breast edema can persist for several years (▶Fig. 46.2).
As edema subsides, the treated breast may become smaller or less ptotic than the normal breast, as can be seen in this woman 3 years after breast conservation therapy with left breast irradiation. If a significant portion of the breast has been removed, this is particularly noticeable after regression of the edema. Healing after local excision with internal derangements can affect the external breast appearance and shape. Decreased sweating, dryness, and loss of hair growth and normal skin lubricants characterize injury to the skin adnexa (▶Fig. 46.3).
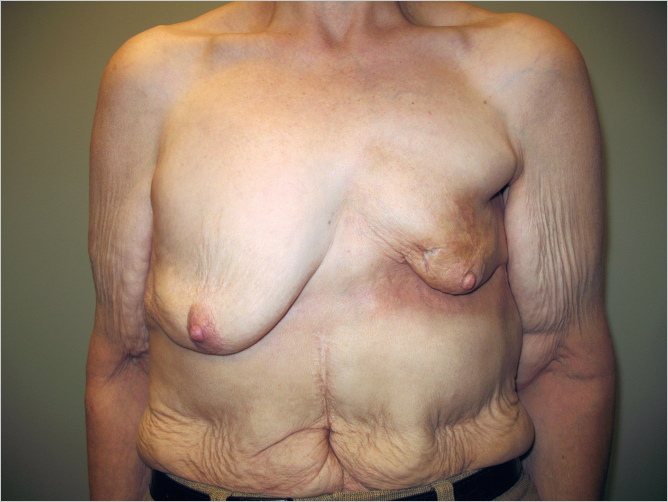
Because the intima and media of blood vessels replicate slowly, the effects of radiation are manifested as a gradual and relatively delayed occlusion of these vessels with an absolute reduction of vascularity to the treated area (▶Fig. 46.4).
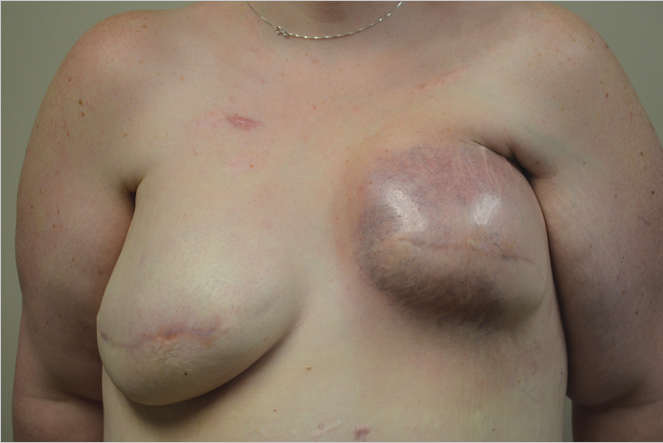
Telangiectasia within the treated field increases over time. The subcutaneous tissues may also be damaged, with fibrotic, atrophic tissue replacing the normal fat and areolar structures, resulting in gradual but progressive breast fibrosis and contraction. These features are clearly illustrated in the young woman shown in ▶Fig. 46.4, who presented 18 months after poorly administered radiation therapy to a submuscular implant. Note the severe contracture, distortion, thinned skin, and telangiectasia from radiation damage.
Radiation can have multiple, persistent, and progressive effects on tissues. Many of these sequelae appear only after many years or even decades. Proper sequencing by the radiologic oncologist will help minimize these after effects.
46.5 Implants and Their Impact on Radiation Delivery
Radiation therapy delivers linear beams of ionizing radiation to a given target. Given that the breast and chest wall are curved, multidimensional structures, radiation beams have to be focused in such a way as to cross the plane of treatment tangentially with the most minimal scatter to the underlying great vessels, heart, and lungs.
Technical Pearl
Radiation oncologists prefer the flattest target possible, the ideal being an unreconstructed mastectomy defect. Implants distort the target field for planning, resulting in potentially greater radiation scatter to adjacent critical structures.
The larger the breast all the more prominent in the implant reconstruction, the more difficult it is to plan radiation to provide adequate saturation of the target tissues without too much scattered to the adjacent uninvolved tissues. The steeper the upslope of the medial breast border, the more difficult the planning becomes. Similarly, if there is a gentle taper of the implant off the chest wall, the easier planning becomes.
46.6 The Dilemma of Implants and Their Impact on the Cold Triangle
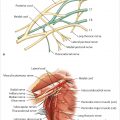
Based on the Danish and Canadian studies published in the 1990s, greater emphasis has been placed on targeting the adjacent nodal basins in addition to the tangential field across the breast. The angle subtended between the internal mammary treatment beam and that across the breast and chest wall is known as the cold triangle. If this angle changes too radically due to the placement of implant or expander, it can cause inadequate dosage delivery to important treatment areas (▶Fig. 46.5).
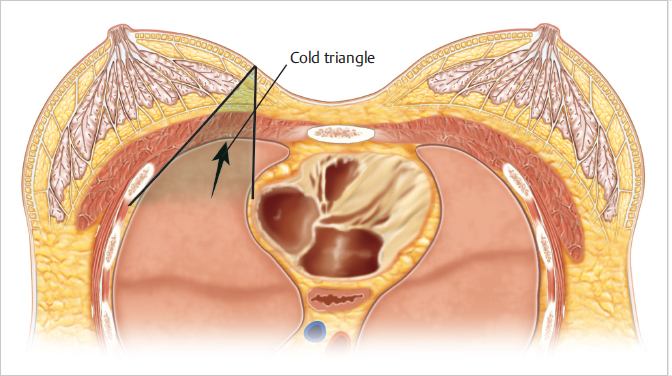
These problems gave rise to the MD Anderson delayed–immediate expander protocol, which will be discussed later in this chapter. We have done some preliminary work, which suggests that when compared with a conventional expander or high-profile, round smooth implant, that the tapered, gentler profile of an anatomical implant appears to allow for slightly easier planning of radiation dosimetry. This remains to be confirmed in a larger study.
It is helpful to approach the patient who has undergone or will be having radiation therapy in a systematic fashion, with treatment plans specifically tailored to the problem.
Radiation Treatment Categories
Patients with failed breast conservation therapy and recurrence.
Patients undergoing mastectomy and immediate reconstruction followed by radiation therapy.
Patients undergoing mastectomy and radiation followed by delayed reconstruction.
Patients who have had radiated implants now experiencing complications.
46.6.1 Patients with Failed Breast Conservation or Recurrence
Breast conservation therapy may fail due to any of the following factors:
Local recurrence requiring mastectomy.
Severe asymmetry.
Ulceration and pain.
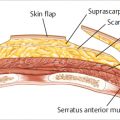
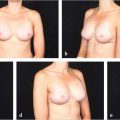
Once the skin has ulcerated or the patient is suffering from intractable pain secondary to radiation neuritis, mastectomy and autologous reconstruction is the obvious choice. The patient shown in ▶Fig. 46.6 illustrates the problem of skin flap necrosis induced by a boost to the upper medial part of the breast following mastectomy with previous transverse rectus abdominis myocutaneous (TRAM) flap reconstruction. Most cases of severe asymmetry develop this problem because the skin envelope has shrunk so badly that it is essentially unusable for further reconstruction without autologous intervention as well as seen in ▶Fig. 46.7. All of this damaged skin requires excision and replacement with healthy nonradiated tissue.
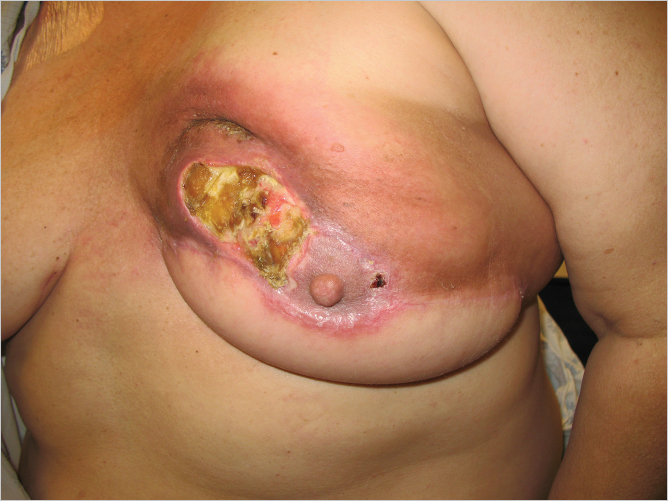
In a patient who has undergone prior breast conservation therapy, and then develops a local recurrence amenable to mastectomy, the question becomes one of whether the mastectomy skin envelope can be utilized safely for implant-based reconstruction as opposed to autologous reconstruction. When examining the patient preoperatively, if the skin appears soft with little to no visible sequelae of radiation therapy, it may be reasonable to proceed with either direct-to-implant or expander-based reconstruction.
Technical Pearl
This would be predicated on the viability of the skin using skin perfusion assessment devices intraoperatively.
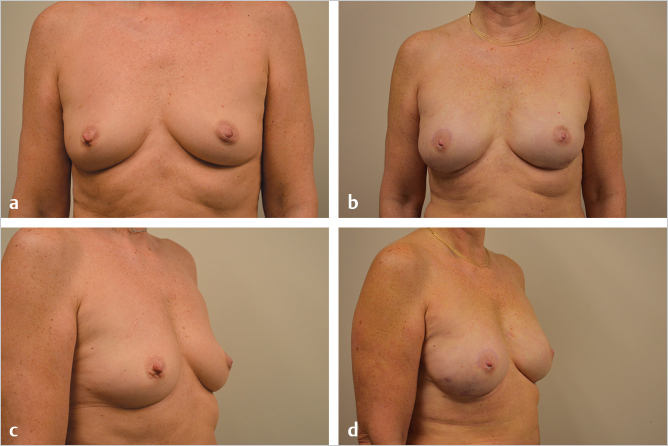
The patient illustrated in ▶Fig. 46.8 shows no obvious impact of prior radiation therapy on the breast and underwent successful single-stage reconstruction due to the adequacy of diffusion assessment intraoperatively. She is shown 1 year following the procedure. Shape and symmetry are excellent.
46.7 Patients Undergoing Mastectomy and Immediately Construction Followed by Radiation Therapy
Most patients presenting to the plastic surgeon fall into this category. They will be undergoing either skin- or nipple-sparing mastectomy with possible postoperative radiation therapy in the treatment plan, based on tumor size and nodal status. In this patient population three possible options are available:
Mastectomy with immediate expander or implant placement followed by radiation therapy if required.
Mastectomy with immediate autologous reconstruction followed by radiation therapy if required.
Mastectomy followed by radiation therapy with delayed reconstruction.
In my hands, option 2 has never been a particularly good one, for the simple reason that most postoperative radiation therapy causes a significant impact on autologous reconstruction resulting in long-term deterioration of the aesthetic result. Although a number of authors have found radiation therapy in their locations to be acceptable with relatively limited impact on autologous reconstruction, this has not been the case in any of the major breast centers in which I have worked. Nearly, all of the autologous reconstructions that I have seen radiated postoperatively have developed significant fibrosis and shrinkage within 5 years.
Technical Pearl
In my opinion, an autologous reconstruction is far too valuable a piece of tissue to lose to radiation fibrosis. I would rather risk radiating an implant-based reconstruction and then convert it to an autologous one in the future if it becomes necessary.
Option 3: Mastectomy with postoperative radiation and subsequent reconstruction commits the patient with almost 100% certainty to an extensive autologous reconstruction.
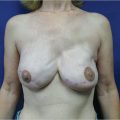
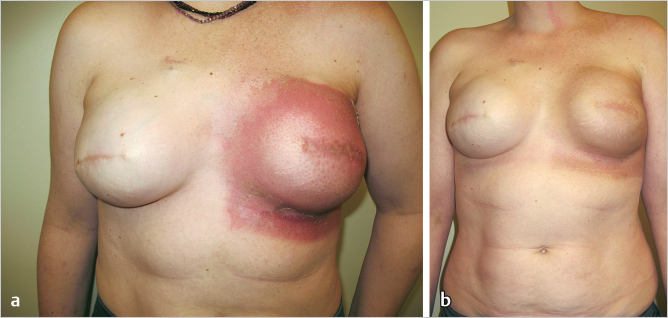
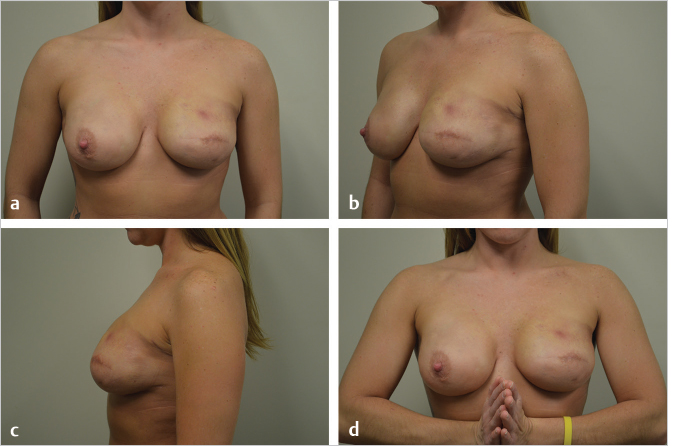
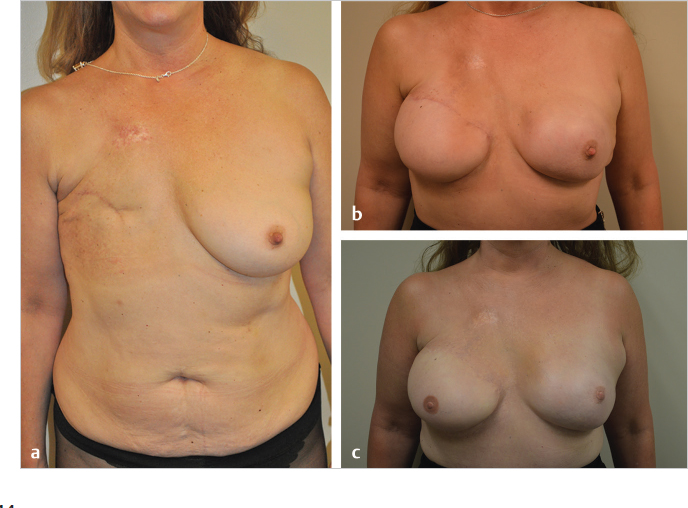
For these reasons option 1 is my personal preference whenever radiation therapy is potential therapeutic modality to be used in the future. Prepectoral implant reconstructions covered with acellular dermal matrices (ADMs) tend to do better than their subpectoral counterparts and may avoid autologous conversion for many years, if not permanently. The patient shown in ▶Fig. 46.9 illustrates the reasonable outcome at 2 years following radiation therapy to a prepectoral direct-to-implant reconstruction. She has no animation deformity and shape and symmetry are quite good.
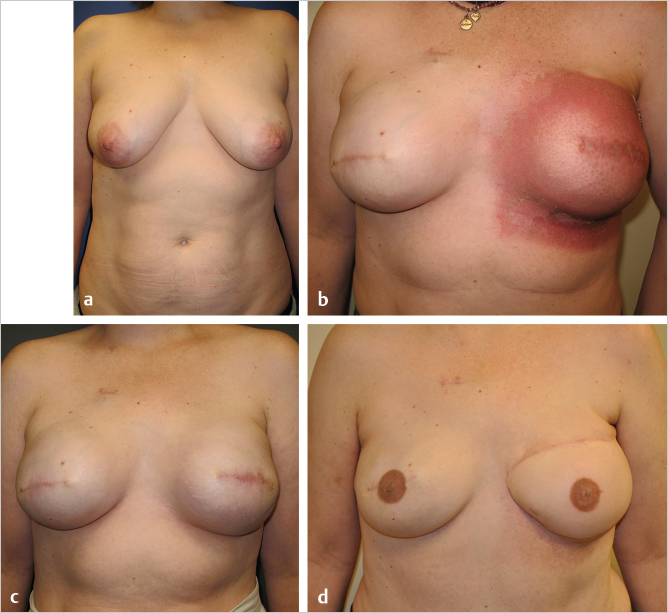
By contrast, the patient in ▶Fig. 46.10 was treated with subpectoral direct-to-implant reconstruction before the era of prepectoral reconstruction. She was radiated and did fairly well for 3 years but developed progressive tightening of her radiated pectoralis major muscle with shoulder discomfort and chest wall tightness. She was converted to a left deep inferior epigastric perforator (DIEP) flap and is now comfortable.
Technical Pearl
This difference in outcome between radiation of a subpectoral implant versus a prepectoral implant is quite striking. The patients with prepectoral reconstructions tend to have relatively minimal shoulder stiffness when compared to their subpectoral counterparts.
The question as to whether single-stage direct-to-implant versus two-stage expand–implant reconstruction fares better with radiation has been answered into studies, one by Colwell in Boston and the other by Nava in Milan.
Technical Pearl
Both studies demonstrated that in immediate reconstruction, expander-based reconstructions had a five- to sixfold increased risk of complications from radiation when compared with single-stage direct-to-implant approaches.
46.8 Patients Undergoing Mastectomy Followed by Radiation with Delayed Reconstruction
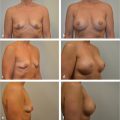
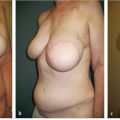
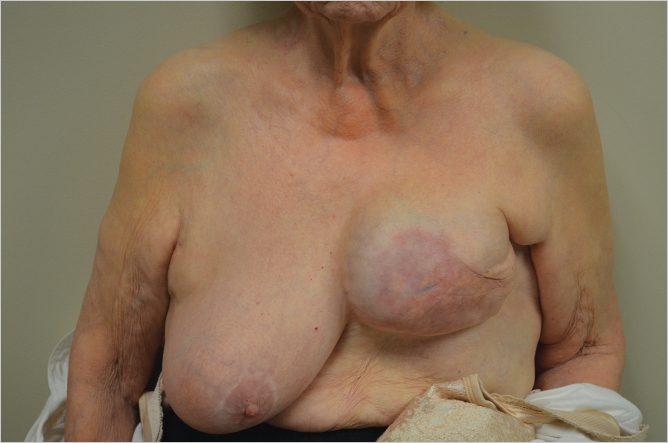
Decision making in this patient subset is easy. Most of these patients will have a relatively tight chest wall after radiation therapy has exerted its effect, rendering the skin stiff and poorly compliant. This does not allow for successful expanding insertion in most cases and even if expanders can be inserted, the amount of expansion tends to be much more limited compared with nonradiated patient. Spear also showed in an extensive series that reconstructing radiated patients created a high percentage of patients requiring salvage. Unless the skin is remarkably healthy after radiation therapy, I offer almost all of these patients delayed autologous reconstruction using abdominal tissue if available. The patient shown in ▶Fig. 46.11 underwent right mastectomy and radiation therapy 20 years previously. She requested reconstruction and an autologous approach with unipedicle TRAM flap was performed with an extremely successful outcome. She is an accomplished equestrian and hunter and was fully functional after surgery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree