51 Transverse Rectus Abdominis Musculocutaneous Flaps
Summary
This chapter describes the unipedicle transverse rectus abdominis musculocutaneous flap and its use in breast reconstruction. The indications for and technique of performing the flap are discussed in detail, outlining important technical tips, which may help with overall success.
Key Topics
Pertinent anatomy.
Assessing tissue requirements.
Donor site considerations.
Selecting the appropriate abdominal flap.
Vertical orientation.
Oblique orientation.
Horizontal or transverse orientation.
Selection of vascularization and mound-shaping technique for TRAM flap.
Breast reconstruction.
Unipedicle TRAM flap reconstruction.
Bipedicle TRAM flap reconstruction.
Bilateral TRAM flap reconstruction.
Special problems and ancillary procedures.
Key Teaching Points
The unipedicle TRAM flap remains a viable option for autologous reconstruction.
It is a useful alternative for surgeons who do not have the expertise or inclination to perform microsurgical reconstruction.
Ipsilateral transfer is safer than contralateral transfer.
Meticulous attention needs to be paid to abdominal wall closure to achieve long-term success.
Preoperative vascular delay can increase the success rate in higher-risk patients.
51.1 Origins of the Pedicle TRAM Flap
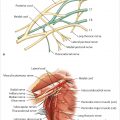
Millard described the use of a tubed lower abdominal pedicle flap for reconstructing the radical mastectomy defect in 1976. The flap was sequentially waltzed onto the chest by way of the forearm to achieve a highly successful autologous tissue reconstruction for the time. In 1979 Robbins used a vertical rectus abdominis flap for breast reconstruction. Independently, Drever, Dinner, and Sakai all refined variations on the use of vertical rectus abdominis myocutaneous flaps for breast reconstruction. Hartrampf and colleagues took the bold step of changing the skin island orientation to a transverse one across the mid-abdomen, making a more sizeable volume of tissue available for breast reconstruction with a cosmetically desirable donor site. Scheflan and colleagues confirmed the dominant inferior epigastric arterial supply to the lower abdominal skin and fat. Blood supply was most robust directly over the muscle belly where perforators were most abundant, whereas the periphery of the flap relied on the superficial epigastric and circumflex iliac terminal branches. Milloy and McAfee had documented the blood supply of the rectus muscles in 1960, and these findings, together with Scheflan’s dissections, found their culmination in the lead oxide injection studies of Taylor, Moon, and Palmer. Their publication of the angiosome concept was an extension of Michel Salmon’s anatomical studies. From these humble beginnings, the transverse rectus abdominis musculocutaneous (TRAM) flap was destined to become the benchmark procedure for breast reconstruction and remains so today. Subsequently, several free flap options have developed as refinements of the original pedicle technique, including the free TRAM, muscle-sparing free TRAM, and the perforator flaps.
51.2 Operative Overview
Flap Options
Pedicle ipsilateral TRAM flap.
Contralateral unipedicle TRAM flap.
Bipedicle TRAM flap.
Vascular delayed TRAM flap.
Turbocharged TRAM flap (a segue between the pedicle and the free TRAM).
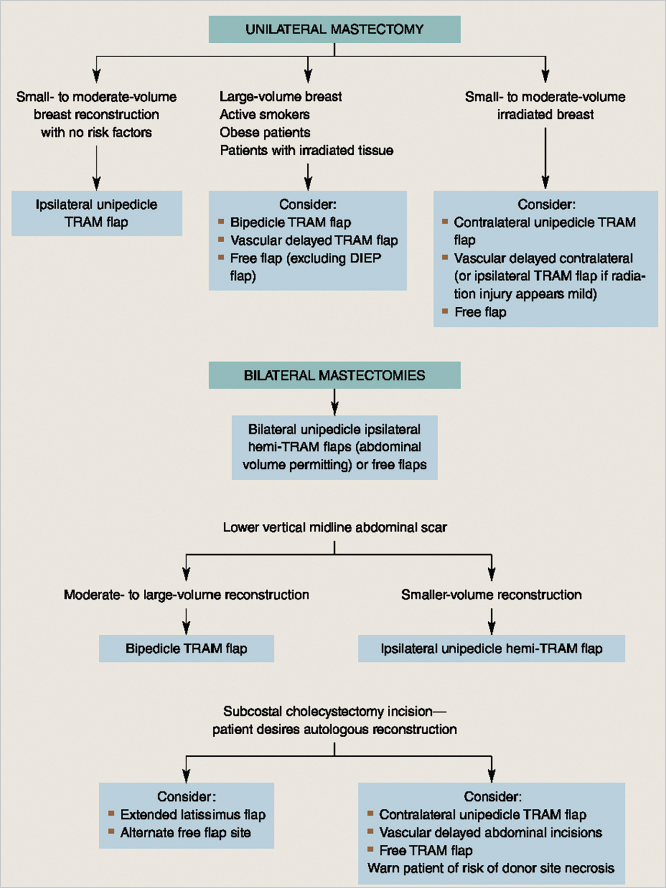
The TRAM flap is based on the concept of elevating lower abdominal skin and fat for breast mound construction while leaving one or both rectus abdominis muscles attached to maintain adequate blood supply for transposition. By definition, a pedicle TRAM flap requires subcutaneous tunneling beneath the upper abdominal skin to be placed into its recipient bed. Several variations of the flap exist and have evolved in response to unique anatomical problems or clinically relevant risk factors. These include such situations as patients who smoke, are obese, have abdominal scars, or have irradiated tissue and surface area volume requirements that demand more extensive blood flow to a potentially compromised area of tissue. A number of flap options are available.
In principle, the higher the patient’s risk, the greater the likelihood that she will require a bipedicle vascular delayed procedure. For the nonmicrosurgeon, this is certainly the case and provides a safe alternative to a complex microsurgical procedure with which he or she might not be comfortable. A surgeon familiar with unipedicle and bipedicle procedures will be able to offer the TRAM flap to a large number of patients with a wide variety of clinical problems. Many patients who are unsuited to this procedure because of associated risk factors are often candidates for latissimus dorsi flaps.
51.3 Patient Assessment and Selection
Careful patient selection is possibly more crucial for the TRAM flap than for any breast reconstruction procedure other than microsurgical transfer.
TRAM flap breast reconstruction is a significant surgical undertaking for both the patient and the surgeon. The first prerequisite for this procedure is that the patient be healthy enough to undergo a 2- to 3-hour operation, a 3- to 5-day hospital stay, and a 4- to 8-week recovery period before she begins to feel that life is returning to some degree of normalcy. The second major requirement is an available donor site.
A thorough history should be taken, including an evaluation of comorbidities such as the following:
Gastroesophageal reflux disease.
Irritable bowel syndrome.
Lumbar spine problems.
Smoking history.
Cardiovascular risk factors.
Diabetes mellitus.
Collagen vascular disease.
History of prior abdominoplasty or abdominal liposuction.
A clinical examination should be performed, noting the following:
Body habitus and weight.
Old scars, particularly cholecystectomy scars or vertical midline incisions. I do not regard the Pfannenstiel incisions as a risk factor. Laparoscopic incisions are rarely a problem, but port sites may have injured the vessels within the rectus muscle in the upper abdomen, and Doppler evaluation is prudent. It is probably unwise to operate within 6 weeks after laparoscopic surgery.
Body habitus is an important factor for assessing the tension of the lower abdomen. Patients with a rotund apple-shaped profile (as opposed to the softer, looser abdominal wall of the pear-shaped physique) tend to have very tight abdominal walls with minimal tension to achieve safe closure. This is particularly true if they have long umbilicus to pubis distance. Final factors in flap selection are the patient’s occupation and lifestyle. Very active young individuals are better served by a perforator flap, and patients who are singers occasionally express concerns about the impact of muscle loss on their ability to sing. This does not appear to be a significant issue in practice.
Hartrampf and colleagues attempted to assign risk scores to patients to determine their eligibility for TRAM flap reconstruction.
Hartrampf Risk Assessment
Risk factors included the following:
Smoking.
Obesity.
Psychological instability.
Autoimmune disease.
Diabetes mellitus.
Severe systemic disease.
Surgeon inexperience.
Borderline risk: two risk factors or a score below 5.
High risk: three or more risk factors or a score of higher than 5.
In our own series, diabetes did not correlate well with complications, but obesity, smoking, abdominal scars, and prior radiation therapy did.
When the procedure is executed expertly in properly selected patients, the TRAM flap produces natural breasts that are shapely, long lasting, warm, and mobile. The results that can be obtained with this flap are exceptional. There are, however, numerous possibilities for complications and disappointment using this complex procedure. For some patients the magnitude of the procedure, prolonged convalescence, postoperative abdominal discomfort, and additional scars outweigh the benefits of breast reconstruction with autologous tissue. The problems associated with convalescence are magnified when there are complications such as major tissue loss, abdominal wall weakness, and hernias. However, these issues face the microsurgical patient as well, although at lower rates.
Indications for TRAM Flap Surgery
Women requesting autologous breast reconstruction who do not want an implant-based procedure.
Women with excess abdominal tissue after childbearing.
Women requesting breast reconstruction and abdominoplasty.
Obese women with large opposite breasts.
Salvage procedures following failed expander–implant reconstruction.
Women having immediate reconstruction after skin- or nipple-sparing mastectomy.
Failed breast conservation patients with a recurrence necessitating mastectomy.
Women with multiple local risk factors, such as irradiation, with limited reconstructive options.
51.3.1 Relative Contraindications
Relative contraindications to TRAM flap surgery include the following:
Serious medical problems.
Psychiatric disorders.
Morbid obesity.
Thin patients who would require augmentation for adequate volume.
Heavy smokers.
Elderly patients.
Given the array of flap options available to surgeons and the variability of the patient population described previously, it is appropriate to have a working algorithm to assist with decision making and flap selection. The algorithm can be used to simplify flap choices for patients with varying risk factors and includes the surgeon’s preference and level of comfort with microsurgery. This flow chart provides a plan for both microsurgeons and nonmicrosurgeons.
51.3.3 Patient Profiles
Women Requesting Autologous Reconstruction Who Do Not Want an Implant-Based Procedure
Despite the fact that the implant crisis of the 1990s has passed, many women still have preconceived objections to reconstruction with implants, either because of fears of implant-related complications or because they do not want a foreign material in their bodies. These individuals frequently request an autologous breast reconstruction option assuming that they have an adequate donor site and would forego reconstruction if this cannot be provided.
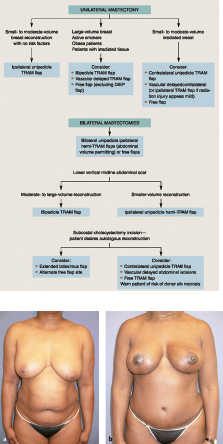
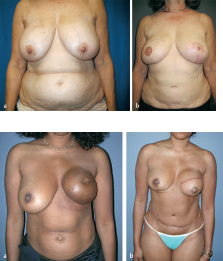
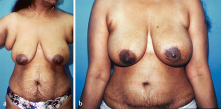
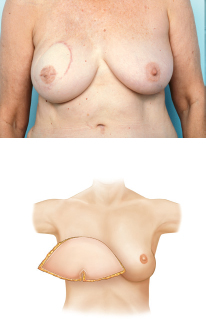
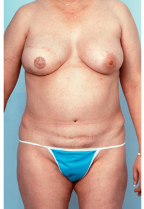
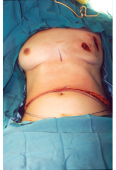
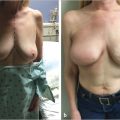
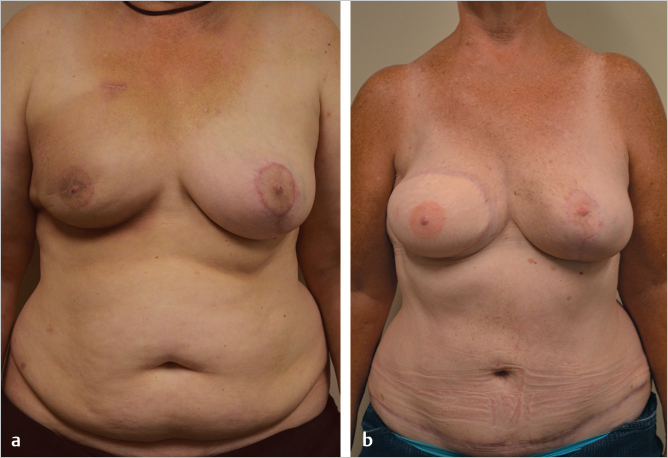
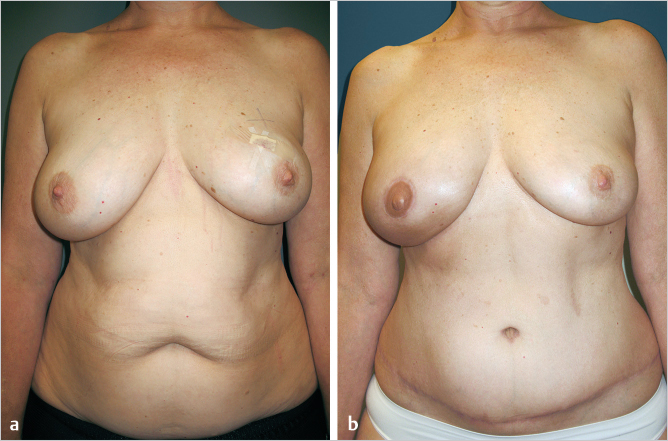
This 40-year-old woman with infiltrating ductal carcinoma of the right breast underwent mastectomy and immediate reconstruction. She wanted autologous tissue reconstruction and was vehemently opposed to any form of implant-based procedure. She had an adequate TRAM flap donor site to provide suitable volume to match her normal breast volume. An ipsilateral unipedicle TRAM flap was performed, combined with immediate left vertical mastopexy for symmetry. A staged nipple reconstruction was performed. She is shown 18 months after her procedure with excellent shape and symmetry (▶Fig. 51.2).
Women with Excess Abdominal Tissue after Childbearing
Multiparous women often have a redundant lower abdominal pannus which are excellent donor sites for autologous reconstruction. Often, they are not active in sports or fitness activities and look to the surgeon to give them a new figure. When these women meet the criteria for a TRAM flap, they are usually satisfied with the result. Their expectations are met by the abdominoplasty and a soft, natural breast reconstruction.
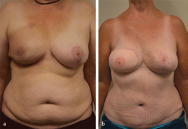
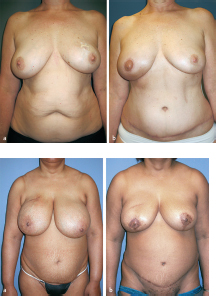
This patient presented having previously had expanded implant reconstruction followed by radiation therapy. She had significant radiation fibrosis with asymmetry. A pedicled TRAM flap salvage procedure was planned. She had a well-developed lower abdominal pannus as a donor site. Her radiated skin was excised and replaced with soft abdominal skin providing better ptosis. She is shown 1 year after reconstruction with a successful outcome in much better shape and symmetry (▶Fig. 51.3).
Women Requesting Breast Reconstruction and Abdominoplasty
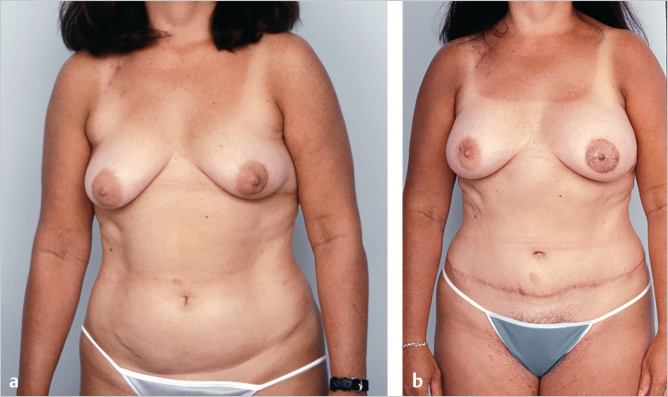
This 40-year-old woman intending to undergo abdominoplasty, then developed multifocal ductal carcinoma in situ (DCIS) of the left breast. She requested autologous reconstruction as well as a modest right breast augmentation.
Left breast reconstruction was performed with an ipsilateral unipedicle TRAM flap, followed by subsequent C-V flap nipple reconstruction and contralateral subpectoral augmentation with a saline-filled implant.
The patient is shown 9 months postoperatively and had returned to her career as a professional horse riding instructor and show jumper (▶Fig. 51.4).
Obese Women with Large Opposite Breasts
Overweight patients with large pendulous breasts require a substantial amount of tissue for satisfactory breast reconstruction. Their donor sites are usually substantial and offer more tissue than is required for reconstruction. A large volume TRAM flap consisting mostly of well-vascularized zone I tissue can be used, achieving a very satisfactory outcome.
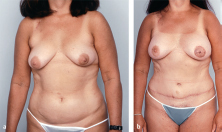
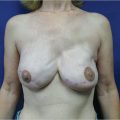
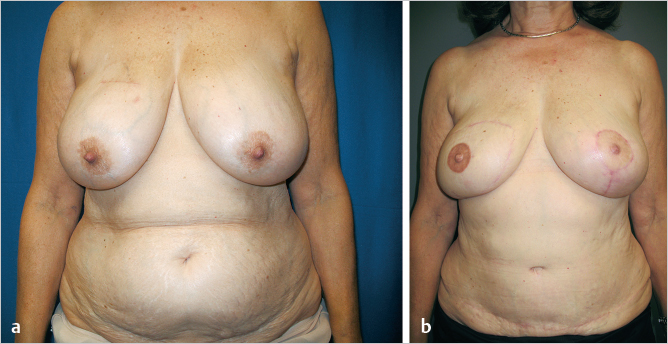
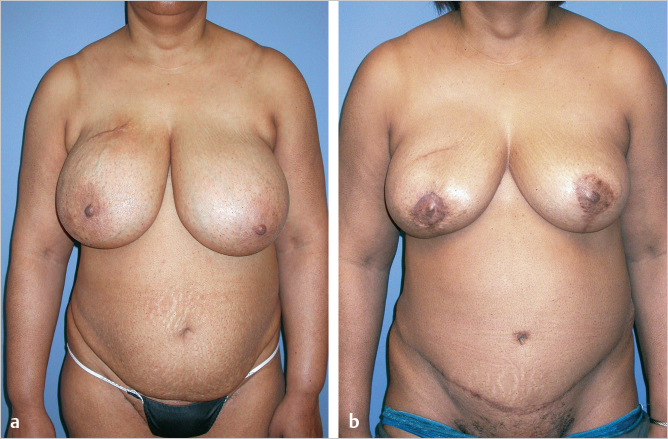
This 58-year-old woman, who wore a 42E bra, developed a 1.2-cm ductal carcinoma in her right breast. She had an ample abdominal pannus and grade 3 ptosis and had wanted to have a breast lift for years. An immediate right ipsilateral unipedicle TRAM flap breast reconstruction utilizing zone I tissue achieved a well-perfused soft result. A conservative 250-g reduction of the left breast was performed using a modified Lassus vertical approach. She is shown 1 year postoperatively with well-shaped, soft breasts and an improved overall breast contour (▶Fig. 51.5).
Salvage Procedures Following Failed Expander–Implant Reconstruction
Radiated expander–implant reconstructions tend to deteriorate with time. Tightness and pain can become disabling, and infection and erosion can also supervene. The patient’s only alternatives are explantation with no further reconstruction, or provision of an autologous alternative. A TRAM flap can provide both surface area and volume fill under these circumstances.
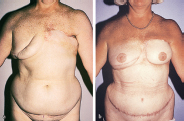
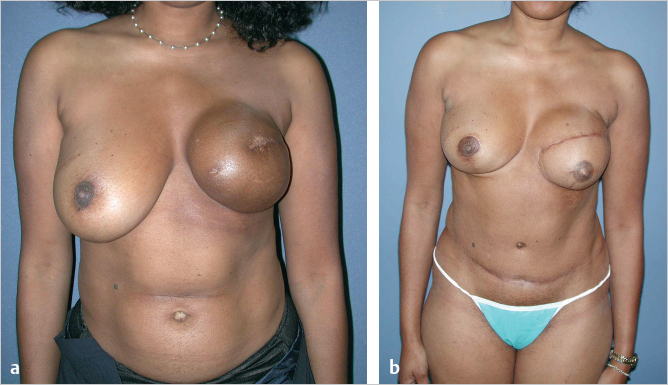
This 38-year-old woman developed invasive ductal carcinoma of the left breast requiring chemotherapy and radiation after mastectomy. As a busy lawyer, she insisted on an expander-based reconstruction, because she was concerned about the magnitude of a TRAM flap procedure and the associated lengthy anesthesia. She is shown 6 months after completion of radiation therapy with a hyperpigmented skin envelope and a tight, uncomfortable grade 4 capsular contracture around the expander. She finally agreed to a bipedicle TRAM flap, because we required the entire lower abdominal skin surface area to reconstruct the defect ensuing after resection of her poor-quality skin envelope as well as providing fill. Her pannus was not adequate to achieve a D cup bra size despite her modest weight gain when she was receiving tamoxifen. A 200-cc saline-filled breast implant was placed at the time of TRAM flap reconstruction. She is shown 2 years after surgery. Despite using her entire TRAM flap skin island, she still has some upper medial pole volume deficiency. She would clearly benefit from fat injection, but she declined further surgery, since she was very satisfied with her result (▶Fig. 51.6).
Women Having Immediate Reconstruction after Skin-Sparing Mastectomy
While nipple-sparing mastectomy is the aesthetic ideal for breast reconstruction, skin-sparing mastectomy can also achieve outstanding results if the areola skin disk is replaced by autologous tissue. This represents an excellent indication for an autologous abdominal reconstruction which provides the appropriate skin circle to replace the areola.
This 55-year-old woman developed a 1-cm diameter invasive ductal carcinoma of the right breast treated with skin-sparing mastectomy and immediate right ipsilateral unipedicle TRAM flap reconstruction. No procedure was required on the contralateral side to maintain symmetry. She returned to her previous activities, including golf and tennis. Her breast is soft and supple, with excellent symmetry without the need for contralateral surgery (▶Fig. 51.7).
Failed Breast Conservation Patients with a Recurrence Necessitating Mastectomy
The TRAM flap is a good choice in patients who have recurrences after lumpectomy and radiation treatment or in whom severe deformity, pain, or tightness develops, necessitating mastectomy. The procedure affords the patient a soft autologous reconstruction which can be accompanied by resection of the entire radiated skin envelope if necessary.
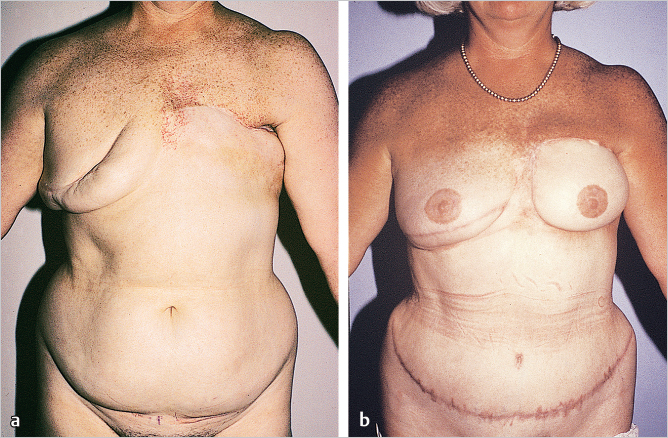
This 44-year-old woman developed breast cancer in the upper outer quadrant of the right breast. She had undergone breast-conservation treatment 3 years previously and had a remarkably symmetrical, soft result. Unfortunately, she subsequently developed a second lesion in the middle of the breast. She underwent a right mastectomy with a vertical-pattern skin envelope reduction. A bipedicle TRAM flap was performed to achieve adequate volume and protect against fat necrosis. Her TRAM flap healed uneventfully with a soft, supple result. A contralateral vertical breast reduction was performed at the time of right nipple reconstruction to achieve symmetry. She is shown 2 years postoperatively with a well-healed, soft breast and a strong abdominal wall with no bulges (▶Fig. 51.8).
Women with Multiple Risk Factors
A patient may present to a surgeon with an adequate abdominal pannus but have associated risk factors such as smoking, obesity, and/or previous irradiation. For the nonmicrosurgeon, patients are often best treated with a pedicle latissimus flap if it is available and appropriate. However, if the patient strongly desires a TRAM flap and her tissue volume would be more appropriate for this procedure, a vascular delay performed before definitive flap transfer is a reasonable alternative. The following case illustrates a high-risk patient where such an alternative provided an excellent outcome despite these mitigating circumstances.
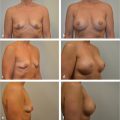
This 58-year-old candidate for bilateral breast reconstruction had multiple risk factors for complications after a TRAM flap reconstruction. She was overweight, smoked cigarettes, and had irradiated tissue at the mastectomy sites. Therefore, a surgical delay was done 2 weeks before flap elevation to increase the blood flow and arterial pressure in the TRAM flap and facilitate safe transfer. The patient is seen 1 year postoperatively; she experienced no complications from her reconstructive surgery (▶Fig. 51.9).
Obese Patients
Breast reconstruction is problematic in overweight women, particularly those with a body mass index (BMI) above 30. Implant reconstructions, however large, may not be able to provide adequate volume, and the TRAM flap, which offers sufficient tissue, is associated with an increased risk of tissue loss, fat necrosis, abdominal wall laxity, and other breast and systemic complications. In my own series of patients while practicing at the Emory Clinic, we identified obesity as one of the major risk factors for both abdominal and flap complications. This has been borne out in numerous studies from other centers and will be discussed in the Outcome Studies section.
However, if they are appropriately counseled preoperatively, I have found these women to be very appreciative of the reconstruction procedure and its associated body contouring. It is certainly better than a poor attempt at implant reconstruction, which is usually doomed to failure in obese patients because of their massive volume requirements.
Technical Pearl
Interestingly, these patients often have large perforators, and a high zone I-based TRAM flap can be remarkably robust and a zone I flap often has enough volume to provide an excellent reconstruction without the need for harvesting additional zones, which carry greater risk of necrosis. However, a pendulous pannus is certainly not safe to use.
The fat below Scarpa’s fascia particularly is subject to fat necrosis in these patients. Although all patients who are overweight have an increased risk of complications after the TRAM flap procedure, I have found the patients who have the best microcirculation and the fewest complications are those who are premenopausal, have relatively normal lower abdominal skin without significant striae, are not smokers, have no other microcirculatory problems, and have a TRAM flap oriented a few centimeters higher above the umbilicus than usual. For women with significant risk factors, TRAM flap reliability can be increased with a surgical delay performed 3 to 10 days before the TRAM flap.
Postmenopausal Patients with Serious Risk Factors such as Smoking Should Be Considered for Autologous Latissimus Dorsi Reconstruction
Although it may be a second choice, it is a safer and more prudent choice for some of these individuals. Also, women who are considerably overweight usually have excess back tissue that can be mobilized for breast reconstruction as a latissimus dorsi flap, thus avoiding the high risk of serious TRAM flap complications.
Patients Who Have Had Radiation Therapy to the Chest Wall or Abdomen
There has been a dramatic increase in patients receiving radiation for breast cancer management. This is a reflection of both the increasing trend toward breast conservation therapy as well as the increased use of adjunctive radiation for patients with early breast cancer as suggested by the Canadian and Danish studies published in the mid-1990s. Patients with failed breast conservation therapy presenting for mastectomy pose a unique set of problems. Such individuals have irradiated mastectomy flaps that may necrose more readily, and internal mammary and thoracodorsal vessels have often been heavily irradiated. Although there are data suggesting that the caliber of these vessels may be unchanged by radiation, our own data at the Emory Clinic demonstrated an increased risk of flap complications when flaps were raised on irradiated pedicles. This was corroborated by the report of Clugston and colleagues, who found ipsilateral TRAM flaps to be no more complicated than those raised on nonradiated pedicles. However, if free flaps are planned, when dissecting the internal mammary vessels as recipient vessels, the veins in particular are certainly more fragile. Radiation to the abdominal wall itself is an absolute contraindication to TRAM flap elevation.
51.4 Planning
51.4.1 The Opposite Breast
As in all reconstructive breast surgery, the opposite breast (or the corrected opposite breast) is the model for the reconstruction. If the TRAM flap breast reconstruction is to match the opposite breast, resected skin, the nipple–areola complex, and breast parenchyma must be replaced with a similar amount of abdominal wall skin and fat. Experience with autologous flaps, especially with skin- or nipple-sparing mastectomy and immediate breast reconstruction, has made it easier to match the opposite breast without altering it. When the breast margins are defined and secure, it becomes primarily a matter of placing the proper TRAM flap volume in the preserved skin envelope. Careful attention to detail can often result in a near-perfect match with the opposite side (assuming this is desirable) thereby eliminating the need for contralateral symmetry procedures.
51.4.2 Skin Deficit
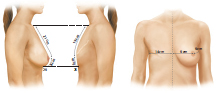
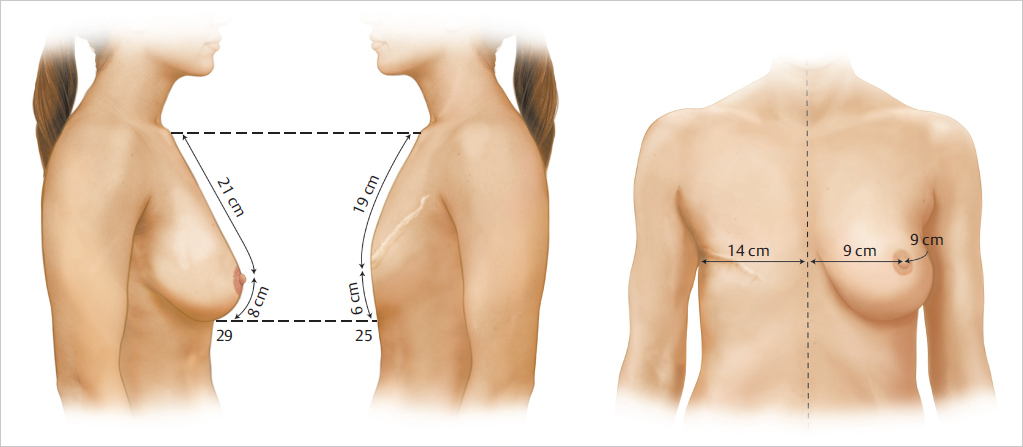
The amount of subcutaneous tissue and skin necessary for final symmetry, axillary, and infraclavicular fill, and breast mound restoration must be determined. Patients with a modified radical mastectomy deformity usually do not need fill for the infraclavicular area unless pectoralis major muscle denervation or significant excision of infraclavicular tissue has created a tissue deficiency.
The approximate amount of skin required for the reconstruction is estimated by measuring the opposite breast both vertically from clavicle to inframammary crease and horizontally from midline to midaxillary line and assessing the deficit. The difference in the two sides indicates the approximate skin requirements. Most of the breast reconstructions I perform now are immediate procedures involving skin-sparing mastectomy. Under these circumstances, a small skin island is preserved on the flap to replace the areola skin and the remainder of the flap is deepithelialized and buried beneath the mastectomy skin for volume replacement. When the excised areola is small, the areola site may be closed as a purse-string suture and the entire flap is buried; however, I do not favor this approach, as I have not been satisfied with the quality of the skin available for subsequent nipple reconstruction. Its scarred surface tends to take up tattoo dye rather poorly and unevenly. If the areola diameter is large, I attempt to reduce it to a more normal level. In patients who smoke or in those with large or irradiated breasts, skin perfusion may be poor. In these cases, it is preferable to resect the dubiously viable skin and replace it with exteriorized TRAM flap skin (▶Fig. 51.10).
51.4.3 Inframammary Crease
It is important to plan and re-create the inframammary crease properly and at the correct level. The surgeon who is inexperienced with TRAM flap breast reconstruction tends to place the inframammary crease too low. There are several reasons for this. An incision at the level of the inframammary crease to inset the flap will tend to drop down, making it lower. When the upper abdominal flap is advanced downward for abdominal wall closure, this pulls the inframammary crease down farther. I only commit to my planned inframammary crease location once the abdominal closure is complete.
For these reasons the inframammary crease is usually planned at least 2 cm above the opposite inframammary crease and can be defined after abdominal skin closure.
51.4.4 Unipedicle Flap
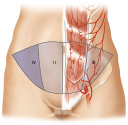
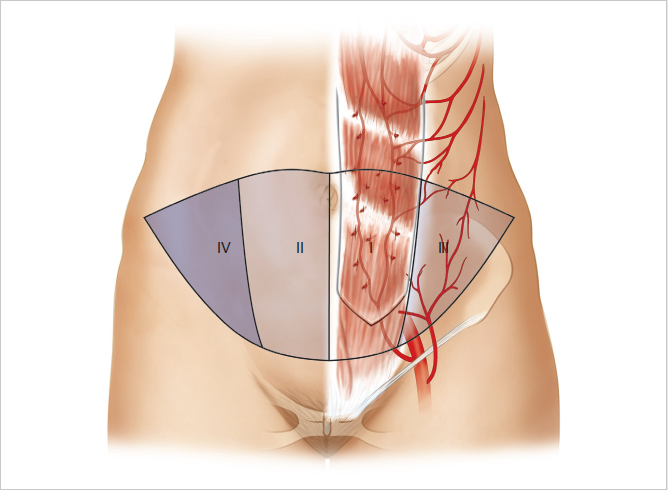
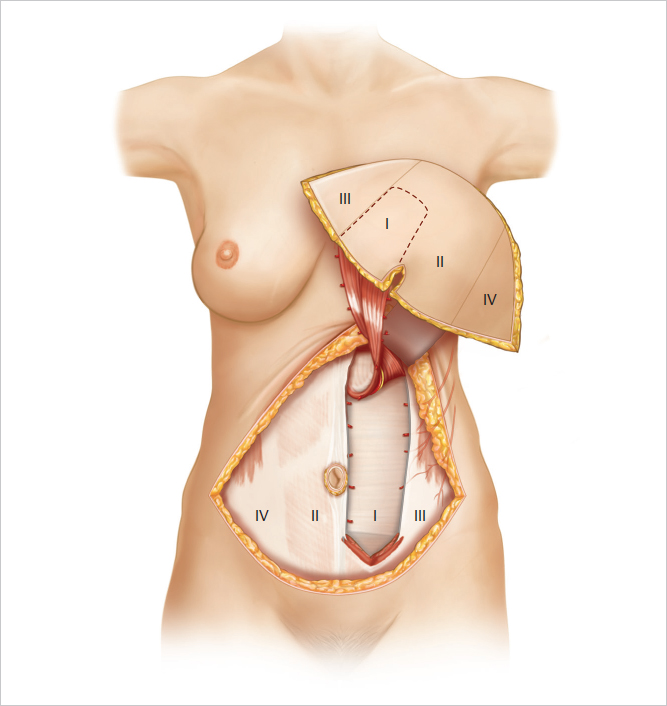
Hartrampf and colleagues divided the skin of the lower abdomen into four distinct zones based on their proximity to the underlying perforator supply and, as a result, their degree of perfusion. The zones are as follows:
Zone I immediately above the vascular pedicle.
Zone II across the midline adjacent to zone I.
Zone III ipsilateral to zone I.
Zone IV contralateral to and adjacent to zone II.
Zone I is most well perfused, since it directly overlies the perforator system. Zones II and III benefit from adjacent flow in the terminal branches of the perforating vessels, whereas zone IV is least reliably perfused, having to rely on flow from zone I, first crossing zone II before reaching the tip of the contralateral side of the flap. Both clinical evaluation and the use of indocyanine green (ICG) perfusion studies have demonstrated extremely unreliable perfusion to zone IV, and I routinely discard this zone in both pedicle and free TRAM flaps (▶Fig. 51.11).
51.4.5 Bilateral Unipedicle Flaps
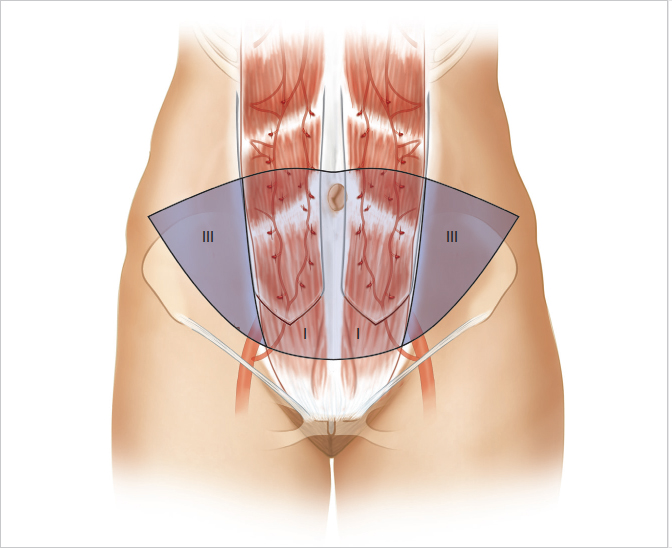
Bilateral unit pedicle flaps are used for bilateral autologous reconstruction as an alternative to microsurgery. They utilize exactly the same principles as the original unipedicle flap, but because both muscles are harvested, they require a far more significant donor site repair. In such cases, each flap consists of zones I and III with none of the risks of cross-midline blood flow complications.
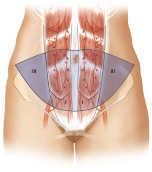
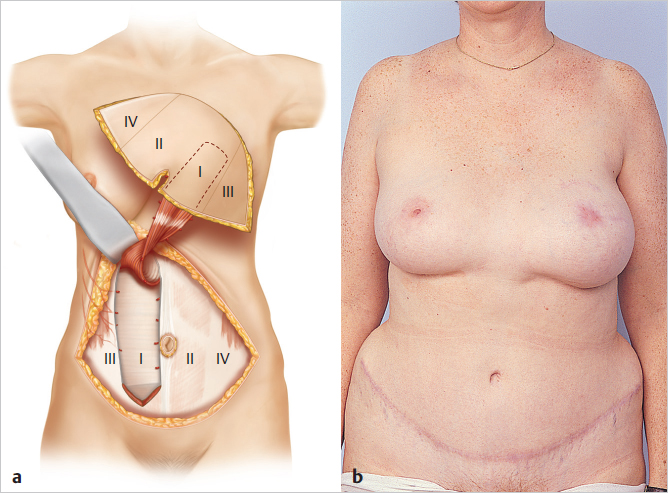
51.4.6 Bipedicle Flap
The bipedicle TRAM flap is a solution to some of the potential perfusion problems encountered with the unipedicle approach. Zones II and IV are absent in the bipedicle flap, increasing its vascularity but adding to its complexity. With this approach the blood flow and venous return to the flap is doubled. It also avoids reliance on tissue perfusion across the midline and has been found to be less prone to fat necrosis. It leaves a much more significant abdominal donor site for repair.
Taylor and colleagues suggested that the bipedicle flap may have a more robust vascular supply than a free TRAM flap in that the entire flap achieves a more stable perfusion. This has certainly been my own experience, and I have yet to see a bipedicle TRAM flap develop clinically apparent fat necrosis. What is gained by increased flap perfusion, vascularity, and safety must not be lost by increased operative and postoperative complications, functional loss, or morbidity. The bipedicle TRAM flap breast reconstruction increases the technical challenge of flap elevation and abdominal wall closure. The surgeon must be well versed in anatomy and have the skills needed for limited fascial harvest to allow safe muscle harvest and abdominal wall repair and closure. The bipedicle flap also restricts the transposition and positioning of the flap somewhat and adds bulk to the pedicle. Either TRAM flap delay with a unilateral TRAM flap or a free TRAM or deep inferior epigastric perforator (DIEP) flap can be used as alternate strategies to avoid the bipedicle TRAM flap. These factors should be balanced by increased flap vascularity, reliability, and safety and decreased risk of fat necrosis (▶Fig. 51.12).
51.4.7 Selecting the Appropriate Abdominal Flap Orientation
Flap positioning profoundly affects the ultimate shape of the breast mound, particularly in delayed reconstruction. There are three basic flap positions and mound-shaping techniques: the vertical, the horizontal or transverse, and the oblique (intermediate between the vertical and horizontal flap orientations).
Vertical Orientation
Vertical orientation is used for patients with a long, narrow chest. These patients are often thin and typically have a narrow breast diameter and require upper pole fill to camouflage a bony chest wall (▶Fig. 51.13).
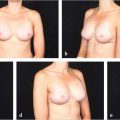
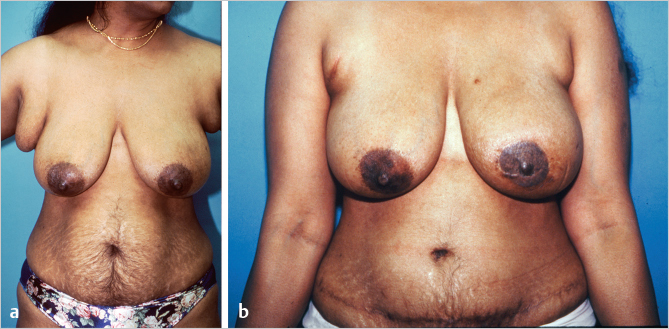
This 42-year-old woman developed cancer of the left breast and underwent immediate bipedicle TRAM flap reconstruction because she did not want a contralateral reduction or mastopexy, and a large volume of tissue was required for reconstruction. Although her transverse breast diameter was fairly wide, her insistence on maintaining a pendulous reconstruction dictated the need for a vertical orientation to her flap. She is shown 1 year postoperatively with excellent symmetry; bilateral resection of adjacent axillary breast tissue was also performed (▶Fig. 51.14).
Oblique Orientation
Oblique orientation is used for inferolateral fullness as well as upper medial fill. The oblique mound-shaping technique is a variant of the technique used for vertical mound shaping. After the flap has been positioned vertically, the lower portion is rotated outward to give additional lower breast fullness. This also ensures good fill in the upper medial portion of the breast and restores upper breast cleavage. This orientation may create a small deficiency laterally toward the axilla; however, this is the natural position for a lateral concavity of the breast profile and can be corrected later with fat injection, if necessary (▶Fig. 51.15).
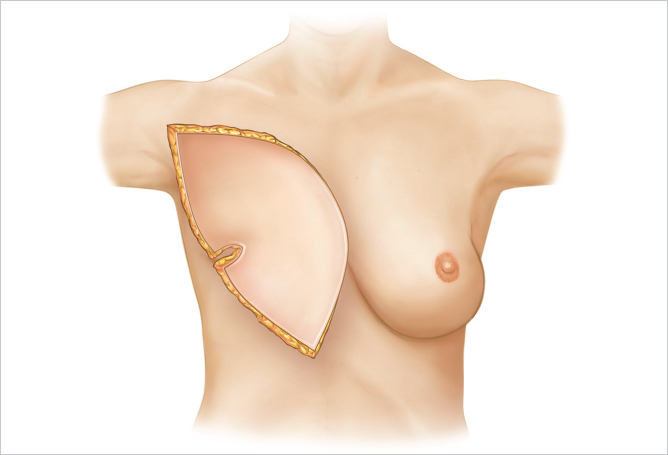
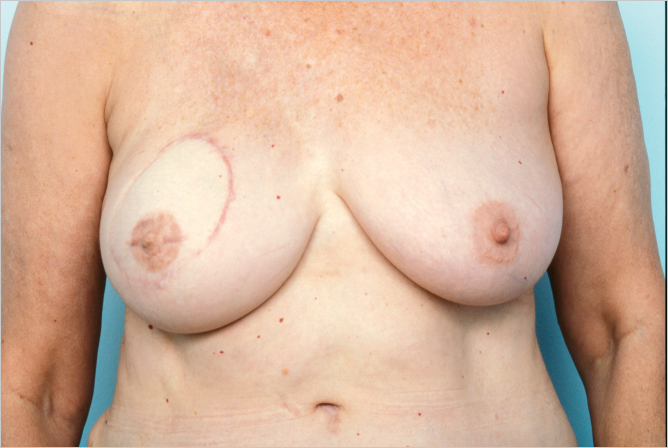
This 58-year-old woman with fairly pendulous breasts and inferolateral fullness had a right mastectomy followed by immediate ipsilateral unipedicle TRAM flap reconstruction. The flap was oriented obliquely to provide her with inferolateral fullness to match the contralateral uncorrected breast as well as providing good superomedial fill (▶Fig. 51.16).
Horizontal or Transverse Orientation
Horizontal or transverse orientation is used for wide breasts with broad underlying chest wall with reduced vertical chest height (▶Fig. 51.17).
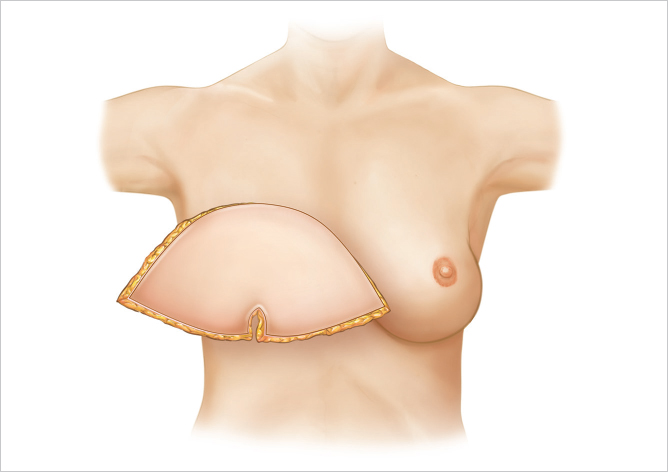
When the patient has significant ptosis, has a wider, broad chest and breast, or does not have a substantial tissue deficiency in the upper portion of her breast, I prefer the horizontal mound-shaping technique. This technique is particularly good for the wider breast and for one with increased fullness in the lower portion. It is also helpful for the woman who has a relatively large abdominal pannus and will have a wide flap—at least 15 cm, as measured from just above the umbilicus to the lower incision at the level of the pubic hair. When the horizontal mound-shaping technique is selected, care must be taken to avoid placing zones II or IV medially. With this approach, the flap is rotated 180 degrees so that in the case of an ipsilateral flap, zones I and II are placed medially. The umbilicus is placed along the inframammary crease and can be closed vertically to give additional breast projection and contour. When the flap is tailored, the subscarpal tissues at the less vascular portions of the flap are trimmed. The tail of the flap can be draped up toward the anterior axillary fold laterally to provide fill in this area.
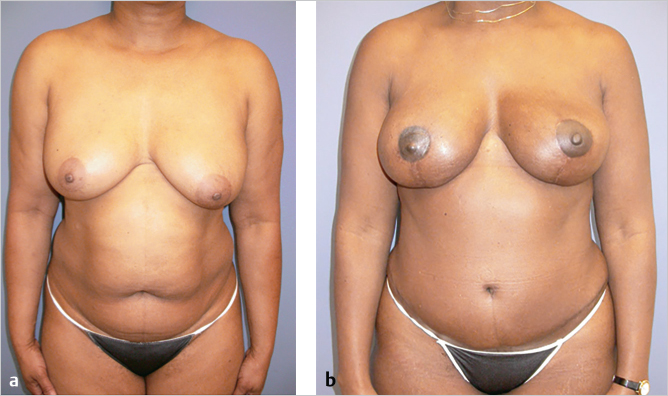
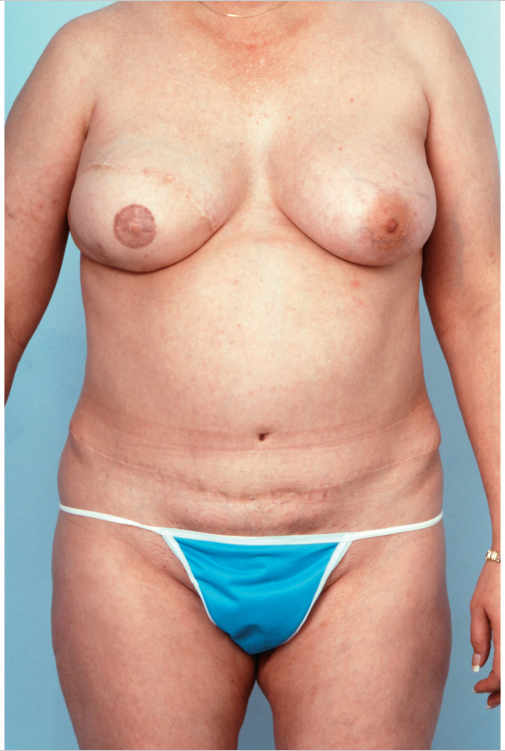
This 45-year-old woman with cancer of the right breast underwent a conventional mastectomy with extensive lower pole skin excision. She was overweight, with a BMI of 31, and had a broad-based breast with a short vertical dimension to her chest. An ipsilateral unipedicle TRAM flap was placed in a transverse orientation, providing her with adequate upper pole fill while addressing her requirement for a wide flap reconstruction (▶Fig. 51.18, ▶Table 51.1).
51.5 Unipedicle TRAM Flap Technique
The unipedicle TRAM flap procedure is selected for lower-risk patients who do not have large volume or surface area requirements. Zone I, the medial part of zone III, and if necessary, a small segment of the medial portion of zone II is used. The flap may be ipsilateral or contralateral. I use the ipsilateral technique now whenever feasible.
Surgical Plan
Preoperative markings to identify tissue deficiency as well as the amount of available lower abdominal tissue.
Recreation of the mastectomy defect.
Incision and elevation of the TRAM flap to the medial and lateral perforators overlying the muscle pedicle.
Incision of overlying fascia as a long vertical ellipse.
Elevation of the TRAM flap to the costal margin, with division of the eighth costal nerve.
Transposition of the flap to the breast.
Direct closure of fascia if it is loose enough, or insertion of mesh or acellular dermal material, followed by abdominal skin closure.
Final shaping and inset of the flap into the mastectomy defect.
51.5.1 Operative Technique
Mastectomy Site Evaluation and Preparation
If the patient is having an immediate reconstruction, in our practice she has usually had a skin-sparing procedure. I evaluate the defect and inspect the bed of the wound to ensure that hemostasis is secure before proceeding with TRAM flap elevation. I also assess the thickness and color of the skin flaps to determine the likelihood of having to resect poorly vascularized tissue in an effort to avoid wound healing problems. If the procedure is delayed, the mastectomy scar is excised and the upper and lower mastectomy flaps are raised off the underlying pectoralis major muscle to create an appropriately sized recipient bed for the flap. I routinely submit the mastectomy scar for pathologic evaluation, although recent data suggest that the likelihood of microscopic recurrence within the scar is less than 1%. In delayed reconstruction I usually resect the entire lower pole mastectomy skin to allow the soft fullness of the TRAM flap to become the new lower pole and inframammary fold.
I used to routinely incorporate ICG laser fluorescence imaging (Novadaq SPY camera system, but other versions such as PDE, PhotonVue exist) in all our skin-sparing mastectomies, because it fairly accurately determines what skin will survive and what is marginally perfused and likely to necrose postoperatively. Any skin that fluoresces poorly is resected back to healthy, bleeding tissue. This is more of a problem in patients with risk factors such as large, pendulous breasts, obesity, and smoking (▶Fig. 51.19). In my current practice I have now replaced ICG imaging completely with near infrared multispectral imaging using the Kent Snapshot hand-held device (Kent Medical Imaging, Calgary, Alberta). The device is less costly and is extremely reliable.
Abdominal Incisions
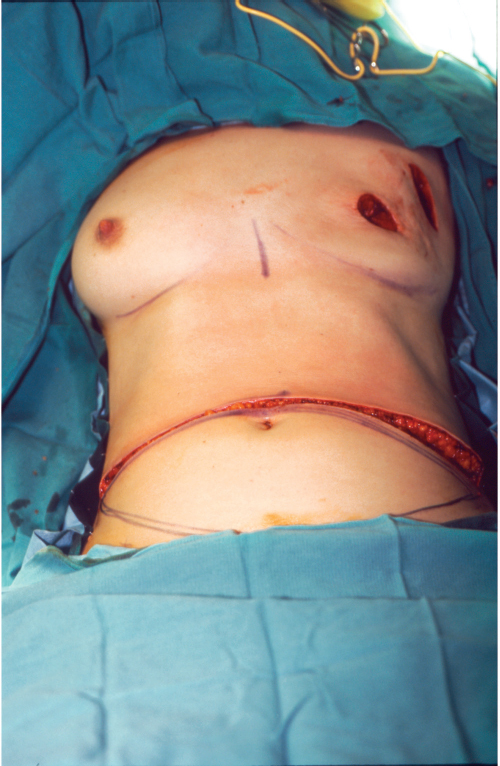
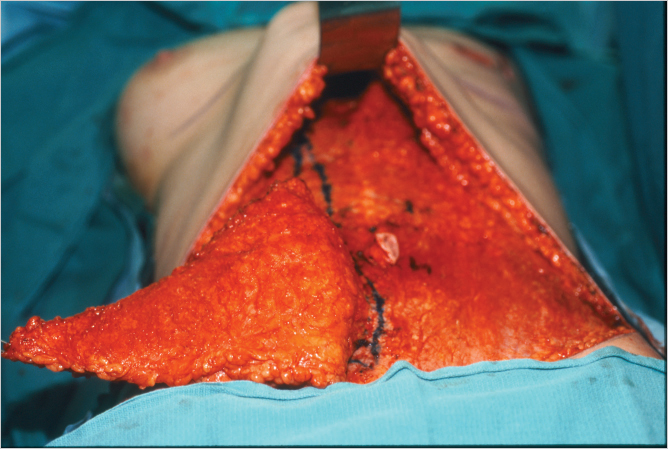
The upper abdominal incision is made first and is beveled in a slightly cephalad direction as dissection is taken down to the rectus fascia (▶Fig. 51.20). This allows potentially more perforators to be incorporated into the flap. The upper abdominal skin flap is elevated to the costal margins laterally and to the xyphoid process centrally (▶Fig. 51.21). The patient is flexed to assess the adequacy of closure to the inferior incision line. The inferior incision may need to be elevated slightly to allow for a less-tense suture line in patients with a long narrow torso.
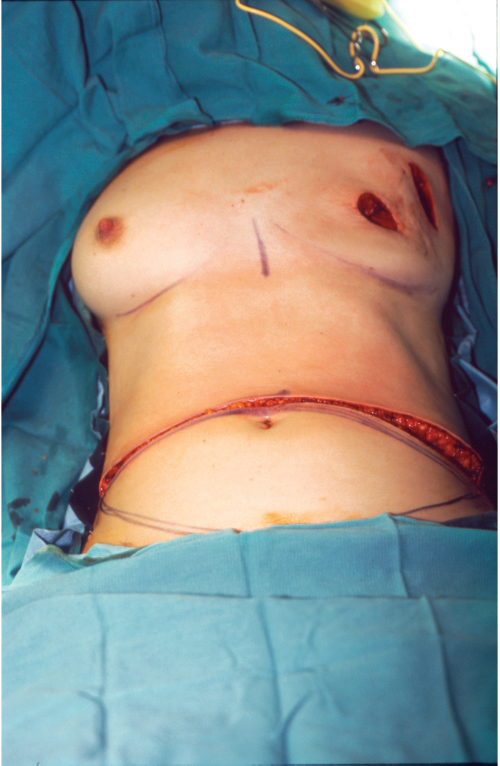
Tight closure can seriously compromise blood flow to the skin edges, causing skin necrosis. Obese patients are particularly at risk of tension-induced necrosis. The Pfannenstiel incisions are routinely ignored. If they happen to fall into the planned lower incision line, I incorporate them, but if they are too low, I simply ignore them. I have never seen the intervening skin between a Pfannenstiel incision and the TRAM donor site closure undergo necrosis. The distal incision is made, and the TRAM flap can be elevated (▶Fig. 51.22).
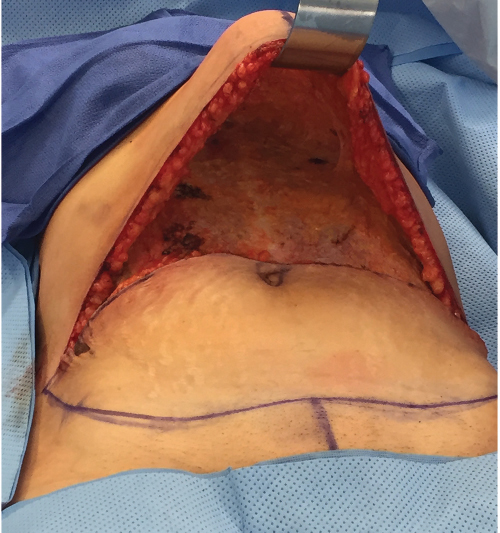
Umbilical Dissection
The umbilicus is incised around its periphery, and I prefer the Avelar technique, using an inverted triangular shape with its base superior. Dissection is carried down around the umbilical stalk, taking care to leave a tapering cuff of fat toward the base to maximize the blood supply to this structure (▶Fig. 51.23).
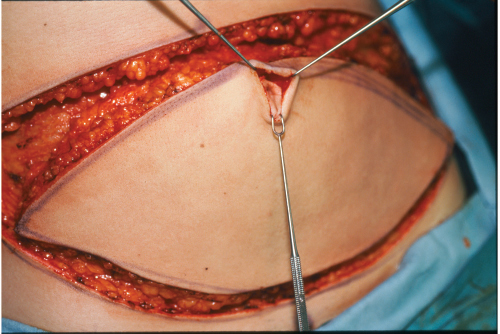
TRAM Flap Elevation
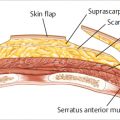
The skin island is elevated from lateral to medial with cautery dissection, identifying first the lateral border of the rectus abdominis muscle, followed by the lateral row of perforators. The perforators are carefully approached with gentle scissor dissection until sizable vessels are reached. Cautery should be used with care in this area, because inadvertent injury to the vessels can mean the difference between success and iatrogenically induced fat necrosis or worse, partial flap loss. The decision as to which side to base the flap on depends on the patient’s abdominal anatomy and the surgeon’s preference (▶Fig. 51.24).
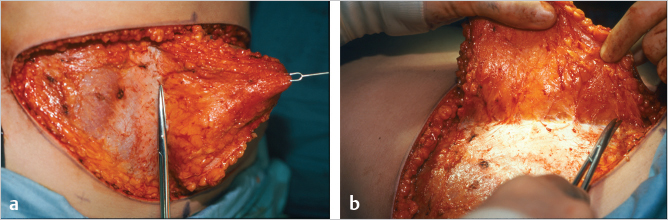
In an unscarred abdomen either side may be used; I prefer the ipsilateral pedicle. Ipsilateral transfer reduces initial intermammary bulging, and the definition of the ipsilateral inframammary crease tends to be excellent. Pedicle tension is reduced and flap positioning is easier. Venous drainage of the flap appears better with ipsilateral transfer, as noted by Clugston and colleagues (▶Fig. 51.25).
The contralateral pedicle tends to create more blunting of the medial inframammary crease and limits the ease of flap positioning laterally. It also places more tenuous cross-midline tissue medially, where reconstructions would be more difficult to correct.
Radiation to the affected breast necessitates either a contralateral unipedicle flap (with or without surgical delay) or preferably a bipedicle or free TRAM flap. Although the ipsilateral irradiated pedicle can be used in many patients, it may be unreliable. Clugston and colleagues suggested that this is not the case, stating that the ipsilateral pedicle performs safely in irradiated pedicles. However, in our series at Emory, we have shown a higher fat necrosis rate in patients with preoperative radiation to the internal mammary supply. A contralateral pedicle is useful in such cases but tends to cause some blunting of the medial inframammary fold and softens the depth of the intermammary space. Careful denervation of the pedicle may ameliorate this problem. A surgical delay performed 5 to 14 days previously improves TRAM flap blood supply and may be considered for a pedicle procedure if a large breast is to be fashioned. If vascular delay is performed, it should include an incision right across the lower inferior end of the TRAM flap with elevation of the flap tips, as described by Taylor and colleagues, and not just two small groin incisions to give access for vascular division. Pedicle selection is based on surgeon preference as well as direct inspection of the visible perforators of the lateral row (▶Fig. 51.26).
Once the flap has been elevated to the lateral perforators on either side, I usually perform another ICG perfusion study to assess the flow to the flap and localize where the best perforator “blooms” exist. Although I usually use the ipsilateral option, on rare occasions, the ipsilateral perforators have clearly been less impressive than those on the contralateral side and then I change to the opposite side for pedicle harvest.
Whichever side is selected, the dissection from the nonpedicle side is then carried across the midline to the medial row of perforators on the pedicle side. This leaves the flap attached only by the perforator rows and a narrow band of fat (▶Fig. 51.27).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree