3 Implant Options
Summary
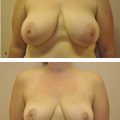
Implant options for breast augmentation have evolved and improved significantly over the last several decades. Plastic surgeons have many different implants from which to choose based on volume, surface type, and shape.
3.1 Implant Background
Breast augmentation is the most common cosmetic surgical procedure performed in the United States, with over 300,000 procedures performed in 2018. 1 Plastic surgeons presently have a large assortment of breast implants to achieve desired aesthetic goals and improve quality of life. We will briefly review the evolution of breast implants and the different implant characteristics available when choosing an implant.
3.2 Implant Evolution
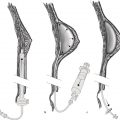
Vincenz Czerny performed the first breast augmentation in 1895, transferring a lipoma to replace a resection defect. 2 Subsequently, surgeons experimented with various solid alloplastic implant materials, such as polyurethane, polytetrafluoroethylene (PTFE), and spongelike material, but patients experienced suboptimal results. 3 , 4 The use of silicone in breast augmentation began disastrously with free silicone injection for breast soft tissue augmentation and resulted in disfiguring long-term effects. 5 In response to complications from free silicone injection, Thomas Cronin and Frank Gerow developed the first silicone implant in 1962 using a thin, smooth silicone elastomer shell surrounding the silicone gel filling materal. 6 , 7 Saline implants were created in 1965 as an alternative to solid silicone implants. Since then, silicone gel–filled implants have undergone several technical and physical modifications. All generations of silicone implants are composed of polydimethylsiloxanes (PDMS), a group of polymers that determines gel cohesiveness with varying degrees of cross-linking.
3.3 Silicone Implants
See Table 3‑1 for an overview of five generations of silicone breast implants.
3.3.1 Generations of Silicone Implant
First Generation (1960s)
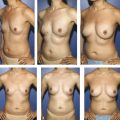
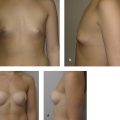
The first silicone implant was an anatomically shaped (teardrop) implant with several Dacron fixation patches on the posterior aspect to maintain implant position in situ. The shell was a thick, smooth two-piece silicone elastomer envelope with a peripheral seam enclosing a moderately viscous shell. This implant generation was associated with a relatively high contracture rate secondary to the shell properties and the lack of gel cohesivity (Fig. 3‑1).
Second Generation (1970s)
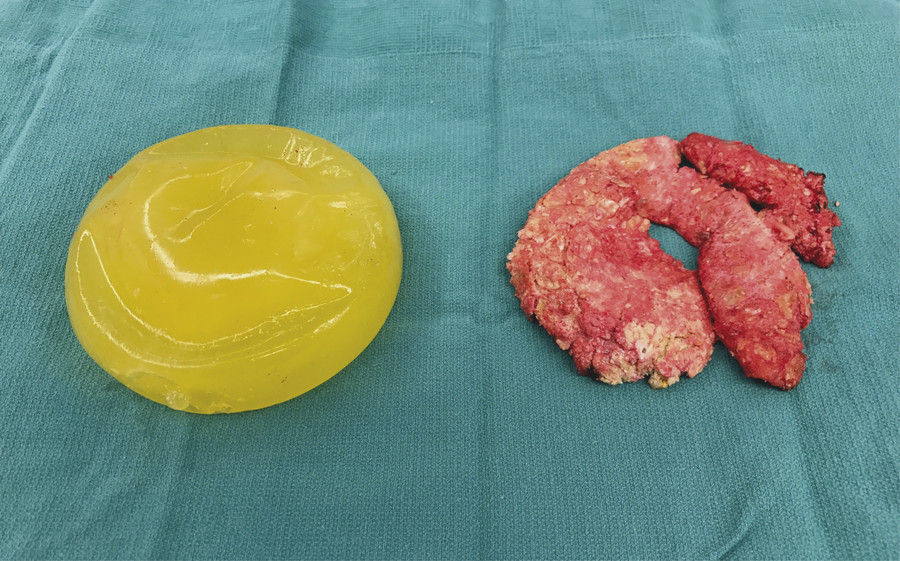
A round implant with a thinner, seamless elastomer shell without Dacron fixation patches was developed to reduce the rate of capsular contracture. 8 A less viscous silicone gel filling of this implant generation provided a “natural feel.” Unfortunately, these implants were associated with “gel-bleed” as the low viscosity of the gel permitted diffusion of microscopic silicone molecules through the thin, permeable shell. 9 An oily, sticky residue surrounding the implant within the periprosthetic capsule is characteristic during explantation of second-generation implants. Device shell failure was also higher in this implant generation. 10 , 13
Third Generation (1980s)
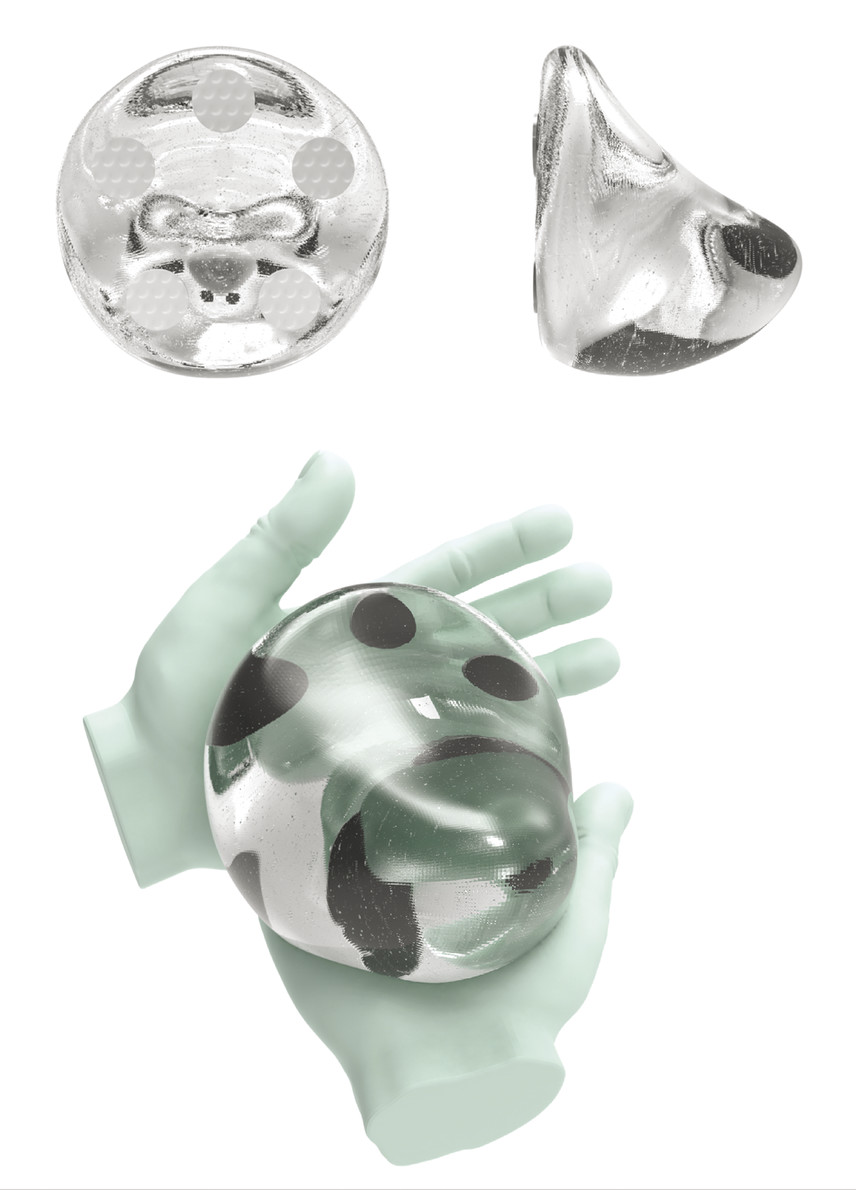
Gel bleed and device shell failure were reduced in third-generation implants, when a more viscous gel and a thicker, multilayer silicone elastomer shell with barrier layer were introduced. For the first time, textured surfacing was added to implants as an attempt to decrease the capsular contracture rate. From 1992 to 2006, the FDA placed a moratorium on third-generation implants due to an unfounded claim that silicone caused cancer. 11 , 12 They were also still subject to rupture and contracture (Fig. 3‑2).
Fourth Generation (1990s to Present)
These implants improved on the previous generation by being manufactured under stringent FDA quality control and criteria for device shell thickness and greater gel viscosity. 13 Implants were also made available in a greater variety of textures and shapes.
Fifth Generation (1990s to Present)
These “form-stable” implants were produced to provide a natural contour, unlike the fullness created with round implants. 14 , 15 , 16 Fifth-generation implants are also defined by greater gel cohesion and textured surface (Fig. 3‑3). 17

3.3.2 Indications
Breast augmentation with permanent silicone implants is indicated in women at least 22 years old who acknowledge the FDA-required recommendation of magnetic resonance imaging (MRI) surveillance 3 years postimplantation and then every 2 years thereafter.
3.3.3 Shape and Projection
The goal of breast augmentation is to achieve aesthetically pleasing proportion, shape, and volume. To that end, implant selection is just one of a number of decisions, which include preoperative evaluation (Chapter 17), selection of surgical approach (Chapter 4), and implant placement (Chapter 5). The decision regarding implant shape selection is influenced by factors that include implant cohesivity, degree of implant overfilling, projection, breast analysis including upper and lower pole breast parenchyma coverage of the implant, and proposed vertical placement of the implant. 18 , 19
Another component of implant shape selection is determined by three additional components: patient’s desires, anatomy, and surgical history. The importance of understanding the subjective appearance the patient desires cannot be underestimated. It may be prudent to show photographs of results of other patients or use three-dimensional imaging tools to align expectations of the patient and the surgeon. A patient who desires a natural appearance, one that has a linear or slightly concave upper pole, may prefer an anatomic-shaped implant to mimic the normal breast. The anatomic-shaped implants provide the desired contour owing to their “form stable” silicone formulations. Generally, the implant is tapered at the top, and there is more projection at the bottom. For patients who request an “augmented” appearance, round implants will provide greater volume for a given base width and tend to provide upper pole fullness. In both cases, the patient’s anatomy must be considered during implant selection.
Anatomic factors that influence implant shape selection are breast shape and soft tissue coverage, lower pole constriction, breast asymmetries, chest wall deformities, and ptosis. Broadly speaking, round implants are indicated when a patient has a good baseline breast shape and adequate soft-tissue coverage (> 2 cm) to camouflage the implant edge. Aesthetic outcomes may not vary greatly according to implant shape, particularly when low- or moderate-profile implants are placed. In comparison, anatomic-shaped implants are indicated when one or more of the following conditions is present: 20
Lack of breast shape or thin soft tissue coverage (< 2 cm).
Lower pole constriction or tuberous breast, to expand the lower pole and minimize double-bubble deformity.
Breast and chest wall asymmetries in which differential customization can correct shape and volume.
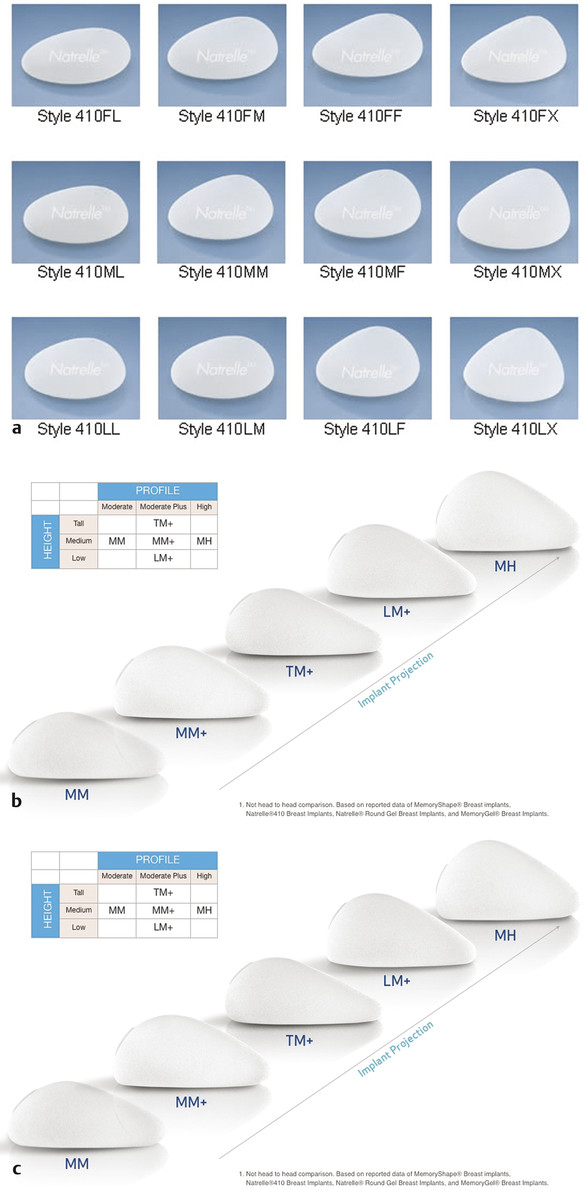
Ptosis that can be corrected with an anatomical implant to elevate the nipple areolar complex (NAC) and augment the lower pole, given the array of implants with different height and projection combinations.
The last consideration is the patient’s surgical history. Round implants should be used in cases of recurrent implant rotation, situations where precise implant pocket control cannot be obtained, and instances that require low-cohesive implants to mold into the implant pocket after capsular contracture correction.
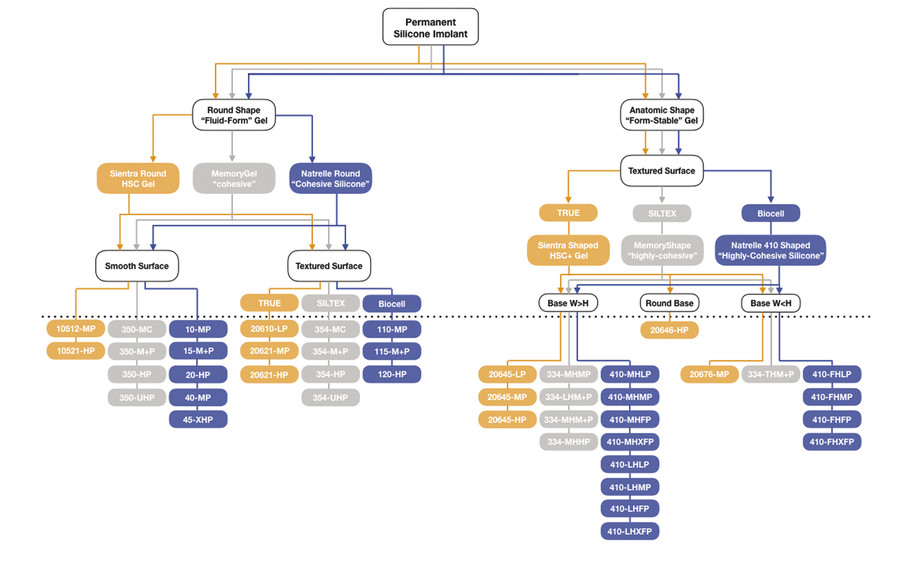
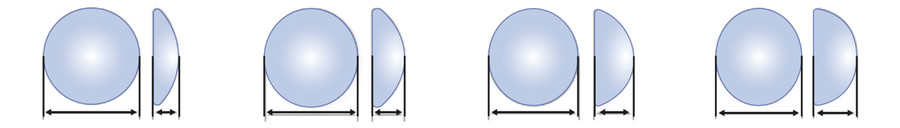
Altogether, implant shape selection is dependent on several factors that rely on preoperative assessment and combined patient- and surgeon-directed goals. In the United States, surgeons may chose round and anatomic-shaped implants made by Allergan, Mentor, and Sientra. Each company offers implants in a range of different base width diameters that permit custom sizing (Fig. 3‑4). As a response to early round implants, higher-profile options were created to achieve greater projection (Fig. 3‑5). Thus, for a given base width diameter, the greater projection profiles allowed fill volume to increase with each implant device. All three companies offer anatomic-shaped implants with different projection profiles (Fig. 3‑6). Similarly, these companies produce implants with taller and lower heights relative to the base width. However, Sientra is the only company that offers a round-based anatomic implant.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree