45 Preventing and Managing Complications of Expander–Implant Reconstruction
Summary
This chapter discusses the main complications of expander–implant reconstruction including tips for preventing complications. Details are provided on handling complications once they occur and how best to salvage the situation.
Key Teaching Points
Skin perfusion should be carefully assessed before proceeding with expander–implant reconstruction.
Expanders should be inflated as much as possible to accommodate healthy perfusion to the overlying skin.
Acellular dermal matrix incorporation may help with capsular contracture reduction.
Tabbed expanders should be the devices of choice.
Device infection is best treated with explantation.
The base of the expander should be placed at the inframammary fold.
Lower pole expansion is more difficult to achieve up a pole expansion.
Prepectoral reconstruction achieves more natural shapes and cleavage than submuscular placement.
45.1 Introduction
As at the time of writing, implant-based reconstruction was almost six times more prevalent in the United States than all other forms of autologous reconstruction combined.
Approximately 85% of those prosthetic reconstructions are performed with expanders while about 15% are direct-to-implant based.
With the retention of skin using skin- and nipple-sparing mastectomies, the original rationale for expansion has largely disappeared, but most surgeons continue to use it routinely.
In my own practice, I have all but eliminated expansion unless skin availability is at a premium to achieve a satisfactory outcome. With the extensive use of both expanders and implants, complications and problems surrounding the use abound. The purpose of this chapter is to discuss the problems that may arise from the use of these devices as well as seeking techniques to reduce the incidence of complications.
45.2 Common Problems Arising from Expander–Implant Usage
Skin necrosis with device exposure.
Hematoma.
Infection.
Seroma.
Extrusion.
Incorrect inframammary fold (IMF) creation.
Poor cleavage formation.
Poor lower pole fullness.
Long-term capsular contracture.
Rippling.
45.2.1 Skin Necrosis with Device Exposure
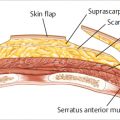
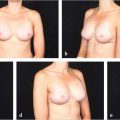
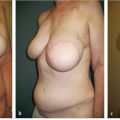
The incidence of skin crisis after mastectomy varies tremendously based on breast surgeon technique and the availability and use of skin perfusion assessment devices. Prior to these devices becoming available, many studies reported necrosis incidences of between 10 and 45% with many surgeons quoting an incidence in the 15 to 20% range. With the advent of skin perfusion assessment devices, the incidence of skin necrosis has dropped to between 3 and 6% in many studies. When implants or expanders are inserted under mastectomy skin flaps, there is always a risk that skin may develop necrosis. Should this occur, the risk of device exposure or extrusion is extremely higher. Furthermore, if skin is closed under tension over an overinflated expander or a sizeable implant, the risk of necrosis may increase (▶Fig. 45.1).
Prevention
Skin perfusion assessment prior to device insertion.
The use of underinflation of an expander.
The use of closed wound suction (e.g., Prevena) to bolster cutaneous blood supply.
Management of Established Necrosis
Debridement and reclosure with reduction in expander volume.
Explantation if inadequate skin is available and delayed reconstruction at a later date.
Salvage with immediate latissimus dorsi myocutaneous flap coverage.
45.2.2 Hematoma
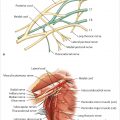
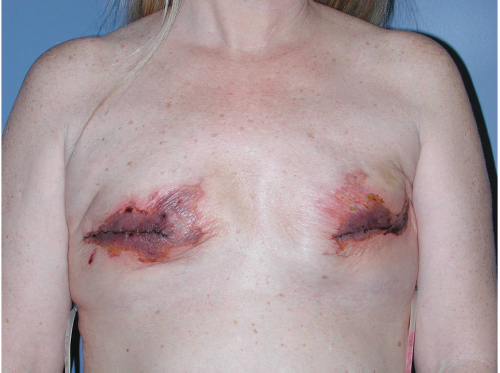
Hematomas are rare following expander–implant reconstruction in my experience. They occur in less than 1% of procedures. They should be a predominantly avoidable occurrence if the reconstructive surgeon pays meticulous attention to hemostasis. Hematomas are serious problems in that they increase the risk of both, infection in the short term and capsular contracture in the longer term (▶Fig. 45.2).
Prevention
Meticulous in hemostasis with electrocautery.
Diffuse small-vessel oozing can be limited with fibrin glue spray to the implant pocket prior to closure.
Management
Immediate exploration and evacuation with control of bleeding and drain insertion.
Lavage with Betadine or triple antibiotic solution or chlorhexidine irrigation (Irrisept).
45.2.3 Infection
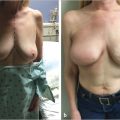
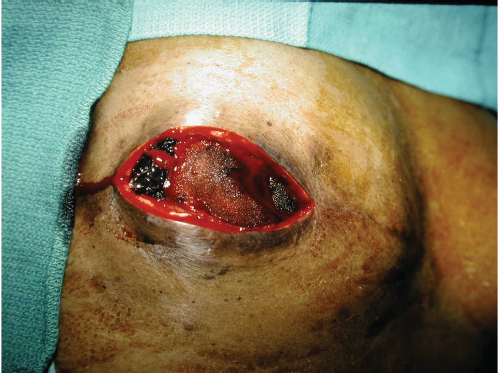
Infection is a catastrophic complication in either expander or implant reconstruction, with an incidence of between 2 and 6% in most current series. It results in high implant loss rates if control is unsuccessful. If salvage is achieved, capsular contracture tends to be a considerable longer-term problem. The problem is more severe when textured surface devices are used, as the interstices of the implant surface tend to harbor bacteria even more aggressively than smooth-surfaced devices (▶Fig. 45.3).
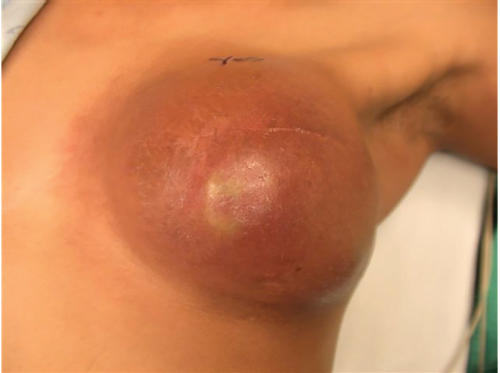
Prevention
Meticulous attention to detail.
Careful pocket preparation with antibacterial lavage techniques.
Surgeon glove change prior to implant handling.
Use of the no touch technique for implant insertion (the Keller funnels, iodine-impregnated drapes, etc.).
Careful drainage of fresh mastectomy pockets.
Management of Established Infection
Patients may be given a trial of oral antibiotics for 24 hours if cellulitis is the only symptom.
Ultrasound should be performed to exclude fluid collections be they pus or seroma. If either is present, drainage is mandatory.
If the entire breast is inflamed, admission with intravenous antibiotics can be tried for 24 to 48 hours.
If full-blown periprosthetic infection with the fluid is present, explantation and drainage is preferable to salvage attempts.
If the device does not look grossly infected, it can be removed and a temporary wound vacuum-assisted closure can be inserted for 72 hours, following which reinsertion of a fresh implant can be performed. This approach has been achieving salvage in the 70 to 85% range in recent years.
Patients should be warned of the increased risk for capsular contracture in the longer term.
45.2.4 Seroma
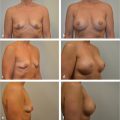
Seroma is a troubling complication of prosthetic reconstruction and has plagued the use of implants as well as acellular dermal matrices (ADMs) associated with their use. Seroma rates vary tremendously, largely to the fact that many authors do not report small seromas aspirated in the office. The incidence ranges between 3% and as high as 40% with most recent studies reporting a 3 to 10% incidence. Seromas can be difficult to treat, may recur, and are a potent source of infection due to their high sugar and protein content (▶Fig. 45.4).
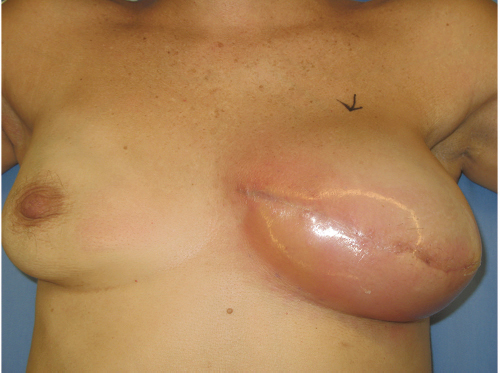
Prevention
Drainage of all mastectomy reconstruction pockets using reliable large-caliber drains.
Train should be left in place until output has been less than 20 to 30 mL per day for at least 48 to 72 hours. This occurs typically, between 2 and 3 weeks after expander or implant insertion.
Management of Established Seroma
Aspiration can be attempted but is likely to result in recurrence.
Reinsertion of a drain in the office using a Seroma-Cath device, or an ultrasound-guided pigtail catheter drain insertion by the interventional radiology department.
Fluid should be cultured.
Doxycycline 100 mg may be injected into the indwelling catheter to reduce drain output.
Technical Pearl
Any patient with a late-onset seroma occurring 1 year or more after reconstruction should be investigated for the possibility of breast implant–associated anaplastic large cell lymphoma if a textured device has been used.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree