Antiviral
Population
Route
Age/weight
Dosing
Acyclovira,b
Eczema herpeticum
PO
All ages
15 mg/kg/day (400 mg maximum) in 3–5 divided doses for 10–14 days or until all mucocutaneous lesions are healed
Acyclovira,b
Eczema herpeticum
IV
<12 years
30 mg/kg/day in 3 divided doses for 10–14 days or until all mucocutaneous lesions are healed
Acyclovira,b
Eczema herpeticum
IV
≥12 years
15 mg/kg/day in 3 divided doses for 10–14 days or until all mucocutaneous lesions are healed
Acyclovira,b
Mucocutaneous; first episode
PO
All ages
40–80 mg/kg/day in 3–4 divided doses for 5–10 days (maximum pediatric dose 1,000 mg/day)
Acyclovira,b
Mucocutaneous; first episode
PO
≥12 years
1,000–1,200 mg/day in 3–5 divided doses for 7–10 days
Acyclovira,b
Mucocutaneous; first episode
IV
All ages
15 mg/kg/day in 3 divided doses for 5–7 days
Acyclovira,b
Mucocutaneous; recurrent
PO
≥12 years
1,000 mg/day in 5 divided doses for 5 days, or 1,600 mg/day in 2 divided doses for 5 days, or 2,400 mg/day in 3 divided doses for 2 days
Acyclovira,b
Mucocutaneous chronic suppressive
PO
All ages
30 mg/kg/day in 3 divided doses for up to 12 months; maximum daily dose 1,000 mg/day; re-evaluate after 6–12 months
Acyclovira,b
Mucocutaneous chronic suppressive
PO
≥12 years
800 mg/day in 2 divided doses for up to 1 year
Famciclovira,b
Recurrent herpes labialis
PO
Adult
1,500 mg as a single dose
Famciclovira,b
Genital herpes; first episode
PO
Adult
750 mg/day in 3 divided doses for 7–10 days
Famciclovira,b
Genital herpes; recurrent
PO
Adult
2,000 mg/day in 2 divided doses for 1 day
Famciclovira,b
Genital herpes; chronic suppression
PO
Adult
500 mg/day in 2 divided doses for up to 1 year, then reassess for recurrence
Valacyclovira,b
Recurrent herpes labialis
PO
>12 years
4,000 mg/day in 2 divided doses for 1 day
Valacyclovira,b
Genital herpes; first episode
PO
Adult
2,000 mg/day in 2 divided doses for 10 days
Valacyclovira,b
Genital herpes; recurrent
PO
Adult
1,000 mg/day in 2 divided doses for 3 days
Valacyclovira,b
Genital herpes; chronic suppression
PO
Adult
500 mg daily for <10 outbreaks/year or 1,000 mg daily for ≥10 outbreaks/year, then reassess for recurrence
Immunocompromised children should also be treated with oral or intravenous acyclovir depending upon the severity of the case (Table 18.2) [5, 6]. Topical acyclovir may accelerate healing of lesions in immunocompromised children (Table 18.3). Acyclovir resistance can occur and foscarnet is a potential treatment [7].
Table 18.2
Antiviral dosing guidelines for herpes simplex virus in the immunocompromised host
Antiviral | Population | Route | Age | Dosing |
|---|---|---|---|---|
Acyclovira,b | Mucocutaneous initial infection | PO | ≥2 years | 1,000 mg/day in 3–5 divided doses for 7–14 days; maximum dose for child 80 mg/kg/day |
Acyclovira,b | Mucocutaneous initial infection | IV | All ages | 30 mg/kg per day in 3 divided doses for 7–14 days |
Acyclovira,b | Mucocutaneous chronic suppresion | PO | ≥2 years | 600–1,000 mg/day in 3–5 divided doses during period of risk |
Acyclovira,b | Mucocutaneous chronic suppresion | IV | All ages | 15 mg/kg in 3 divided doses during period of risk |
Famciclovira,b | Herpes labialis; recurrent in setting of HIV infection | PO | Adult | 1,000 mg/day in 2 divided doses for 7 days |
Famciclovira,b | Genital herpes; recurrent in setting of HIV infection | PO | Adult | 1,000 mg/day in 2 divided doses for 7 days |
Famciclovira,b | Genital herpes; Chronic suppression in setting of HIV infection | PO | Adult | 1,000 mg/day in 2 divided doses during period of risk |
Foscarneta,b | Infection resistant to acyclovir | IV | Adult | 80–120 mg/kg per day in 2–3 divided doses until infection resolves |
Valacyclovira,b | Genital herpes; recurrent in setting of HIV infection | PO | Adult | 2,000 mg/day in 2 divided doses until all mucocutaneous lesions are healed |
Valacyclovira,b | Genital herpes; chronic suppression in setting of HIV infection | PO | Adult | 1,000 mg/day in 2 divided doses during period of risk |
Table 18.3
Topical antiviral dosing guidelines for herpes simplex virus
Antiviral | Population | Route | Age | Dosing |
|---|---|---|---|---|
Acyclovir | Recurrent herpes labialis | 5 % topical cream | ≥12 years | Apply 5 times daily |
Acyclovir | Initial genital herpes; localized HSV | 5 % ointment | Adult | Apply 6 times daily |
Docosanol | Recurrent herpes labialis | 10 % cream | ≥12 years | Apply 5 times daily |
Ganciclovir | Ocular herpes | 0.15 % gel | ≥2 years | Apply 1 drop 5 times daily until epithelial healing occurs, then 3 times daily for 1 week |
Penciclovir | Recurrent herpes labialis | 1 % cream | ≥12 years | Apply every 2 h while awake |
Trifluridine | Ocular herpes | 1 % solution | ≥6 years | Instill 1 drop every 2 h for a total of 8–9 doses daily for 1–2 weeks |
Specific Investigations Recommended
For diagnosis |
Viral culture |
Polymerase chain reaction (PCR) |
Direct fluorescent antibody (DFA) |
Tzanck smear |
Serologic testing |
Herpes Labialis
Clinical Features
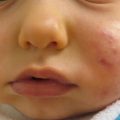
Herpes labialis generally refers to HSV infection on the lips and is the most common form of recurrent herpes infection. Primary infection may be subclinical or appear as gingivostomatitis (refer to above section “Herpetic Gingivostomatisis”). Symptoms typically begin with a prodrome of tingling, burning, or itching. Around 1–2 days later, a localized cluster of small vesicles on an erythematous base appear. As the vesicles rupture, they form painful erosions, which crust over in about 8 days. Other areas of the face may be involved (Fig. 18.1). Immunosuppressed children can develop severe, chronic disease, which may become resistant to antivirals. The diagnosis is generally determined clinically, but can be confirmed by laboratory investigations. The sensitivity of these tests is generally regarded as lower with recurrent episodes since viral shedding is intermittent and declines rapidly as lesions heal.
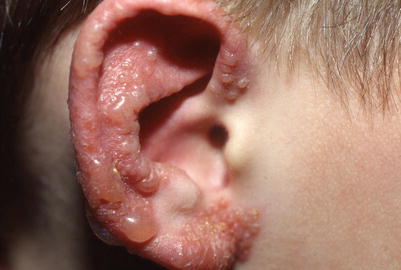

Fig. 18.1
Clustered vesicles of HSV-1 on the ear
Management Strategies
Herpes labialis may self-resolve and require no intervention. Symptomatic treatment includes oral analgesics, petroleum jelly, and topical antibiotics to decrease risk of bacterial superinfection. As with herpes gingivostomatitis, other topical treatments such as “magic mouthwash” or topical anesthetics are not routinely recommended in children. Zilactin, a nonprescription topical medication containing lidocaine and hydroxypropyl cellulose, can protect lesions from trauma and irritants, but studies regarding its efficacy and safety in young children are lacking. Topical benzocaine may cause methemoglobinemia and should not be used in children younger than 2 years.
For recurrent herpes labialis, episodic therapy with antivirals may be beneficial if initiated at first sign of symptoms. Topical antivirals, such as acyclovir, penciclovir, and docosanol can modestly decrease the time to lesion healing, although are not approved in children <12 years (Table 18.3) [8–10]. Oral acyclovir may provide greater benefit than topical antivirals if treatment is initiated during the prodromal stage; benefits include decreased duration of symptoms and time to healing (Table 18.1) [11, 12]. For those ≥18 years with recurrent disease, valacyclovir or famiciclovir can be considered [13, 14]. There are no clinical trials directly comparing antiviral medications. For immunocompromised children, oral acyclovir can be considered; however, those with spreading or persistent infection should be treated with intravenous acyclovir (Table 18.2) [15–17].
Although there are no studies for prophylactic therapy in children, antivirals should be considered for frequent recurrences, immunosupression, and serious systemic complications such as erythema multiforme and eczema herpeticum (Table 18.1 and 18.2) [18, 19]. To reduce frequency and severity, antiviral treatment should be continuous for several weeks to months. After approximately 6 months to 1 year of treatment, acyclovir should be discontinued and the recurrence rate re-evaluated. UV protection may also help reduce viral reactivation [20].
Genital Herpes
Clinical Features
Genital herpes is a widespread sexually transmitted disease primarily caused by HSV-2, but HSV-1 is increasing in prevalence, especially amongst young adults. Primary genital herpes demonstrates painful vesicles on an erythematous base 2–20 days after exposure. Distribution includes the vulva, labia, vagina, perineum, penile shaft, glans penis, urethra, and less often the scrotum; extragenital involvement may also be seen. Painful erosions or ulcers occur following rupture of vesicles and can be associated with burning, pruritus, vaginal or urethral discharge, and regional lymphadenopathy. Influenza-like systemic symptoms are also common and include fever, malaise, myalgias, and headache. Symptoms generally improve over 5–7 days, and cutaneous lesions heal over 2–4 weeks. Aseptic meningitis and sacral radiculomyelitis are potential complications.
The diagnosis is generally determined clinically, but can be confirmed by laboratory investigations. Viral culture, direct fluorescent antibody (DFA) and Polymerase chain reaction (PCR) have varying sensitivities and specificities. PCR is currently regarded as both a rapid, sensitive, and specific method and is increasingly used when available. The diagnosis of genital HSV in a child should prompt question of sexual abuse.
Recurrent genital herpes can be trigged by menstruation, febrile illness, or stress. Outbreaks are typically preceded by a prodrome of pain, itching, and paresthesia. Compared to primary genital herpes, recurrent lesions are less severe, with fewer vesicles. Shedding of the virus can occur even without active lesions (subclinical shedding) and increases with frequency of symptomatic recurrences.
Management Strategies
The treatment of genital herpes includes supportive care and antivirals to promote rapid healing, decrease recurrences, decrease viral shedding, and minimize complications [21, 22]. There is no curative therapy.
For primary genital infections, supportive care includes analgesics and Sitz baths. Systemic antiviral therapy should be initiated promptly, even before laboratory confirmation. Antiviral dosing for immunocompotent patients is provided in Table 18.1. Oral acyclovir initiated within 6 days of onset of disease shortens the duration of illness and viral shedding by 3–5 days [23]. Acyclovir is the only antiviral approved for genital herpes in children, but famciclovir and valacyclovir are well studied in adults; they do not seem to be more effective than acyclovir, but allow for less frequent dosing [24, 25]. Topical acyclovir should not be used, as it is less effective than oral acyclovir.
Antivirals can also be used for recurrent episodes (Table 18.1) and have been shown to decrease duration of lesions and viral shedding and increase proportion of aborted episodes [26–29]. The prescription should instruct patients to initiate treatment immediately when symptoms begin.
Prophylactic therapy has not been studied in children, but in adults, daily oral antiviral suppressive therapy is effective in decreasing the frequency of symptomatic recurrences and improving quality of life [30–32]. Thus, children with frequent recurrences may benefit from continuous oral acyclovir, and a reasonable starting dose in provided in Table 18.1. After approximately 6 months to 1 year of treatment, acyclovir should be discontinued and the recurrence rate re-evaluated.
Immunocompromised patients have more frequent recurrences, develop more severe lesions, and require longer treatment periods (Table 18.2). Intravenous acyclovir should be administered for severe or disseminated cases. Long-term suppressive therapy has been used, although may lead to selection of resistant strains of the virus. In acyclovir-resistant cases, intravenous foscarnet may be required [33].
Evaluation for other sexually transmitted diseases should be considered, and potential sexual abuse cases must be referred to the appropriate investigative agencies. The recurrent nature of the disease can have significant psychological effects on patients; education about the risk of transmitting genital herpes and safe sex practices should be provided [34].
Specific Investigations Recommended
For diagnosis |
Viral culture |
Polymerase chain reaction |
Direct fluorescent antibody |
Tzanck smear |
Serologic testing |
Skin biopsy of atypical lesion |
Ocular Herpes Infection
Clinical Features
Ocular herpes infection can result in numerous complications including blepharitis, conjunctivitis, keratitis, uveitis, and acute retinal necrosis. HSV keratitis typically appears as an infection of the superficial layer of the cornea (epithelial keratitis) while deeper involvement of the cornea (stromal keratitis), uvea, and retina are associated with a greater risk of visual loss.
HSV keratitis has an acute onset with pain, photophobia, blurred vision, and watery discharge. Physical exam is notable for conjunctival injection, decreased corneal sensation, and characteristic dendritic lesions of the cornea. The eyelid may also be involved, leading to blepharitis (Figs. 18.2 and 18.3). Infections may be unilateral or bilateral, and bacterial superinfection is not uncommon. The diagnosis is generally determined clinically, but can be confirmed by laboratory investigations. Ocular samples should be taken from scrapings of epithelial lesions.
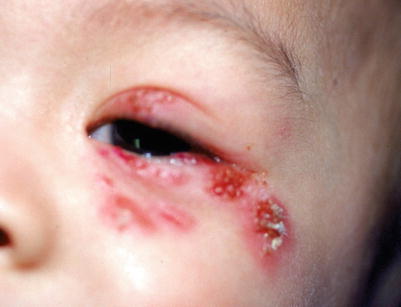
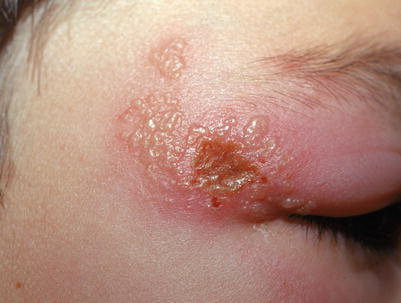

Fig. 18.2
Grouped erosions in a periocular distribution in a patient with recurrent herpes keratitis

Fig. 18.3
Clustered vesicles with central erosion and dramatic eyelid edema from HSV
Children typically have more severe cases then adults, with higher levels of inflammation, risk of amblyobia, and estimated recurrence rates of 50 %. Recurrent HSV keratitis continues to be a leading cause of corneal blindness in the United States.
Management Strategies
Management depends upon whether the infection is primary or recurrent and the location of the infection within the eye [35]. From a dermatologist’s perspective, the most important action is to promptly consult an ophthalmologist, especially if there is facial involvement in a child with disseminated mucocutaneous herpes.
For primary infection involving the superficial layer of the cornea (epithelial keratitis), topical antiviral therapy was previously the standard of care, but randomized trials in adults have also shown oral antivirals to be equally effective [36, 37]. Oral antivirals may be superior, given difficulty of topical antiviral application with small children. Topical ganciclovir 0.15 % gel and trifluridine 1 % drops are potential antiviral treatments (Table 18.3). Oral acyclovir has been reported to be effective in children with ocular herpes, although the study did not evaluate if oral acyclovir should be used in lieu of topical antiviral medications [36]. Topical glucocorticoids should be avoided as they can exacerbate epithelial lesions.
For deeper involvement of the cornea (stromal keratitis), a combination of topical corticosteroids and antivirals is currently the standard of care [38]. The addition of oral acyclovir to these two treatments has not been shown to provide additional benefit. Topical cyclosporine may be considered for those non-responsive to topical prednisolone, although this has only been studied in adults [39].
The most important management issue in HSV ocular infection is prevention of recurrences, as these can lead to corneal scarring and blindness. Suppressive acyclovir has been successful and well tolerated in children and should be considered, given the high rate of recurrence.
Specific Investigations Recommended
For diagnosis |
Viral culture |
Polymerase chain reaction |
Direct fluorescent antibody |
Enzyme-linked virus inducible system (ELVIS) |
Table 18.15
First line therapies
Epithelial Keratitis | |
Topical ganciclovir | A* [37] |
Topical trifluridine | A* [37] |
Oral acyclovir | |
Stromal Keratitis | |
Topical corticosteroids | A* [38] |
Topical trifluridine | D [38] |
Oral acyclovir | D [35] |
Eczema Herpeticum
Clinical Features
Eczema herpeticum or Kaposi’s varicelliform eruption is a severe, disseminated HSV infection in patients with atopic dermatitis or other chronic skin conditions such as pemphigus, Darier disease, or burns. Patients typically have abrupt onset of fever, malaise, lymphadenopathy, and widespread monomorphic vesicles and erosions. Often “punched-out” erosions with hemorrhagic crust are more evident than intact vesicles (Fig. 18.4). Lesions are most prominent in areas of chronic skin disease, but also favor the head, neck, and trunk. If eczema herpeticum is suspected, treatment should be initiated while awaiting diagnostic results such as viral culture, DFA, and PCR. Complications include ocular involvement, secondary bacterial super infections (Staphylococcus aureus and/or group A streptococci), fluid loss, and viremia.
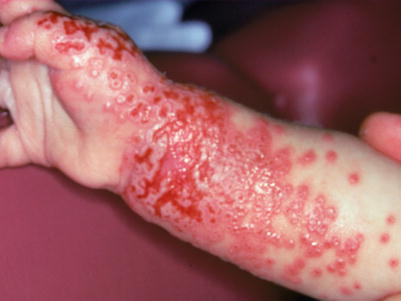

Fig. 18.4
Punched out erosions in the setting of atopic dermatitis representing eczema herpeticum
Management Strategies
The mainstay of treatment is systemic antiviral therapy (Table 18.1) [40, 41]. For most pediatric patients, hospital admission and intravenous acyclovir is the standard of care [42]. There are limited studies available evaluating acyclovir therapy, but one large multicenter retrospective cohort study showed that delayed acyclovir initiation was associated with increased hospital length of stay [43].
Other treatment considerations include intravenous hydration with attention to electrolyte balance, pain control, and antibiotic therapy for secondary bacterial infection [44]. Empiric antibiotic therapy has not been associated with shorter hospital length of stay, but it is critical to recognize systemic bacterial illness early [44]. Topical therapy includes bland emollients and topical corticosteroids as needed [45]. Facial involvement necessitates an ophthalmologic evaluation to monitor for ocular involvement.
Specific Investigations Recommended
For diagnosis |
Viral culture |
Polymerase chain reaction |
Direct fluorescent antibody |
Tzanck smear |
Skin biopsy of atypical lesion |
Serologic testing |
Table 18.17
First line therapies
Intravenous acyclovir | |
Oral acyclovir | |
Topical corticosteroids | D [45] |
Herpetic Whitlow
Clinical Features
Herpetic whitlow is a deep, painful, vesicular or bullous eruption with surrounding erythema involving the pulp of the distal fingertip(s) (Fig. 18.5). It can be a complication of primary oral or genital herpes. The prodrome includes a tingling and burning sensation of the finger(s) and influenza-like symptoms including fever and localized lymphadenopathy. Blister fluid is initially clear, but then appears purulent, mimicking a bacterial infection. Diagnostic studies such as viral culture, PCR, DFA, and Tzanck should be considered to confirm diagnosis. If untreated, symptoms generally spontaneously resolve in 1–3 weeks, but can recur.
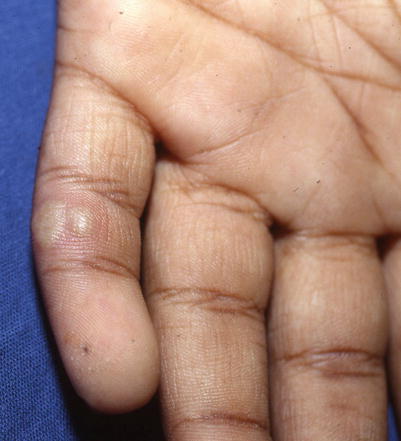

Fig. 18.5
Tense grouped vesicles in a patient with herpetic whitlow
Management Strategies
Treatment is typically supportive including anti-inflammatory agents, immobilization, and dry dressings to prevent transmission [46].
Oral antiviral therapy has been accepted as an effective means of reducing the duration of symptoms in primary infection and recurrent episodes (Table 18.1) [47]. Intravenous acyclovir is recommended for immunocompromised patients or those with severe infections (Table 18.2). Topical acyclovir is not believed to be helpful, and surgical intervention should be avoided. Prophylactic oral acyclovir may be effective in suppressing recurrent infections.
Herpes Gladiatorum
Clinical Features
Herpes gladiatorum is the term used when herpes simplex virus is transmitted during contact sports such as wrestling and rugby. It is characterized by widespread grouped vesicles on an erythematous base, and typically occurs on the head, neck, and upper extremities. Because lesions may become abraded during competition, they may have an atypical appearance. Systemic symptoms include fever, malaise, headache, and regional lymphadenopathy. Diagnosis may be made clinically, but when possible, laboratory confirmation should be performed. There is a general consensus in the literature that herpes gladiatorum is misdiagnosed, and diagnostic tests are underutilized.
Management Strategies
Herpes gladiatorum can be treated with oral antivirals. Depending upon the age of the patient and immunosupression status, antivirals include acyclovir, valacyclovir, and famiciclovir (Tables 18.1 and 18.2). No clinical studies have been performed on patients with primary herpes gladiatorum. Return-to-play criteria has been published by the National Collegiate Athletic Association and National Federation of State High School Associations Sports Medicine Advisory Committee and is summarized in Table 18.20 [48–50].
Table 18.20
Guidelines for return to competition for wrestlers with herpes gladiatorum
National Collegiate Athletic Association | National Federation of State High School Associations | |
|---|---|---|
Primary infection | (1) Skin lesions must be surmounted by a firm adherent crust at competition time and have no evidence of secondary bacterial infection | (1) All lesions must be scabbed over with no oozing or discharge and no new lesions should have occurred in the preceding 48 h |
(2) Wrestlers must have developed no new blisters for 72 h before the examination | (2) Wrestlers should be treated and not allowed to compete for a minimum of 10 days. If general body signs and symptoms like fever and swollen lymph nodes are present, that minimum period of treatment should be extended to 14 days | |
(3) Wrestlers must be free of signs and symptoms like fever, malaise, and swollen lymph nodes | ||
(4) Wrestlers must have been on appropriate dosage of systemic antiviral therapy for at least 120 h before and at the time of the competition | ||
Recurrent infection | (1) Skin lesions must be surmounted by a firm adherent crust at competition time, and have no evidence of secondary bacterial infection | (1) All lesions must be scabbed over with no oozing or discharge and no new lesions should have occurred in the preceding 48 h |
(2) Wrestlers require a minimum of 120 h of oral anti-viral treatment, again so long as no new lesions have developed and all lesions are scabbed over | (2) Wrestlers require a minimum of 120 h or five full days of oral anti-viral treatment, again so long as no new lesions have developed and all lesions are scabbed over | |
(3) Active herpetic infections shall not be covered to allow participation |
Recurrent infections should also be treated with antivirals (Tables 15.1 and 15.2). In a double-blind, placebo-controlled prospective study of 20 wrestlers and coaches with confirmed recurrent HSV, a 7-day regimen of valacyclovir 500 mg twice daily reduced the length of time until clinical clearance [51].
Varicella Zoster Virus
Clinical Features
Varicella or chickenpox is caused by varicella zoster virus (VZV), a member of the herpesvirus family. It is a highly communicable disease transmitted by aerosolized droplets or direct person-to-person contact. The typical incubation period is 10–14 days and it is infectious from 48 h prior to the onset of rash until skin lesions have crusted over.
Clinical features include a prodrome of fever, chills, malaise, headache, and arthralgias, with subsequent development of a generalized vesicular exanthem within 24–48 h. These pruritic vesicles initially appear on the scalp, face, and trunk, and then spread to the extremities (Figs. 18.6 and 18.7). New vesicle formation generally stops within 4 days, and most lesions have fully crusted by day 6. “Breakthrough varicella” can occur after a single dose of the varicella vaccine and subsequent exposure to VZV. Clinical features include absent to low fever, <50 macules and papules, and a shorter duration of illness.
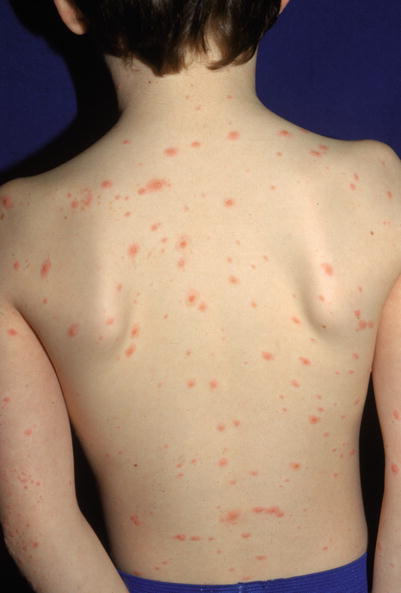
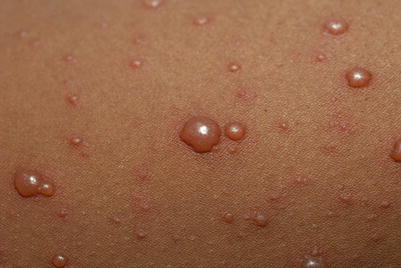

Fig. 18.6
Numerous truncal edematous papules in a child with varicella zoster

Fig. 18.7
Characteristic “dewdrop on a rose petal” demonstrating a vesicle on an erythematous base seen with chickenpox
Varicella tends to be more severe in infants, adolescents, and the immunocompromised. Potential complications include secondary bacterial superinfection of skin lesions, pneumonia, nervous system involvement (encephalitis, cerebellar ataxia, aseptic meningitis), thrombocytopenia, glomerulonephritis, and hepatitis. Reye syndrome was a common complication before the association with salicylates was understood. Herpes zoster (shingles) represents delayed sequelae of varicella and will be discussed in a subsequent section.
Management Strategies
In the United States, a two-dose live, attenuated vaccine program is recommended for all healthy individuals 12 months of age or older without evidence of immunity [53]. For children 12 months to 12 years, a first dose is recommended at age 12–15 months and a second dose at age 4–6 years. The second dose may be administered at an earlier age, provided the time interval between the first and second dose is >3 months. For adolescents >13 years without evidence of varicella immunity, two doses of the vaccine should be administered at least 4–8 weeks apart.
In healthy children below the age of 12 years, treatment is usually symptomatic for itch, pain, and prevention of secondary superinfection. This includes antihistamines, acetaminophen, and topical care including cool compresses, oatmeal baths, calamine lotion, menthol-camphor (Sarna) lotion, and mupirocin. Fingernails should be clipped to avoid excoriation and bacterial superinfection. Salicylates should be avoided given the risk of Reye syndrome. Antiviral therapy is not recommended for routine use in otherwise healthy children [54].
According to the American Academy of Pediatrics, oral acyclovir or valacyclovir should be considered in (1) children older than 12 years without evidence of immunity, and in children of any age with (2) chronic cutaneous or cardiopulmonary disorders, (3) on long-term salicylate therapy, (4) on short, intermittent systemic, or aerosolized courses of corticosteroids, and (5) secondary infections by a household member, as the disease is usually more severe than the index case [55, 56]. Table 18.22 contains antiviral dosing recommendations [57]. Antivirals have a limited window of opportunity to affect clinical course, and should be started within 24 h of the rash developing.
Table 18.22
Varicella anti-viral dosing recommendations for immunocompetent and immunocompromised
Antiviral | Population | Route | Age | Dosing |
|---|---|---|---|---|
Acyclovira,b | Varicella in immunocompetent host | Oral | ≥2 years | 80 mg/kg per day in 4 divided doses for 5 days; maximum dose 3,200 mg/day |
Acyclovira,b | Varicella in immunocompetent host requiring hospitalization | IV | ≥2 years | 30 mg/kg/day in 3 divided doses for 7–10 days; maximum dose 3,200 mg/day |
Acyclovira,b | Varicella in immunocompromised host | IV | <1 year | 30 mg/kg per day in 3 divided doses for 7–10 days |
Foscarneta,b | VZV infection resistant to acyclovir | IV | Adult dose | 120 mg/kg/day, divided every 8 h, up to 3 weeks |
Valacyclovira,b | Varicella in immunocompetent or immunocompromised host | Oral | 2 to <18 years | 20 mg/kg per dose 3 times daily for 5 days, not to exceed 1 g per dose 3 times daily |
Intravenous acyclovir is recommended for immunocompromised children, including those treated with chronic steroids [58, 59]. Therapy initiated within 24 h of rash onset maximizes efficacy, but individuals should be treated even if this time window has passed. Some experts have used valacyclovir in selected immunocompromised patients believed to be at lower risk of developing severe varicella, but this should be reviewed on a case-by-case basis. Foscarnet should be used to treat infections caused by acyclovir-resistant VZV strains, which are typically limited to immunocompromised hosts (Table 18.22) [60].
In the setting of VZV exposure, immunization may prevent or modify disease if given to otherwise healthy children within 72–120 h after varicella exposure [61]. Acyclovir is not recommended for prophylactic use in either the immunocompetent or immunocompromised. Varicella-Zoster Immune Globulin (VZIG; 125 units/10 kg, maximum 625 units) is recommended in immunocompromised children with no history of varicella and/or negative serologic testing as soon as possible within 10 days of exposure [62]. If VZIG is not available, IVIG (400 mg/kg administered once) is a potential alternative [63]. Immune globulin preparations are not effective once symptoms are present. Potential varicella exposures include an infected member of the household, playmate (face-to-face indoor contact), hospital roommate, or healthcare worker. Varicella can be contracted from those with zoster, but requires intimate contact (touch or hugging) before the lesions are crusted [55].
In neonates, varicella infection has a higher case-fatality rate when the mother develops varicella from 5 days before to 2 days after delivery. This is secondary to immature neonatal cellular immunity and insufficient development and transplacental transfer of VZV-specific maternal immunoglobulin G antibodies. VZIG is recommended in this population and is also recommended in a hospitalized preterm infant (28 weeks or more gestation) whose mother lacks evidence of immunity against varicella, and a hospitalized preterm infant (less than 28 weeks gestation or birth weight of 1000 g or less) regardless of maternal immunity [55, 62].
VZIG is not indicated for healthy term infants exposed postnatally to varicella, even in those whose mother develops a rash more than 2 days after delivery. However, some would advise VZIG for exposed newborns within the first 2 weeks of life whose mothers do not have evidence of VZV immunity.
Herpes Zoster
Clinical Features
Herpes zoster or shingles is caused by reactivation of varicella zoster virus (VZV) in the dorsal root ganglia. VZV establishes latency during primary varicella infection or following varicella vaccine. Although pediatric zoster is most common among immunocompromised children and those exposed in utero or within the first year of life, it can occur in children without these risk factors. Childhood zoster tends to be milder and less associated with postherpetic neuralgia. The classic presentation is an acute, unilateral vesicular eruption in a dermatomal distribution along one or more sensory nerves (Figs. 18.8 and 18.9). Patients may complain of pain, hyperesthesia, and tenderness before cutaneous findings. Crusting of the vesicles and healing occurs within a few days to 1 week. Zoster may become disseminated in immunocompromised children, with visceral complications including encephalitis, pneumonia, and hepatitis.
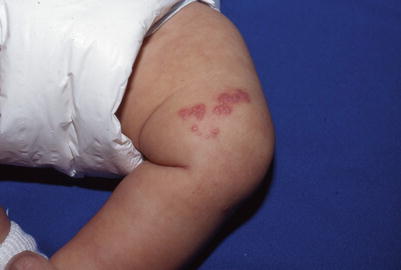
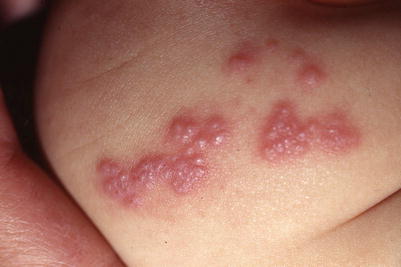

Fig. 18.8
Clustered vesicles in a dermatomal distribution in an infant with herpes zoster. The mother had chicken pox late in pregnancy

Fig. 18.9
Close-up of the infant in Fig. 1.8, showing vesicles grouped on an erythematous base with shingles
Management Strategies
Treatment includes symptomatic therapy and antivirals [64]. Symptomatic care includes cool compresses, calamine lotion, antihistamines, and analgesics. Fingernails should be kept short to avoid superinfection and excoriation.
Acyclovir is the only antiviral agent for zoster with specific pediatric dosing guidelines; famciclovir and valacyclovir do not have sufficient clinical data for pediatric zoster (Table 18.26). Ideally, antiviral therapy should be given within 72 h of exanthem onset to decrease acute neuritis, vesicle formation, and time to crusting [65]. Immuncompromised patients as well as those with serious complications or disseminated disease should be treated with intravenous acyclovir [66].
Table 18.26
Oral and intravenous antiviral dosing guidelines for herpes zoster in the immunocompetent and immunocompromised host
Antiviral | Population | Route | Age | Dosing |
|---|---|---|---|---|
Acyclovira,b | Herpes zoster in immunocompetent host | Oral | ≥12 years | 4,000 mg/day in 5 divided doses for 5–7 days |
Acyclovira,b | Herpes zoster in immunocompetent host | IV (if requiring hospitalization) | <1 year | 30 mg/kg per day in 3 divided doses for 7–10 days |
Acyclovira,b | Herpes zoster in immunocompromised host | IV | <12 years | 30 mg/kg per day in 3 divided doses, for 7–10 days |
Acyclovira,b | Herpes zoster in immunocompromised host | IV | ≥12 years | 30 mg/kg per day in 3 divided doses, for 7–10 days |
Famciclovira,b | Herpes zoster | Oral | Adult dose | 1,500 mg/day in 3 divided doses for 7 days |
Valacyclovira,b | Herpes zoster | Oral | Adult dose | 3 g/day in 3 divided doses for 7 days |
Epstein-Barr Virus/Infectious Mononucleosis
Clinical Features
Infectious mononucleosis (IM) is one of the most recognized syndromes of Epstein-Barr virus (EBV), also known as human herpesvirus 4 (HHV4). IM is caused by primary EBV infection in older children or adolescents, and consists of fever, pharyngitis, lymphadenopathy, and fatigue. Cutaneous manifestations are nonspecific and usually appear as a morbilliform copper-colored eruption on the trunk and extremities (Figs. 18.10 and 18.11). Administration of antibiotics, particularly penicillins such as ampicillin and amoxicillin, will lead to development of a morbilliform, scarlatiniform, or petechial eruption in the majority of patients with IM. This is thought to represent an enhanced reaction to drugs or their metabolites rather than a true allergy. Systemic complications include encephalitis, upper airway obstruction (due to tonsillar hypertrophy), mild acute hepatitis, lymphocytosis with atypical lymphocytes, thrombocytopenia, hemolytic anemia, splenomegaly and, rarely, splenic rupture.
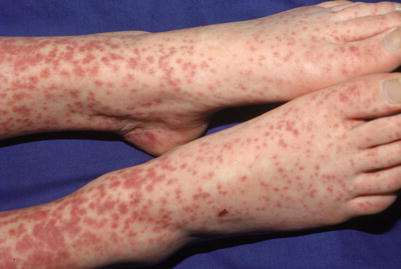
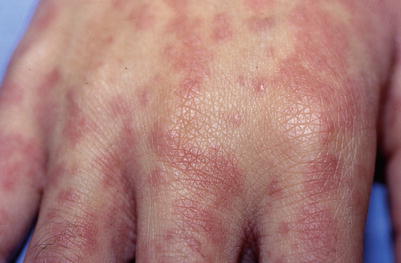

Fig. 18.10
Brownish red or copper-colored macules and papules seen with the rash of infectious mononucleosis from EBV

Fig. 18.11
Purple-red macules and papules of EBV infection
Another dermatologic manifestation of EBV involves large, painful, symmetric genital ulcers, particularly in adolescent females, on the labia minora and majora. Most patients then develop symptoms of infectious mononucleosis.
Initial laboratory evaluation should include a complete blood count with differential, hepatic function panel, and heterophile antibody test (or Monospot test). Monospot may be negative in the early stages and in children less than 4 years of age. Further serologic testing including EBV IgM, and IgG antibodies against viral capsid antigens, early antigens, and nuclear antigen proteins may be pursued in those who are Monospot-negative.
Management Strategies
Management is mainly supportive. Rest, hydration, antipyretics, and analgesics are recommended. Patients should avoid exertion and participation in sports for a minimum of 3 weeks. Amoxicillin and ampicillin should not be prescribed. Systemic corticosteroids may be helpful in managing more severe complications including upper airway obstruction, hemolytic anemia, and thrombocytopenia [68–70].
Specific Investigations Recommended
For diagnosis |
Complete blood count with differential |
Hepatic function panel |
Heterophile antibody test/Monospot test (may be negative in early infection and in children under age 4) |
EBV PCR |
Viral serologies: EBV IgM and IgG against viral capsid antigens, early antigens, nuclear antigen proteins |
Table 18.28
First line therapies
Antipyretics | E |
Analgesics | E |
Hydration | E |
Avoidance of sports for 3 weeks (due to risk of splenic rupture) | E |
Table 18.29
Second line therapies
Systemic corticosteroids (especially for complications such as upper airway obstruction, hemolytic anemia) | B |
Warts
Clinical Features
Warts or verrucae are a common dermatologic condition in children and adults, with a variety of presentations. They may be cobblestoned or filiform as in the common wart (verruca vulgaris) (Fig. 18.12); flesh-colored, flat-topped plane warts (verruca plana); or hyperkeratotic warts on soles (verruca plantaris) or palms (verruca palmaris). A study of wart incidence by age range showed warts were most common among 9- and 10-year-olds, with 8.61 % having warts [71]. In children, verrucae are typically benign and more of a cosmetic concern. Pain and functional impairment may occur if warts proliferate on soles, hands, or periorificially.
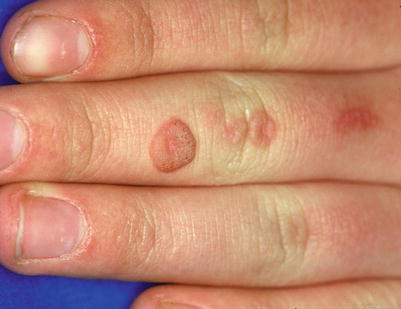

Fig. 18.12
Verrucous cobblestoned papule of verruca vulgaris
Verrucae are caused by the human papillomavirus (HPV), with different strains of HPV being more common in different types of warts. Verruca vulgaris is most commonly associated with HPV strains 1, 4, 27, and 57 while palmoplantar warts are thought to be caused by HPV 2 [72].
Management Strategies
Warts are benign, and immunocompetent children will eventually clear the infection [73]. Treatment strategies for warts in children should focus on minimizing discomfort and progression of lesions, while avoiding painful treatment modalities. Verruca plantaris should be treated to minimize functional impairment and discomfort. Management is similar to verruca vulgaris, with possible need to move to second- and third-line therapies sooner, given the potential symptomatic nature of plantar warts. With any painful treatment modality, encouraging the patient’s family to pretreat with acetaminophen or ibuprofen can help lessen the patient’s pain and make him or her a more willing treatment partner.
HPV is ubiquitous in the environment, and children with warts do not represent a significant infectious risk to others. Children are more likely to auto-inoculate and spread the virus to other parts of their skin. Families should be counseled on this potential for auto-inoculation. Children should be dissuaded from their natural tendencies to touch, bite, pick, or otherwise manipulate warts.
First-line therapies to treat warts include observation, salicylic acid, and occlusion with duct tape [73–75]. While requiring patience on the part of the patient and family, these options are typically well-tolerated by children and are a good initial choice for asymptomatic verrucae that are not cosmetically disturbing to the patient.
More advanced wart therapies involve various degrees of local irritation and immunomodulation to the wart. Topical prescription creams such as 5-fluorouracil and imiquimod and in-office application of a combination of tricholoroacetic acid, podophyllin, and Cantharidin 0.7 %, are well-tolerated [76, 77]. Liquid nitrogen cryotherapy can be effective, and is better tolerated in older children and teenagers [75] (Fig. 18.13). Isolated case reports have described clearance of recalcitrant verrucae after patients received the HPV vaccination, despite the fact the serotypes covered in the vaccine are not those typically associated with common warts [72]. Squaric acid, diphenylcyclopropenone, intralesional bleomycin, intralesional candida antigen, and laser treatments are other advanced wart therapies that may require additional cooperation and pain tolerance on the part of the patient [78–87].
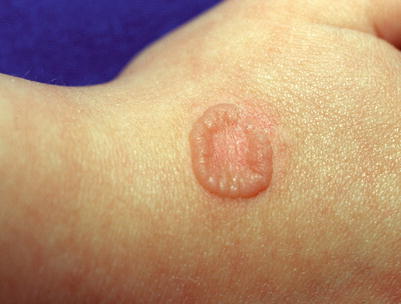

Fig. 18.13
Ring wart after cryotherapy
Specific Investigations Recommended
For diagnosis |
Scraping with #15 blade to visualize pinpoint capillaries and interrupted dermatoglyphics |
Biopsy if appears atypical |
Table 18.30
First line therapies
Observation only | Children will typically clear warts without sequelae in 1–2 years | B [73] |
Salicylic acid | OTC impregnated bandages, 17 % liquid, or stick applicator | B* [75] |
Surrounding skin may become macerated | ||
Duct tape occlusion | May require frequent reapplication if patient has significant sweating | B [74] |
Table 18.31
Second line therapies
5-fluorouracil cream/solution | Daily application of thin layer followed by occlusion | B [77] |
In-office liquid nitrogen cryotherapy | Two freeze-thaw cycles | B* (adults and children older than 12) [75] |
OTC cryotherapy with dimethyl ether and propane | Cannot achieve same level of cooling as liquid nitrogen | E [75] |
35 % tricholoroacetic acid, 25 % podophyllin, cantharidin 0.7 % | Painless application | E (author’s experience) |
Thin layers applied in office | ||
Patient washes off in 6–8 h |
Table 18.32
Third line therapies
Oral cimetidine | 40 mg/kg/day divided BID or TID, maximum 2.4 g/day | A [104] |
Randomized controlled trials have not shown efficacy over placebo | ||
HPV vaccine | FDA approved for females and males ages 9–26 | D* [72] |
Squaric acid dibutyl ester | Requires formulation by compounding pharmacy | |
Pre-treatment sensitization on upper arm required, with 2 % squaric acid | ||
Treatment concentration 0.2 % squaric acid and adjusted based on clinical response | ||
Pre-treatment with 50 % trichloroacetic acid may increase the efficacy of squaric acid | ||
Diphenylcyclopropenone (DPCP) | Requires sensitization prior to treatment with 0.1–0.25 % | B [79] |
Candida antigen, intralesionally | 0.1 mL of candida antigen injected into wart | B [81] |
May treat up to 3 warts at one time with maximum dose 0.3 mL | ||
May require 2–3 treatments spaced 4–6 weeks apart | ||
Topical cidofovir | 1–3 % cidofovir applied daily to every other day | B (children and adults) [83] |
Long-pulse Nd:YAG laser | May require multiple sessions for clearance | B* (one patient in study was a child) [80] |
Pulsed dye laser | Requires patient cooperation and may leave post-treatment ecchymosis | B* [78] |
Bleomycin, intralesionally | Requires pre-treatment with topical or intralesional anesthesia | B* (adults and children older than 12) [86] |
Requires multiple injections into one wart | ||
Patient likely will experience discomfort |
Condyloma
Clinical Features
Condyloma or anogenital warts are pink-gray, polypoid verrucous-surfaced papules along vulvar, penile, perineum, or peri-rectal skin. Condyloma result from infection with human papillomavirus (HPV) on genital skin, primarily with HPV strains 6 and 11 with the incubation period ranging from a few weeks to several months [88]. Adjacent skin folds may have “kissing” lesions representing spread of the virus through skin-on-skin contact. Multiple studies have suggested peri-rectal is the most common location for condyloma. Lesions may be asymptomatic or associated with pruritus, discomfort, or bleeding.
A child may acquire condyloma through several modes of transmission: vertically from mother to child perinatally, auto-inoculation by the child from a non-genital verruca, direct transmission from a caregiver during diapering or bathing, or inoculation from fomites [89]. Suspicion for transmission via sexual abuse is higher in older children (3 years and above) who do not have non-genital verruca in themselves, relatives, or other caregivers. When sexual abuse is a concern, careful history-taking by trained providers and the involvement of child protection agencies are the crucial first steps. A detailed physical exam for any other signs of abuse is key, but many patients who have experienced sexual abuse have no pertinent findings on exam.
Management Strategies
The location and persistence of lesions can make treating condyloma a prolonged and potentially uncomfortable process. Condyloma may be treated with topical preparations applied in-office or at home. Destructive modalities, such as cryotherapy, excision, or laser treatments, are alternative treatment strategies that may be more painful and require more cooperation [90, 91].
With the growing use of HPV vaccines, condyloma management now starts with prevention [92–94]. The quadrivalent HPV vaccine available in the US (Gardasil™) covers HPV strains 6, 11, 16, and 18 [92, 95]. Gardasil-9 is a new expanded version of the original vaccine and covers HPV strains 6, 11, 16, 18, 31, 33, 45, 52, and 58. Both are FDA-approved for females and males aged 9–26 years. A bivalent vaccine (Cervarix™) covers HPV strains 16 and 18 and is FDA-approved for girls aged 9–26 years [92, 95]. The vaccines are given as a series of three intramuscular injections into the deltoid muscle.
Cryotherapy has a clearance rate that varies from 27 % to 88 % depending on technique [88]. Two applications of liquid nitrogen—delivered from cryo-canister, cold forceps, or cotton-tipped application—for at least 15 s or until a rim of frost covers the entire lesion with a 1 mm border, should be separated by sufficient time to allow the lesion to thaw without external re-warming. Cryotherapy should be repeated every 2–4 weeks. Podofilox (Condylox) and podophyllotoxin are plant-derived anti-mitotic medications that may be used topically on condyloma [90, 91, 96, 97].
For second-line therapies, imiquimod applied three to five nights a week is usually well-tolerated by patients [90–98]. Symptomatic smaller condyloma may be excised under local or general anesthesia. Initial clearance rates for excision range from 35 % to 70 %, with recurrence rates around 20 % [88].
Numerous additional destructive and immunomodulatory treatments have been tried for condyloma [99–102]. Cimetidine is an H2 receptor antagonist whose efficacy against condyloma and verruca vulgaris as demonstrated in open-label studies and case series has not been duplicated in randomized controlled trials [103, 104]. In adults, the application of 5-aminolevulinic acid (ALA) following by photodynamic therapy has produced clearance of condyloma, and could be considered for older children able to tolerate the procedure and discomfort [105]. Sinecatechins (VEREGEN or Polyphenon E) are green tea-based polyphenols with immunomodulatory properties, and may appeal to families who prefer a more plant-derived, less medicalized approach [106]. Cidofovir and interferon are treatments used for extensive condyloma in immunosuppressed patients [107].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree