39 Prepectoral Breast Reconstruction: Direct-to-Implant versus Two-Stage
Summary
Prepectoral breast reconstruction has made significant comeback in recent years. Two approaches are available: single-stage direct-to-implant versus more traditional two-stage expander implant reconstruction.
Key Teaching Points
Reconstructive prepectoral contraindications: poorly perfused flaps, HbA1c > 7.5, smokers.
Oncologic prepectoral contraindications: inflammatory breast cancer, deep tumors with muscle involvement.
Candidates for direct to implant: small breasts (A cup–C cup), minimal ptosis (grade 1 or 2), no prior radiation.
Be prepared for two-stage reconstruction even if direct to implant is planned. Final determination is made intraoperatively following tissue perfusion assessment.
39.1 Introduction
Prepectoral implant-based breast reconstruction is in many ways an “ideal” reconstructive strategy. Compared with subpectoral breast reconstruction, the current standard, prepectoral reconstruction is anatomically less invasive (pectoralis muscle sparing), technically less challenging, less time consuming, associated with less pain, reduces the requirement for intravenous pain management, eliminates animation deformity, and results in a more natural breast shape. Not surprisingly, this mode of breast reconstruction is rapidly gaining popularity. Careful patient selection is, however, critical for achieving successful outcomes as in the subpectoral approach.
In a previous publication, recommendations regarding reconstructive and oncologic indications/contraindications for prepectoral reconstruction were provided.
Technical Pearl
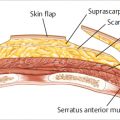
From a reconstructive standpoint, well-perfused viable mastectomy skin flaps are an absolute requirement.
Patient characteristics/comorbidities that compromise skin flap vascularity, such as tobacco use, uncontrolled diabetes (hemoglobin A1c [HbA1c] > 7.5%), and prior breast irradiation (unless a latissimus flap muscle is utilized) can increase the risk of failure in this procedure. Good candidates for the prepectoral approach include the following:
Normal body mass index (BMI).
Minimal comorbidity.
Well-perfused mastectomy flaps.
Obesity, per se, is not a contraindication for implant-based breast reconstruction—both subpectoral and prepectoral. Immediate prepectoral reconstruction may be precluded in obese patients in the presence of other comorbidities such as uncontrolled diabetes or tobacco use, which are contraindications for the prepectoral approach. As diabetes is highly prevalent among obese patients, we routinely assess HbA1c levels in these patients prior to surgery. Immediate reconstruction is not offered if HbA1c is greater than 7.5% and patients are advised to undergo medical therapy to control their blood glucose level before reconstructive surgery.
Once the decision is made that the patient is prepectoral candidate, then a decision has to be made about the candidacy of the patient for a direct-to-implant approach.
The ideal candidate for direct to implant is a patient with small breasts (A–C cup) and minimal ptosis (grades 1 and 2). Once this candidacy is assessed, then the final eligibility of patients for immediate direct-to-implant prepectoral reconstruction is determined intraoperatively following mastectomy.
Technical Pearl
Flap thickness is not a criterion for the prepectoral approach; rather, skin perfusion determines eligibility.
Thus, objective assessment of skin perfusion using a tissue perfusion system is critical and required in these cases. If a tissue perfusion system is not available, clinical assessment of skin viability should be performed before proceeding with the prepectoral approach. Visible dermis on the underside of the skin flap is a contraindication for any reconstruction as it is too tenuous.
The detailed operative techniques of both direct-to-implant and expander-based reconstructions have been described in Chapter 40 and Chapter 42.
When creating the prepectoral pocket for both direct-to-implant and expander-based reconstructions, meticulous attention is focused on eliminating dead space to prevent seroma formation. Seroma formation is a concern in the prepectoral approach as in the subpectoral approach when using acellular dermal matrices. Dead space is eliminated as much as possible by repositioning excess subcutaneous tissue to fill the spaces and collapse the pocket.
Challenging patients generally are not good candidates for direct-to-implant reconstruction. One example is obese patients. Obesity, defined by a BMI > 30 kg/m2, is a global epidemic. In the United States, more than one in three individuals are obese. With the widening girth of the U.S. population, breast reconstructive surgeons are likely to encounter an increasing number of obese patients. These patients present a number of challenges to reconstructive surgeons, including longer operative times, technical difficulties associated with mammary hypertrophy, and an increased risk of postoperative complications, including seroma, compromised wound healing, infection, and reconstructive failure. BMI, in fact, is a powerful predictor of complications.
Technical Pearl
Every unit increase in body mass index increases the odds of complication by 5.9% and reconstructive failure by 7.9%.
The technical difficulty of performing prepectoral reconstruction in obese patients relates to the management of skin flap redundancy due to mammary hypertrophy. Management of skin redundancy begins with skin-reducing mastectomy via an inverted-T incision or extended transverse/oblique skin excision. Following mastectomy, objective assessment of flap perfusion is performed to ensure adequate flap viability for prepectoral reconstruction. Flap vascularity should not be assumed by the thickness of the flaps.
Redundant flaps are utilized as much as possible in reconstructing the new breast. Redundant flaps are excised only if they are poorly vascularized. After device placement and dermal matrix coverage, redundant medial and lateral flaps are deepithelialized. The resulting dermal and subcutaneous fat layers are arranged over the acellular dermal matrix to provide additional soft tissue coverage. The flap layer also helps prevent device exposure in the event of incisional dehiscence.
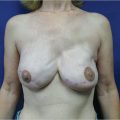
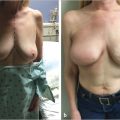
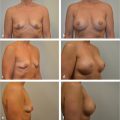
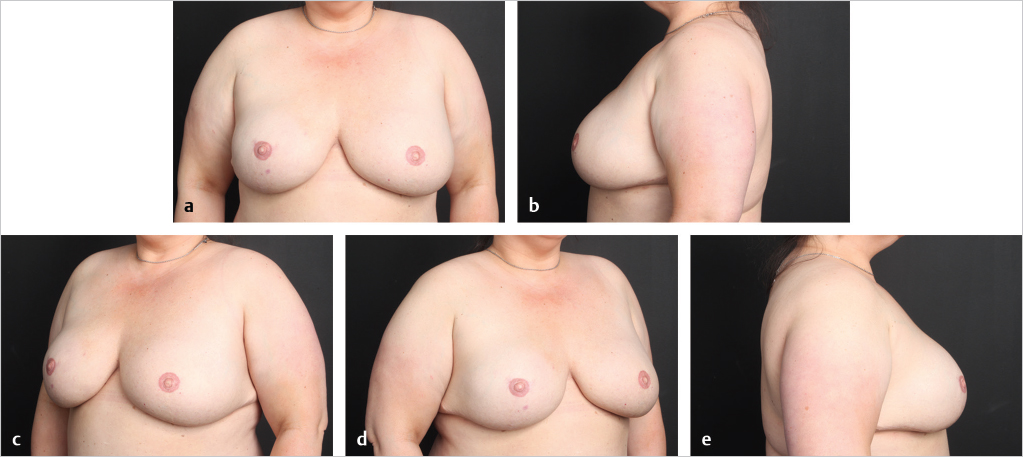
Direct-to-implant reconstruction is rarely performed in obese patients, despite having adequate subcutaneous tissue (▶Fig. 39.1, ▶Fig. 39.2, ▶Fig. 39.3, ▶Fig. 39.4, ▶Fig. 39.5, ▶Fig. 39.6, ▶Fig. 39.7). Poor breast projection and contour problems are common in these patients and the second stage provides the opportunity to make adjustments to improve the overall aesthetic outcome. Despite a thick subcutaneous layer, fat grafting may still be required in these patients, particularly in the upper poles to create a smoother transition.
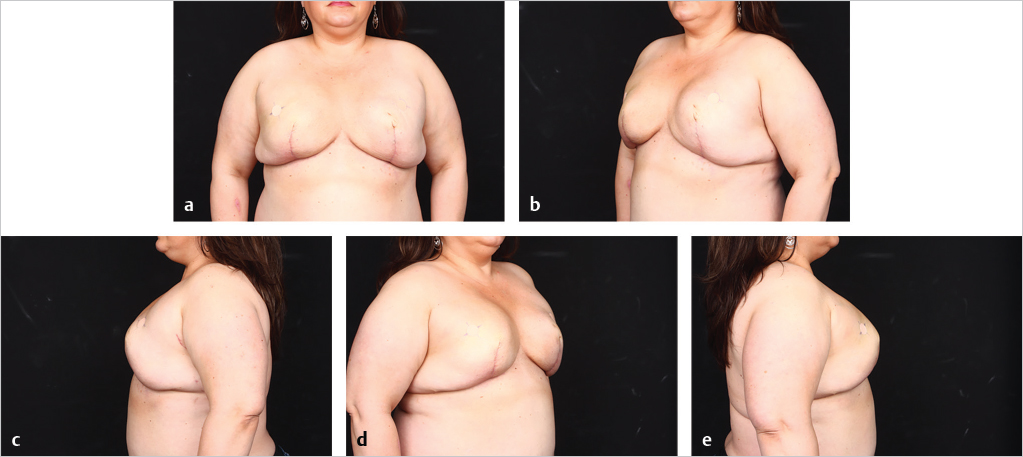
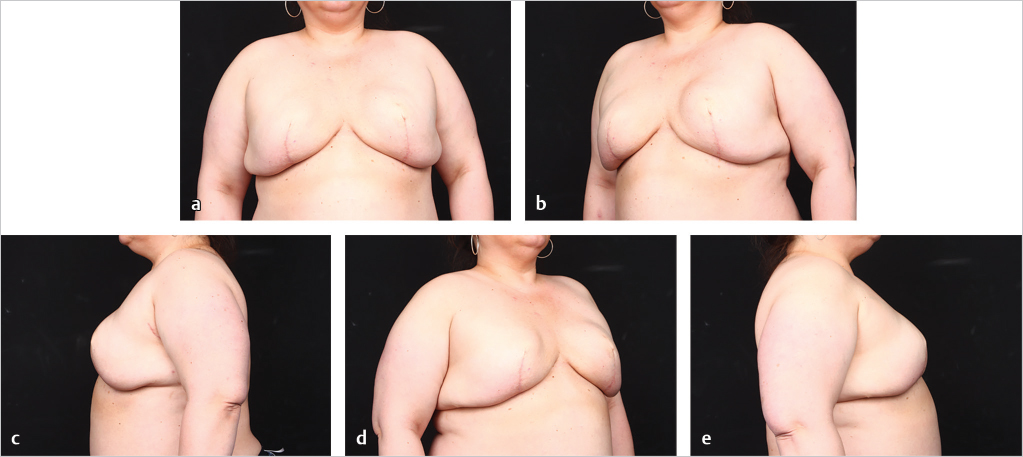
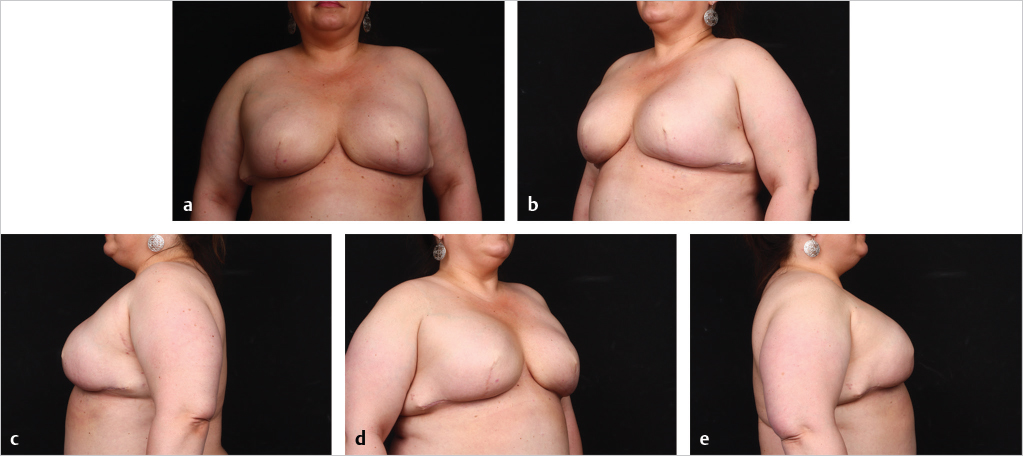
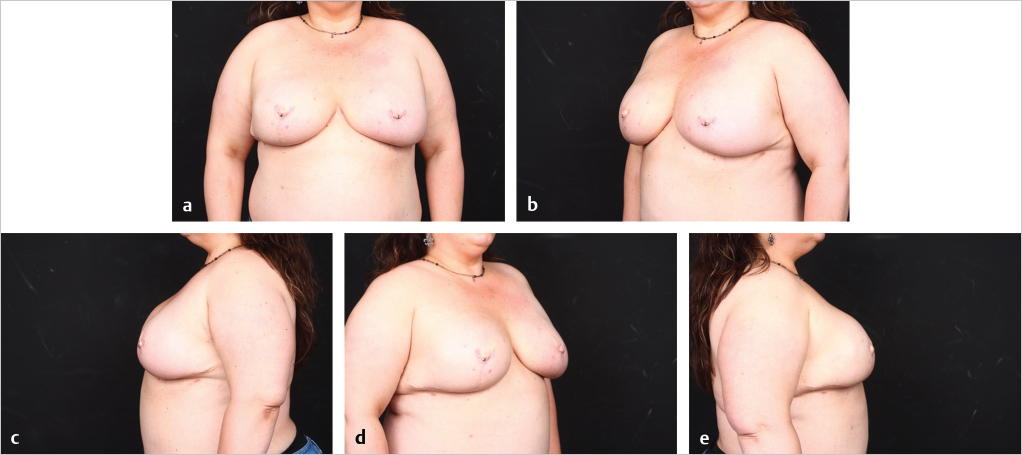
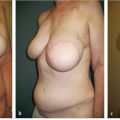
In cases where direct-to-implant reconstruction is not utilized, tissue expanders, when used, are inflated intraoperatively with saline or air (off-label) to 50 to 70% of capacity depending on skin flap perfusion. Underfilling prevents undue tension on the overlying mastectomy skin flap, which can compromise flap perfusion. Delicate flaps, in particular, are more sensitive to compression than thick flaps. Objective reassessment of flap perfusion after intraoperative expansion ensures perfusion is not compromised. Underfilling of expanders also ensures the future implants will fit snugly into the prepectoral pockets. For this reason, slightly smaller expanders are selected with a base width that is 0.5 to 1.5 cm smaller than the future implants. Underfiling, however, should not compromise the elimination of dead space, which is critical for the prevention of seroma development, or the apposition of the acellular matrix to the skin flap, which is critical for the revascularization of the matrix. Direct apposition of the skin to the acellular matrix facilitates neovascularization and recellularization of the matrix, permitting its incorporation into the surrounding subcutaneous tissue. Direct apposition also ensures the elimination of dead space and reduces the risk of seroma. The use of perforated acellular matrix may further facilitate its regeneration and drainage from under the matrix.
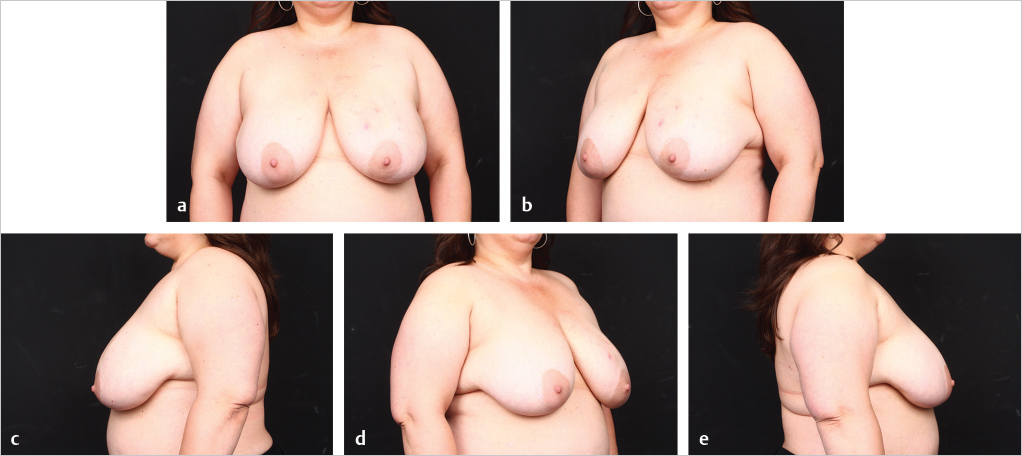
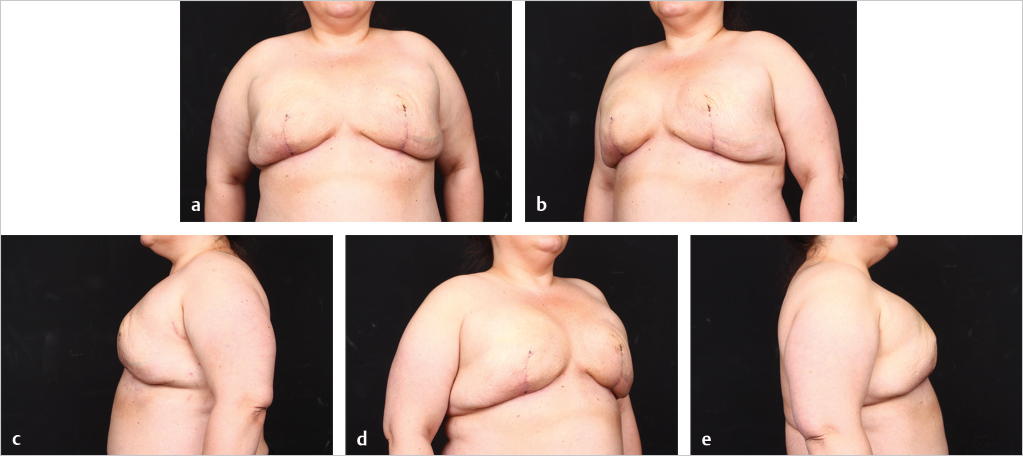
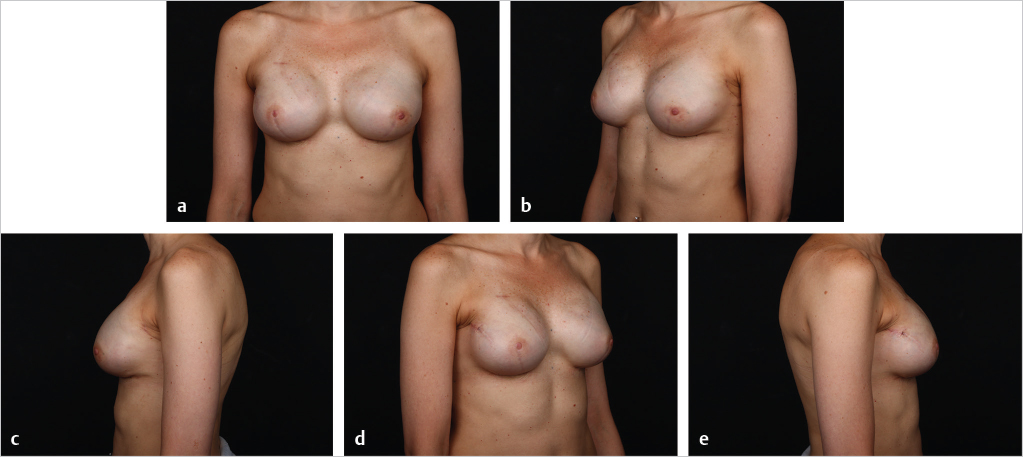
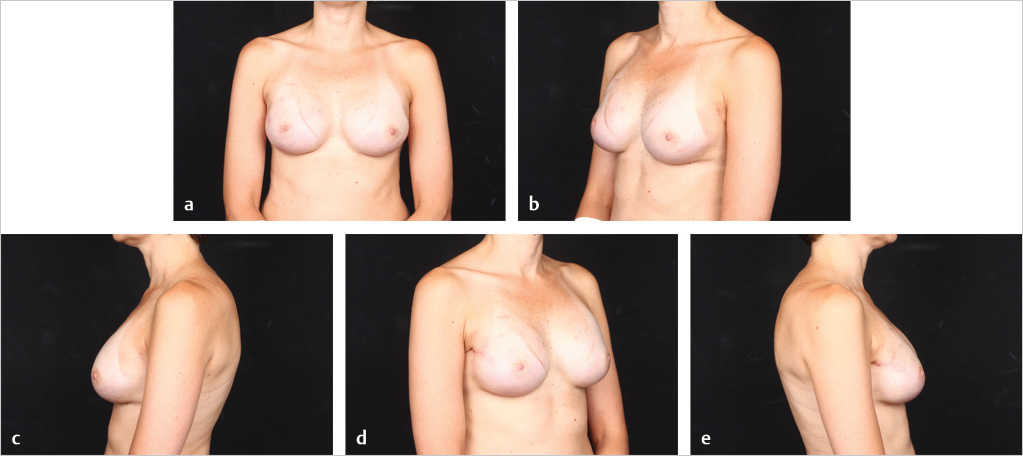
Fat grafting is sometimes needed in the prepectoral setting to enhance breast volume; mask implant palpability, visibility, and rippling/wrinkling; and improve functionality of the reconstructed breast (facilitates gliding of the subcutaneous pocket) and the overall aesthetic outcome. Fat grafting is usually performed as a separate stage. Risk of rippling is discussed with thin patients prior to surgery and patients often prefer the avoidance of animation deformity with the prepectoral approach at the expense of some rippling (▶Fig. 39.8, ▶Fig. 39.9, ▶Fig. 39.10, ▶Fig. 39.11, ▶Fig. 39.12, ▶Fig. 39.13, ▶Fig. 39.14).
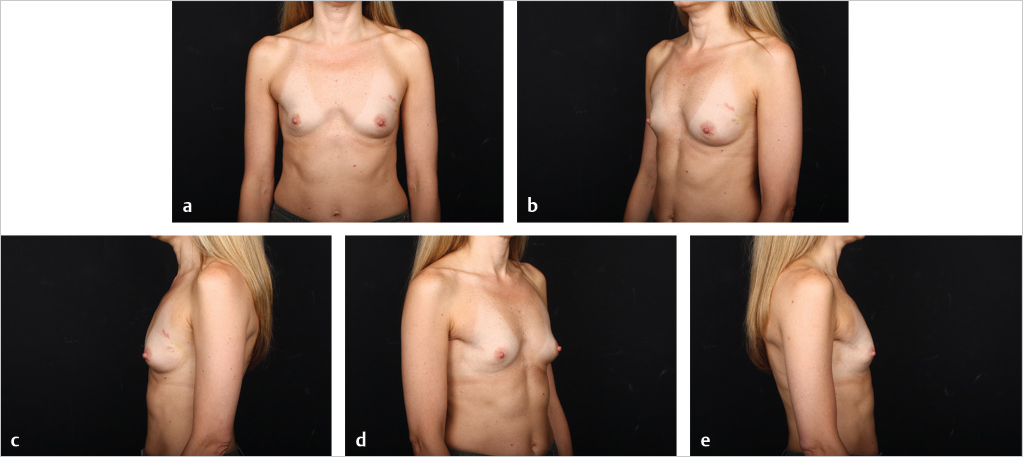
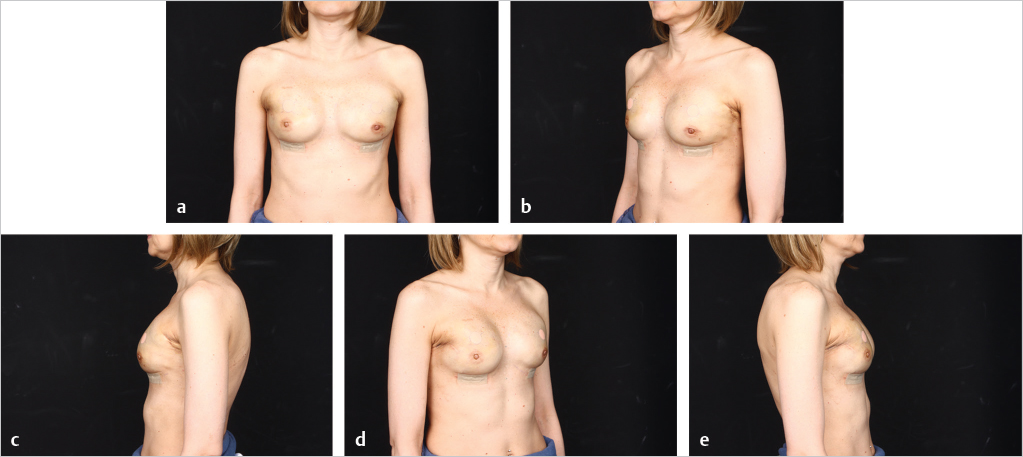
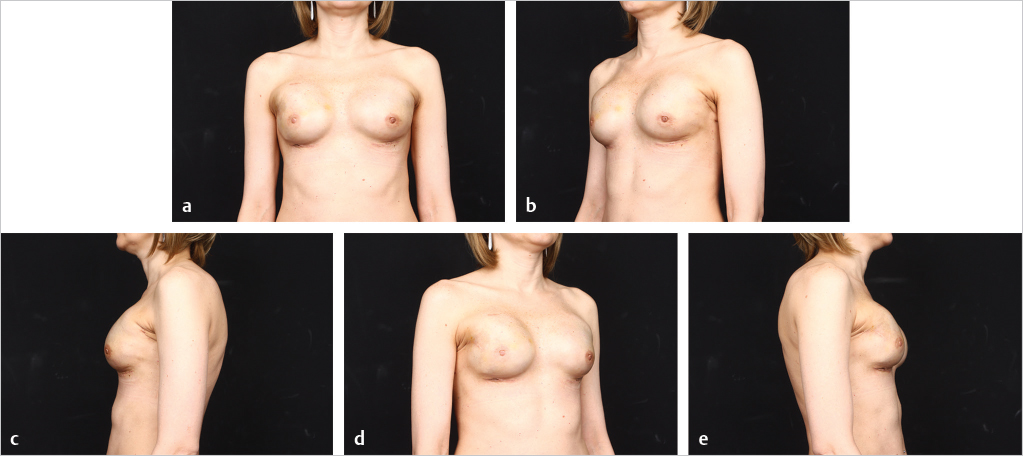
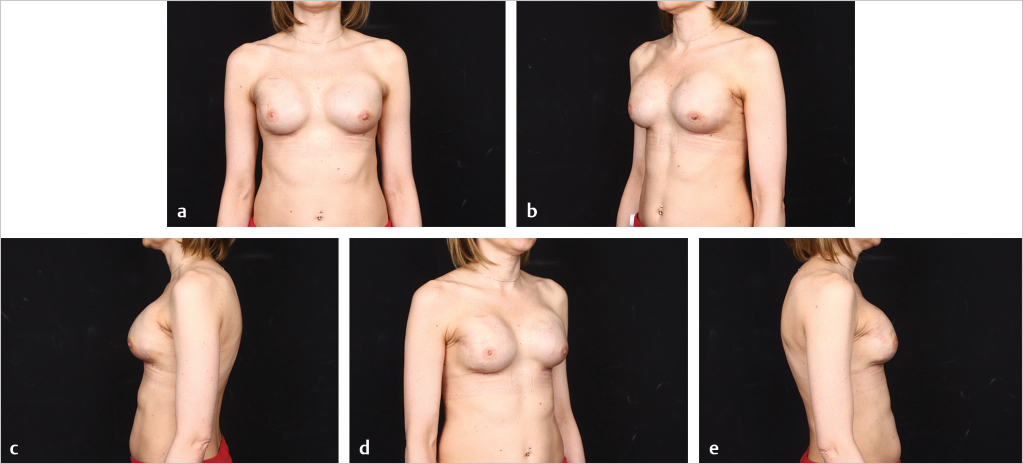
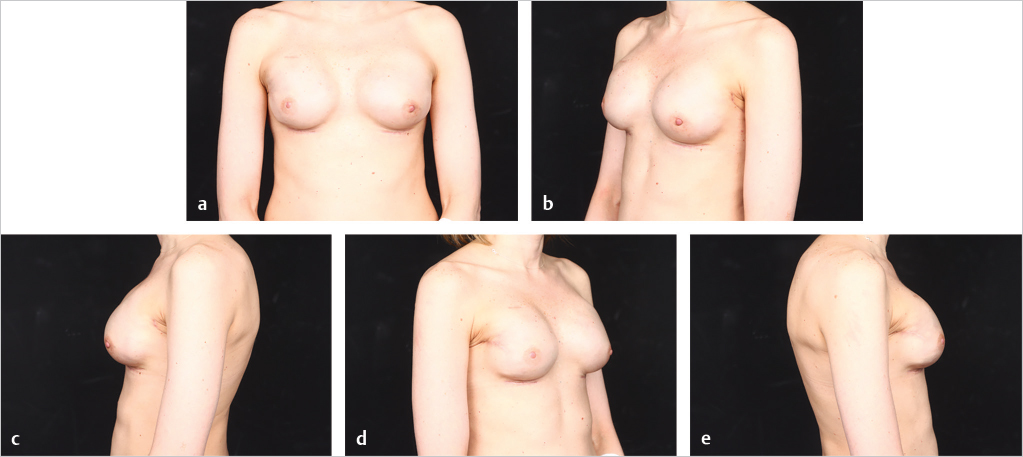
The author’s’ experience with prepectoral expander/implant reconstruction in obese patients involves 39 patients, including 18 with a BMI ≥ 35 kg/m2 and 12 with a BMI ≥ 40 kg/m2. A total of 73 breasts were reconstructed. Complications occurred in 12 breasts and included incisional dehiscence (4 breasts), skin necrosis (5 breasts), fat necrosis (2 breasts), seroma (2 breasts), infection (1 breast), and hematoma (1 breast). Three of the complications required return to the operating room for corrective surgery. There was one reconstructive failure with conversion to a latissimus flap. Aesthetic outcome was good to acceptable in all patients (▶Fig. 39.8, ▶Fig. 39.9, ▶Fig. 39.10, ▶Fig. 39.11, ▶Fig. 39.12, ▶Fig. 39.13, ▶Fig. 39.14). These results are comparable to, if not better than, those reported with subpectoral reconstruction in obese patients. Employing a similar technique of skin reduction mastectomy followed by prepectoral reconstruction using an acellular matrix in conjunction with a dermal flap, Caputo et al have also reported low complication rates in patients with large, ptotic breasts.
One of the most difficult patient populations that we as plastic surgeons are faced with are radiated patients. Radiotherapy is an essential treatment modality for breast carcinoma. Ionizing radiation, however, is associated with short- and long-term sequelae, which may adversely impact implant-based breast reconstruction. Acutely, irradiation causes direct damage to the skin, underlying soft tissue, blood vessels, and nerves. The influx of inflammatory cells and macrophages following tissue damage leads to erythema, desquamation, and ulceration of the skin and soft tissue. The ensuing fibroblast, including myofibroblast, activation, and proliferation results in the deposition of fibrous tissue. Radiation-induced fibrosis is a chronic process, which progressively leads to dermal thickening and chest muscle fibrosis and atrophy. Muscle fibrosis/atrophy impairs mobility, causing tenderness and spasms. In the context of implant-based breast reconstruction, the acute effects of radiation are manifested as incisional dehiscence, infection, delayed healing, seroma, and hematoma, while the effects of radiation-induced fibrosis are manifested as capsular contracture, implant malposition/asymmetry, and reconstructive failure. Elevation of the pectoralis major muscle for subpectoral breast reconstruction in the setting of radiotherapy further compromises the muscle and its function and in retrospect is probably not a good idea.
All published literature on breast reconstruction in combination with radiation does not include the duration, the extent of the field, and the amount of radiation that was applied. In addition, no information on boosting the incisions is mentioned as some centers continue to utilize this concept even though there is no evidence that there are recurrence benefits in mastectomy patients. Given the lack of evidence on correlation between radiation therapy dose, field, and duration and success of reconstruction, immediate prepectoral direct-to-implant, acellular matrix–assisted breast reconstruction is not normally offered to patients in my practice. Immediate expander-based reconstruction is offered to some of the patients and the author utilizes the following criteria. If the lumpectomy scar is in the upper pole, then immediate expander-based reconstruction is a reasonable option in this case if the remaining breast envelope is not damaged and scarred (▶Fig. 39.8, ▶Fig. 39.9, ▶Fig. 39.10, ▶Fig. 39.11, ▶Fig. 39.12, ▶Fig. 39.13, ▶Fig. 39.14). The patient also has to understand that if the reconstruction fails, then a conversion to a latissimus flap or full autologous approach may be needed. On the other hand, when the lumpectomy scar is on the lower pole, where maximal expansion is needed, then the author approaches the reconstruction with an immediate, expander and latissimus-based, in a prepectoral plane. In this approach, the latissimus muscle replaces the acellular dermal matrix for expander/implant coverage and support on the irradiated side. Soft tissue is introduced with the flap in the areas that soft tissue is lacking, otherwise, at times, this is performed as muscle-only flap. Reconstruction is typically performed in two stages. The expander stage is critical to allow for skin and subcutaneous expansion. The lower pole is the most critical area for tissue expansion, and if the scar lies on the lower pole, then expansion will not be successful.
Technical Pearl
PMRT is not a contraindication for prepectoral breast reconstruction; it is actually the author’s preferred method when radiation is planned postoperatively.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree