Viruses are obligatory intracellular parasites. The structural components of a viral particle (virion) consist of a central core of nucleic acid, a protective protein coat (capsid), and (in certain groups of viruses only) an outermost membrane or envelope. The capsid of the simplest viruses consists of many identical polypeptides (structural units) that fold and interact with one another to form morphologic units (capsomeres). The number of capsomeres is believed to be constant for each virus with cubic symmetry, and it is an important criterion in the classification of viruses. The protein coat determines serologic specificity, protects the nucleic acid from enzymatic degradation in biologic environments, controls host specificity, and increases the efficiency of infection. The outermost membrane of the enveloped viruses is essential for the attachment to, and penetration of, host cells. The envelope also contains important viral antigens.
Two main groups of viruses are distinguished: deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). The DNA virus types are parvovirus, papovavirus, adenovirus, herpesvirus, and poxvirus. RNA viruses are picornavirus, togavirus, reovirus, coronavirus, orthomyxovirus, retrovirus, arenavirus, rhabdovirus, and paramyxovirus. Some viruses are distinguished by their mode of transmission: arthropod-borne viruses, respiratory viruses, fecal-oral or intestinal viruses, venereal viruses, and penetrating-wound viruses.
Herpesvirus Group
The herpes viruses are medium-sized viruses that contain double-stranded (ds) DNA and replicate in the cell nucleus. They are characterized by the ability to produce latent but lifelong infection by infecting immunologically protected cells (immune cells and nerves). Intermittently, they have replicative episodes with amplification of the viral numbers in anatomic sites conducive to transmission from one host to the next (genital skin, orolabial region). The vast majority of infected persons remain asymptomatic. Viruses in this group are varicella-zoster virus (VZV; human herpesvirus type 3 [HHV-3]); herpes simplex virus types 1 and 2 (HSV-1 and HSV-2); cytomegalovirus (CMV); Epstein-Barr virus (EBV); human herpesviruses types 6, 7, and 8 (HHV-6, HHV-7, and HHV-8); Herpesvirus simiae (B virus); and other viruses of animals.
Herpes Simplex Virus (Human Herpesvirus Types 1 and 2)
Infection with HSV is one of the most prevalent infections worldwide. HSV-1 infection, the cause of most cases of orolabial herpes, is more common than infection with HSV-2, the classical cause of most cases of genital herpes, though rates of HSV-1 in genital herpes has been rising, particularly in younger patients. Between 30% and 95% of adults (depending on the country and group tested) are seropositive for HSV-1, although seroprevalence of HSV-1 has decreased in the United States among adolescents (age 14–19 years) more recently. Seroprevalence for HSV-2 is lower, and it appears at the age of onset of sexual activity. About 2.4% of adults become infected annually with HSV-2 in their third decade of life. In the United States about 25% of adults are infected with HSV-2, with black men and women twice as likely to be HSV-2 infected as whites. In sexually transmitted disease (STD) clinic patients, the infection rate is 30%–50%. In sub-Saharan Africa, infection rates are 60%–95%. Worldwide, the seroprevalence is higher in persons infected with human immunodeficiency virus (HIV). Serologic data show that many more people are infected than give a history of clinical disease. For HSV-1, about 50% of infected persons give a history of orolabial lesions. For HSV-2, 20% of infected persons are completely asymptomatic (latent infection), 20% have recurrent genital herpes they recognize, and 60% have clinical lesions that they do not recognize as genital herpes (subclinical or unrecognized infection). Most persons with HSV-2 infection are symptomatic, but the majority do not recognize that their symptoms are caused by HSV. All persons infected with HSV-1 and HSV-2 are potentially infectious even if they have no clinical signs or symptoms. Routine screening for HSV in asymptomatic patients, including those who are pregnant, is not currently recommended; testing is appropriate in patients with signs or symptoms of the disease.
Herpes simplex infections are classified as either “first episode” or “recurrent.” Most patients have no lesions or findings when they are initially infected with HSV. When patients have their first clinical lesion, this is usually a recurrence. Because the initial clinical presentation is not associated with a new infection, the previous terminology of “primary” infection has been abandoned. Instead, the initial clinical presentation is called a first episode and may represent a true primary infection or a recurrence. Persons with chronic or acute immunosuppression may have prolonged and atypical clinical courses.
Infections with HSV-1 or HSV-2 are diagnosed by specific and nonspecific methods. Bedside, in-office diagnosis can be made using the Tzanck smear. It is nonspecific because both HSV and VZV infections result in the formation of multinucleate epidermal giant cells. Although the technique is rapid, its success depends heavily on the skill of the interpreter. The accuracy rate is 60%–90%, with a false-positive rate of 3%–13%. The direct fluorescent antibody (DFA) test is more accurate and will identify virus type; results can be available in hours if a virology laboratory is nearby. Both Tzanck, which is neither sensitive nor specific, and DFA, which is nonspecific, are less useful than viral culture or polymerase chain reaction (PCR) testing. Viral culture is very specific and relatively rapid, compared with serologic tests, because HSV is stable in transport and grows readily and rapidly in culture, and can be used for antiviral sensitivity testing if necessary, though it can be expensive to culture virus. Results are often available in 48–72 hours. PCR is as specific as viral culture but four times more sensitive and can be performed on dried or fixed tissue, and is becoming increasingly viewed as the test of choice. Skin biopsies of lesions can detect viropathic changes caused by HSV, and with specific HSV antibodies, immunoperoxidase (IP) techniques can accurately diagnose infection. The accuracy of various tests depends on lesion morphology. Only acute, vesicular lesions are likely to be positive with Tzanck smears. Crusted, eroded, or ulcerative lesions are best diagnosed by viral culture, DFA, histologic methods, or PCR.
Serologic tests are generally not used in determining whether a skin lesion is caused by HSV infection. A positive serologic test indicates only that the individual is infected with that virus, not that the viral infection is the cause of the current lesion. In addition to determining the infection rate in various populations, serologic tests are most useful in evaluating couples in which only one partner gives a history of genital herpes (discordant couples), in couples (if childbearing) at risk for neonatal herpes infection, and for possible HSV vaccination (when available).
Orolabial Herpes
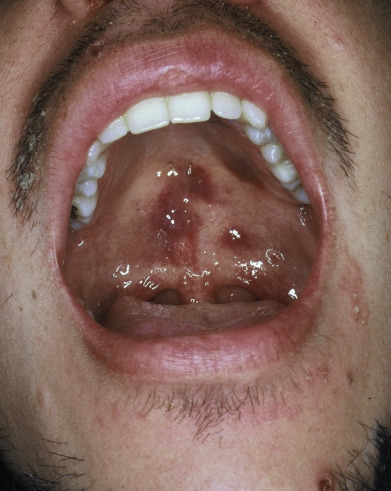
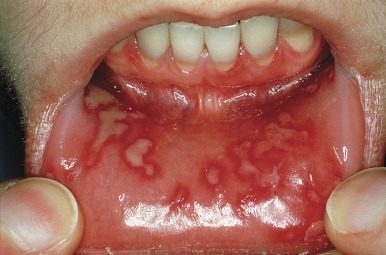
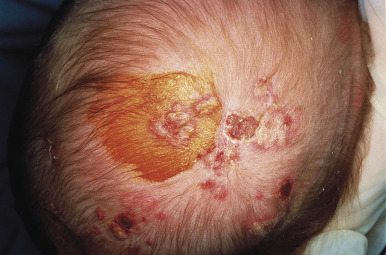
Orolabial herpes is virtually always caused by HSV-1. In 1% or less of newly infected persons, herpetic gingivostomatitis develops, mainly in children and young adults ( Fig. 19.1 ). The onset is often accompanied by high fever, regional lymphadenopathy, and malaise. The herpetic lesions in the mouth are usually broken vesicles that appear as erosions or ulcers covered with a white membrane. The erosions may become widespread on the oral mucosa, tongue, and tonsils, and the gingival margin is usually eroded. Herpetic gingivostomatitis produces pain, foul breath, and dysphagia. In young children, dehydration may occur. It may cause pharyngitis, with ulcerative or exudative lesions of the posterior pharynx. The duration, untreated, is 1–2 weeks. If the initial episode of herpetic gingivostomatitis or herpes labialis is so severe that intravenous (IV) administration is required, IV acyclovir, 5 mg/kg three times daily, is recommended. Oral therapeutic options include acyclovir suspension, 15 mg/kg five times daily for 7 days; valacyclovir, 1 g twice daily for 7 days; or famciclovir, 500 mg twice daily for 7 days. This therapy reduces the duration of the illness by more than 50%.
The most frequent clinical manifestation of orolabial herpes is the “cold sore” or “fever blister.” Recurrent HSV-1 is the cause of 95% or more of cases and typically presents as grouped blisters on an erythematous base. The lips near the vermilion are most frequently involved ( Fig. 19.2 ), although lesions may occur wherever the virus was inoculated or proliferated during the initial episode ( Fig. 19.3 ). Recurrences may be seen on the cheeks, eyelids, and earlobes. Oral recurrent HSV usually affects the keratinized surfaces of the hard palate and attached gingiva. Outbreaks are variable in severity, partly related to the trigger of the outbreak. Some outbreaks are small and resolve rapidly, whereas others may be severe, involving both the upper and the lower lip. In severe outbreaks, lip swelling is often present. Patient symptomatology is variable. A prodrome of up to 24 hours of tingling, itching, or burning may precede the outbreak. Local discomfort, as well as headache, nasal congestion, or mild flulike symptoms, may occur. Ultraviolet (UV) exposure, especially UVB, is a frequent trigger of recurrent orolabial HSV, and severity of the outbreak may correlate with intensity of the sun exposure. Surgical and dental procedures of the lips (or other areas previously affected with HSV) may trigger recurrences, and a history of prior HSV should be solicited in all patients in whom such procedures are recommended (see next section).
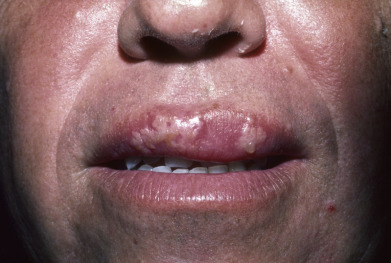
In most patients, recurrent orolabial herpes represents more of a nuisance than a disease. Because UVB radiation is a common trigger, use of a sunblock daily on the lips and facial skin may reduce recurrences. All topical therapies for the acute treatment of recurrent orolabial herpes have limited efficacy, reducing disease duration and pain by 1 day or less. Tetracaine cream, penciclovir cream, and acyclovir cream (not ointment) have some limited efficacy. Topical acyclovir ointment and docosanol cream provide minimal to no reduction in healing time or discomfort. The minimal benefit from these topical agents suggests that they should not be recommended for significant symptomatic orolabial herpes outbreaks. If oral therapy is contemplated for patients with severely symptomatic recurrences of orolabial HSV, it must be remembered that much higher doses of oral antivirals are required than for treatment of genital herpes. Intermittent treatment with valacyclovir, 2 g twice daily for 1 day, or famciclovir, 1.5 g as a single dose, starting at the onset of the prodrome, are simple and effective oral, 1-day regimens. Because the patient’s own inflammatory reaction against the virus contributes substantially to the severity of lesions of orolabial herpes simplex, topical therapy with a high-potency topical corticosteroid (fluocinonide gel 0.05%, three times daily) in combination with an oral antiviral agent more rapidly reduces pain and reduces maximum lesion area and time to healing. In nonimmunosuppressed patients, if episodic treatment for orolabial HSV is recommended and an oral agent is used, the addition of a high-potency topical corticosteroid should be considered. In patients with six or more outbreaks per year, chronic suppressive daily antiviral therapy can be used. Squaric acid dibutyl ester has been used as an immunosensitizer in one small study to reduce recurrent outbreaks.
Although most patients with orolabial herpes simplex do not require treatment, certain medical and dental procedures may trigger outbreaks of HSV. If the cutaneous surface has been damaged by the surgical procedure (e.g., dermabrasion, chemical peel, laser resurfacing), the surgical site can be infected by the virus and may result in prolonged healing and possible scarring. Prophylaxis is regularly used before such surgeries in patients with a history of orolabial herpes simplex. Famciclovir, 250 mg twice daily, and valacyclovir, 500 mg twice daily, or oral acyclovir 400 mg three times daily, are prophylactic options, to be begun 24 hours before the procedure. Duration of treatment in part depends on severity of the skin insult and rate of healing but should be at least 1 week and could be as long as 14 days. For routine surgeries at sites of HSV recurrences (upper or lower lip), acyclovir, 200 mg five times daily; famciclovir, 250 mg three times daily; or valacyclovir, 1 g twice daily, starting 2–5 days before the procedure and continuing for 5 days, can be considered. Prophylaxis could also be considered before skiing or tropical vacations and before extensive dental procedures, at the same dosages. Reactivation of orolabial herpes has also been associated with hyaluronic acid filler injections in about 1.5% of patients; and extensive facial HSV-1 infection has followed intense inhaled corticosteroid therapy. Kissing of the penis during circumcision can lead to penile HSV-1 infection, which can present acutely, or even years after the initial exposure. Some of these infants have died of disseminated or central nervous system HSV infection.
Herpetic Sycosis
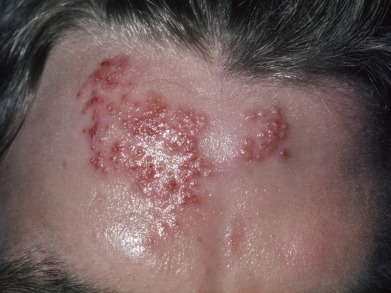
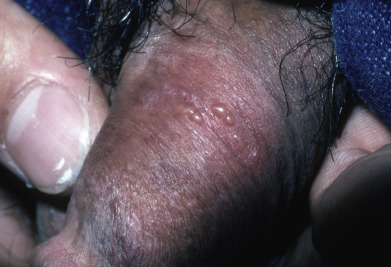
Recurrent or initial herpes simplex infections (usually from HSV-1) may primarily affect the hair follicle. The clinical appearance may vary from a few eroded follicular papules (resembling acne excoriée) to extensive lesions involving the whole beard area in men ( Fig. 19.4 ). Close razor blade shaving immediately before initial exposure or in the presence of an acute orolabial lesion may be associated with a more extensive eruption. The onset may be acute (over days) or more subacute or chronic. Diagnostic clues include the tendency for erosions, a self-limited course of 2–3 weeks, and an appropriate risk behavior. The diagnosis may be confirmed by biopsy. Although the herpes infection is primarily in the follicle, surface cultures of eroded lesions will usually be positive in the first 5–7 days of the eruption.
Herpes Gladiatorum
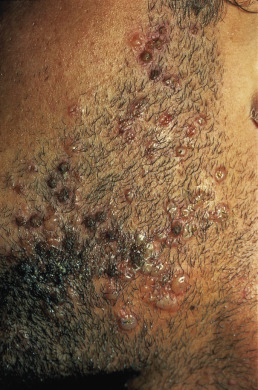
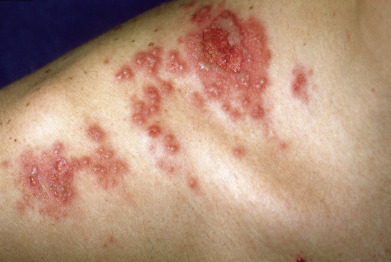
Infection with HSV-1 is highly contagious to susceptible persons who wrestle with an infected individual with an active lesion. One third of susceptible wrestlers will become infected after a single match. In tournaments and wrestling camps, outbreaks can be epidemic, affecting up to 20% of all participants. Lesions usually occur on the lateral side of the neck, the side of the face, and the forearm, all areas in direct contact with the face of the infected wrestler ( Fig. 19.5 ). Vesicles appear 4–11 days after exposure, often preceded by 24 hours of malaise, sore throat, and fever. Ocular symptoms may occur. Lesions are frequently misdiagnosed as a bacterial folliculitis. Any wrestler with a confirmed history of orolabial herpes should be taking suppressive antiviral therapy during all periods of training and competition. Rugby players (especially forwards who participate in scrums), mixed–martial arts fighters, and even boxers are also at risk.
Herpetic Whitlow
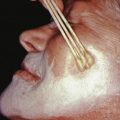
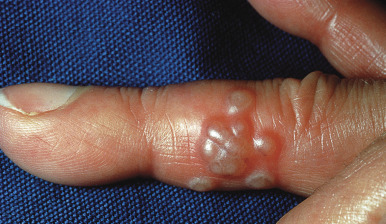
Herpes simplex infection may occur infrequently on the fingers or periungually. Lesions begin with tenderness and erythema, usually of the lateral nailfold or on the palm. Deep-seated blisters develop 24–48 hours after symptoms begin ( Fig. 19.6 ). The blisters may be tiny, under the thick epidermis, and require careful inspection to detect them. Deep-seated lesions that appear unilocular may be mistaken for a paronychia or other inflammatory process. Lesions may progress to erosions or may heal without ever impairing epidermal integrity because of the thick stratum corneum in this location. Herpetic whitlow may simulate a felon. Swelling of the affected hand can occur. Lymphatic streaking and swelling of the epitrochlear or axillary lymph nodes may occur, mimicking a bacterial cellulitis. Repeated episodes of herpetic lymphangitis may lead to persistent lymphedema of the affected hand. Herpetic whitlow has become much less common among health care workers since the institution of universal precautions and glove use during contact with the oral mucosa. Currently, most cases are seen in persons with herpes elsewhere. Children may be infected while thumb sucking or nail biting during their initial herpes outbreak or by touching an infectious lesion of an adult. Herpetic whitlow is bimodal in distribution, with about 20% of cases occurring in children younger than 10 years and 55% of cases in adults between ages 20 and 40. Virtually all cases in children are caused by HSV-1, and there is often a coexisting herpetic gingivostomatitis. In adults, up to three quarters of cases are caused by HSV-2. Among adults, herpetic whitlow is twice as common in women. Herpetic whitlow in health care workers can be transmitted to patients. In patients whose oropharynx is exposed to the ungloved hands of health care workers with herpetic whitlow, 37% develop herpetic pharyngitis.
Herpetic Keratoconjunctivitis
Herpes simplex infection of the eye is a common cause of blindness in the United States. It occurs as a punctate or marginal keratitis or as a dendritic corneal ulcer, which may cause disciform keratitis and leave scars that impair vision. Topical corticosteroids in this situation may induce perforation of the cornea. Vesicles may appear on the lids, and preauricular nodes may be enlarged and tender. Recurrences are common. Ocular symptoms in any person with an initial outbreak of HSV could represent ocular HSV, and an ophthalmologic evaluation should be performed to exclude this possibility.
Genital Herpes
Genital herpes infection is usually caused by HSV-2. In the mid-1980s the prevalence of genital herpes caused by HSV-1 began to increase because of changes in sexual habits and decreasing prevalence of orolabial HSV-1 infection in developed nations. In women under age 25, HSV-1 represents more than 50% of cases of genital herpes, whereas in women over 25 and in men of all ages, HSV-2 remains the most common cause of genital herpes. HSV-1 in the genital area is much less likely to recur. Only 20%–50% of patients have a recurrence; when it does recur, the average patient experiences only about one outbreak per year.
Genital herpes is spread by skin-to-skin contact, usually during sexual activity. The incubation period averages 5 days. Active lesions of HSV-2 contain live virus and are infectious. Persons with recurrent genital herpes shed virus asymptomatically between outbreaks (asymptomatic shedding). Even persons who are HSV-2 infected but have never had a clinical lesion (or symptoms) shed virus, so everyone who is HSV-2 infected is potentially infectious to a sexual partner. Asymptomatic shedding occurs simultaneously from several anatomic sites (penis, vagina, cervix, rectum) and can occur through normally appearing intact skin and mucosae. In addition, persons with HSV-2 infection may have lesions they do not recognize as being caused by HSV (unrecognized outbreak) or have recurrent lesions that do not cause symptoms (subclinical outbreak). Most transmission of genital herpes occurs during subclinical or unrecognized outbreaks, or while the infected person is shedding asymptomatically.
The risk of transmission in monogamous couples, in which only one partner is infected, is about 5%–10% annually, with women being at much greater risk than men for acquiring HSV-2 from their infected partner. Prior HSV-1 infection does not reduce the risk of being infected with HSV-2 but does make it more likely that initial infection will be asymptomatic. There is no strategy that absolutely prevents herpes transmission. All prevention strategies are more effective in reducing the risk of male-to-female transmission than female-to-male transmission. Condom use for all sexual exposures and avoiding sexual exposure when active lesions are present have been shown to be effective strategies, as has chronic suppressive therapy of the infected partner with valacyclovir.
The symptomatology during acquisition of infection with HSV-2 has a broad clinical spectrum, from totally asymptomatic to severe genital ulcer disease (erosive vulvovaginitis or proctitis). Only 57% of new HSV-2 infections are symptomatic. Clinically, the majority of symptomatic initial herpes lesions are classic, grouped blisters on an erythematous base. At times, the initial clinical episode is that of typical grouped blisters, but with a longer duration of 10–14 days. Although uncommon and representing 1% or fewer of new infections, severe first-episode genital herpes can be a significant systemic illness. Grouped blisters and erosions appear in the vagina, in the rectum, or on the penis, with continued development of new blisters over 7–14 days. Lesions are bilaterally symmetric and often extensive, and the inguinal lymph nodes can be enlarged bilaterally. Fever and flulike symptoms may be present, but in women the major complaint is vaginal pain and dysuria (herpetic vulvovaginitis). The whole illness may last 3 weeks or more. If the inoculation occurs in the rectal area, severe proctitis may occur from extensive erosions in the anal canal and on the rectal mucosa. The initial clinical episode of genital herpes is treated with oral acyclovir, 200 mg five times or 400 mg three times daily; famciclovir, 250 mg three times daily; or valacyclovir, 1000 mg twice daily, all for 7–10 days. It is clinically difficult to distinguish true initial (or primary) HSV-2 infection from a recurrence, so all patients with their initial clinical episode receive the same therapy. Only serology can determine whether the person is totally HSV naïve and experiencing a true primary episode, is partially immune from prior HSV-1 infection, or is already HSV-2 infected with first clinical presentation actually a recurrence. In fact, 25% of “initial” clinical episodes of genital herpes are actually recurrences.
Virtually all persons infected with HSV-2 will have recurrences, even if the initial infection was subclinical or asymptomatic. HSV-2 infection results in recurrences in the genital area six times more frequently than HSV-1. Twenty percent of persons with HSV-2 infection are truly asymptomatic, never having had either an initial lesion or a recurrence. Twenty percent of patients have lesions they recognize as recurrent genital herpes, and 60% have clinical lesions that are culture positive for HSV-2, but that are unrecognized by the patient as being caused by genital herpes. This large group of persons with subclinical or unrecognized genital herpes are infectious, at least intermittently, and represent one factor in the increasing number of new HSV-2 infections.
Typical recurrent genital herpes begins with a prodrome of burning, itching, or tingling. Usually within 24 hours, red papules appear at the site, progress to blisters filled with clear fluid over 24 hours, form erosions over the next 24–36 hours, and heal in another 2–3 days ( Fig. 19.7 ). The average total duration of a typical outbreak of genital herpes is 7 days. Lesions are usually grouped blisters and evolve into coalescent grouped erosions, which characteristically have a scalloped border. Erosions or ulcerations from genital herpes are usually very tender and not indurated (unlike chancre of primary syphilis). Lesions tend to recur in the same anatomic region, although not at exactly the same site (unlike fixed drug eruption). Less classic clinical manifestations are tiny erosions or linear fissures on the genital skin. Lesions occur on the vulva, vagina, and cervical mucosa, as well as on the penile and vulval skin. The upper buttock is a common site for recurrent genital herpes in both men and women. Intraurethral genital herpes may present with dysuria and a clear penile discharge and is usually misdiagnosed as a more common, nongonococcal urethritis such as Chlamydia or Ureaplasma infection. Inguinal adenopathy may be present. Looking into the urethra and culturing any erosions will establish the diagnosis. Recurrent genital herpes usually heals without scarring.
The natural history of untreated recurrent genital herpes is not well studied. Over the first few years of infection, the frequency of recurrences usually stays the same. Over periods longer than 3–5 years, the frequency of outbreaks decreases in at least two thirds of patients treated with suppressive antiviral therapy.
Recurrent genital herpes is a problematic disease because of the associated social stigma. Because it is not curable, patients frequently have a significant emotional response when first diagnosed, including anger (at presumed source of infection), depression, guilt, and feelings of unworthiness. During the visit, the health care worker should ask about the patient’s feelings and any psychological complications. This psychological component of genital herpes must be recognized, addressed directly with the patient, and managed properly for the therapy of recurrent genital herpes to be successful. Education regarding timing of outbreak and exposure, including potential for initial asymptomatic infection, is important.
Management of recurrent genital herpes should be individualized. A careful history, including a sexual history, should be obtained. Examination should include seeing the patient during an active recurrence so that the infection can be confirmed. The diagnosis of recurrent genital herpes should not be made on clinical appearance alone because of the psychological impact of the diagnosis. The diagnosis is best confirmed by viral culture, PCR, or DFA, allowing for typing of the causative virus. Genital HSV is a risk factor for HIV infection, and patients with genital HSV should be evaluated for and counseled about other STDs.
Treatment depends on several factors, including the frequency of recurrences, severity of recurrences, infection status of the sexual partner, and psychological impact of the infection on the patient. For patients with few or mildly symptomatic recurrences, treatment is often unnecessary. Counseling regarding transmission risk is required. In patients with severe but infrequent recurrences and in those with severe psychological complications, intermittent therapy may be useful. To be effective, intermittent therapy must be initiated at the earliest sign of an outbreak. The patient must be given the medication before the recurrence so that treatment can be started by the patient when the first symptoms appear. Intermittent therapy only reduces the duration of the average recurrence by about 1 day. However, it is a powerful tool in the patient who is totally overwhelmed by each outbreak. The treatment of recurrent genital herpes is acyclovir, 200 mg five times daily or 800 mg twice daily, or famciclovir, 125 mg twice daily, for 5 days. Shorter regimens that are equally effective include valacyclovir, 500 mg twice daily for 3 days; acyclovir, 800 mg three times daily for 2 days; or famciclovir, 1 g twice daily for 1 day.
For patients with frequent recurrences (>6–12 yearly), suppressive therapy may be more reasonable. Acyclovir, 400 mg twice daily, 200 mg three times daily, or 800 mg once daily, will suppress 85% of recurrences, and 20% of patients will be recurrence free during suppressive therapy. Valacyclovir, 500 mg/day (or 1000 mg/day for those with >10 recurrences/year), or famciclovir, 250 mg twice daily, is an equally effective alternative. Up to 5% of immunocompetent patients will have significant recurrences on these doses, and the dose of the antiviral may need to be increased. Chronic suppressive therapy reduces asymptomatic shedding by almost 95%. After 10 years of suppressive therapy, many patients can stop treatment, with substantial reduction in frequency of recurrences. Chronic suppressive therapy is safe, and laboratory monitoring is not required.
Intrauterine and Neonatal Herpes Simplex
Neonatal herpes infection occurs in 1 : 3000 to 1 : 20,000 live births, with an estimate of 14,000 cases annually worldwide and 1500–2200 cases of neonatal herpes annually in the United States. Eighty-five percent of neonatal herpes simplex infections occur at delivery; 5% occur in utero with intact membranes; and 10%–15% occur from nonmaternal sources after delivery. In utero infection may result in fetal anomalies, including skin lesions and scars, limb hypoplasia, microcephaly, microphthalmos, encephalitis, chorioretinitis, and intracerebral calcifications. It is either fatal or complicated by permanent neurologic sequelae.
Seventy percent of neonatal herpes simplex infections are caused by HSV-2 worldwide; however, in the United States recently HSV-1 was reported more frequently than HSV-2. Neonatal HSV-1 infections are usually acquired postnatally through contact with a person with orolabial disease, but can also occur intrapartum if the mother is genitally infected with HSV-1. The clinical spectrum of perinatally acquired neonatal herpes can be divided into the following three forms:
- 1.
Localized infection of the skin, eyes, and/or mouth (SEM)
- 2.
Central nervous system (CNS) disease
- 3.
Disseminated disease (encephalitis, hepatitis, pneumonia, and coagulopathy)
The pattern of involvement at presentation is important prognostically. With treatment, localized disease (skin, eyes, or mouth) is rarely fatal, whereas brain or disseminated disease is fatal in 15%–50% of neonates so affected. In treated neonates, long-term sequelae occur in 10% of infants with localized disease. More than 50% of patients with CNS or disseminated neonatal herpes have neurologic disability.
In 68% of infected babies, skin vesicles are the presenting sign and are a good source for virus recovery. However, 39% of neonates with disseminated disease, 32% with CNS disease, and 17% with SEM disease never develop vesicular skin lesions. Because the incubation period may be as long as 3 weeks and averages about 1 week, skin lesions and symptoms may not appear until the child has been discharged from the hospital.
The diagnosis of neonatal herpes is confirmed by viral culture or preferably immediate DFA staining of material from skin or ocular lesions. CNS involvement is detected by PCR of the cerebrospinal fluid (CSF). PCR of the CSF is negative in 24% of neonatal CNS herpes infections, so pending other testing, empiric therapy may be required. Neonatal herpes infections are treated with IV acyclovir for 14 days for SEM disease and for 21 days for CNS and disseminated disease.
Seventy percent of mothers of infants with neonatal herpes simplex are asymptomatic at delivery and have no history of genital herpes. Thus extended history taking is of no value in predicting which pregnancies may be complicated by neonatal herpes. The most important predictors of infection appear to be the nature of the mother’s infection at delivery (first episode vs. recurrent) and the presence of active lesions on the cervix, vagina, or vulvar area. The risk of infection for an infant delivered vaginally when the mother has active recurrent genital herpes infection is 2%–5%, whereas it is 26%–56% if the maternal infection at delivery is a first episode. One strategy to prevent neonatal HSV would be to prevent transmission of HSV to at-risk women during pregnancy, eliminating initial HSV episodes during pregnancy. To accomplish this, pregnant women and their partners would be tested to identify discordant couples for HSV-1 and HSV-2. If the woman is HSV-1 negative and the man is HSV-1 positive, orogenital contact during pregnancy should be avoided and a condom used for all episodes of sexual contact. Valacyclovir suppression of the infected male could also be considered but might have limited efficacy. If the woman is HSV-2 seronegative and her partner is HSV-2 seropositive, barrier protection for sexual contact during gestation is recommended, and valacyclovir suppression of the man could be considered. Abstinence from intercourse during the third trimester would also reduce the chances of an at-risk mother acquiring genital herpes that might first present perinatally. These strategies have not been tested and could not be guaranteed to prevent all cases of neonatal HSV. At a minimum, discordant couples should be informed of the increased risk to the fetus from the mother’s acquisition of HSV during pregnancy.
The appropriate management of pregnancies complicated by genital herpes is complex and still controversial. Routine prenatal cultures are not recommended for women with recurrent genital herpes because they do not predict shedding at delivery. Such cultures may be of value in women with primary genital herpes during pregnancy. Scalp electrodes should be avoided in deliveries where cervical shedding of HSV is possible; they can increase the risk of neonatal infection by up to sevenfold ( Fig. 19.8 ). Vacuum-assisted delivery also increases the relative risk of neonatal transmission of HSV 2–27 times. Genital HSV-1 infection appears to be much more frequently transmitted intrapartum than HSV-2. The current recommendation is still to perform cesarean section in the mother with active genital lesions or prodromal symptoms. This will reduce the risk of transmission of HSV to the infant from 8% to 1% for women who are culture positive from the cervix at delivery. However, this approach will not prevent all cases of neonatal herpes, is expensive, and has a high maternal morbidity. Because the risk of neonatal herpes is much greater in mothers who experience their initial episode during pregnancy, antiviral treatment of all initial episodes of genital HSV in pregnancy is recommended (except in the first month of gestation, when there may be an increased risk of spontaneous abortion). Standard acyclovir doses for initial episodes, 400 mg three times daily for 10 days, are recommended. This is especially true for all initial episodes in the third trimester. Chronic suppressive therapy with acyclovir has been used from 36 weeks of gestation to delivery in women with an initial episode of genital HSV during pregnancy, to reduce outbreaks and prevent the need for cesarean section. This approach has been recommended by the American College of Obstetrics and Gynecology and may also be considered for women with recurrent genital herpes.
The condition of extensive congenital erosions and vesicles healing with reticulate scarring may represent intrauterine neonatal herpes simplex. The condition is rare because intrauterine HSV infection is rare and usually fatal. Probably only a few children survive to present later in life with the characteristic widespread reticulate scarring of the whole body. This may explain the associated CNS manifestations seen in many affected children. One author treated a child with this condition who developed infrequent widespread cutaneous blisters from which HSV could be cultured. Modern obstetric practices, which screen for herpes in pregnant women, and prophylactic treatment with acyclovir in the third trimester may prevent the condition, explaining the lack of recent cases.
Eczema Herpeticum (Kaposi Varicelliform Eruption)
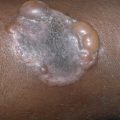
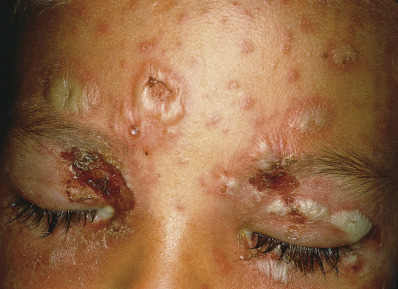
Infection with herpesvirus in patients with atopic dermatitis (AD) may result in spread of herpes simplex throughout the eczematous areas, called eczema herpeticum (EH) ( Fig. 19.9 ) or Kaposi varicelliform eruption (KVE). In a large series, development of EH was associated with more severe AD, higher IgE levels, elevated eosinophil count, food and environmental allergies (as defined by radioallergosorbent testing [RAST]), and onset of AD before age 5 years. EH patients are also more likely to have Staphylococcus aureus and molluscum contagiosum infections. All these features identify AD patients who have significant T-helper type 2 cell (Th2) shift of their immune system. The use of topical calcineurin inhibitors (TCIs) has been repeatedly associated with EH development. Bath or hot tub exposure has been reported as a risk factor. The Th2 shift of the immune system and TCIs are both associated with a decrease in antimicrobial peptides in the epidermis, an important defense against cutaneous HSV infection. Increased interleukin-10 (IL-10)–producing proinflammatory monocytes lead to local expansion of regulatory T cells and may contribute to the development of EH. HLA-B7 and local IL-25 expression are also associated with EH. In Japan, polymorphisms in the gene for IL-19 are associated with EH complicating TCI treatment. The repair of the epidermal lipid barrier with physiologic lipid mixtures reverses some of the negative effects of the TCIs and may reduce the risk of EH. One sequencing study suggested that IFNGR1 variants may confer susceptibility.
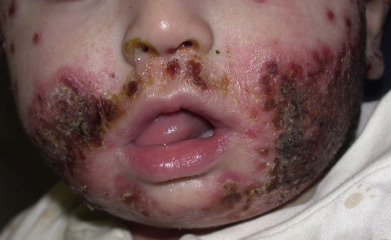
Cutaneous dissemination of HSV-1 or HSV-2 may also occur in severe seborrheic dermatitis, scabies, Darier disease, benign familial pemphigus, pemphigus (foliaceus or vulgaris), pemphigoid, cutaneous T-cell lymphoma, Wiskott-Aldrich syndrome, allergic and photoallergic contact dermatitis, burns, and other states with widespread damage to the epidermis. In its severest form, hundreds of umbilicated vesicles may be present at the onset, with fever and regional adenopathy. Although the cutaneous eruption is alarming, the disease is often self-limited in healthy individuals. Much milder cases are considerably more common and probably go unrecognized and untreated. They present as a few superficial erosions or even small papules ( Fig. 19.10 ). In patients with systemic immunosuppression in addition to an impaired barrier, such as patients with pemphigus and cutaneous T-cell lymphoma, KVE can be fatal, usually from S. aureus septicemia, but also from visceral dissemination of herpes simplex.
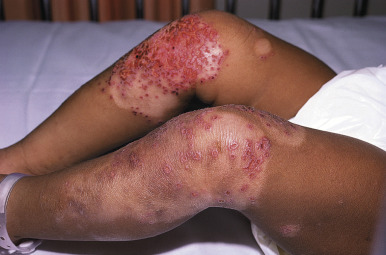
Psoriasis patients treated with immunosuppressants may develop KVE as well, although this is less common. It usually occurs in the setting of worsening disease or erythroderma. Patients present with erosive lesions in the axilla and erosions of the psoriatic plaques. Lesions extend cephalad to caudad, and the development of large, ulcerated, painful plaques can occur. The lesions are often coinfected with bacteria and yeast. Cultures positive for other pathogens do not exclude the diagnosis of KVE, and specific viral culture, DFA, and biopsy should be done if diagnosis of KVE is suspected. Given the limited toxicity of systemic antiviral therapy, treatment should be started immediately, pending the return of laboratory confirmation. Depending on the severity of the disease, either IV or oral antiviral therapy should be given for KVE patients. In children with EH, bacterial superinfection is not uncommon, and early antibiotics in patients with bacteremia are important, though empiric antibiotics are not indicated. Systemic steroids should be avoided, but topical corticosteroids during an eruption do not appear to prolong hospital stay and in some cases may be helpful, though further study is necessary.
Immunocompromised Patients
In patients with suppression of the cell-mediated immune system by cytotoxic agents, corticosteroids, or congenital or acquired immunodeficiency, primary and recurrent cases of herpes simplex are more severe, persistent, and symptomatic and more resistant to therapy. In some settings, such as in bone marrow transplant recipients, the risk of severe reactivation is so high that prophylactic systemic antivirals are administered. In immunosuppressed patients, any erosive mucocutaneous lesion should be considered to be herpes simplex until proved otherwise, especially lesions in the genital and orolabial regions. Atypical morphologies are also seen. HSV reactivation is common with institution of effective antiretroviral therapy (ART) and can be part of the immune reconstitution inflammatory syndrome (IRIS). Oral antivirals prevent this reactivation and can be considered in the HIV-infected patient who will receive ART.
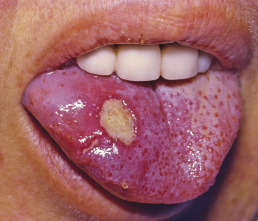
Typically, lesions appear as erosions or crusts ( Fig. 19.11 ). The early vesicular lesions may be transient or never seen. The three clinical hallmarks of HSV infection are pain, an active vesicular border, and a scalloped periphery. Untreated erosive lesions may gradually expand, but they may also remain fixed and even become papular or vegetative, mimicking a wart or granulation tissue. In the oral mucosa, numerous erosions may be seen, involving all surfaces, unlike the hard, keratinized surfaces usually involved by recurrent oral herpes simplex in the immunocompetent host. The tongue may be affected with geometric fissures on the central dorsal surface ( Fig. 19.12 ). Symptomatic stomatitis associated with cancer chemotherapy may be caused or exacerbated by HSV infection. Herpetic whitlow presents as a painful paronychia that is initially vesicular and involves the lateral or proximal nailfolds. Untreated, it may lead to loss of the nail plate and ulceration of a large portion of the digit.
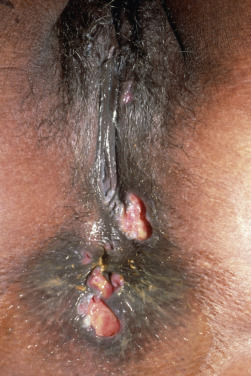
Despite the frequent and severe skin infections caused by HSV in the immunosuppressed patient, visceral dissemination is unusual. Extension of oral HSV into the esophagus or trachea may develop spontaneously or as a complication of intubation through an infected oropharynx. Ocular involvement can occur from direct inoculation, and if lesions are present around the eye, careful ophthalmologic evaluation is required.
In an immunosuppressed host, most herpetic lesions are ulcerative and not vesicular. Viral cultures or PCR from the ulcer margin are positive. At times, these tests are negative, but a skin biopsy will show typical herpetic changes in the epithelium adjacent to the ulceration. If an ulceration does not respond to treatment in 48 hours and cultures are negative, a biopsy is recommended, because it may be the only technique that demonstrates the associated herpesvirus infection.
Therapy often can be instituted on clinical grounds pending confirmatory tests. Acyclovir, 400 mg orally three times daily; famciclovir, 500 mg twice daily; or valacyclovir, 1 g twice daily, all for a minimum of 5–10 days, is used. Therapy should continue until lesions are essentially healed. In severe infection, or in the hospitalized patient with moderate disease, IV acyclovir (5 mg/kg) can be given initially to control the disease. In patients with acquired immunodeficiency syndrome (AIDS) and those with persistent immunosuppression, consideration should be given to chronic suppressive therapy with acyclovir, 400–800 mg two or three times daily, or valacyclovir or famciclovir, 500 mg twice daily.
In the immunosuppressed host (but not in the immunocompetent host), long-term treatment with acyclovir and its analogs, or treatment of large herpetic ulcerations, may be complicated by the development of acyclovir resistance. This resistance may be caused by selection of acyclovir-resistant wild-type virus, which is present in large numbers on the surface of such large herpes lesions. In the immunocompetent host, these acyclovir-resistant mutants are few in number and eradicated by the host’s immune system. The immunosuppressed host has much more HSV in the lesions, and the host’s immune system is ineffective in killing the virus. These acyclovir-resistant viral strains may be difficult to culture and may be identified only by skin biopsy or PCR of the ulceration. Antiviral resistance is suspected if maximum oral doses of acyclovir, valacyclovir, or famciclovir do not lead to improvement. IV acyclovir, except if given by constant infusion, will also invariably fail in such patients. Resistance to one drug is associated with resistance to all three of these drugs, usually from loss of the viral thymidine kinase. HSV isolates can be tested for sensitivity to acyclovir and some other antivirals. The standard treatment of acyclovir-resistant herpes simplex is IV foscarnet. In patients intolerant of or resistant to foscarnet, IV cidofovir may be used; brincidofovir is being investigated and may be an option in the future. Smaller lesions can sometimes be treated with topical trifluorothymidine (Viroptic) with or without topical or intralesional interferon (IFN) alpha, or topical or intralesional cidofovir. Imiquimod may sometimes be of benefit in healing these lesions, perhaps through activation of cystatin A. Destruction of small lesions by desiccation, followed by the previous therapies, may also be curative. If an HIV-infected patient with previous acyclovir-resistant genital herpes has a recurrence, at least half will be acyclovir sensitive, as the latent virus within the nerve that is reactivating is usually the initial virus from first infection, so a trial of standard antivirals is acceptable. If an AIDS patient has a nonhealing genital ulcer that harbors HSV, there may be dual infection with cytomegalovirus, and only treatment with an agent active against both HSV and CMV will lead to improvement in those cases.
Histopathology
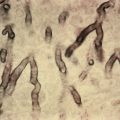
The vesicles of herpes simplex are intraepidermal. The affected epidermis and adjacent inflamed dermis are infiltrated with leukocytes. Ballooning degeneration of the epidermal cells produces acantholysis. The most characteristic feature is the presence of multinucleated giant cells, which tend to mold together, forming a crude jigsaw puzzle appearance. The steel-gray color of the nucleus and peripheral condensation of the nucleoplasm may be clues to HSV infection, even if multinucleate cells are not seen. IP stains can detect HSV and differentiate from VZV even in paraffin-fixed tissue, allowing the diagnosis to be absolutely confirmed from histologic material.
Differential Diagnosis
Herpes labialis most often must be differentiated from impetigo. Herpetic lesions are composed of groups of tense, small vesicles, whereas in bullous impetigo, the blisters are unilocular, occur at the periphery of a crust, and are flaccid. A mixed infection is not unusual and should especially be suspected in immunosuppressed hosts and when lesions are present in the typical herpetic regions around the mouth. Herpes zoster presents with clusters of lesions along a dermatome, but early on, if the number of zoster lesions is limited, it can be relatively indistinguishable from herpes simplex. In general, herpes zoster will be more painful and over 24 hours will progress to involve more of the affected dermatome.
A genital herpes lesion, especially on the glans or corona, can be mistaken for a syphilitic chancre or chancroid. Darkfield examination, multiplex PCR, and cultures for Haemophilus ducreyi on selective media will aid in making the diagnosis, as will diagnostic tests for HSV (Tzanck, culture, or DFA). Combined infections occur in up to 20% of patients, so finding a single pathogen may not complete the diagnostic evaluation, and patients are often empirically treated for multiple ulcer-forming STDs and screened for other STDs when one is diagnosed.
Herpetic gingivostomatitis is often difficult to differentiate from aphthosis, streptococcal infections, diphtheria, coxsackievirus infections, and oral erythema multiforme. Aphthae tend to occur mostly on the buccal and labial mucosae. They usually form shallow, grayish erosions, generally surrounded by a prominent ring of hyperemia. Aphthae typically occur on nonattached mucosa, whereas recurrent herpes of the oral cavity primarily affects the attached gingiva and palate.
Aronson PL, et al: Empiric antibiotics and outcomes of children hospitalized with eczema herpeticum. Pediatr Dermatol 2013; 30: 207.
Aronson PL, et al: Topical corticosteroids and hospital length of stay in children with eczema herpeticum. Pediatr Dermatol 2013; 30: 215.
Ashack KA, et al: Skin infections among US high school athletes. J Am Acad Dermatol 2016; 74: 679.
Beck LA, et al: Phenotype of atopic dermatitis subjects with a history of eczema herpeticum. J Allergy Clin Immunol 2009; 124: 260.
Bernstein DI, et al: Epidemiology, clinical presentation, and antibody response to primary infection with herpes simplex virus type 1 and type 2 in young women. Clin Infect Dis 2013; 56: 344.
Bibbins-Dombingo K, et al: Serologic screening for genital herpes infection. JAMA 2016; 316: 2525.
Bradley H, et al: Seroprevalence of herpes simplex virus types 1 and 2—United States, 1999–2010. J Infect Dis 2014; 209: 325.
Brown ZA, et al: Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant. JAMA 2003; 289: 203.
Chen CY, Ballard RC: The molecular diagnosis of sexually transmitted genital ulcer disease. Methods Mol Biol 2012; 903: 103.
Chosidow O, et al: Valacyclovir as a single dose during prodrome of herpes facialis. Br J Dermatol 2003; 148: 142.
Corey L, et al: Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med 2004; 354: 11.
De SK, et al: Herpes simplex virus and varicella zoster virus. Curr Opin Infect Dis 2015; 28: 589.
Dickson N, et al: HSV-2 incidence by sex over four age periods to age 38 in a birth cohort. Sex Transm Infect 2014; 90: 243.
Fatahzadeh M, Schwartz RA: Human herpes simplex virus infections. J Am Acad Dermatol 2007; 57: 737.
Gao L, et al: Targeted deep sequencing identifies rare loss-of-function variants in IFNGR1 for risk of atopic dermatitis complicated by eczema herpeticum. J Allergy Clin Immunol 2015; 136: 1591.
Gazzola R, et al: Herpes virus outbreaks after dermal hyaluronic acid filler injections. Aesthet Surg J 2012; 32: 770.
Hirokawa D, et al: Treatment of recalcitrant herpes simplex virus with topical imiquimod. Cutis 2011; 88: 276.
Hollier LM, et al: Third trimester antiviral prophylaxis for preventing maternal genital herpes simplex virus (HSV) recurrences and neonatal infection. Cochrane Database Syst Rev 2008; 1: CD004946.
Jiang YC, et al: New strategies against drug resistance to herpes simplex virus. Int J Oral Sci 2016; 8: 1.
Johnston C, et al: Current concepts for genital herpes simplex virus infection. Clin Microbiol Rev 2016; 29: 149.
Jones CA, et al: Antiviral agents for treatment of herpes simplex virus infection in neonates. Cochrane Database Syst Rev 2009; 3: CD004206.
Kan Y, et al: Imiquimod suppresses propagation of herpes simplex virus 1 by upregulation of cystatin A via the adenosine receptor A 1 pathway. J Virol 2012; 86: 10338.
Katz K: Screening for genital herpes. JAMA Dermatol 2017; 153: 265.
Kim BE, et al: IL-25 enhances HSV-1 replication by inhibiting filaggrin expression, and acts synergistically with Th2 cytokines to enhance HSV-1 replication. J Invest Dermatol 2013; 133: 2678.
Kortekangas-Savolainen O, et al: Epidemiology of genital herpes simplex virus type 1 and 2 infections in southwestern Finland during a 10-year period (2003–2012). Sex Transm Dis 2014; 41: 268.
Kotzbauer D, et al: Clinical and laboratory characteristics of central nervous system herpes simplex virus infection in neonates and young infants. Pediatr Infect Dis J 2014; 33: 1187.
Leas BF, et al: Neonatal HSV-1 infection and Jewish ritual circumcision with oral suction. J Pediatric Infect Dis Soc 2015; 4: 126.
Leung DY: Why is eczema herpeticum unexpectedly rare? Antiviral Res 2013; 98: 153.
Looker KJ, et al: First estimates of the global and regional incidence of neonatal herpes infection. Lancet Glob Health 2017; 5: e300.
Mathais RA, et al: Atopic dermatitis complicated by eczema herpeticum is associated with HLA B7 and reduced interferon-γ-producing CD8+ T cells. Br J Dermatol 2013; 169: 700.
Muluneh B, et al: Successful clearance of cutaneous acyclovir-resistant, foscarnet-refractory herpes virus lesions with topical cidofovir in an allogeneic hematopoietic stem cell transplant patient. J Oncol Pharm Pract 2013; 19: 181.
Palli MA, et al: Immunotherapy of recurrent herpes labialis with squaric acid. JAMA Dermatol 2017; 153: 828.
Pichler M, et al: Premature newborns with fatal intrauterine herpes simplex virus-1 infection. J Eur Acad Dermatol Venereol 2015; 29: 1216.
Robinson JL, et al: Prevention, recognition and management of neonatal HSV infections. Expert Rev Anti Infect Ther 2012; 10: 675.
Rojek NW, Norton SA: Diagnosis of neonatal infection with herpes simplex virus. JAMA 2014; 311: 527.
Schoenfeld J, et al: Cutaneous co-infected cytomegalovirus and herpes simplex virus perigenital ulcers in human immunodeficiency virus patients. J Clin Aesthet Dermatol 2013; 6: 41.
Seang S, et al: Long-term follow-up of HIV-infected patients once diagnosed with acyclovir-resistant herpes simplex virus infection. Int J STD AIDS 2014; 25: 676.
Shenoy R, et al: Eczema herpeticum in a wrestler. Clin J Sport Med 2015; 25: e18.
Takahashi R, et al: Pathological role of regulatory T cells in the initiation and maintenance of eczema herpeticum lesions. J Immunol 2014; 192: 969.
Tobian AA, et al: Reactivation of herpes simplex virus type 2 after initiation of antiretroviral therapy. J Infect Dis 2013; 208: 839.
Wanat KA, et al: Intralesional cidofovir for treating extensive genital verrucous herpes simplex virus infection. JAMA Dermatol 2013; 149: 811.
Wanat KA, et al: Bedside diagnostics in dermatology. J Am Acad Dermatol 2017; 77: 197.
Whitley RJ: Changing epidemiology of herpes simplex virus infections. Clin Infect Dis 2013; 56: 352.
Wollenberg A, et al: Predisposing factors and clinical features of eczema herpeticum. J Am Acad Dermatol 2003; 49: 198.
Workowski KA, et al: Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep 2015; 64: 1.
Varicella
Varicella, commonly known as chickenpox, is the primary infection with the VZV. In temperate regions, 90% of cases occur in children younger than 10 years, with the highest age-specific incidence in ages 1–4 in unvaccinated children. More than 90% of adults in temperate countries have evidence of prior infection and are “immune” to varicella by age 15. In tropical countries, however, varicella tends to be a disease of teenagers, and only 60% of adults are “immune” serologically. Outbreaks among non–U.S.-born crew members have occurred on cruise ships.
The incubation period of VZV is 10–21 days, usually 14–15 days. Transmission is by the respiratory route and less often by direct contact with the lesions. A susceptible person may develop varicella after exposure to the lesions of herpes zoster. Infected persons are infectious from 5 days before the eruption appears and are most infectious 1–2 days before the rash appears. Infectivity usually ceases 5–6 days after the eruption appears. There is an initial viral replication in the nasopharynx and conjunctiva, followed by viremia and infection of the reticuloendothelial system (liver, spleen) between days 4 and 6. A secondary viremia occurs at days 11–20, resulting in infection of the epidermis and the appearance of the characteristic skin lesions. Low-grade fever, malaise, and headache are usually present but slight. The severity of the disease is age dependent, with adults having more severe disease and a greater risk of visceral disease. In healthy children, mortality from varicella is 1.4 in 100,000 cases; in adults, 30.9 in 100,000 cases. Pregnant women have five times greater risk of an adverse outcome. As with most viral infections, immunosuppression may worsen the course of the disease. Lifelong immunity follows varicella, and second episodes of “varicella” indicate either immunosuppression or another viral infection such as coxsackievirus.
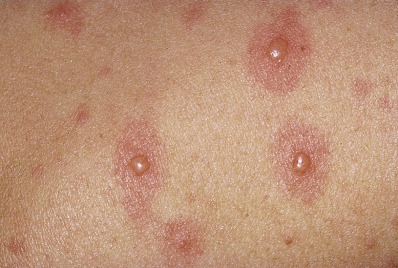
Varicella is characterized by a vesicular eruption consisting of delicate “teardrop” vesicles on an erythematous base ( Fig. 19.13 ). The eruption starts with faint macules that develop rapidly into vesicles within 24 hours. Successive fresh crops of vesicles appear for a few days, mainly on the trunk, face, and oral mucosa. Initially, the exanthem may be limited to sun-exposed areas, the diaper area of infants, or sites of inflammation. The vesicles quickly become pustular and umbilicated, then crusted. Because the lesions appear in crops, lesions of various stages are present at the same time, a useful clue to the diagnosis. Lesions tend not to scar, but larger lesions and those that become secondarily infected may heal with a characteristic round, depressed scar.
Secondary bacterial infection with S. aureus or a streptococcus is the most common complication of varicella ( Fig. 19.14 ). Rarely, it may be complicated by osteomyelitis, other deep-seated infections, or septicemia. Other complications are rarer. Pneumonia is uncommon in normal children but is seen in 1 in 400 adults with varicella. It may be bacterial or caused by VZV, a difficult differential diagnosis. Cerebellar ataxia and encephalitis are the most common neurologic complications. VZV can rarely infect cerebral arteries and cause a vasculopathy leading to stroke, sinus thromboses, and giant cell arteritis. VZV may cause stroke in both children and adults. Asymptomatic myocarditis and hepatitis may occur in children with varicella, but rarely are significant and resolve spontaneously with no treatment. Reye syndrome, with hepatitis and acute encephalopathy, is associated with the use of aspirin to treat the symptoms of varicella. Aspirin is absolutely contraindicated in patients with varicella. Any child with varicella and severe vomiting should be referred immediately to exclude Reye syndrome. Symptomatic thrombocytopenia is a rare manifestation of varicella, which can occur either with the exanthem or several weeks after. Purpura fulminans, a form of disseminated intravascular coagulation associated with low levels of proteins C and S, may complicate varicella.
The diagnosis of varicella is easily made clinically. Tzanck smear from a vesicle will usually show characteristic multinucleate giant cells, and DFA can confirm the infection and type the virus. PCR is increasingly used due to high sensitivity and specificity. VZV grows poorly and slowly in the laboratory, so viral culture is rarely indicated.
Treatment
Both immunocompetent children and adults with varicella benefit from acyclovir therapy if started early, within 24 hours of the eruption’s appearance. Therapy does not seem to alter the development of adequate immunity to reinfection. Because the complications of varicella are infrequent in children, routine treatment is not recommended; therapeutic decisions are made on a case-by-case basis. Acyclovir therapy appears mainly to benefit secondary cases within a household, which tend to be more severe than the index case. In this setting, therapy can be instituted earlier. Therapy does not return children to school sooner, however, and the impact on parental workdays missed is not known. The dose of acyclovir is 20 mg/kg, maximum 800 mg per dose, four times daily for 5 days. Aspirin and other salicylates should not be used as antipyretics in varicella because their use increases the risk of Reye syndrome. Topical antipruritic lotions, oatmeal baths, dressing the patient in light, cool clothing, and keeping the environment cool may all relieve some of the symptomatology. Children living in warm homes and kept very warm with clothing have anecdotally been observed to have more numerous skin lesions. Children with AD, Darier disease, congenital ichthyosiform erythroderma, diabetes, cystic fibrosis, conditions requiring chronic salicylate or steroid therapy, and inborn errors of metabolism should be treated with acyclovir, because they may have more complications or exacerbations of their underlying illness with varicella.
Varicella is more severe and complications are more common in adults. Between 5% and 14% of adults will have pulmonary involvement. Smokers and those with preexisting lung disease (but not asthma) are at increased risk. The pneumonitis can progress rapidly and be fatal. Adults with varicella and at least one other risk factor should be evaluated with physical examination, pulse oximetry, and chest radiography. Antiviral treatment is recommended in all adolescents and adults (13 and older) with varicella. The dose is 800 mg four or five times daily for 5 days. Severe, fulminant cutaneous disease and visceral complications are treated with IV acyclovir, 10 mg/kg every 8 hours, adjusted for creatinine clearance. If the patient is hospitalized for therapy, strict isolation is required. Patients with varicella should not be admitted to wards with immunocompromised hosts or to pediatric wards, but rather are best placed on wards with healthy patients recovering from acute trauma.
Pregnant Women and Neonates
Maternal VZV infection may result in severe illness in the mother, and if the infection occurs before 28 weeks of gestation, and especially before 20 weeks, a small but significant risk of infection of the fetus (congenital varicella syndrome). In one study, 4 of 31 women with varicella in pregnancy developed varicella pneumonia. The risk for spontaneous abortion by 20 weeks is 3%; in an additional 0.7% of pregnancies, fetal death occurs after 20 weeks. The risk of preterm labor, as reported in various studies, has varied from no increased risk to a threefold increase. Severe varicella and varicella pneumonia or disseminated disease in pregnancy should be treated with IV acyclovir. All varicella in pregnancy should be treated with oral acyclovir, 800 mg five times daily for 7 days, (adjusted for renal function) except perhaps during the first month, when a specialist should be consulted. In all women past 35 weeks of gestation or with increased risk of premature labor, admission and IV acyclovir, 10 mg/kg three times daily, (“adjusted for renal function”) should be recommended. The patient should be evaluated for pneumonia, renal function should be carefully monitored, and the patient should be switched to oral therapy once lesions stop appearing (usually in 48–72 hours).
Varicella-zoster immune globulin (VZIG) should not be given once the pregnant woman has developed varicella. VZIG should be given for significant exposures (see next section) within the first 72–96 hours to ameliorate maternal varicella and prevent complications. Its use should be limited to seronegative women because of its cost and the high rate of asymptomatic infection in U.S. patients. The lack of a history of prior varicella is associated with seronegativity in only 20% or fewer of the U.S. population.
Congenital varicella syndrome is characterized by a series of anomalies, including hypoplastic limbs (usually unilateral and lower extremity), cutaneous scars, and ocular and CNS disease. This may not be identified until months after infection. Repeated sonographic examination can be used to monitor at-risk pregnancies. Female fetuses are affected more often than males. The overall risk for this syndrome is about 0.4%; the highest risk, about 2%, is from maternal varicella between weeks 13 and 20. Infection of the fetus in utero may result in zoster occurring postnatally, often in the first 2 years of life. This occurs in about 1% of varicella-complicated pregnancies, and the risk for this complication is greatest in varicella occurring in weeks 25–36 of gestation. The value of VZIG in preventing or modifying fetal complications of maternal varicella is unknown. In one study, however, of 97 patients with varicella in pregnancy treated with VZIG, none had complications of congenital varicella syndrome or infantile zoster, suggesting some efficacy for VZIG. Although apparently safe in pregnancy, acyclovir’s efficacy in preventing fetal complications of maternal varicella is unknown.
If the mother develops varicella between 5 days before and 2 days after delivery, neonatal varicella can occur and may be severe because of inadequate transplacental delivery of antivaricella antibody. These neonates develop varicella at 5–10 days of age. In such cases, administration of VZIG is warranted, and IV acyclovir therapy should be considered.
Varicella Vaccine
Live attenuated viral vaccine for varicella is a currently recommended childhood immunization. Two doses are now recommended, one between age 12 and 15 months and the second at 4–6 years. This double-vaccination schedule is recommended because epidemics of varicella still occurred in children ages 9–11 in well-immunized communities, suggesting a waning of immunity by this age. The introduction of widespread vaccination has led to a dramatic decrease in varicella-related hospitalizations. Complications of varicella vaccination are uncommon, and severe complications are rare. A systematic literature review suggested that there were approximately 50–60 breakthrough varicella cases with disseminated infection associated with over 30 million doses of virus. A mild skin eruption from which virus usually cannot be isolated, occurring locally at the injection site within 2 days or generalized 1–3 weeks after immunization, occurs in 6% of children. Many of the breakthrough cases in vaccinated children are mild, and many reported skin lesions were not vesicular (see next section, Modified Varicella-like Syndrome). Prevention of severe varicella is virtually 100%, even when the vaccine is given within 36 hours of exposure. Immunized children with no detectable antibody also have reduced severity of varicella after exposure. Secondary complications of varicella, including scarring, are virtually eliminated by vaccination. One theoretical population-based consequence of widespread vaccination and reduced rates of infection is whether there will be an observed loss of natural “booster” exposures to the virus, and perhaps a changing epidemiology of the disease and increased incidence of zoster (shingles).
Household exposure of immunosuppressed children to recently immunized siblings does not appear to pose a great risk. Children whose leukemia is in remission are also protected by the vaccine but may require three doses. Leukemic children still receiving chemotherapy have a complication rate from vaccination (usually a varicella-like eruption) approaching 50%. They may require acyclovir therapy. Unprotected close contacts develop varicella 15% of the time. In leukemic children, adequate immunization results in complete immunity in some and partial immunity in the others, protecting them from severe varicella. Immunization also reduces the attack rate for zoster in leukemic children. As a live vaccine, administration is contraindicated during pregnancy and in immunosuppressed patients.
Modified Varicella-Like Syndrome.
Children immunized with live attenuated varicella vaccine may develop varicella of reduced severity on exposure to natural varicella. This has been called modified varicella-like syndrome (MVLS). The frequency of MVLS is between 0% and 2.7% per year, and children with lower antibody titers are more likely to develop the illness. MVLS occurs an average of 15 days after exposure to varicella and consists primarily of macules and papules with relatively few vesicles. The average number of lesions is about 35–50, compared with natural varicella, which usually has about 300 lesions. The majority of patients are afebrile and the illness is mild, lasting fewer than 5 days on average.
Immunocompromised Patients
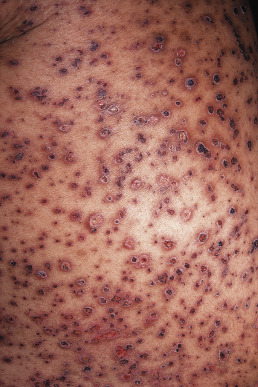
Varicella cases can be extremely severe and even fatal in immunosuppressed patients, especially in individuals with impaired cell-mediated immunity. Before effective antiviral therapy, almost one third of children with cancer developed complications of varicella and 7% died. In this setting, varicella pneumonia, hepatitis, and encephalitis are common. Prior varicella does not always protect the immunosuppressed host from multiple episodes. The skin lesions in the immunosuppressed host are usually identical to varicella in the healthy host; however, the number of lesions may be numerous ( Fig. 19.15 ). In an immunosuppressed patient, the lesions more frequently become necrotic, and ulceration may occur. Even if the lesions are few, the size of the lesion may be large, up to several centimeters, and necrosis of the full thickness of the dermis may occur. In patients with HIV infection, varicella may be severe and fatal. Atypical cases of a few scattered lesions without a dermatomal distribution usually represent reactivation disease with dissemination. Chronic varicella may complicate HIV infection, resulting in ulcerative (ecthymatous) or hyperkeratotic (verrucous) lesions. These patterns of infection may be associated with acyclovir resistance.
The degree of immunosuppression likely to result in severe varicella has been debated. There are case reports of severe and even fatal varicella in otherwise healthy children given short courses of oral corticosteroids or even using only inhaled corticosteroids. In a case-control study, however, corticosteroid use did not appear to be a risk factor for development of severe varicella. In the United Kingdom, any patient receiving or having received systemic corticosteroids in the prior 3 months, regardless of dose, is considered at increased risk for severe varicella. Inhaled steroids are not considered an indication for prophylactic antiviral treatment. A “high-risk” or significant exposure is defined as follows:
- 1.
Household contact (i.e., living in same house as a patient with chickenpox or zoster)
- 2.
Face-to-face contact for at least 5 minutes with a patient who has chickenpox
- 3.
Contact indoors for more than 1 hour with a patient who has chickenpox or herpes zoster or, in a hospital setting, a patient with chickenpox or herpes zoster in an adjacent bed or the same open ward
Immunosuppressed children with no prior history of varicella and a high-risk exposure should be treated with VZIG as soon as possible after exposure (within 96 hours). Preengraftment bone marrow transplant patients should receive the same therapy. VZIG treatment does not reduce the frequency of infection, but it does reduce the severity of infection and complications. The value of prophylactic antivirals is unknown. Parents of immunosuppressed children and their physicians should be aware that severe disease can occur and counseled to return immediately after significant exposure or if varicella develops.
An unusual variant of recurrent varicella is seen in elderly patients with a history of varicella in childhood, who have a malignancy of the bone marrow and are receiving chemotherapy. They develop a mild illness with 10–40 widespread lesions and usually no systemic findings. This type of recurrent varicella tends to relapse. It is different from typical varicella because all the lesions are in a single stage of development, and thus it could be easily confused with smallpox.
Ideally, management of varicella in the immunocompromised patient would involve prevention through varicella vaccination before immunosuppression. Vaccination is safe if the person is more than 1 year from induction chemotherapy, if chemotherapy is halted about the time of vaccination, and if lymphocyte count is higher than 700/mm 3 . IV acyclovir, 10 mg/kg three times daily (or 500 mg/m 2 in children) is given as soon as the diagnosis of varicella is suspected. IV therapy is continued until 2 days after all new vesicles have stopped. Oral antivirals are continued for a minimum of 10 days of treatment. VZIG is of no proven benefit once clinical disease has developed, but may be given if the patient has life-threatening disease and is not responding to IV acyclovir.
In HIV-infected adults, treatment is individualized. Persons with typical varicella should be evaluated for the presence of pneumonia or hepatitis. Valacyclovir, 1 g three times daily; famciclovir, 500 mg three times daily; or acyclovir, 800 mg 5 times daily, may be used if no visceral complications are present. Valacyclovir and famciclovir may be preferable to acyclovir because of their enhanced oral bioavailability. Visceral disease mandates IV therapy. If the response to oral antiviral agents is not rapid, IV acyclovir therapy should be instituted. The optimal duration of oral antiviral treatment is unknown but must be at least until all lesions are crusted and have no elevated or active borders. Given the safety and efficacy of oral antivirals, treatment duration of at least 10 days and perhaps longer should be considered. Most cases of chronic or acyclovir-resistant VZV infection are associated with initial inadequate oral doses of acyclovir (too short in duration, too low a dose, or in patients with gastrointestinal [GI] disease, in whom reduced GI absorption may be associated with inadequate blood levels of acyclovir). Patients with atypical disseminated disease must be treated aggressively until all lesions resolve. The diagnosis of acyclovir-resistant VZV infection may be difficult. Acyclovir-resistant VZV strains may be difficult to culture, and sensitivity testing is still not standardized or readily available for VZV. Acyclovir-resistant varicella is treated with foscarnet or, in nonresponsive patients, with cidofovir.
Amlie-Lefond C, et al: Varicella zoster virus. J Stroke Cerebrovasc Dis 2016; 25: 1561.
Bate J, et al: Varicella postexposure prophylaxis in children with cancer. Arch Dis Child 2012; 97: 853.
Baxter R, et al: Long-term effectiveness of varicella vaccine. Pediatrics 2013; 131: e1389.
Bollaerts K, et al: A systematic review of varicella seroprevalence in European countries before universal childhood immunization. Epidemiol Infect 2017; 145: 2666.
Centers for Disease Control and Prevention (CDC): FDA approval of an extended period for administering VariZIG for postexposure prophylaxis of varicella. MMWR Morb Mortal Wkly Rep 2012; 61: 212.
Creed E, et al: Varicella zoster vaccines. Dermatol Ther 2009; 22: 143.
Gabutti G, et al: Varicella-zoster virus. Minerva Pediatr 2016; 68: 213.
Hirose M, et al: The impact of varicella vaccination on varicella-related hospitalization rates. Rev Paul Pediatr 2016; 34: 359.
Kao CM, et al: Child and adolescent immunizations. Curr Opin Pediatr 2014; 26: 383.
Leung J, et al: Severe varicella in persons vaccinated with varicella vaccine. Expert Rev Vaccines 2017; 16: 391.
Marin M, et al: Varicella prevention in the United States. Pediatrics 2008; 122: e744.
Marin M, et al: Global varicella vaccine effectiveness. Pediatrics 2016; 137: e20153741.
Nagel MA, et al: Varicella zoster virus vasculopathy. J Neuroimmunol 2017; 308: 112.
Sauerbrei A: Diagnosis, antiviral therapy, and prophylaxis of varicella-zoster virus infections. Eur J Clin Microbiol Infect Dis 2016; 35: 723.
Sugiura K, et al: Varicella zoster virus-associated generalized pustular psoriasis in a baby with heterozygous IL36RN mutation. J Am Acad Dermatol 2014; 71: e216.
Wilson DA, et al: Should varicella-zoster virus culture be eliminated? A comparison of direct immunofluorescence antigen detection, culture, and PCR with a historical review. J Clin Microbiol 2012; 50: 4120.
Varicella-Zoster Virus (Zoster, Shingles, Herpes Zoster), Human Herpesvirus Type 3
Zoster is caused by reactivation of VZV. Following primary infection or vaccination, VZV remains latent in the sensory dorsal root ganglion cells. The virus begins to replicate at some later time, traveling down the sensory nerve into the skin. Immunosuppression, including use of tumor necrosis factor (TNF) inhibitors, Janus kinase Janus kinase (JAK)-inhibitors, and some cancer therapies, HIV, and age-related deficiency of cell-mediated immunity are some of the most common causes of zoster. A family history of zoster is associated with an increased risk of developing zoster, suggesting a genetic risk component. Patients on hemodialysis and those with comorbidities have increased risk of zoster, possibly related to the association between zoster risk and cholesterol level. Statin use also slightly increases the risk for zoster. Zoster patients are more likely to be subsequently diagnosed with a malignancy, especially a hematologic malignancy, with an absolute risk of any cancer of 0.7%–1.8% at 1 year after zoster.
The incidence of zoster increases with age. Under age 45, the annual incidence is less than 1 in 1000 persons. Among patients older than 75, the rate is more than four times greater. For white persons older than 80, the lifetime risk of developing zoster is 10%–30%. Women appear to have higher risk than men. Overall, about one in three unvaccinated persons will develop herpes zoster. For unknown reasons, being nonwhite reduces the risk for herpes zoster, with African Americans one half to one fourth less likely to develop zoster. Autoimmune diseases, asthma, diabetes, and chronic obstructive pulmonary disease are also associated with increased risk. Immunosuppression, especially hematologic malignancy and HIV infection, dramatically increases the risk for zoster. In HIV-infected patients, annual incidence is 30 in 1000 persons, or an annual risk of 3%. With the universal use of varicella vaccination and decrease in pediatric and adolescent varicella cases, older persons will no longer have periodic boosts of the anti-VZV immune activity. Theoretically this could result in an increase in the incidence of zoster.
Herpes zoster classically occurs unilaterally within the distribution of a cranial or spinal sensory nerve, often with some overflow into the dermatomes above and below. The dermatomes most frequently affected are the thoracic (55%), cranial (20%, with the trigeminal nerve being the most common single nerve involved), lumbar (15%), and sacral (5%). The cutaneous eruption is frequently preceded by one to several days of pain in the affected area, although the pain may appear simultaneously or even after the skin eruption, or the eruption may be painless. The eruption initially presents as papules and plaques of erythema in the dermatome ( Fig. 19.16 ). Within hours the plaques develop blisters. Lesions continue to appear for several days. The eruption may have few lesions or reach total confluence in the dermatome. Lesions may become hemorrhagic, necrotic, or bullous. Rarely, the patient may have pain, but no skin lesions (zoster sine herpete). Pain severity correlates with extent of the skin lesions, and elderly persons tend to have more severe pain. In patients under age 30, the pain may be minimal. Scattered lesions can occur outside the dermatome, usually fewer than 20. In the typical case, new vesicles appear for 1–5 days, become pustular, crust, and heal. The total duration of the eruption depends on three factors: patient age, severity of eruption, and presence of underlying immunosuppression. In younger patients, the total duration is 2–3 weeks, whereas in elderly patients, the cutaneous lesions of zoster may require 6 weeks or more to heal. Scarring is more common in elderly and immunosuppressed patients. Scarring also correlates with the severity of the initial eruption. Lesions may develop on the mucous membranes within the mouth in zoster of the maxillary division ( Fig. 19.17 ) or mandibular division of the facial nerve, or in the vagina with zoster in the S2 or S3 dermatome. Zoster may appear in recent surgical scars and may follow injections of botulinum toxin.
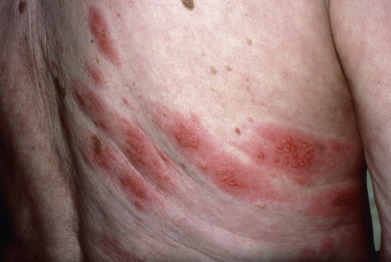
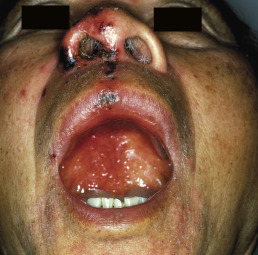
Zoster may rarely be seen in children under age 1 year. This can result from intrauterine exposure to VZV or exposure to VZV during the first few months of life. The maternal antibodies still present result in muted expression of varicella—subclinical or very mild disease. The immaturity of the infant’s immune system results in poor immune response to the infection, allowing for early relapse in the form of zoster.
Disseminated Herpes Zoster
Disseminated herpes zoster is defined as more than 20 lesions outside the affected dermatome. It occurs chiefly in older or debilitated individuals, especially in patients with lymphoreticular malignancy or AIDS. Low levels of serum antibody against VZV are a highly significant risk factor in predicting dissemination of disease. The dermatomal lesions are sometimes hemorrhagic or gangrenous. The outlying vesicles or bullae, which are usually not grouped, resemble varicella and are often umbilicated and may be hemorrhagic. Visceral dissemination to the lungs, liver, and/or CNS may occur in the patient with disseminated zoster. Disseminated zoster requires careful evaluation and systemic antiviral therapy. Initially, IV acyclovir is given, which may be changed to an oral antiviral agent once visceral involvement has been excluded and the patient has received at least 2–3 days of IV therapy.
Ophthalmic Zoster
In herpes zoster ophthalmicus, the ophthalmic division of the fifth cranial nerve is involved. If the external division of the nasociliary branch is affected, with vesicles on the side and tip of the nose (Hutchinson sign), the eye is involved 76% of the time, compared with 34% when it is not involved ( Fig. 19.18 ). Vesicles on the lid margin are virtually always associated with ocular involvement. In any case, the patient with ophthalmic zoster should be seen by an ophthalmologist. Systemic antiviral therapy should be started immediately, pending ophthalmologic evaluation. Ocular involvement is most often in the form of uveitis (92%) and keratitis (50%). Less common but more severe complications include glaucoma, optic neuritis, encephalitis, hemiplegia, and acute retinal necrosis. These complications are reduced from 50% of patients to 20%–30% with effective antiviral therapy. Unlike the cutaneous lesions, ocular lesions of zoster and their complications tend to recur, sometimes as long as 10 years after the zoster episode.
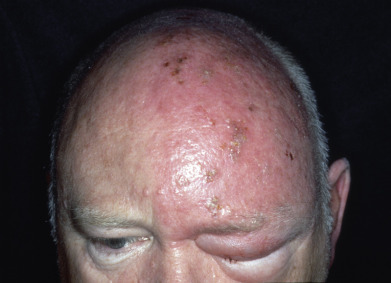
Other Complications
Motor nerve neuropathy occurs in about 3% of patients with zoster and is three times more common if zoster is associated with underlying malignancy. About 75% of patients slowly recover, leaving 25% with some residual motor deficit. Thoracic zoster may be associated with motor neuropathy of the abdominal muscles resulting in a bulge on the flank or abdomen, called a “postherpetic pseudotumor.” If the sacral dermatome S3, or less often S2 or S4, is involved, urinary hesitancy or actual urinary retention may occur. Hematuria and pyuria may also be present. The prognosis is good for complete recovery. Similarly, pseudoobstruction, colonic spasm, dilation, obstipation, constipation, and reduced anal sphincter tone can occur with thoracic (T6–T12), lumbar, or sacral zoster. Recovery is complete. Maxillary and mandibular alveolar bone necrosis may occur an average of 30 days after zoster of the maxillary or mandibular branches of the trigeminal nerve. Limited or widespread loss of teeth may result.
Ramsay Hunt syndrome (herpes zoster oticus) results from involvement of the facial and auditory nerves by VZV. Herpetic inflammation of the geniculate ganglion is thought to be the cause of this syndrome. The presenting features include zoster of the external ear or tympanic membrane; herpes auricularis with ipsilateral facial paralysis; or herpes auricularis, facial paralysis, and auditory symptoms. Auditory symptoms include mild to severe tinnitus, deafness, vertigo, nausea and vomiting, and nystagmus.
Herpes zoster can be associated with delayed complications, many of which are caused by vasculopathies affecting the CNS or even the peripheral arteries. There appears to be an increased risk of both ischemic and hemorrhagic stroke within the first 3 months after zoster, which fades by 1 year. Delayed contralateral hemiparesis, simulating cerebrovascular accident (stroke), is a rare but serious complication of herpes zoster that occurs weeks to months (mean 7 weeks) after an episode of zoster affecting the first branch of the trigeminal nerve. By direct extension along the intracranial branches of the trigeminal nerve, VZV gains access to the CNS and infects the cerebral arteries. Patients present with headache and hemiplegia. Arteriography is diagnostic, demonstrating thrombosis of the anterior or middle cerebral artery. This form of vasculopathy can also occur after varicella and may be the cause of up to one third of ischemic strokes in children. The recognized vasculopathic complications of VZV have been expanded to include changes in mental status, aphasia, ataxia, hemisensory loss, and both hemianopia and monocular visual loss. Monocular vision loss can occur up to 6 months after zoster. Aneurysm, subarachnoid or cerebral hemorrhage, carotid dissection, and even peripheral vascular disease are other recognized forms of VZV vasculopathy. The vasculopathy may be multifocal and involve both large and small arteries. In more than one third of cases, VZV vasculopathy occurs without a rash. Magnetic resonance imaging (MRI) is virtually always abnormal. The diagnosis is confirmed by VZV PCR and anti-VZV IgG antibody testing of the CSF. Because this is caused by active viral replication in the vessels, the treatment is IV acyclovir, 10–15 mg/kg three times daily for a minimum of 14 days. In some patients, months of oral antivirals are given if symptoms are slow to resolve. A short burst of systemic corticosteroids is also given in some cases. VZV has been linked to giant-cell arteritis as well, theoretically through similar mechanisms. One recent meta-analysis has also suggested an increased risk of cardiac events associated with herpes zoster as well.
Treatment
Middle-age and elderly patients with herpes zoster are urged to restrict their physical activities or even stay home in bed for a few days. Bed rest may be of paramount importance in the prevention of neuralgia. Younger patients may usually continue with their customary activities. Local applications of heat, as with an electric heating pad or a hot water bottle, are recommended. Simple local application of gentle pressure with the hand or with an abdominal binder often gives great relief.
Antiviral therapy is the cornerstone in the management of herpes zoster. Because antiviral therapy does not reduce the rate of zoster-associated pain (ZAP), clinicians may underappreciate the tremendous benefit that antivirals provide. The main benefit of therapy is reduction of the duration and severity of ZAP. Therefore treatment in immunocompetent patients is indicated for those at highest risk for persistent pain—those over age 50. It is also recommended to treat all patients with painful or severe zoster, ophthalmic zoster, Ramsay Hunt syndrome, immunosuppression, cutaneous or visceral dissemination, and motor nerve involvement. In the most severe cases, especially in ophthalmic zoster and disseminated zoster, initial IV therapy may be considered. Therapy should be started as soon as the diagnosis is suspected, pending laboratory confirmation. It is preferable for treatment to be instituted within the first 3 or 4 days. In immunocompetent patients, the efficacy of starting treatment beyond this time is unknown. Treatment leads to more rapid resolution of the skin lesions and, most importantly, substantially decreases the duration of ZAP. Valacyclovir, 1000 mg, and famciclovir, 500 mg, may be given three times daily. These agents are as effective as or superior to acyclovir, 800 mg five times daily, probably because of better absorption or medication compliance with simpler regimens. Side-by-side trials have demonstrated valacyclovir and famciclovir to be of similar efficacy, but one study from Japan showed famciclovir to result in more rapid reduction in acute zoster pain.
In the immunocompetent host, a total of 7 days of treatment has been as effective as 21 days. These antivirals must be dose-adjusted in patients with renal impairment. In an elderly patient, if the renal status is unknown, the valacyclovir and famciclovir may be started at twice-daily dosing (which is almost as effective), pending evaluation of renal function, or acyclovir can be used. For patients with renal failure (creatinine clearance <25 mL/min), acyclovir is preferable. In the patient with known or acquired renal failure, acyclovir neurotoxicity can occur from IV acyclovir or oral valacyclovir therapy. This can present in the acute setting as hallucinations or with prolonged elevated blood levels, disorientation, dizziness, loss of decorum, incoherence, photophobia, difficulty speaking, delirium, confusion, agitation, and death delusion. Because acyclovir can reduce renal function, the patient’s baseline renal function may have been normal, but high doses of acyclovir may have reduced renal function, leading to neurotoxic acyclovir levels.
In the immunosuppressed patient, an antiviral agent should always be given because of the increased risk of dissemination and zoster-associated complications. The doses are identical to those used in immunocompetent hosts. Immunosuppressed patients with ophthalmic zoster, disseminated zoster, or Ramsay Hunt syndrome and those failing oral therapy should receive IV acyclovir, 10 mg/kg three times daily, adjusted for renal function.
Because some of the pain during acute zoster (acute zoster neuritis) may have an inflammatory component, corticosteroids have been used during the acute episode. The use of corticosteroids in this setting is controversial. In selected older patients, corticosteroid use is associated with better quality-of-life measures, reduction in time to uninterrupted sleep, quicker return to usual activities, and reduced analgesic use. A tapering dose of systemic corticosteroids, starting at about 1 mg/kg and lasting 10–14 days, is adequate to achieve these benefits. Systemic corticosteroids should not be used in immunosuppressed patients or when there is a contraindication. All factors considered, the benefits of corticosteroid therapy during acute zoster appear to outweigh the risks in treatment-eligible patients. Reduction in postherpetic neuralgia (PHN) by corticosteroids has never been documented despite multiple studies, but this is also true of antiviral therapy, which reduces the severity and duration but not the prevalence of postherpetic neuralgia.
Zoster-Associated Pain (Postherpetic Neuralgia)
Pain is the most troublesome symptom of zoster; 84% of patients over age 50 will have pain preceding the eruption, and 89% will have pain with the eruption. Various terminologies are used to classify the pain. The simplest approach is to refer to all pain occurring immediately before or after zoster as “zoster-associated pain” (ZAP). Another classification system separates acute pain (within first 30 days), subacute pain (30–120 days), and chronic pain (>120 days).
Two different mechanisms are proposed to cause ZAP: sensitization and deafferentation. Nociceptors (sensory nerves mediating pain) become sensitized after injury, resulting in ongoing discharge and hyperexcitability (peripheral sensitization). Prolonged discharge of the nociceptor enhances the dorsal horn neurons to afferent stimuli and expands the dorsal horn neuron’s receptive field (central sensitization), leading to allodynia and hyperalgesia. In addition, neural destruction causes spontaneous activity in deafferented central neurons, generating constant pain. The spinal terminals of mechanoreceptors may contact receptors formerly occupied by C fibers, leading to hyperalgesia and allodynia. The loss of function or death of dorsal horn neurons, which have an inhibitory effect on adjacent neurons, contributes to increased activity transmitted up the spinal cord. The central sensitization is initially temporary (self-limited) but may become permanent.
The quality of the pain associated with herpes zoster varies, but three basic types have been described: the constant, monotonous, usually burning or deep, aching pain; the shooting, lancinating (neuritic) pain; and triggered pain. The last is usually allodynia (pain with normal nonpainful stimuli such as light touch) or hyperalgesia (severe pain produced by a stimulus normally producing mild pain). The character and quality of acute zoster pain are identical to the pain that persists after the skin lesions have healed, although these may be mediated by different mechanisms.
The rate of resolution of pain after herpes zoster is reported over a wide range. The following data are from a prospective study and do not represent selected patients, as are recruited in drug trials for herpes zoster. The tendency to have persistent pain is age dependent, occurring for longer than 1 month in only 2% of persons under age 40. Fifty percent of persons over age 60 and 75% of those over 70 continue to have pain beyond 1 month. Although the natural history is for gradual improvement in persons over age 70, 25% have some pain at 3 months and 10% have pain at 1 year. Severe pain lasting longer than 1 year is uncommon, but 8% of persons over 60 have mild pain and 2% still have moderate pain at 1 year.
The ZAP, especially that of long duration, is very difficult to manage. Adequate medication should be provided to control the pain from the first visit. Once established, neuropathic pain is difficult to control. Every effort should be made to prevent neuronal damage. In addition, chronic pain may lead to depression, complicating pain management. Patients with persistent, moderate to severe pain may benefit from referral to a pain clinic. With this background, the importance of early and adequate antiviral therapy and pain control cannot be overemphasized.
Oral antiviral agents are recommended in all patients over age 50 with pain who still have blisters, even if the drugs are not given within the first 96 hours of the eruption. Oral analgesia should be maximized using acetaminophen, nonsteroidal antiinflammatory drugs (NSAIDs), and opiate analgesia as required. The combination of oral gabapentin and valacyclovir was reported to reduce PHN in one study criticized due to the lack of a control group, but gabapentin alone has not been shown to be highly effective in other zoster trials. Capsaicin applied topically every few hours may reduce pain, but the application itself may cause burning, and the benefits are modest. Local anesthetics, such as 10% lidocaine in gel form, 5% lidocaine-prilocaine, or lidocaine patches (Lidoderm), may acutely reduce pain. Combined topical amitriptyline 4% and ketamine 2% has been reported for PHN and other neuropathic pain conditions. These topical measures may provide some short-term analgesic effect, but do not appear to have any long-term benefit in reducing the severity or prevalence of ZAP. Patients with PHN have lower vitamin C levels than controls, and vitamin C supplementation intravenously (not orally) has been associated with PHN reduction. Sublesional anesthesia, epidural blocks (with or without ketamine), and sympathetic blocks with and without corticosteroids are reported in large series (but rarely studied in controlled trials) to provide acute relief of pain. Although the benefit of nerve blocks in preventing or treating persistent ZAP remains to be proved, these are a reasonable consideration in the acute setting if the patient is having severe pain (unable to eat or sleep) and if oral therapy has yet to be effective. Nerve blocks may also be used in patients who have failed the standard therapies. A transcutaneous electrical nerve stimulation (TENS) unit may be beneficial for persistent neuralgia. Ultrasound-guided spinal nerve radiofrequency treatment has been utilized as well. Botulinum toxin, 100 U, spread out over the affected area in a checkerboard or fanlike pattern with 5 U per route, has dramatically improved PHN in anecdotal reports.
Despite this vast array of medication options, PHN is typically difficult to treat for two reasons. First, the recommended medications are simply often not effective. Second, in elderly patients, who are most severely affected by PHN, these medications have significant and often intolerable side effects, limiting the dose that can be prescribed. If multiple agents are combined to reduce the toxicity of any one agent, their side effects overlap (sedation, depression, constipation) and drug-drug interactions may occur, limiting combination treatment options.
Three classes of medication are used as standard therapies to manage ZAP and PHN: tricyclic antidepressants (TCAs), antiseizure medications, and long-acting opiates. If opiate analgesia is required, it should be provided by a long-acting agent, and the duration of treatment should be limited and the patient transitioned to another class of agent. Constipation is a major side effect in elderly persons. During painful zoster, these patients ingest less fluid and fiber, enhancing the constipating effects of the opiates. Bulk laxatives should be recommended. Tramadol is an option for acute pain control, but drug interactions with the TCAs must be monitored. TCAs such as amitriptyline (or nortriptyline) and desipramine are well tested and documented as effective for the management of PHN and are considered first-line agents. The TCAs are dosed at 25 mg/night (or 10 mg for those over age 65–70). The dose is increased by the same amount nightly until pain control is achieved or the maximum dose is reached. The ultimate dose is between 25 and 100 mg in a single nightly dose. The early use of amitriptyline was able to reduce the pain prevalence at 6 months, suggesting that early intervention is optimal. Venlafaxine (Effexor) may be used in patients who do not tolerate TCAs, at a starting dose of 25 mg/night, gradually titrated upward as required. Gabapentin (Neurontin) and pregabalin (Lyrica) have been documented as helping to reduce ZAP. The starting dose of gabapentin is usually 300 mg three times daily, escalating up to 1800mg/day. A minimum total dose of 600 mg or more is needed to obtain optimal benefit. One meta-analysis suggests that higher-dose gabapentin (1800 mg/day) can reduce PHN pain, though somnolence, dizziness, and other side effects were common. Pregabalin has improved pharmacokinetics and is given at 300 mg or 600 mg daily, depending on renal function, with better absorption and steadier blood levels. The anticonvulsants diphenylhydantoin, carbamazepine, and valproate; neuroleptics such as chlorprothixene and phenothiazines; and H 2 blockers such as cimetidine cannot be recommended because they have been not been studied critically, many are poorly tolerated by elderly patients, and some are associated with significant side effects. If the patient fails to respond to local measures, oral analgesics (including opiates), TCAs, gabapentin, and venlafaxine, referral to a pain center is recommended.
Immunosuppressed Patients
Immunosuppression increases the risk of zoster, though studies vary as to the impact. The use of TNF inhibitors was initially reported to increase the risk of development of zoster by 1.6 times (or 60%), though a large cohort study suggested no increased risk with monotherapy, only an elevated risk with combination TNF-I and methotrexate. The risk is significant enough that prophylactic immunization should be considered, if not contraindicated, before a patient starts a TNF inhibitor. VZV immunization of patients already receiving TNF inhibition who have had prior varicella has not led to increased zoster or adverse events, and it has reduced the rate of zoster with anti-TNF therapy. This strategy should be considered on a case-by-case basis, perhaps in consultation with an infectious disease specialist. This may be more of a risk with newer classes of drugs, particularly the JAK inhibitors, which appear to carry an elevated risk for zoster.
Patients with malignancy, especially Hodgkin disease and leukemia, are five times more likely to develop zoster than their age-matched counterparts. Patients who also have a higher incidence of zoster include those with deficient immune systems, such as individuals who are immunosuppressed for organ transplantation or by connective tissue disease, or by the agents used to treat these conditions, especially corticosteroids, chemotherapeutic agents, cyclosporine, sirolimus, and tacrolimus. After stem cell transplantation for leukemia, up to 68% of patients will develop herpes zoster in the first 12 months (median 5 months). The cumulative incidence of VZV reactivation in this group may exceed 80% in the first 3 years. Antiviral prophylaxis has been shown to reduce zoster in this population. Because zoster is 30 times more common in HIV-infected persons, the zoster patient under age 50 should be questioned about HIV risk factors. In pediatric patients with HIV infection and in other immunosuppressed children, zoster may rapidly follow primary varicella.
The clinical appearance of zoster in the immunosuppressed patient is usually identical to typical zoster, but the lesions may be more ulcerative and necrotic and may scar more severely. Dermatomal zoster may appear, progress to involve the dermatome, and persist without resolution. Multidermatomal zoster is more common in immunosuppressed patients, including the rare variant “herpes zoster duplex bilateralis,” with involvement of two different contralateral dermatomes. Visceral dissemination and fatal outcome are extremely rare in immunosuppressed patients (about 0.3%), but cutaneous dissemination is possible, occurring in 12% of cancer patients, especially those with hematologic malignancies. In bone marrow transplant patients with zoster, 25% develop disseminated zoster and 10%–15%, visceral dissemination. Disseminated zoster may be associated with the syndrome of inappropriate antidiuretic hormone secretion (SIADH) and present with hyponatremia, abdominal pain, and ileus. This later presentation has been reported in stem cell transplant patients. Despite treatment with IV acyclovir, the SIADH can be fatal. In this patient, the number of skin lesions may be small, and the lesions resemble “papules” rather than vesicles. The mortality rate in patients with zoster who have undergone bone marrow transplantation is 5%. VZV IgG serostatus is determined before transplant, and all seropositive patients receive prophylaxis with either acyclovir, 800 mg twice daily, or valacyclovir, 500 mg twice daily, for 1 year or longer if the patient is receiving immunosuppressive therapy. In AIDS patients, ocular and neurologic complications of herpes zoster are increased. Immunosuppressed patients often have recurrences of zoster, up to 25% in patients with AIDS ( Fig. 19.19 ).
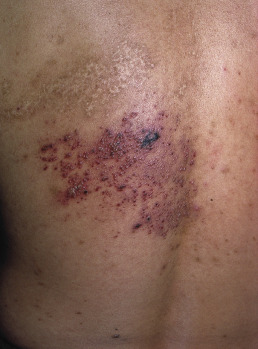
Two atypical patterns of zoster have been described in AIDS patients: ecthymatous lesions, which are punched-out ulcerations with a central crust, and verrucous lesions ( Fig. 19.20 ). These patterns were not reported before the AIDS epidemic. Atypical clinical patterns, especially the verrucous pattern, may correlate with acyclovir resistance.
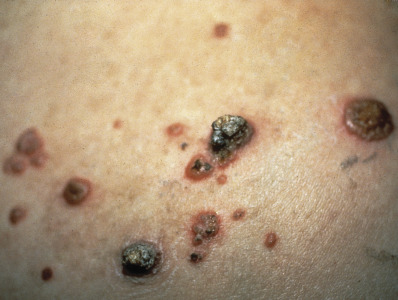
Diagnosis
The same techniques used for the diagnosis of varicella are used to diagnose herpes zoster. The clinical appearance is often adequate to suggest the diagnosis, and an in-office Tzanck smear can rapidly confirm the clinical suspicion. Zosteriform herpes simplex can also have a positive Tzanck smear, but the number of lesions is usually more limited and the degree of pain substantially less than with zoster. Beyond Tzanck preparation, PCR or DFA testing is preferred to a viral culture because it is rapid, types the virus, and has a higher yield than culture. Compared with documented VZV infections, Tzanck smear was 75% positive (with up to 10% false-positives and high variability, depending on skill of examiner), and culture only 44% positive. PCR testing is 97% positive and is generally the test of choice. In atypical lesions, biopsy may be necessary to demonstrate the typical herpes virus cytopathic effects. IP stain tests can then be performed on paraffin-fixed tissue to identify VZV specifically. When acyclovir fails clinically, viral culture may be attempted and acyclovir sensitivity testing performed. It is not as standardized for VZV as it is for HSV, and availability is limited.
Histopathology
As with herpes simplex, the vesicles in zoster are intraepidermal. Within and at the sides of the vesicle are large, swollen cells called balloon cells, which are degenerated cells of the spinous layer. Acidophilic inclusion bodies similar to those seen in HSV are present in the nuclei of the cells of the vesicle epithelium. Multinucleated keratinocytes, nuclear molding, and peripheral condensation of the nucleoplasm are characteristic and confirmatory of an infection with either HSV or VZV. In the vicinity of the vesicle, there is marked intercellular and intracellular edema. In the upper part of the dermis, vascular dilation, edema, and perivascular infiltration of lymphocytes and polymorphonuclear leukocytes (PMNs) are present. Atypical lymphocytes may also be found. An underlying leukocytoclastic vasculitis suggests VZV infection over HSV. Inflammatory and degenerative changes are also noted in the posterior root ganglia and in the dorsal nerve roots of the affected nerve. The lesions correspond to the areas of innervation of the affected nerve ganglion, with necrosis of the nerve cells.
Differential Diagnosis
The distinctive clinical picture of zoster permits a diagnosis with little difficulty. A unilateral, painful eruption of grouped vesicles along a dermatome, with hyperesthesia and on occasion regional lymph node enlargement, is typical. Occasionally, segmental cutaneous paresthesias or pain may precede the eruption by 4 or 5 days. In such patients, prodromal symptoms are easily confused with the pain of angina pectoris, duodenal ulcer, biliary or renal colic, appendicitis, pleurodynia, or early glaucoma. The diagnosis becomes obvious once the cutaneous eruption appears. Herpes simplex and herpes zoster are confused if the lesions of HSV are linear (zosteriform HSV), or if the number of zoster lesions is small and localized to one site (not involving whole dermatome). DFA testing or viral culture will distinguish them; DFA is generally preferred because it is rapid and sensitive.
Prevention of Zoster
A vaccine using the same attenuated virus as in the varicella vaccination, but at much higher titers, was licensed for the prevention of herpes zoster (Zostavax), and initially recommended in all persons 60 years or older. This vaccination reduced the incidence of zoster by 50%. In addition, PHN was 67% lower in the vaccine recipients, and duration of ZAP was shortened. Those vaccinated between ages 60 and 69 had a greater reduction in zoster incidence than those over 70, but in both groups, PHN and burden of illness were reduced similarly. Vaccination has led to a reduced rate of ED visits and costs associated with zoster in populations recommended for vaccination. Because it is a live virus vaccine, persons taking antiviral medications must stop them 24 hours before immunization and not take them for 14 days after immunization. Immunosuppressed patients can be safely immunized following specific guidelines. A second zoster vaccine has been developed (Shingrix), which is now recommended for healthy adults 50 years and older, including adults who previously received the first generation shingles vaccine (Zostavax), and is now the preferred vaccine for shingles. This vaccine demonstrated a moderately high rate of side effects, including 81% of subjects with injection-site reactions (pain, myalgia) and 66% with systemic reaction (myalgias), in clinical trials.
Institutionalized patients who develop herpes zoster are infectious to other patients and the health care team. Prior immunization with varicella vaccine and “adequate serologic titers” may not prevent acquiring infection, especially in immunosuppressed patients. Covering the cutaneous lesions specifically with semipermeable dressings (not just gauze bandages) appears to reduce transmission. Patients with active disease should avoid unvaccinated, nonimmunized, immunosuppressed, or pregnant patients if possible.
Inflammatory Skin Lesions after Zoster Infection (Isotopic Response)
Following zoster, inflammatory skin lesions may rarely occur within the affected dermatome. Lesions usually appear within 1 month, or rarely, longer than 3 months, after the zoster. Clinically, the lesions are usually flat-topped or annular papules in the dermatome. Histologically, such papules most frequently demonstrate various patterns of granulomatous inflammation from typical granuloma annulare to sarcoidal reactions or even granulomatous vasculitis ( Fig. 19.21 ). Persistent viral genome has not been detected in these lesions, suggesting that continued antiviral therapy is not indicated. Persistent VZV glycoproteins may be the triggering antigens. Topical and intralesional therapy with corticosteroids is beneficial, but the natural history of these lesions is generally spontaneous resolution. Less often, other inflammatory skin diseases have been reported in areas of prior zoster, including lichen planus, lichen sclerosus, vitiligo, Kaposi sarcoma (KS), graft-versus-host disease (GVHD), morphea, and benign or even atypical lymphoid infiltrates. Leukemic infiltrates and lymphomas may affect zoster scars, as can metastatic carcinomas (inflammatory oncotaxis) or nonmelanoma skin cancers (NMSCs).
Ansaldi F, et al: Real-world effectiveness and safety of a live-attenuated herpes zoster vaccine. Adv Ther 2016; 33: 1094.
Antoniou T, et al: Statins and the risk of herpes zoster. Clin Infect Dis 2014; 58: 350.
Beal B, et al: Gabapentin for once-daily treatment of post-herpetic neuralgia. Clin Interv Aging 2012; 7: 249.
Cacciapaglia F, et al: Varicella-zoster virus infection in rheumatoid arthritis patients in the anti-tumour necrosis factor era. Clin Exp Rheumatol 2016; 33: 917.
Chakravarty EF: Incidence and prevention of herpes zoster reactivation in patient with autoimmune diseases. Rheum Dis Clin North Am 2017; 43: 111.
Cohen JI: Herpes zoster. N Engl J Med 2013; 369: 255.
Dommasch ED, et al: Trends in nationwide herpes zoster emergency department utilization from 2006-2013. JAMA Dermatol 2017; 153: 874.
Erskine N, et al: A systematic review and meta-analysis on herpes zoster and the risk of cardiac and cerebrovascular events. PLoS One 2017; 12: e0181565.
Fan H, et al: Efficacy and safety of gabapentin 1800 mg treatment for post-herpetic neuralgia. J Clin Pharm Ther 2014; 39: 334.
Fett N: Gabapentin not shown to prevent postherpetic neuralgia. Arch Dermatol 2012; 148: 400.
Friesen KJ, et al: Cost of shingles. BMC Infect Dis 2017; 17: 69.
Gabutti G, et al: Prevention of herpes zoster and its complications. Hum Vaccin Immunother 2017; 13: 391.
Gagliardi AM, et al: Vaccines for preventing herpes zoster in older adults. Cochrane Database Syst Rev 2016; 3: CD008858.
Gilden D, et al: Varicella zoster virus and giant cell arteritis. Curr Opin Infect Dis 2016; 29: 275.
Green CB, Stratman EJ: Prevent rather than treat postherpetic neuralgia by prescribing gabapentin earlier in patients with herpes zoster. Arch Dermatol 2011; 147: 908.
Hadley GR, et al: Post-herpetic neuralgia. Curr Pain Headache Rep 2016; 20: 17.
Hales CM, et al: Update on recommendations for use of herpes zoster vaccine. MMWR Morb Mortal Wkly Rep 2014; 63: 729.
Han Y, et al: Corticosteroids for preventing postherpetic neuralgia. Cochrane Database Syst Rev 2013; 3: CD005582.
Kawai K, et al: Risk factors for herpes zoster. Mayo Clin Proc 2017; 92: 1806.
Keating GM: Shingles (Herpes zoster) vaccine (Zostavax). BioDrugs 2016; 30: 243.
Kim SR, et al: Varicella zoster. Expert Opin Pharmacother 2014; 15: 61.
Lal H, et al: Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N Engl J Med 2015; 372: 2087.
Lapolla W, et al: Incidence of postherpetic neuralgia after combination treatment with gabapentin and valacyclovir in patients with acute herpes zoster. Arch Dermatol 2011; 147: 901.
Lian Y, et al: Herpes zoster and the risk of ischemic and hemorrhagic stroke. PLoS One 2017; 12: e0171182.
Neuzil KM, et al: Preventing shingles and its complications in older persons. N Engl J Med 2016; 375: 1079.
Pi ZB, et al: Randomized and controlled prospective trials of ultrasound-guided spinal nerve posterior ramus pulsed radiofrequency treatment for lower back post-herpetic neuralgia. Clin Ter 2015; 166: e301.
Pickering G: Antiepileptics for post-herpetic neuralgia in the elderly. Drugs Aging 2014; 31: 653.
Sawynok J, et al: Topical amitriptyline and ketamine for post-herpetic neuralgia and other forms of neuropathic pain. Expert Opin Pharmacother 2016; 17: 601.
Schmidt SA, et al: Herpes zoster as a marker of occult cancer. J Infect 2017; 74: 215.
Schuster AK, et al: Valacyclovir versus acyclovir for the treatment of herpes zoster ophthalmicus in immunocompetent patients. Cochrane Database Syst Rev 2016; 11: CD011503.
Seo HM, et al: Antiviral prophylaxis for preventing herpes zoster in hematopoietic stem cell transplant recipients. Antiviral Res 2017; 140: 106.
Shalom G, et al: Systemic therapy for psoriasis and the risk of herpes zoster. JAMA Dermatol 2015; 151: 533.
Shreberk-Hassidim R, et al: Janus kinase inhibitors in dermatology. J Am Acad Dermatol 2017; 76: 745.
Snedecor SJ, et al: Systematic review and meta-analysis of pharmacological therapies for pain associated with postherpetic neuralgia and less common neuropathic conditions. Int J Clin Pract 2014; 68: 900.
Tran CT, et al: Herpes zoster. Joint Bone Spine 2017; 84: 21.
Tyring SK: Management of herpes zoster and postherpetic neuralgia. J Am Acad Dermatol 2007; 57: S136.
Vrcek I, et al: Herpes zoster ophthalmicus. Am J Med 2017; 130: 21.
Wise J: Shingles is linked to increased risk for cardiovascular events. BMJ 2015; 351: h6757.
Yamaoka K: Benefit and risk of tofacitinib in the treatment of rheumatoid arthritis. Drug Saf 2016; 39: 823.
Yang SY, et al: Risk of stroke in patients with herpes zoster. J Stroke Cerebrovasc Dis 2017; 26: 301.
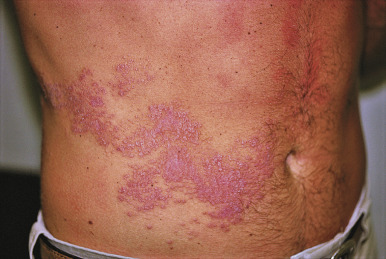
Epstein-Barr Virus (Human Herpesvirus Type 4)
Epstein-Barr virus (EBV) is a γ-herpesvirus. It infects human mucosal epithelial cells and B lymphocytes, and infection persists for the life of the host. EBV infection may be latent—not producing virions, but simply spread from mother cell to both daughter cells by copying the viral DNA with each host cell replication. Intermittently, infection may be productive, resulting in production and release of infectious virions. EBV infection may transit between latent and productive infection many times. The ability of EBV to maintain persistent infection is aided by the expression of the EBV nuclear antigen 1 (EBNA-1) viral gene product, which prevents cytotoxic T-lymphocyte response to the virus.
Initial infection with EBV occurs in childhood or early adulthood, so that by their early twenties, 95% of the population has been infected. The virus is shed into the saliva, so contact with oral secretions is the most common route of transmission. Primary infection may be asymptomatic or may produce only a mild, nonspecific febrile illness, especially in younger children. In young adults, primary infection is more likely to be symptomatic and in 50% of cases produces a syndrome termed infectious mononucleosis (IM). The incubation period is 3–7 weeks. IM is characterized by a constellation of findings: fever (up to 40°C), headache, lymphadenopathy, splenomegaly, and pharyngitis (sore throat).
Cutaneous and mucous membrane lesions are present in about 10% of IM patients. Exanthems occur in less than 5%, more often in children. Edema of the eyelids and a macular or morbilliform eruption are most common. The eruption is usually on the trunk and upper extremities. Other, less common eruptions are urticarial, vesicular, bullous, petechial, erythema multiforme, and purpuric types. Cold urticaria transiently occurs in 5% of patients with IM. Leukocytoclastic vasculitis and large-vessel arteritis have been seen in chronic EBV infection, often in the setting of immunodeficiency. The mucous membrane lesions consist of distinctive pinhead-sized petechiae, 5–20 in number, at the junction of the soft and hard palate (Forchheimer spots). Gianotti-Crosti syndrome (GCS) and the papular-purpuric glove-and-stocking (or gloves-and-socks) syndrome are two specific viral exanthem patterns that may occur in the patient with asymptomatic primary EBV infection. EBV is now the leading cause of GCS worldwide. EBV reactivation has been infrequently associated with drug-induced hypersensitivity syndrome. EBV is also associated with enhanced insect bite reactions.
Painful genital ulcerations may precede the symptomatic phase of IM, especially in premenarcheal girls. The ulcerations are up to 2 cm in diameter, single or multiple, and may be accompanied by marked swelling of the labia. Lesions last several weeks and heal spontaneously, often as the patient is developing symptoms of IM. Transmission to patients through orogenital sex has been proposed, but the virus may also reach the vulvar mucosa hematogenously. EBV may be the cause of some nonsexually related genital ulcers (Lipschutz ulcers). The lesions closely resemble herpetic ulcerations and fixed drug eruption, which must be considered in the differential diagnosis.
Laboratory evaluation in patients with IM frequently shows an absolute lymphocytosis of greater than 50% and monocytosis with abnormally large, “atypical” lymphocytes. The white blood cell (WBC) count ranges from 10,000–40,000 cells/mm 3 . Liver function tests (LFTs) may be elevated. Heterophile antibodies will be present in 95% or more of cases. In acute primary EBV infection, immunoglobulin M (IgM) antibodies to early antigen (EA) and viral capsid antigen (VCA) are found in high titer and decrease during recovery. Antibodies to VCA and EBNA appear in the recovering phase and persist for years after primary infection. There is no specific therapy, and in most IM patients, no treatment is required. Acyclovir is not effective in altering the length or severity of IM, although it is active against EBV in doses used for VZV. A recent Cochrane review concluded there is a lack of quality evidence to support antiviral therapy, and treatment decisions depend on the clinical scenario. If patients have severe pharyngeal involvement with encroachment on the airway, or cytopenias or liver failure, oral corticosteroid therapy (40–60 mg/day of prednisone) is useful to induce a prompt response, but are not generally indicated for symptom control in more mild cases. Most patients recover completely. Patients who participate in contact sports should have abdominal ultrasonography to rule out splenomegaly if planning to participate within 1 month of IM diagnosis.
Almost all patients with IM treated with ampicillin, amoxicillin, or other semisynthetic penicillins typically develop a generalized, pruritic, erythematous to copper-colored macular exanthem on the 7th–10th day of therapy. The eruption starts on the pressure points and extensor surfaces, generalizes, and becomes confluent. The eruption lasts about 1 week and resolves with desquamation. The eruption often does not recur when these medications are given after the acute mononucleosis has resolved, and this is not a true drug allergy.
Oral hairy leukoplakia (OHL) is a distinctive condition initially reported in strong association with HIV, though now has been recognized in patients with other forms of immunocompromise. It appears as poorly demarcated, corrugated white plaques seen on the lateral aspects of the tongue ( Fig. 19.22 ). Lesions on the other areas of the oral mucosa are simply white plaques without the typical corrugations. OHL can be distinguished from thrush by the fact that OHL cannot be removed by firm scraping with a tongue blade. More than one third of patients with AIDS have OHL, but is not restricted to patients with HIV infection; it also occurs in other immunosuppressed hosts, especially renal and bone marrow transplant recipients, and those using inhaled steroids for chronic obstructive pulmonary disease. OHL can be a part of the IRIS. EBV does not establish infection in the basal cell layer of the oral epithelium but is maintained by repeated direct infection of the epithelium by EBV in the oral cavity. Only chronically immunosuppressed patients continuously shed EBV in their oral secretions, thus explaining the restriction of OHL to immunosuppressed hosts. In normal persons, a similar morphologic and histologic picture can be seen (pseudo-OHL), but EBV is not found in these patients’ lesions. Thus the finding of OHL warrants immunologic evaluation. A biopsy of the OHL lesions searching for EBV in the epithelium can be useful in this setting. OHL is usually asymptomatic and requires no treatment. If treatment is requested in immunosuppressed patients, podophyllin, applied for 30 seconds to 1 minute to the lesions once each month, is the simplest approach. Tretinoin gel, applied topically twice daily, or oral acyclovir, 400 mg five times daily, is also effective. Lesions recur when treatment is discontinued.
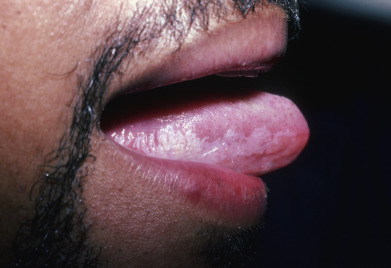
In immunosuppressed and immunocompetent hosts, EBV may be responsible for benign and malignant disorders, some of which can be fatal. These include Kikuchi disease (histiocytic necrotizing lymphadenitis), hydroa vacciniforme (HV) and HV-like lymphoma, hypereosinophilic syndrome, leiomyomas and leiomyosarcomas, lymphomatoid granulomatosis, erythema multiforme, and multiple types of lymphoma and lymphoproliferative disorders, especially in organ transplant recipients (posttransplant lymphoproliferative disorders, immunosuppression-associated lymphoproliferative disorders). Richter syndrome (development of lymphoma in the patient with chronic lymphocytic leukemia) can present in the skin and may be associated with fludarabine treatment of EBV infection. EBV and other HHV reactivation has been demonstrated in drug reaction with eosinophilia and systemic symptoms (DRESS), and viral replication and host-antiviral response is felt to play a role in the pathophysiology. Ongoing studies are exploring whether there is a connection between EBV and autoimmune disorders, including multiple sclerosis and systemic lupus erythematosus.
Cytomegalovirus (Cytomegalic Inclusion Disease); Human Herpesvirus Type 5
Congenital cytomegalovirus (CMV) infection, as documented by CMV excretion, is found in 1% of newborns, 90% of whom are asymptomatic. Clinical manifestations in infants may include jaundice, hepatosplenomegaly, cerebral calcifications, chorioretinitis, microcephaly, mental retardation, and deafness. Cutaneous manifestations may result from thrombocytopenia, with resultant petechiae, purpura, and ecchymoses. Purpuric lesions, which may be macular, papular, or nodular, may show extramedullary hematopoiesis (dermal erythropoiesis), producing the “blueberry muffin baby.” A generalized vesicular eruption may rarely occur. Most symptomatic cases occur within the first 2 months of life. Neonatal disease is more severe and sequelae are more frequent in neonates of mothers with primary rather than recurrent CMV disease in pregnancy.
Between 50% and 80% of immunocompetent adults and up to 100% of HIV-infected men who have sex with men (MSM) are infected with CMV. Infection in adults may be acquired by exposure to infected children, sexual transmission, and transfusion of CMV-infected blood. Symptomatic primary infection in adults is unusual and is identical to IM caused by EBV. An urticarial or morbilliform eruption or erythema nodosum may occur in primary CMV infection in immunocompetent adults. Ampicillin and amoxicillin administration will often result in a morbilliform eruption in acute CMV infection, similar to that seen in acute EBV infection.
Infection with CMV is common in AIDS patients, most frequently causing retinitis (20% of patients), colitis (15%), cholangitis, encephalitis, polyradiculomyopathy, and adrenalitis. It occurs in the setting of very advanced HIV infection, usually with CD4 counts below 50, and has become much less common in the era of highly active antiretroviral therapy (HAART).
CMV infection can manifest in other immunosuppressed hosts. CMV viremia is routinely monitored after stem cell transplant depending on host/donor CMV serostatus, and some cases are treated with prophylactic antivirals or preemptive therapy based on serial CMV PCR testing. Infection will usually affect the GI tract or lungs. Skin disease is rare but may have a wide range of manifestations, from nonspecific morbilliform exanthems that can easily be mistaken for drug reactions, to ulcerated plaques.
Cytomegalovirus infection in tissues is usually identified by the histologic finding of a typical CMV cytopathic effect. In a very small percentage of AIDS patients with CMV infection, skin lesions may occur that contain such cytopathic changes. In most cases, CMV is found in association with another infectious process, and the treatment of that other infection will lead to resolution of the CMV in the skin without its treatment. This is especially true of perianal HSV ulcerations. CMV may even be found in totally normal skin in CMV-viremic AIDS patients, suggesting that finding the CMV cytopathic effect is insufficient alone to imply a causal relationship of the CMV to any cutaneous lesion. Only in the case of perianal and oral ulcerations has the pathogenic role of CMV been documented. In unusual cases of extremely painful genital ulcerations, only CMV infection is found histologically, or the ulceration persists after effectively treating HSV, with CMV identified histologically. The CMV cytopathic changes may be noted in the nerves at the base of these ulcerations, suggesting that CMV neuritis may be producing the severe pain that characterizes these cases. The diagnosis of CMV ulceration is one of exclusion. CMV cytopathic changes must be seen in the lesion and cultures, and histologic evidence of any other infectious agent must be negative. In these ulcerations, clinically suggested by their location (genital or oral) and painful nature, specific treatment with valganciclovir, foscarnet, or cidofovir will lead to healing of the ulceration and dramatic resolution of the pain.
The CMV infecting endothelial cells may produce a vasculopathy in CMV-viremic or partially reactivating patients. This can lead to prominent vasculopathic changes, including Raynaud phenomenon, deep venous thrombosis, digital gangrene, and reticulated purpura. Anticardiolipin antibodies may be positive. Anti-CMV therapy can reverse the syndrome and clear the anticardiolipin antibodies.
Ascherio A, Munger KL: EBV and autoimmunity. Curr Top Microbiol Immunol 2015; 390: 365.
Chambers AE, et al: Twenty-first-century oral hairy leukoplakia–a non-HIV-associated entity. Oral Surg Oral Med Oral Pathol Oral Radiol 2015; 119: 326.
Chawla JS, et al: Oral valganciclovir versus ganciclovir as delayed pre-emptive therapy for patients after allogeneic hematopoietic stem cell transplant. Transpl Infect Dis 2012; 14: 259.
De Paor M, et al: Antiviral agents for infectious mononucleosis. Cochrane Database Syst Rev 2016; 12: CD011487.
Elqui de Oliveira D, et al: Viral carcinogenesis beyond malignant transformation. Trends Microbiol 2016; 24: 649.
Ferguson NN, et al: Primary cutaneous polymorphic EBV-associated posttransplant lymphoproliferative disorder after a renal transplant and review of the literature. Am J Dermatopathol 2015; 37: 790.
Fernandez-Flores A: Epstein-Barr virus in cutaneous pathology. Am J Dermatopathol 2013; 35: 763.
Gabrib G, et al: Atypical presentation of exophytic herpes simplex virus type 2 with concurrent cytomegalovirus infection: a significant pitfall in diagnosis. Am J Dermatopathol 2013; 35: 371.
Garcia JG, et al: Lipschutz ulcer. Am J Emerg Med 2016; 34: 1326e1.
Klion AD, et al: Chronic active Epstein-Barr virus infection. Blood 2013; 121: 2364.
Lee HY, et al: Primary Epstein-Barr virus infection associated with Kikuchi’s disease and hemophagocytic lymphohistiocytosis. J Microbiol Immunol Infect 2010; 43: 253.
Lehloenya R, Meintjes G: Dermatologic manifestations of the immune reconstitution inflammatory syndrome. Dermatol Clin 2006; 24: 549.
Lennon P, et al: Infectious mononucleosis. BMJ 2015; 350: h1825.
Maffini E, et al: Treatment of CMV infection after allogeneic hematopoietic stem cell transplantation. Expert Rev Hematol 2016; 9: 585.
Marty FM, et al: Letermovir prophylaxis for CMV in hematopoietic-cell transplantation. N Engl J Med 2017; 377: 2433.
Milano F, et al: Intensive strategy to prevent CMV disease in seropositive umbilical cord blood transplant recipients. Blood 2011; 118: 5689.
Ozoya OO, et al: EBV-related malignancies, outcomes, and novel prevention strategies. Infect Disord Drug Targets 2016; 16: 4.
Shabani M, et al: Primary immunodeficiencies associated with EBV-induced lymphoproliferative disorders. Crt Rev Oncol Hematol 2016; 108: 109.
Rezk E, et al: Steroids for symptom control in infectious mononucleosis. Cochrane Database Syst Rev 2015; 11: CD004402.
Ryan, C et al: Cytomegalovirus-induced cutaneous vasculopathy and perianal ulceration. J Am Acad Dermatol 2011; 64: 1216.
Tetzlaff MT, et al: Epstein-Barr virus–associated leiomyosarcoma with cutaneous involvement in an African children with human immunodeficiency virus. J Cutan Pathol 2011; 38: 731.
Thompson DF, et al: Antibiotic-induced rash in patients with infectious mononucleosis. Ann Pharmacother 2017; 51: 154.
Human Herpesviruses 6 and 7
Infection with HHV-6 is almost universal in adults, with seropositivity in the 80%–85% range in the United States, and seroprevalence almost 100% in children. There are intermittent periods of viral reactivation throughout life; persistent infection occurs in several organs, particularly in the CNS. Acute seroconversion to HHV-6 and to HHV-7 each appears to be responsible for about one third of roseola cases, and in the remaining third, neither is found. HHV-6 infection occurs earlier than HHV-7, and second episodes of roseola in HHV-6–seropositive children may be caused by HHV-7. Primary infection with HHV-6 is associated with roseola in only 9% of cases, and 18% of children with seroconversion have a rash. Primary infection may occur with only fever and no rash, or rash without fever. Other common findings include otitis media, diarrhea, and bulging fontanelles, sometimes with findings of meningoencephalitis and seizures. Infrequently, hepatitis, intussusception, and even fatal multisystem disease may occur. In adults, acute HHV-6 infection resembles acute mononucleosis. Some have suggested that HHV-6 and HHV-7 may be involved in the development of pityriasis rosea. HHV-6 and HHV-7 reactivation has been demonstrated during DRESS syndrome (along with less commonly EBV and CMV), with many feeling that the drug trigger then allows latent virus replication followed by a host-antiviral response leading to the clinical phenotype observed, which explains the long time course and prolonged treatment required, and the potential for delayed autoimmune sequelae.
As with other herpesviruses, the pattern of disease in HHV-6 may be different in immunosuppressed hosts. HHV-6 reactivation is common after transplantation: 32% after solid-organ and 48% after bone marrow transplantation, peaking on day 21. These patients can be quite ill, with fever, diarrhea, and elevated LFTs, simulating GVHD, with some developing pneumonitis and CNS manifestations as well. Engraftment can be delayed. HHV-6 viremia, detected by PCR, can confirm the diagnosis. Chronic macular erythema has been reported in several cases. Careful histologic evaluation has identified HHV-6–infected lymphocytes and histiocytes in the macular erythema, confirming the diagnosis. Antiviral therapy with ganciclovir, foscarnet, or valganciclovir can lead to improvement, with broad-acting antiviral T cells under investigation.
Roseola Infantum (Exanthem Subitum, Sixth Disease)
Roseola infantum is a common cause of sudden, unexplained high fever in young children between 6 and 36 months of age. Prodromal fever is usually high and may be accompanied by convulsions and lymphadenopathy. Suddenly, on about the fourth day, the fever drops. Coincident with the decrease in temperature, a morbilliform erythema of discrete, rose-colored macules appears on the neck, trunk, and buttocks and sometimes on the face and extremities. Often, there is a blanched halo around the lesions. The eruption may also be papular or rarely even vesicular. The mucous membranes are spared. Complete resolution of the eruption occurs in 1–2 days. A case of spontaneously healing, generalized eruptive histiocytosis has been reported after exanthem subitum.
Human Herpesvirus 8
A γ-herpesvirus, HHV-8 is most closely related to EBV. HHV-8 has been found in all patients with KS, including those who have AIDS ( Fig. 19.23 ), in African cases; in elderly men from the Mediterranean basin; and in transplant patients. In addition, the seropositivity rate (infection rate) for this virus correlates with the prevalence of KS in a given population.