18 Evaluation and Surgical Management of Gynecomastia
Summary
Gynecomastia is very common, with an incidence of up to 50–70% in the general population, and is thus one of the most common reasons for men to seek help from a plastic surgeon. The surgeon must be adept at the evaluation, workup, and surgical management of the disease. Given recent advances in technology, plastic surgeons have more in their armamentarium than ever before. This chapter reviews the disease process and recent evidence to modify the workup, and present a “reconstructive ladder” approach to gynecomastia, which will help the reader assimilate the new technologies into a more cohesive methodology.
18.1 Definitions and Epidemiology
Gynecomastia is defined as a benign proliferation of glandular tissue of the male breast. Pseudogynecomastia, often seen in obese men, refers to fat deposition without glandular proliferation. Gynecomastia is a relatively common entity, with a reported prevalence between one-third and two-thirds of the male population, 1 , 2 and occurs transiently in most infant males, 14–65% of adolescent males, and 57–72% of men aged 50–80 years. 3 , 4 , 5 , 6 It is most commonly bilateral, and it persists beyond 12 months in about 10–20% of males, at which time fibrosis occurs and regression is unlikely. 1 , 7 , 8 , 9 Gynecomastia can be quite anguishing and has been shown to disturb the mental and social health of adolescent and adult males. 10 , 11 , 12 , 13
18.2 Etiology
Imbalances between estrogens, which stimulate mammary gland growth, and androgens, which inhibit it, are responsible for the pathologic changes that occur in gynecomastia. These imbalances can result from a multitude of systemic conditions (Box 18.1) and medications. It is important to note that many historical references identifying drugs as contributing to gynecomastia were based on poor-quality evidence, though medications have been found culpable in up to 25% of cases. A recent systematic review of drug-induced gynecomastia elucidates which medications are supported by the best data and clearest pathophysiology as causing gynecomastia. 14 These medications are summarized in Table 18‑1.
Box 18.1 Pathologic Classification of Gynecomastia
Increased serum estrogen
Increased endogenous production
Leydig or Sertoli cell tumors
Eutopic or ectopic human chorionic gonadotropin–secreting tumors
Adrenocortical tumors
Higher aromatization
Aging
Obesity
Hyperthyroidism
Liver disease
Familial or sporadic aromatase excess syndrome
Klinefelter’s syndrome
Testicular tumors
Refeeding after starvation
Exogenous sources
Topical estrogen creams
Oral estrogen ingestion
Displacement of estrogen from sex hormone–binding globulin
Medications such as spironolactone and ketoconazole
Decreased estrogen metabolism
Cirrhosis
Decreased testosterone synthesis
Primary gonadal failure
Trauma
Radiation
Drugs
Klinefelter’s syndrome
Congenital anorchia
Secondary hypogonadism
Hypothalamic diseases
Pituitary failure
Kallmann’s syndrome
Decreased androgen action
Androgen receptor defect
Antiandrogen drugs
Miscellaneous
Chronic renal failure
Liver disease
Human immunodeficiency virus (HIV)
Chronic illness
Enhanced breast tissue sensitivity
Environmental agents
Embalming agents
Lavender and tea tree oils
Phenothrin in delousing agents
From Beck Do, Rios JL. Gynecomastia. In: Janis JE. Essentials of Plastic Surgery. 2nd ed. New York, NY: Thieme Medical Publishers; 2014; 573–579
18.3 Pathology
On histologic review, gynecomastia is initially described as “florid,” characterized by hyperplasia of ductal epithelium, and increased inflammatory cells, subareolar fat, and blood vessels. This process typically either regresses within the first year or transitions to the “fibrous” state, noted for a predominance of stromal fibrosis with minimal ductal proliferation.
18.3.1 Diagnosis
A careful history must be obtained, focusing on the age of onset of breast enlargement, symptoms of discomfort or tenderness, recreational drug and alcohol use, and the effect that chest feminization has had on the patient’s psychological and social well-being. A review of systems should focus on symptoms from systemic pathologies that contribute to gynecomastia including thyroid, liver, kidney, and adrenal disease, in addition to any malignancies. Physical exam should assess the amount of glandular versus adipose tissue, ptosis, skin access, masses. Abnormal findings on testicular, thyroid, or liver exam should prompt workup specific to that organ system. Overall feminizing characteristics should also raise concern for an underlying endocrinologic derangement. Routine endocrinology workup is not necessary, as a recent study evaluating the utility of endocrinologic referral in the workup of gynecomastia found that only 7.6% of patients had secondary gynecomastia. Referral led to expensive blood tests, which were positive in only 1.7% of cases. The authors concluded that a referral to endocrinology should be considered if an endocrine disorder is suspected on initial evaluation. 8 Findings suggestive of such a disorder include galactorrhea, abnormal testicular examination, features of Klinefelter’s syndrome (small, firm testes, gynecomastia, above-average height, long limbs, and abdominal obesity), visual disturbances, and rapid gynecomastia progression. 15 Men with Klinefelter’s syndrome are not candidates for liposuction, as they have a 58-fold increase in breast cancer risk and thus should be managed with excisional procedures. 16
Finally, a primary goal at the initial evaluation is to screen for characteristics consistent with breast cancer. Tissue in gynecomastia is soft, firm, or elastic, but generally not hard. The area affected arises beneath and extends radially from below the nipple areolar complex (NAC) and is bilateral in most cases. Breast carcinoma, on the other hand, is more typically firm in nature, often outside the NAC, and is most commonly unilateral. Also, skin dimpling, nipple retraction, nipple bleeding, and nipple discharge are factors associated more commonly with breast cancer and should prompt investigation, starting with a diagnostic mammogram and biopsies as indicated. 1
18.3.2 Staging
Simon, Hoffman, and Kahn originally divided gynecomastia into four grades: grade 1 (small enlargement, no skin excess); grade 2a (moderate enlargement, no skin excess); grade 2b (moderate enlargement with extra skin); grade 3 (marked enlargement with extra skin). The authors recommended skin resection in grades 2b and 3. 17 Rohrich et al later submitted a revised classification scheme after the advent of ultrasound-assisted liposuction: grade I: minimal hypertrophy (< 250 g of breast tissue) without ptosis; grade II: moderate hypertrophy (250–500 g of breast tissue) without ptosis; grade III: severe hypertrophy (> 500 g breast tissue) with grade I ptosis; grade IV: severe hypertrophy with grade II or III ptosis. 18 Assessing the stage of the disease is helpful in planning nonoperative and surgical management.
18.4 Management
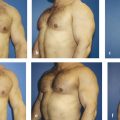
Gynecomastia that persists beyond 12 months in general has progressed to the fibrous state just described and is unlikely to regress. 1 , 17 , 18 Prior to that time, a trial of observation is appropriate. If there is a component of pseudogynecomastia, an attempt at weight loss should also be considered, and if the gynecomastia is found to be medication-induced, stopping the offending agent for a period of time is prudent. The surgical treatments for gynecomastia increase in their level of invasiveness with increasing stage of gynecomastia. Fig. 18‑1 illustrates a stepwise approach to gynecomastia similar to the reconstructive ladder, in which options are evaluated beginning with the simplest and progressing up the ladder to the more complex.
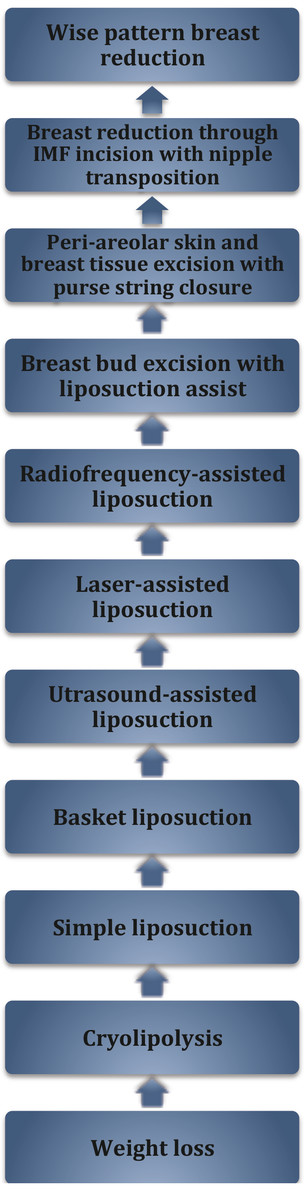
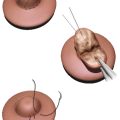
If there is absence of or a very minimal component of glandular hypertrophy in Grade I or II pseudogynecomastia (no skin excess), cryolipolysis (Coolsculpting, ZELTIQ Aesthetics, Pleasanton, CA) can achieve marked improvement. 19 However, most patients will require two sessions, and thus, patient education and managing expectations are key when considering cryolipolysis in pseudogynecomastia. In patients with pseudogynecomastia but with more significant volume (i.e., Grade II or III), liposuction is a better option than cryolipolysis, as liposuction allows for removal of greater volume in one session. The addition of a basket-type cannula may allow more aggressive resection of the subareolar dense, fibrous, glandular tissue in patients with more of a glandular component.
Ultrasound-assisted liposuction (VASER, Solta, Bothell, WA), initially introduced for gynecomastia in the mid 1990s, has gained popularity in many centers due to several advantages. 18 , 20 Compared to traditional liposuction, ultrasound-assisted liposuction has been shown to be associated with lower intraoperative conversion to open excision, improved postoperative pain scores and bruising, smoother breast contour, improved skin retraction, and release of more triglycerides per mL of hemoglobin loss. 18 , 21 , 22 Even in Grade IV gynecomastia (> 500 g breast tissue with grade II or III ptosis), Rohrich and colleagues reported that less than half (43%) required further surgery. 18 Since that study, their group has found that with the addition of a pull-through excision of the glandular tissue through a stab incision at the lateral inframammary fold (IMF), almost no patients require further surgery. 23
Laser-assisted lipolysis (LAL) for gynecomastia, in addition to decreasing breast volume, provides significant dermal tightening and has yielded very good results. 24 , 25 Laser light energy is converted to thermal energy in fat cells, causing them to rupture. Coagulation effects of the laser decrease bleeding complications and stimulate collagen formation in the dermis. 26 In a study of 28 patients, Trelles and colleagues described their method of approach. 27 Initial fibrotic attachments are broken down in cases where the breast is more conical and firm. This is then followed by insertion of a 980-nm wavelength diode laser, with delivery of 8–12 kJ of total average accumulated energy per breast, depending on the breast size. The cannula is first passed in a deep plane, then by a quicker movement in a more superficial plane, aiming for equal disbursement of energy to the dermis. During the laser use, the skin is cooled using an external air flow device, and skin temperature is monitored using an infrared thermometer. Traditional liposuction is then carried out. SmartLipo, a 1064-nm Nd:YAG laser (Cynosure, Langen, Germany), has shown significant improvement in gynecomastia. The thermal treatment can induce skin tightening in addition to more effective tissue separation with liposuctioning following the prescribed amount of energy deployed to break up the dense gynecomastia tissue. 28
Similar to laser-assisted lipolysis, radiofrequency-assisted liposuction (RFAL) also offers the benefit of liposuction with additional soft tissue contraction. 29 RFAL also distributes energy over a greater volume of tissue than LAL, also allowing for contracture of soft tissues, but over a more uniform area, and possibly a lower risk of focal burns. 30 , 31 RFAL has shown promising results for some patients with gynecomastia in one study in Europe. 32
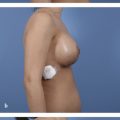
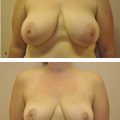
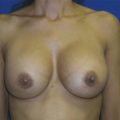
If there is a substantial glandular component that cannot be treated effectively with the techniques just described, a small, crescent-shaped incision at the inferior border of the NAC can be utilized to remove the underlying gland with adjunctive liposuction. 33 Keeping the glandular incision at the edge of the NAC renders the resultant scar practically invisible, and most patients will be satisfied with this. Sometimes a crescent-shaped incision is insufficient to resect enough skin. In such cases, with moderate skin excess, a complete periareolar eccentric skin resection with purse string closure is useful to remove excess skin and correct ptosis while continuing to hide the scar at the edge of the NAC. 34 , 35
If skin excess is severe, a reduction through an incision at the IMF may be required. 36 , 37 The NAC is preserved on a de-epithelialized dermoglandular pedicle and delivered to its new location. The new nipple location is placed at the junction of the fifth rib and the lateral border of the pectoralis major muscle, or at 0.33 times the distance from the sternal notch to the pubis. 38 , 39 Finally, in extreme cases, a breast reduction through a Wise-pattern incision may be necessary (Chapter 12, Reduction Mammaplasty). However, in the senior author’s experience, Wise-pattern reductions or other techniques that produce vertical scars on the chest are not ideal and should be avoided.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree