5 Implant Placement Options
Summary
Current approaches to breast augmentation are diverse and controversial. A long-standing topic of discussion is whether to place a breast implant in the subglandular, subfascial, or subpectoral position. Selection of the ideal location is often based on patient and breast characteristics but, interestingly, has geographic variability from a global perspective. Decision making often depends on the size of the natural breast, the patient’s degree of physical activity, and an appreciation of patient expectations. With all options, breast enhancement is readily achieved, adverse events are infrequent, and patient satisfaction is high. This chapter will review many of the salient aspects of subglandular, subfascial, and subpectoral breast augmentation with an emphasis on technique and outcomes.
5.1 Introduction
Deciding upon the ideal plane for breast augmentation has been a long-standing topic of debate and controversy. Options for implant placement include the subglandular, subpectoral, and subfascial locations. Each of these planes has advantages and disadvantages. Clearly, enhancement of volume and contour are achieved with all; however, the plane of implant placement can affect other outcomes such as capsular contracture, animation deformity, rippling, and wrinkling, as well as malposition. The intent of this chapter is to review the options for implant position in cosmetic breast augmentation.
5.2 Anatomy
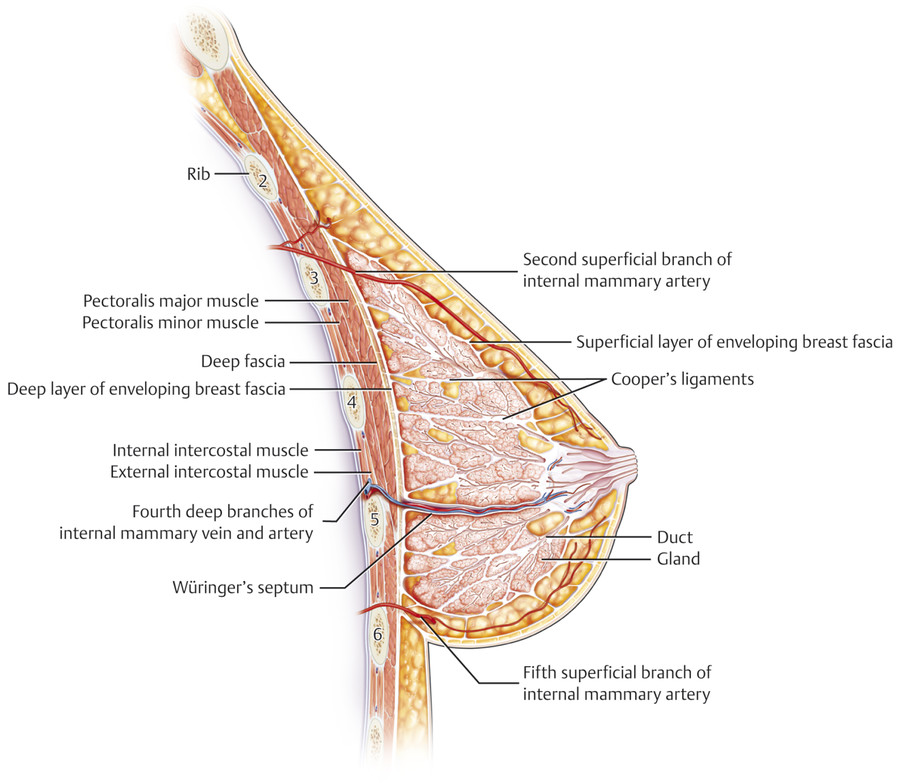
The relevant anatomy of the breast as it relates to breast augmentation will be reviewed. The natural borders of the breast include the inframammary fold (IMF), anterior axillary line, medial sternal border, and the clavicle. The breast is composed of parenchyma, which includes the lobules and ducts, as well as the subcutaneous layer, which ranges from 1 mm to 3 cm in thickness with a median thickness of 1 cm. 1 The subcutaneous layer gradually and naturally becomes thinner as it approaches the nipple areolar complex (NAC). Other components of the breast include the ligaments of Cooper (pectineal ligaments), the deep and superficial layers of the superficial fascia, as well as the pectoralis major fascia. The diameter of the areola is highly variable and can range from 3 to 8 cm.
The vascularity of the breast is another important concern in considering the planes of dissection. The primary blood supply to the breast is derived from the perforating branches of the internal mammary artery (IMA). Van Deventer has demonstrated that of the six perforators emanating from the medial costal interspaces and arising from the IMA, the third perforator was most commonly seen, followed by the second, the first, and the fourth. 2 Many of these perforators form an anastomotic cluster around the periareolar region and enhance the vascularity to the NAC. These anastomoses are primarily formed from the lateral thoracic and internal mammary perforators. Le Roux demonstrated that the IMA perforators traverse superficially 1 cm under the skin surface. 3 A dominant vessel originates from either the third or the fourth intercostal space with a depth of 10.3 mm at the NAC boundary and 14.2 mm at 3 cm medially. Würinger demonstrated that the second, third, and fourth perforating branches of the IMA coursed within the medial vertical ligament of the breast toward the nipple. 4 The remainder of the vascular supply to the breast is derived from the lateral thoracic, anterior intercostal, thoracoacromial, posterior intercostal, and superficial thoracic arteries. It should be noted that although the vascularity of the breast is generally predictable, embryologic variation could result in vascular unpredictability of the NAC.
Anatomic studies have demonstrated that the innervation to the breast is derived from the first through seventh intercostal nerves and that the innervation to the NAC is derived primarily from the fourth intercostal nerve. 5 , 6 The dominant innervation of the NAC is derived from the lateral and anterior cutaneous branches of the third, fourth, and fifth intercostal nerves (Fig. 5‑1). Schlenz has demonstrated that the most constant innervation patterns were from the fourth lateral (79%) and the third and fourth anterior cutaneous branches (57%). 5 The specific pathways within the breast for these sensory nerves were relatively constant. The anterior cutaneous nerve branch coursed superficially within the subcutaneous tissue and terminated at the medial border of the NAC, whereas the lateral cutaneous branches coursed deep within the pectoral fascia and approached the NAC from its posterior surface.
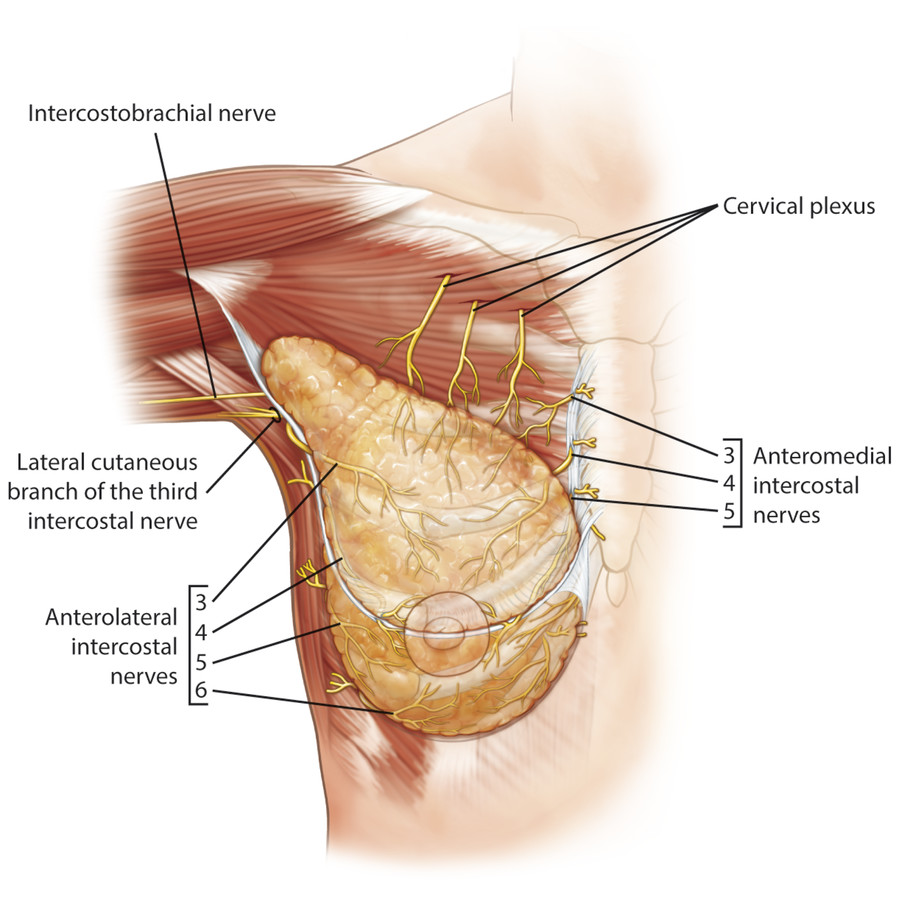
Beneath the breast parenchyma are the pectoralis fascia and the pectoralis major muscle (Fig. 5‑2). The origin of the pectoralis major muscle is from the sternal segment of the clavicle, the anterior surface of the sternum extending to the sixth and seventh ribs, and the aponeurosis of the external oblique muscle. The insertion is to the bicipital groove of the humerus. The medial and lateral pectoral nerves innervate the pectoralis major muscle. Understanding of this innervation is especially important when breast implant–associated animation deformity becomes problematic. The medial and lateral pectoral nerves enter the pectoral space from lateral to medial. The medial pectoral nerve pierces the pectoralis minor muscle before innervating the pectoralis major muscle. The lateral pectoral nerve pierces the pectoralis major muscle directly. Additional anatomic studies have demonstrated that the branches of the lateral and medial pectoral nerves penetrate the pectoralis major several centimeters from the lateral edge of the sternum, thus minimizing the risk of injury to the medial pectoral nerve during breast augmentation. 7
Anatomic studies depicting the relationship between the pectoralis major and the external oblique muscles have demonstrated that the costal portion of the pectoralis major originates from the fifth (25%), sixth (70%), or seventh rib (5%). 8 The distances from the midclavicular line to the lateral border of the pectoralis major muscle at the fourth, fifth, and sixth ribs were 49.8 mm, 30.5 mm, and 6.3 mm, respectively. The pectoralis major and the external oblique muscles overlapped near the midclavicular line in 90% of patients, and the width of the overlap was about 25 mm.
With the advent of subfascial breast augmentation, the anatomy of the pectoral fascia has become important. The benefits of preserving the pectoral fascia–parenchyma interface are several but include retention of the Cooper’s ligament attachments as well as the horizontal septum of Würinger. Anatomic studies have demonstrated that the pectoral fascia originates superiorly and medially from the clavicle and sternum and extends over the surface of the muscle. 9 Lin and others have demonstrated that the thickness of the pectoral fascia ranges from 0.2 to 1.1 mm. 9 Tebbetts demonstrated that the thickness ranged from 0.1 to 0.5 mm, 10 whereas Graf found that the thickness of the pectoral fascia was approximately 0.2 mm centrally and 1.0 mm laterally and medially. 11
5.3 Patient Selection
Patient selection for breast augmentation begins with a complete history and physical examination. Included in this are a thorough assessment of patient expectations and an understanding of the risks and benefits. These issues were explained in detail in prior chapters. The discussion relating to the ideal plane of dissection will depend in part on physical examination and surgeon preference. Patient selection for glandular and subfascial device placement are essentially the same because the device is superficial to the pectoralis major muscle for both planes. However, selection criteria for subpectoral placement may differ in some cases. In general, the subpectoral position is typically recommended in patients with a thin body habitus and an upper pole pinch test < 2 cm, whereas the subglandular or subfascial plane can be considered in patients with an upper pole pinch thickness > 2 cm.
5.4 Advantages and Disadvantages
The subglandular position of a breast implant results in ideal projection; however, in thin patients, implant visibility in the form of rippling and wrinkling as well as edge visibility may be problematic. 12 , 13 When the thickness of the upper pole of the natural breast is > 2 cm, the likelihood of implant visibility and rippling is minimized. The subglandular approach is sometimes recommended in women with larger breasts or in women who are more athletic to minimize distortion and compression associated with pectoralis contraction. With a subglandular dissection plane Cooper’s ligaments are divided, and the likelihood of glandular ptosis over time may be increased.
With the advent of the newest generation of silicone gel implants, including the optimally filled and highly cohesive implants, visibility in thin patients can be minimized. There are a variety of anatomic implants that can minimize upper pole visibility and reduce the incidence of rippling and wrinkling, even when in the subglandular position. Because these devices have different handling characteristics than the traditional less cohesive silicone gel implants, their use in the subglandular position may be considered in thinner patients.
The subpectoral position of a breast implant will generally improve the contour of the upper pole of the breast, especially in patients with a pinch test < 2 cm, because the upper pole of the device is naturally compressed by the muscle and less visible. In addition, studies have demonstrated that the incidence of capsular contracture is least when implants are placed in the subpectoral plane; however, the use of a textured-surface device placed in the subglandular position will mimic that of subpectoral position. 14 Studies have also demonstrated that breast imaging, namely mammography, is facilitated when implants are in the subpectoral position because the parenchyma–pectoral muscle interface is more clearly visualized. 15 A limitation of subpectoral implant placement is the development of animation deformity associated with voluntary contraction of the pectoralis major muscle. 16 Other disadvantages associated with subpectoral placement of implants include malposition, distortion, asymmetry, and distortion. 13 Banbury evaluated the function of the pectoralis major muscle in 47 patients following subpectoral augmentation and did not detect any change in muscle flexion, extension, or adduction from preoperative to 3 and 6 months postoperative. 17
The subfascial position has the advantage of providing improved projection, similar to the subglandular but without the edge visibility, because the upper pole of the implant is under the pectoral fascia. This plane has the additional benefit in that it separates the implant from the breast parenchyma and is associated with a reduced incidence of capsular contracture than in subglandular positioning, with an incidence similar to subpectoral placement. 18 In addition, it has been theorized that since Cooper’s ligaments are retained, glandular ptosis over time may be minimized; however, there have been no studies to corroborate this.
5.5 Technique
With all three planes, the markings and the incision locations can be the same. Typical markings include delineation of the sternal midline and the peripheral outline of the breast footprint. The level of the IMF is delineated and extended to the midline to ensure equal position. The distance from the NAC to IMF on and off stretch, as well as the sternal notch–to-NAC distance, are measured. The upper pole pinch test is performed to assess the thickness of the soft tissues and facilitate the decision between subglandular/subfascial and subpectoral placement. In a recent review of 1,067 plastic surgeons in the United States, Hidalgo has demonstrated that the most common pocket locations are the partial submuscular or dual plane (79.5%), complete submuscular (12.7%), subglandular (5.4%), and subfascial (2.4%). 19 These percentages vary globally, as subfascial placement is more common in Europe and South America. The incision choices for subpectoral, subglandular, and subfascial include the inframammary, periareolar, and transaxillary locations. In the Hidalgo review, the most common incision choice in the United States was the inframammary (83.9%), followed by the periareolar (12.6%), transaxillary (3.3%), and periumbilical (0.2%). 19
5.5.1 Subglandular Augmentation
Cronin and Gerow performed the first breast augmentation procedure with silicone gel implants in 1962 and them in the subglandular position. 20 Although the subglandular placement of implants often results in excellent volume and contour with an easier recovery, disadvantages include implant visibility, especially in patients with thin tissues, and an increased incidence of capsular contracture. 21 Some surgeons have questioned the longevity of the breast shape with implants in the subglandular space, because implant ptosis is a potential problem, as the majority of the connective fibers between the breast parenchyma and the pectoralis fascia are disrupted with the subglandular dissection (Hunstad). 7
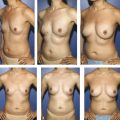
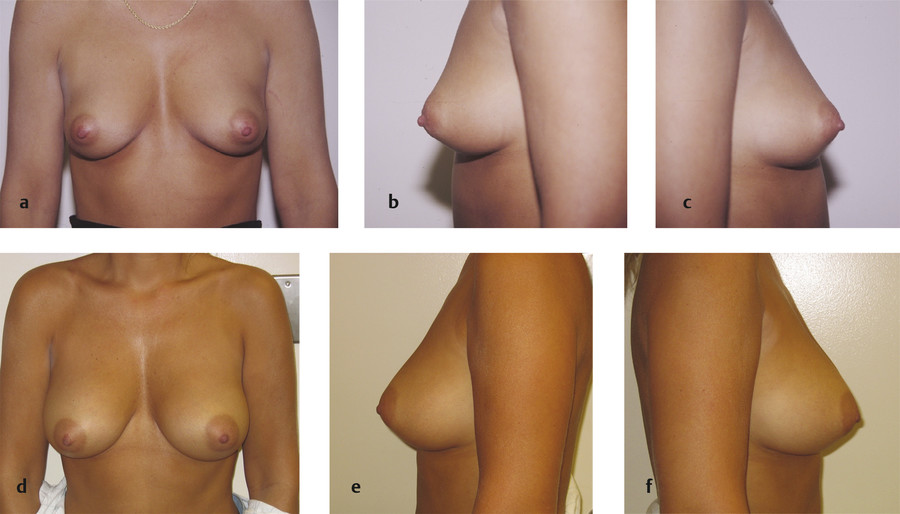
The salient details of the subglandular augmentation technique will be described. With the patient standing, the relevant breast landmarks are delineated and include the breast footprint and the proposed incisions. The incisional approach can be through the periareolar, inframammary, or transaxillary regions. The incision length varies from 3 to 5 cm, depending upon the size of the implant. The inframammary approach is the most common and will be profiled. A scalpel is used to create the incision through the dermis to the subcutaneous fat. Double-pronged skin hooks are used to separate the dermal edges, and electrocautery is used to dissect to the level of the fascia. At the level of the fold, the fascia may be that of the rectus abdominis or the pectoralis major. A narrow 90° retractor is used to elevate the upper soft tissue, and the dissection is continued in a cephalad direction. The inferior edge of the pectoralis major is identified. The dissection continues medially, superiorly, and laterally above the pectoralis major muscle and the pectoralis fascia. The borders of the dissection plane include the medial border of the pectoralis, the upper footprint of the breast parenchyma, and the lateral border of the pectoralis major. All perforating branches within the plane of dissection are identified and cauterized before bleeding occurs. It is important not to expand the pocket excessively but rather to create a space that will result in a hand-in-glove fit with the implant itself. Hemostasis is achieved, and the subglandular space is irrigated with an appropriate solution. Fig. 5‑3 illustrates a patient before and after subglandular augmentation.
5.5.2 Subfascial Augmentation
Graf et al initially described the subfascial technique in 2002. 11 It is important to recognize that the benefit of subfascial placement is not to provide additional thickness between the skin and the implant; its real benefit is twofold, as it provides a barrier function between the breast parenchyma and the implant in addition to providing additional support to the implant by preserving Cooper’s ligaments, which attach breast parenchyma to the chest wall. The pectoral fascia essentially functions as an internal bra to support the implant and minimize its inferior migration, which is typically associated with subglandular placement. Advocates claim to achieve an enhanced aesthetic outcome with less upper pole visibility of the implant, a reduced incidence of capsular contraction, and less animation of the implant. 11 , 21 , 22 As previously mentioned, the pectoralis fascia is thin and varies in thickness based on the specific segment. Thickness measurements are 0.49 mm superiorly, 0.68 mm laterally at the margin of the muscle and axillary fascia, and 0.52 mm at its attachments to the sternal margin over the second to fourth ribs. Inferiorly, it condenses into the fascia overlying the latissimus dorsi muscle. Despite the thin nature of the pectoral fascia, it can be dissected as a firm and distinct layer with structural integrity. The dense attachment of the pectoral fascia at the upper aspect of the dissection can prevent upward displacement of the implant, provide compression of the upper pole of the breast, and reduce superior implant visibility. 23
Graf has described the technical aspects of subfascial breast augmentation based on the location of the incision, which includes the transaxillary, periareolar, and inframammary. 24
With axillary access, a 4-cm, S-shaped incision is created along the axillary fold approximately 1 cm behind the lateral border of the pectoralis major. Care must be exercised to avoid damage to the lymphatics in the region by directing the dissection upward toward the superolateral edge of the pectoralis major. The pectoralis fascia is incised, and the subfascial pocket is dissected using electrocautery. The fascial incision should be created perpendicular to the direction of the pectoral muscle fibers. 21
When the periareolar incision is selected, it is important to mark the exact location of the proposed incision along the inferior areolar edge with the patient standing. 21 , 24 Once the patient is supine and prepped and infiltrated with local anesthetic, the areolar edge may be less distinct. The fascial incision is created at the vertical level of the midclavicle a few centimeters away from the NAC to minimize the risk of injury to the sensory nerves. 21 The dissection toward the IMF is directed along the parenchyma–subcutaneous fat plane. The transglandular approach is avoided to minimize injury to the ductal elements of the breast, which often harbor bacteria. The dissection extends to the chest wall and then extends cephalad toward the pectoralis major muscle. The pectoralis fascia is incised at the level of the NAC.
When the inframammary approach is selected, a 3- to 5-cm incision is created along the crease. 24 In cases where the level of the IMF needs adjustment, the location of the incision is modified accordingly such that the final incision lies within the crease. The dissection progresses toward the inferior edge of the pectoralis major muscle. Once the pectoralis fascia is identified, it is incised and the subfascial plane is dissected.
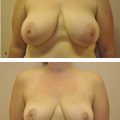
The actual creation of the subfascial pocket requires attention to several salient points. 24 During the undermining process, care should be exercised to avoid fascial injury such as thermal injury and perforation. In cases where it is very thin, inclusion of thin slips of pectoral muscle fibers may be elevated with the fascia. The dissection is performed using a low-set electrocautery device in coagulation mode to minimize any potential bleeding. Finger dissection can also be used to develop the subfascial plane and eventually to allow the insertion of an endoscope to facilitate the dissection. 21 Stoff-Khalili advocates for the dissection of breast tissue to begin at the lateral border of the pectoralis muscle and continue to the level of the pectoral fascia. 18 Once the fascia is identified, it is separated from the muscle using cauterizing scissors. It was noted that the thickness of the fascia increased from approximately 0.1 mm inferiorly to approximately 0.5 mm in the upper third of the pectoralis major muscle. According to Stoff-Khalili, this increase in thickness is sufficient to minimize implant rippling. Other maneuvers to facilitate the dissection include upward traction of the breast. The typical boundaries for the dissection include the second intercostal space superiorly, 1–2 cm from the midsternum medially, 5–8 cm below the NAC, and the anterior axillary line laterally. It is important to avoid dissection beyond the lateral border of the pectoralis major muscle to prevent lateral displacement of the implant. 21 It is also important to ensure hemostasis and to irrigate the pocket prior to implant insertion. Fig. 5‑4 illustrates a patient before and after subfascial augmentation.
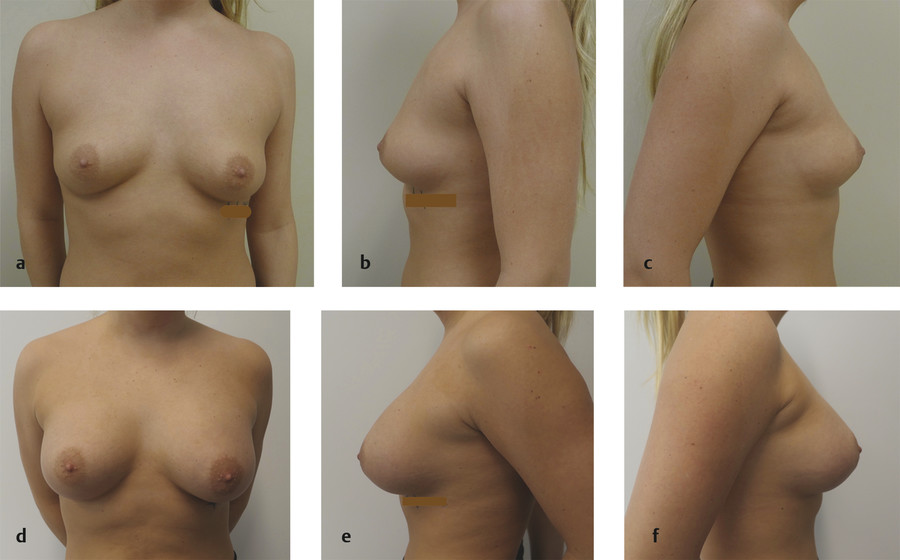
5.5.3 Subpectoral Augmentation
Dempsey and Latham were the first to consider the subpectoral position and performed the first subpectoral breast augmentation in 1968. 23 The advantages included less visibility, decreased ptosis, reduced capsular contracture, and increased longevity compared to subglandular placement. Like the subglandular and subfascial techniques, the technique of subpectoral augmentation can also be performed through a variety of access incisions that include the periareolar, transaxillary, and inframammary. Subpectoral augmentation can be performed using a total muscle coverage approach as well as partial muscle coverage. The total submuscular plane has been performed less frequently, primarily because the natural contour of the breast may be compromised and because it sometimes involves elevation of the serratus anterior muscle as well for lateral coverage. Indications for total muscle coverage are few. Partial subpectoral coverage has become far more common. The release of the inferior origin of the pectoralis major muscle allows improved lower pole projection and contour. Another benefit of the subpectoral positioning is that capsular contracture rates are generally lower compared to subglandular placement; however, this will also be dependent upon the surface texturing of the implant. 14
The evolution of partial subpectoral coverage has been refined and more accurately defined by Tebbetts and is now commonly referred to as dual-plane breast augmentation. 28 Dual-plane augmentation involves the release of the inferior origin of the pectoralis major muscle without release of the muscle along the sternal border, as well as varying degrees of subglandular dissection. The defining characteristics of dual-plane augmentation, according to Tebbetts 26 :
The breast implant is partially behind the pectoralis major muscle and partially behind the breast parenchyma.
A specific portion of the origin of the pectoralis major muscle is divided to alter the position of the pectoral muscle relative to the implant, with the ultimate goal of altering the relationship between the implant and breast parenchyma.
The parenchyma–pectoralis major interface is sufficiently altered to change the dynamic nature of the parenchyma–implant relationship.
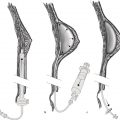
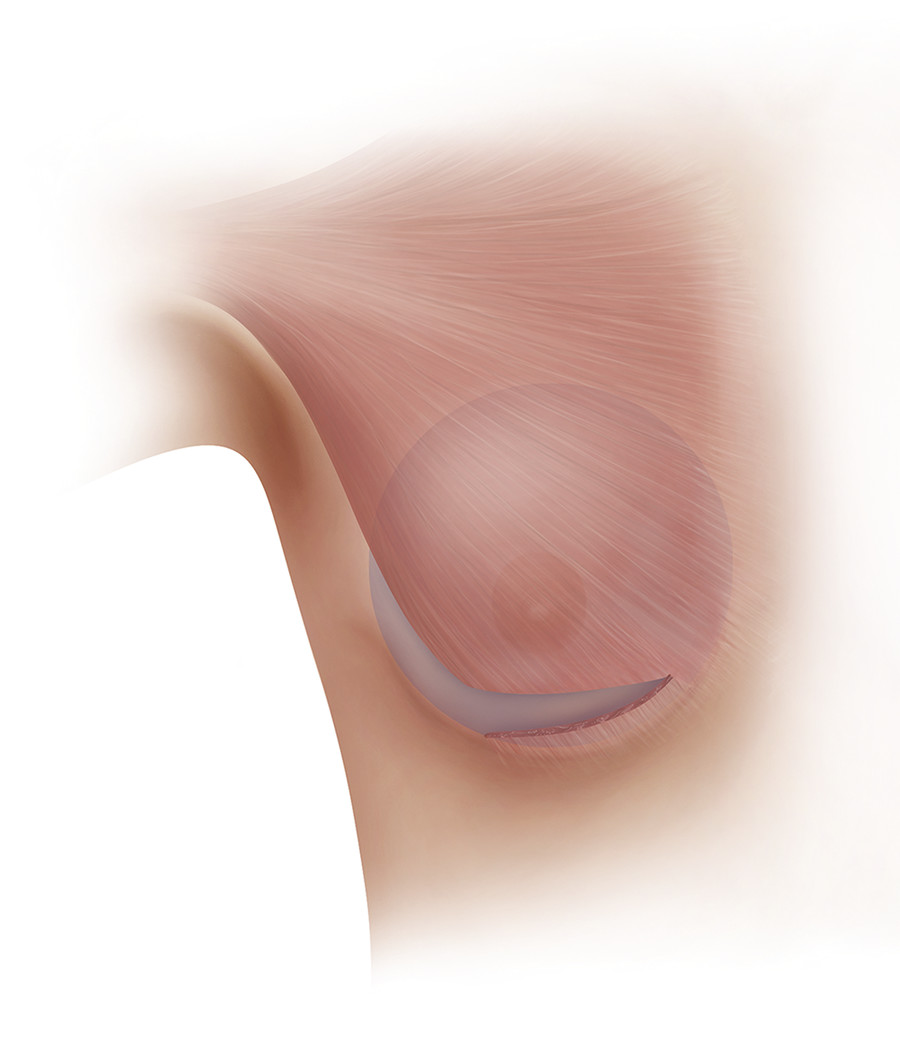
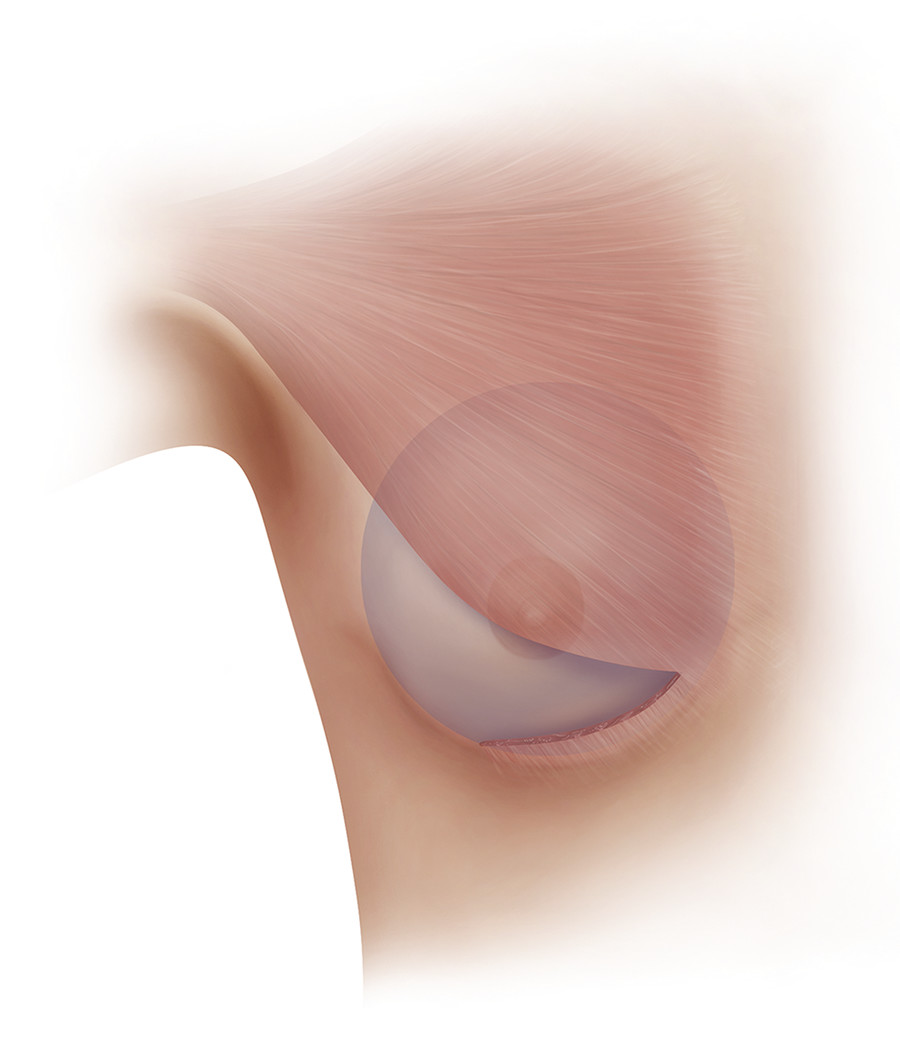
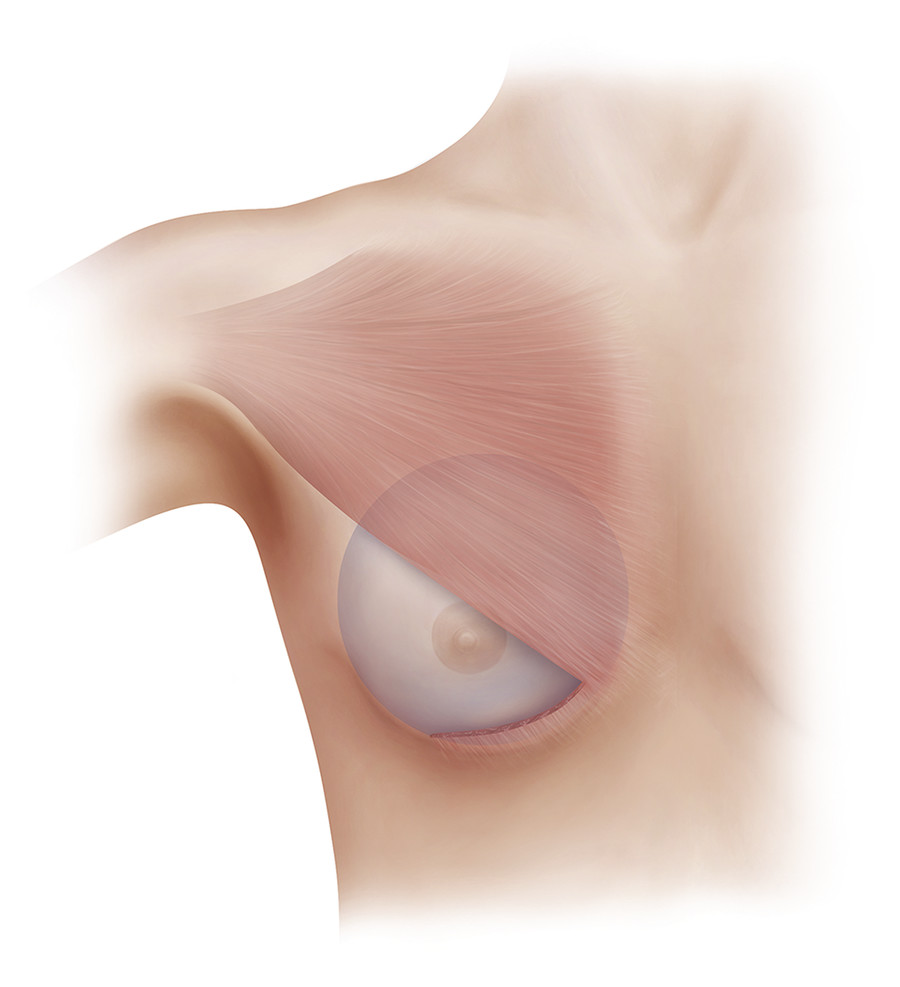
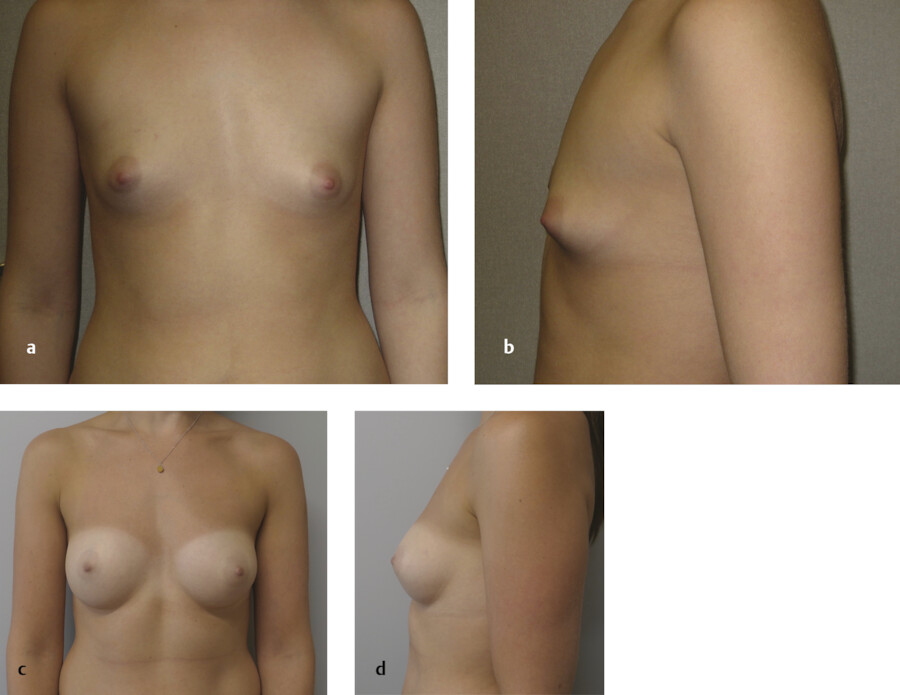
Three types of dual-plane breast augmentation have been described. 26 Dual-plane 1 is defined as the release of the inferior pectoral origin of the pectoralis major muscle only and is indicated for patients without glandular ptosis, tight attachments of the parenchyma–muscle interface, and a minimally stretched lower pole (Fig. 5‑5). Dual-plane 2 is defined as a release of the inferior origin of the pectoralis major and a subglandular dissection to the level of the inferior border of the areola and is indicated for patients with minimal glandular ptosis, looser attachments of the parenchyma–muscle interface, and a lower pole that has more stretch (Fig. 5‑6). Dual-plane 3 is defined as a release of the inferior origin of the pectoralis major and a subglandular dissection to the level of the superior border of the areola and is indicated for patients with glandular ptosis in which more than one-third of the parenchyma is below the desired IMF, loose attachments of the parenchyma–muscle interface, a markedly stretched lower pole, as well as a constricted lower pole (Fig. 5‑7). Extensive medial and superior release of the pectoralis major muscle can result in upward retraction of the muscle, known as window-shading, and may lead to animation deformity. 27 Overdissection along the medial origin of the pectoralis major may also result in symmastia as the soft-tissue attachments over the sternum gradually weaken, eventually resulting in implant–implant contact. 27
The salient aspects of the subpectoral technique are as follows. Preoperative markings are made with the patient standing. As with other implant planes, the incisional approach can be periareolar, transaxillary, or inframammary. When the inframammary approach is selected, the skin and subcutaneous fat are incised and dissected to the level of the deep fascia. Often this is the fascia of the rectus abdominis muscle. The suprafascial dissection is advanced to the inferior edge of the pectoralis major muscle, which is then incised extending from the inferomedial origin to the midclavicular line. Fig. 5‑8 illustrates a patient preceding and following subpectoral augmentation. At this point the three variations of the dual plane dissections are considered.
Dual-Plane 1
The subpectoral plane is entered, leaving a 1-cm cuff of the inferior pectoralis major on the chest wall. The pectoral incision extends from the inferior sternal border of the pectoralis major to the lateral inferior border. The pectoralis major–parenchymal interface is not separated. It is best to enter the subpectoral space along the medial aspect of the areola. The subpectoral dissection is performed with electrocautery. Care must be exercised to identify and cauterize the perforating branches of the internal mammary artery and vein as they pierce the pectoral fascia and the costal interspaces. The pectoralis major is separated from the pectoralis minor and the serratus anterior muscle using blunt dissection technique. The dimensions or footprint of the created subpectoral pocket should approximate the dimensions of the desired implant. The original description of the dual-plane technique recommended that the superior extent of the dissection proceed to the level of the thoracoacromial vascular pedicle; however, this degree of dissection is no longer recommended. The lateral dissection should not extend beyond the lateral edge of the pectoralis major. The medial dissection should extend to the medial origins of the pectoralis major without complete division to avoid future symmastia.
Dual-Plane 2 and 3
The technique for the dual-plane 1 and 2 procedures begins exactly as it does for dual-plane 1. Once the subpectoral pocket is created, upward traction on the parenchyma is applied and the pectoral–parenchymal interface is identified. An electrocautery device is then used to separate the interface extending from medial to lateral. The lateral limit is the lateral edge of the pectoralis major muscle. With the type 2 dual-plane, the dissection extends approximately to the inferior border of the areola, whereas for the type 3 dual-plane the dissection extends approximately to the superior border of the areola. The muscle–parenchymal interface should be released until no muscle banding is appreciated internally that could potentially restrict implant projection.
5.6 Complications
Adverse outcomes associated with breast augmentation, based on the plane of implant placement, are varied and include capsular contracture, animation deformity, implant malposition, compromised sensation, and soft tissue alterations. The incidence of these adverse outcomes can be influenced by the plane of insertion and must be appreciated. This ensuing section will review these adverse events and how devices placed in the subpectoral, subfascial, and subglandular positions can influence the incidence of these adverse events.
5.6.1 Capsular Contracture
When comparing the subglandular and subpectoral position of breast implants, one of the proposed advantages of subpectoral placement is a reduction in the incidence of capsular contracture. Although implant location is considered by many to be the most important predictor of this event, the surface characteristics of the implant are also important. In a review of 5,109 breast implants placed in the setting of breast augmentation, the incidence of capsular contracture varied based on plane of insertion and surface texture of the implant. 16 When textured devices were placed, the incidence of capsular contracture was 4.9% for subglandular and 2.1% for subpectoral. When smooth-surfaced implants were used, the incidence of capsular contracture was 21% when subglandular and 5.1% when subpectoral. Numerous studies have demonstrated that subpectoral placement of implants is associated with a reduced incidence of capsular contracture. 9 , 21 , 28 , 29 Although this relation is generally accepted, Codner et al were unable to demonstrate any difference in capsular contracture rates between subglandular and subpectoral placement. 30
Complications related to subglandular and subpectoral placement of implants have been studied. In a review of 328 patients following breast augmentation, Stoff-Khalili demonstrated that postoperative complications occurred in 49% of patients with subglandular augmentation and in 16% of patients following subpectoral augmentation (p < 0.001). On the other hand, from the subpectoral cohort to the subfascial cohort the incidence of complications decreased from 16% to 6.7% (p < 0.031). The incidence of Baker grade 3 capsular contracture was 14.35% with textured subglandular implants, 4.5% with textured subpectoral implants, 1.9% with smooth subpectoral implants, and 1.5% with smooth subfascial implants. In the subglandular cohort, revisions were performed in 37% of patients. Eight patients with either a Baker 3 or 4 capsular contracture had explantation, capsulotomy, and no reaugmentation. Also within this subglandular cohort, fourteen patients with Baker grade 3 capsular contracture (13.3%) had explantation, capsulectomy, and a new smooth implant placed in the subpectoral position. In the subpectoral cohort, the rate of secondary revisions was 9.1% compared with 37% for the subglandular cohort (p < 0.05). 18 A criticism of this study was that for any comparative study such as this to have scientific validity, patient cohort samples must be similar preoperatively. 31
The correction of capsular contracture following breast augmentation in the subglandular position has been described. In a review of 85 patients with capsular contracture (54 subpectoral, 31 subglandular) by Spear et al, correction was achieved using a dual-plane strategy. 16 The subglandular contracture patients were managed by pocket conversion to a partial subpectoral space, with the inferior edge of the pectoralis major muscle being stabilized to the overlying skin with marionette sutures. The subpectoral contracture patients were managed by releasing the inferior origin of the pectoralis major muscle and creating a partial subglandular plane that extended to the inferior border of the areola. Three of the 85 patients (3.5%) required reoperation for complications due to implant malposition. There were no Baker grade 3 or 4 capsular contractures noted in patients who were converted to the dual plane position; however, 2% were graded as Baker grade 2.
Mofid et al described a technique whereby the subglandular implant is removed and a capsulectomy is performed. 32 A dual-plane or subpectoral pocket is created by incising the inferior origin of the pectoralis major muscle, dissecting the subpectoral plane, using an acellular dermal matrix (ADM) to stabilize the position of the pectoral muscle, and placing a new implant. They compared outcomes with this technique to the traditional pocket conversion without ADM described by Spear and demonstrated that adverse events using ADM were significantly fewer based on implant displacement, pain, and need for revision.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree