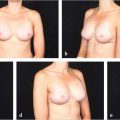
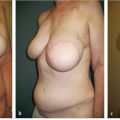
42 Prepectoral Direct-to-Implant Breast Reconstruction
Summary
Single-stage direct-to-implant reconstruction with anterior acellular dermal matrix coverage has recently emerged as an outstanding, safe, and reliable technique for immediate breast reconstruction. It completely eliminates animation deformity and has absolutely no negative impact on upper extremity motion. It provides unparalleled opportunity to create cleavage, which has always been an elusive goal in implant-based reconstruction. It eliminates the need for mandatory second operation and can be performed as an outpatient procedure in select patients.
Key Teaching Points
Single-stage prepectoral direct-to-implant (DTI) reconstruction is a safe procedure.
It prevents animation deformity.
It obviates the need for an expander.
DTI allows significant volume augmentation without expansion if mastectomy skin is well perfused.
Prepectoral DTI is less painful than subpectoral DTI.
It preserves pectoral muscle function.
42.1 Introduction
In 2016, over 245,000 women were diagnosed with breast cancer in the United States. Of these, just over 109,000 women underwent mastectomy with reconstruction; 86,000 of them were treated with prosthetic-based reconstruction, 90% of which were two-stage expander–implant procedures with 10% being direct-to-implant (DTI) approaches. With respect to implant positioned, 83% where subpectoral while 17% were prepectoral.
Implant-based reconstruction has undergone significant changes in the last decade. Three technological advances have ushered in a new era in breast reconstruction:
Improvements in implant design.
Availability of skin perfusion techniques.
Acellular dermal matrices (ADMs).
In addition, there has been the recent focus once more on the prepectoral plane for breast reconstruction.
42.2 The Original Rationale for Subpectoral Breast Reconstruction
When the first Cronin–Gerow gel-filled implants were introduced in 1962, these were used for breast augmentation in the subglandular plane. Subsequently, these were used for breast reconstruction following subcutaneous mastectomy for chronic pain associated with fibrocystic disease. It soon became apparent, however, that complications where significant, including capsular contracture, rippling, severe asymmetry with malposition, deformity, and chronic breast pain.
At that time, the Halsted radical mastectomy had reigned supreme in breast cancer management and it was only as the procedure was altered as modified radical mastectomy to preserve more muscle that alternative techniques of breast reconstruction became feasible.
In breast augmentation, the implant pocket was moved into the subpectoral plane in order to ameliorate the complications of the subglandular approach. This change was ported into the reconstructive arena and subpectoral reconstruction became the gold standard for breast reconstruction.
The move to the subpectoral plane was motivated by several perceived potential benefits.
Benefits
Subpectoral implant positioning appeared to reduce contracture possibly due to constant prosthetic massage by the muscle.
The muscle provided padding over the implant to reduce upper pole rippling.
The tapering effect of the muscle over the upper pole of the implant at its juncture with the chest wall was thought to soften the contour of the upper pole of the reconstruction.
The muscle was thought to provide additional vascularity to the skin flaps.
While capsular contracture rates did appear to drop slightly and implant visibility problems were softened, the subpectoral position introduced a new set of complications to breast reconstructive surgeons.
Complications
Adhesion of the pectoralis major muscle to the overlying mastectomy skin flap causing significant animation deformity.
Tightness across the reconstruction due to tension on the pectoralis major muscle.
Chronic discomfort in the upper chest and shoulder girdle.
Window-shading deformity of pectoralis major.
Implant deformation and displacement during upper extremity exercise.
A profound impact on the ability to create cleavage due to the mechanical barrier of the medial border of the pectoralis major muscle in preventing medial positioning of the implant.
Hollowing of the medial aspect of the reconstruction at the site of inferomedial release of the pectoralis major origin.
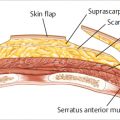
The introduction of total submuscular coverage followed on as a natural progression; if partial muscle coverage was superior to subcutaneous cover, then total muscle coverage must be better! The concept involved raising pectoralis major, the anterior border of serratus anterior as well as the superior origin of the rectus abdominis muscle and its associated fascia in an effort to create a complete submusculofascial envelope to cover the implant. The major benefit to this approach was that if skin necrosis of the inferior flap supervened following mastectomy, muscle present at the base of the wound allowed for granulation and delayed healing to occur. This was a positive development at a time when perfusion assessment was limited to fluorescein injection at best.
The problem with this approach was the intense tightness of the confluence of pectoralis major, serratus anterior, and rectus abdominis creating a point of maximal pressure at exactly the area where the surgeon needed elasticity for lower pole projection (▶Fig. 42.1). The result was loss of lower pole projection, forcing displacement of the reconstruction in a cephalad direction with bulging of the implant below the clavicle (▶Fig. 42.2).
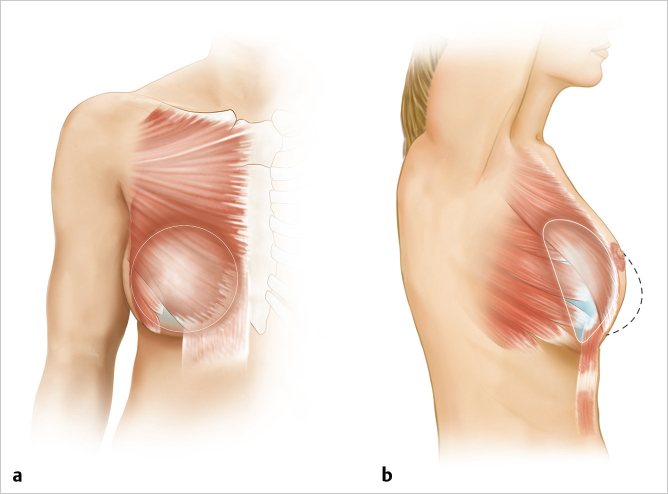
Given the negatives of total submuscular coverage, many surgeons sought to move away from this approach into a dual-plane technique incorporating pectoralis major above and the lower pole breast skin below. This left a significant amount of the implant covered only by skin, exacerbated by the problem of window-shading deformity occurring when the lower portion of the pectoralis major was released from its origin to accommodate the expander–implant volume.
42.3 Acellular Dermal Matrices
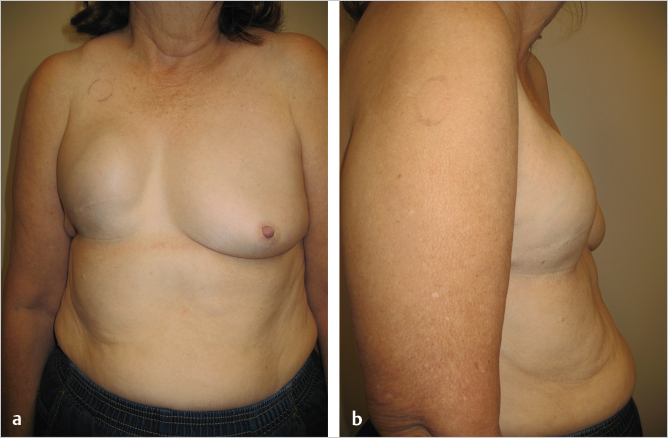
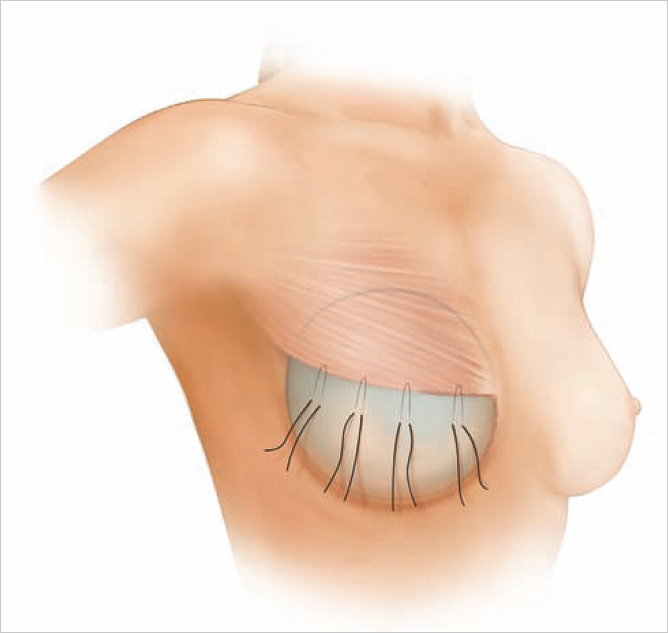
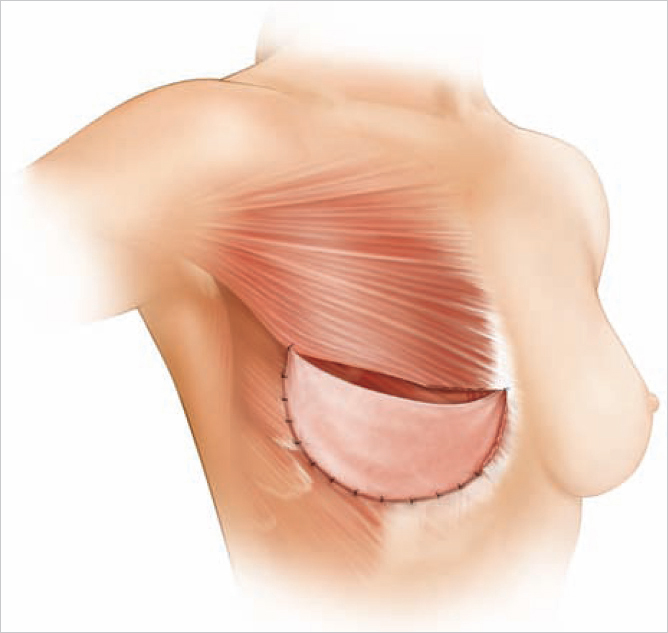
Spear offered the first solution to window-shading deformity by suggesting the use of marionette sutures between the pectoralis major and the overlying mastectomy skin envelope (▶Fig. 42.3). While this helped to alleviate the problem to some extent, it was also capable of inducing ischemic changes in the mastectomy skin envelope. A more permanent solution became apparent when ADMs were introduced. These products were sutured from the caudal border of the pectoralis major over the lower pole of the implants to the chest wall creating an anchor between the pectoralis major and the inframammary crease, thereby minimizing window shading. The ADM also acted as an internal bra to hold the implant in place and enable control of the subpectoral pocket to a degree never before achieved (▶Fig. 42.4). Given our more recent understanding of biofilms and their role in capsular contracture, as well as the fact that ADM modifies capsular biology to reduce capsular contracture development, this troublesome complication has been dramatically reduced.
42.4 Implant Shape
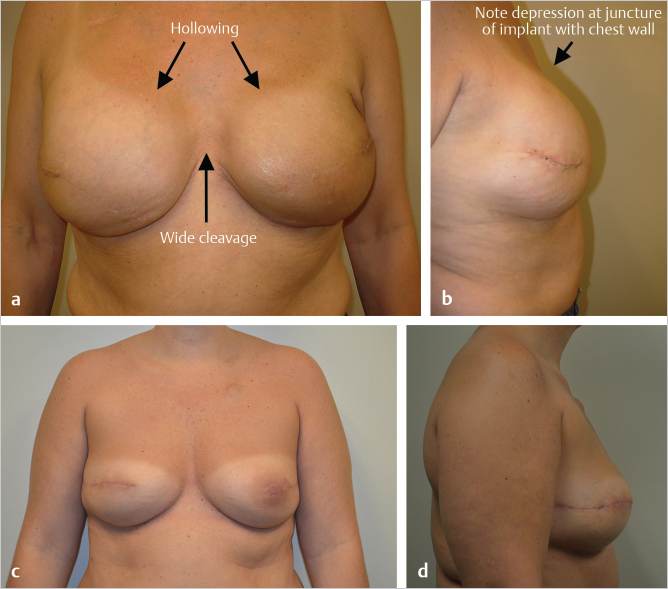
The use of more cohesive gel implants aided in reducing rippling and improving shape. Shaped anatomical implants have almost eliminated the upper pole hollowing seen so commonly with round devices and have, as a consequence, caused a reduction in the need for upper pole fat grafting (▶Fig. 42.5).
42.5 Perfusion Assessment Devices
Wound healing complications and skin necrosis after breast reduction occur in between 6 and 45% of patients depending on the series reported. Reduction in wound healing complications and skin necrosis has been achieved most successfully through the use of perfusion assessment devices. While fluorescein perfusion enabled flap assessment to some degree, it tends to overread potential necrosis resulting in unnecessarily large amounts of skin being resected. Laser-induced fluorescence of indocyanine green (ICG) dye provided a much more accurate, real-time assessment of skin perfusion that could be repeated multiple times during a given procedure. Floor-mounted and handheld devices are available at varying costs, but they still remain very expensive. New technologies including thermography (FLIR device) and various forms of spectral imaging are coming onto the market and it will be interesting to watch the development of this niche technology subset.
When teaching this procedure to surgeons, it has often been my experience that they mention the thickness of skin flaps as a limiting factor in being able to perform the procedure.
Technical Pearl
It is not the thickness of mastectomy skin flap that matters, but rather it is adequate vascularity that allows it to accommodate an implant safely beneath it in one stage.
I have performed this procedure in dozens of patients with extremely thin skin flaps who have had highly successful outcomes. This has been based not on their skin flap thickness but on their skin viability. Skin perfusion assessment devices have been invaluable in providing this level of safety during the procedure.
Given the development of these three different technologies, the stage was set for the successful launch of a prepectoral approach to breast reconstruction for the first time since the 1960s. While technologically feasible at this point, many surgeons ask the question “Why change to the prepectoral location when subpectoral reconstruction has provided reasonable success?” The answer to that question lies in the way in which we define success in the past. If success is defined as creating a reasonably soft round mound on the chest wall, yes it has been achieved. The problem is we have always tended to examine our patients with their arms at their sides and tend not to ask penetrating questions about aesthetics, cleavage, function, and the daily struggles with tightness and animation. If we factor into the assessment natural shape as well as the behavior of the breast during arm motility and activities of daily living, we suddenly find our supposedly successful reconstructions failing miserably. Patients find animation deformity to be embarrassing and the tightness of subpectoral implant placement to cause chronic discomfort during activity and at rest.
42.6 The Impact of The Pectoralis Major Muscle on Animation Deformity
This can be defined as adhesion between the pectoralis major and the overlying mastectomy skin flap resulting in bizarre and often uncomfortable contraction of the skin envelope during upper extremity emotion. The only reasonable cure for the problem is to separate the muscle completely from the skin with a relatively inanimate object in the form of a breast implant or flap reconstruction.
42.7 The Impact of the Pectoral Muscle on Cleavage Formation
One of the difficulties of placing breast implants in patients, whether for reconstruction or for cosmetic augmentation, has been the creation of attractive cleavage. Many implant-based procedures are marred by the presence of a wide, flat expanse between the two breasts even when the latter have reasonable shape and volume. This absence of cleavage is an important factor in the aesthetic outcome of any breast procedure whether cosmetic or reconstructive and for many women it is one of the crucial elements in their assessment of success.
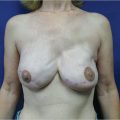
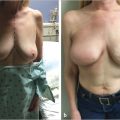
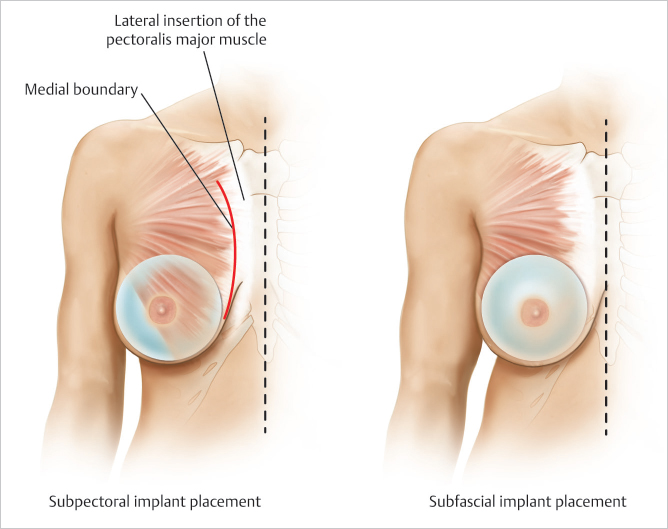
The location of the medial pectoral origin varies tremendously between different women. In some patients the medial borders almost approximate each other on the anterior aspect of the sternum, while in other women the muscles can arise as far lateral as the costal cartilages and even rib surfaces. When performing either subpectoral breast augmentation or breast reconstruction, the inferomedial origin of pectoralis major is divided to allow easier accommodation of the implants beneath the muscle. Despite this release, the remaining superomedial attachment of the pectoralis major acts as a physical barrier to medial implant placement. No amount of increasing implant volume can impact this physical limitation in a positive way. The wider the spacing of the pectoral origins, the wider, and less well-defined will be the ultimate cleavage created.
Technical Pearl
The only way to overcome this problem is to place the implant in the prepectoral position, which allows the surgeon to determine the location of the medial breast border rather than the muscle being the defining limiting factor.
This is clearly demonstrated in ▶Fig. 42.6.
These aesthetic and functional considerations prompted the author to move toward performing only prepectoral breast reconstruction. With the accuracy of skin perfusion assessment, the better shapes of our implants coupled with the use of ADM, I can no longer justify performing subpectoral reconstruction and find expanders necessary in only the most extreme situations or for delayed reconstruction.
Surgical Plan
Inspection of mastectomy skin flaps to assess thickness, perfusion, and areas of exposed dermis.
Assessment of mastectomy weight in grams, and placement of temporary sizer in the prepectoral space.
Indocyanine green perfusion imaging to assess skin flap perfusion with sizer in place.
Removal of sizer and shaping of acellular dermal matrix (ADM) to implant.
Suturing of ADM to create prepectoral ADM pocket.
Pocket lavage.
Insertion of implant beneath ADM using biofilm reduction technique.
Completion of closure of ADM pocket.
Drain insertion and closure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree