4 Three-Dimensional Imaging
Summary
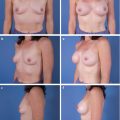
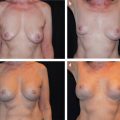
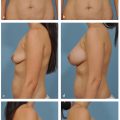
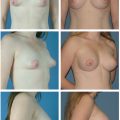
This chapter outlines the utility of three dimensional (3D) surface imaging in primary breast augmentation to enhance preoperative planning and highlight postoperative outcomes.
Key Points
Three-dimensional imaging.
Patient education.
Breast augmentation.
4.1 Introduction
Three-dimensional (3D) surface imaging continues to gain popularity in the field of plastic and reconstructive surgery; this technology quantifies surface topography to enhance preoperative planning and postoperative outcomes. Additionally, this analytical tool identifies and incorporates standardized objective measurements in a preoperative 3D model to simulate postoperative results. This information enhances patient–surgeon communication and satisfaction, reduces operative costs and times, and improves conversion to surgery rates. Although this technology has been validated with various anatomical regions, this chapter focuses on the clinical utility of 3D imaging and analysis in primary breast augmentation.
4.2 The History of Three-Dimensional Imaging
Since the 1800s, clinicians have used two-dimensional photography to document patient features; however, this modality lacks shape and depth. Other imaging tools such as computed tomography (CT) and magnetic resonance imaging (MRI) have attempted to supplement these deficiencies; however, these instruments poorly depict soft-tissue topography; additionally, they are timely, invasive, and cost-prohibitive. 1 Three-dimensional imaging supplements two-dimensional technology and triangulates measurements from surface x, y, and z coordinates to project a 3D image.
Thalmaan first applied 3D technology in 1944 to diagnose orthodontologic conditions. 2 Subsequently, Tanner and Weiner validated 3D imaging by comparing anthropometric measurements to 3D measurements, 3 and Karlan established its use in plastic surgery in 1979. 4 As clinicians continued to develop 3D imaging and analysis, investigators extrapolated qualitative measurements from these photographs and validated the precision and accuracy of this imaging modality. In 1988, Cutting et al developed software to rotate facial images to various views and perform surface measurements at various locations. 5 In the early 2000s, the same concepts were successfully applied to breast imaging to determine breast projection, volume, and contour. 6 After the initial application to various anatomical locations, investigators have since developed a standardized approach to 3D imaging and analysis. 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18
The advent of 3D has paved a foundation to objectively assess postoperative outcomes following aesthetic and reconstructive procedures. Ultimately, 3D imaging may eventually provide a blueprint to influence future surgical techniques. Concurrently, nationwide, clinicians are encouraged to contribute 3D images to a central database. Although in its infancy and thousands of patients are needed to make definitive conclusion, this database could revolutionize outcomes in plastic surgery and predict postoperative trends over time, ethnicity, gender, and so on.
Additionally, software has developed to incorporate objective measurements and implant dimensions into 3D models to simulate postoperative results. Patients may select the size and width of their implants and are able to visualize how different implants would affect their outcomes.
Creasman et al documented that breast augmentation patients felt that their 3D preoperative simulation consultations were essential in helping them select their surgeon and believed the simulations to accurately predict the postoperative results. 19 Not only could patients visualize their simulated outcome but they could also compare this simulation to their true postoperative results at follow-up visits. In a follow-up questionnaire, a majority of respondents indicated that 3D imaging was the “main reason” or “very important” in helping them select their surgeon and believed the simulation to be accurate. 20 Of note, surgical simulations are only an estimate of postoperative results based on the collaboration of engineers and plastic surgeons; the software lacks evidence-based data, specifically the impact of age, body mass index, ethnicity, and gender on surgical outcomes. Ultimately, with nationwide databases and these factors integrated, outcome simulations will improve patient consultation and enhance preoperative planning.
4.3 Imaging Devices
Various 3D cameras are commercially available today including CAM3D, C3D, Axisthree, Canfield, Crisalix, Di3D, and 3dMDface. These imaging modalities use stereo-photography and/or structured light to capture 3D images. Of note, stereo-photography uses multiple cameras arranged as a stereo-pair and merges points of intersection between various images into a 3D breast model. Conversely, structured-light uses a projector to shine a light pattern onto the targeted surface, which distorts and bends the light at an angle that is captured by a camera system and translated into 3D coordinates. CAM3D, C3D, and Axisthree utilize structured-light to create 3D models; Canfield, Crisalix, and Di3D use stereophotogrammetry; and 3dMD combines both. 21 , 22 In addition to the different imaging modality, all systems have variable camera size/shape, capture time, radiographic compatibilities, and cost. Since 3D surface imaging systems differ widely in technique and technology, selection is based on intended application.
Independent of the device used, the clinician should have a standardized approach for image acquisition. Poor standardization will affect reproducible objective measurements. The patient should stand at the recommended distance from the camera lens, and this distance should be recorded for each patient. Hands should be placed at the anterior superior iliac spine; the shoulders should be level and parallel with the hips. Additionally, elbows should be level and tangential with the mid-axillary line. Images should be captured after inspiration with a patient holding his or her breath for 2 seconds. Any irregularities of the spine and chest should be documented. Ideally, a room should be dedicated to image acquisition; appropriate room lighting should be confirmed with the specifications of the device. 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18
Of note, the authors have the most clinical experience with the Canfield system and the remainder of the chapter will be based on the application and utility of this device.
4.4 Imaging Software
After capturing images from various angles, the camera transfers the images to the software, which uses overlapping points to merge the multiple images into one 3D wire frame. Any imaging defects are filled by triangulation and subsequently skin color is projected onto a fluid image. Of note, multiple cameras may be required to capture a 360-degree image of a patient. This model can be rotated, manipulated, and magnified based on the clinical information required. 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 Additionally, images can be cropped to isolate the area of interest. After image acquisition and processing, various measurements (volume, surface area, vector distance, curvature, etc.) can be generated almost instantaneously. Volumetric calculations may have increased variability, as the software must generate an individualized chest wall, and thus a closed object to calculate volume. 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 The software creates the chest wall based on the boundaries of the breast: superiorly where the breast projects from the chest wall, laterally at the most lateral extent of the inframammary fold (IMF), medially at the most medial extent of the IMF, and inferiorly at the lowest pole of the breast. 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 Of note, the chest wall must be inspected by the technician to determine appropriate contour. Devices and software continue to evolve improving the accuracy and precision of these measurements.
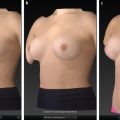
Once the surface of the breast is defined and its volume is known, simulation of breast augmentation with standard implants can be accomplished. Together, the surgeon and patient should use tissue-based preoperative planning to select an implant, and the two can visualize the implant in the 3D model. Of note, a library of different implant shapes and volumes from several manufacturers are typically incorporated in the software. Based on the dimensions of the implant selected, the underlying device can morph the overlying breast to simulate augmentation. The technician or surgeon can reposition the implant in the surgical pocket or select alternate implants based on patient’s preference and surgical expertise. While visualizing the simulation, the surgeon and patient can confirm expectations based on a scientifically proven technology or make appropriate adjustments in the patient’s own picture based on volume and shape to affirm the ideal implant. Previous investigators, including our clinical practice, have verified the accuracy of the preoperative simulation and postoperative images for patient consultation. Three-dimensional imaging enhances the communication between surgeon and patient and allows the patient to choose an implant based on an actual image of her body. 23 In addition, with the resurgence of anatomic silicone implants in the U.S. market, 3D imaging may serve as an adjunct to educate patients in different volume and surface characteristics. This technology has been particularly effective in demonstrating different outcomes between a round and shaped implants for a given patient.
A dedicated patient care coordinator should be designated to review the images with the patient. Minimal but necessary training is required to appropriately use the software to generate accurate measurements and facilitate the consultation. A photography checklist may be useful to guide the discussion; both the coordinator and patient can sign the document to reinforce informed consent. Fig. 4‑1 represents a template photography checklist. The clinical coordinator again should reiterate that 3D photography and analysis by no means guarantees results but rather supplements patient/surgeon discussion.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree