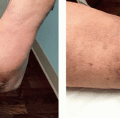
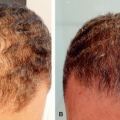
TUMESCENT TECHNIQUE FOR LOCAL ANESTHESIA
The term tumescent describes the firm and swollen quality of tissue when infiltrated with large volumes of fluid. The technique allows for significant amounts of dilute lidocaine, epinephrine, and sodium bicarbonate solution to be delivered to the skin and subcutaneous tissues. By diluting the concentration of anesthetic, systemic absorption is slowed.
4,5 The large volume of interstitial fluid creates a local tissue reservoir, which prolongs the
duration of action and reduces the need for postoperative narcotics. Another benefit of tumescence is that it physically lifts the targeted fat, creating a hydrodissection effect. This enables a uniform, precise removal during aspiration.
Prior to the use of epinephrine in the anesthetic solution, the amount of whole blood in the liposuction aspirate approached half of the volume. This is reduced to 1% to 3% when epinephrine is used. As a result, there is decreased bruising, decreased postoperative pain, and a vastly reduced need for intraoperative fluid replacement.
As with other locally injected anesthetics, sodium bicarbonate is added to reduce discomfort on infiltration. Unbuffered lidocaine has a pH of 3.5 to 5.5, which can result in significant burning and stinging when infused into the skin. Adding bicarbonate raises the pH to a physiologic range, minimizing these side effects.
While the principles of tumescence can apply to many anesthetics, lidocaine is the most commonly used and has the greatest safety record.
4,6,7,8,9,10 According to the full prescribing information, the maximum recommended dose of lidocaine is 7 mg/kg in adults. In general, a total dose of 500 mg should not be exceeded.
11 However, up to 35 to 55 mg/kg can be safely used in tumescent anesthesia.
4,5 The pharmacokinetics can be explained by the effect of concentration gradient on the rate of diffusion.
Lidocaine is a hydrophobic molecule that diffuses quickly across cell membranes. Blood levels correlate with signs of systemic toxicity. The amount of lidocaine that moves from the infiltrated tissue into the vascular space is proportional to the concentration gradient. In other words, higher concentrations of lidocaine lead to higher blood levels and increased risk of toxicity. In a study of almost 10,000 nerve blocks, all eight toxicity events were attributed to inadvertent intravascular injection.
12 In mice studies, the higher the concentration injected subcutaneously, the lower the lethal dose (
Table 7.1).
13 Thus, dilute concentrations allow for the use of a higher total dose of lidocaine.
Lidocaine is metabolized by the cytochrome P450 system in the liver. Those patients with a history of liver disease or abnormal liver function may be at increased risk for lidocaine toxicity. Medications which are known to inhibit the cytochrome P450 system should be reviewed. If these medications cannot be safely discontinued, the maximum dose of lidocaine should be adjusted. Initial manifestations of lidocaine toxicity include circumoral paresthesias, lightheadedness, and euphoria. As lidocaine levels increase, symptoms progress to include nausea, vomiting, blurred vision, seizures, and cardiac and respiratory depression (
Table 7.2).
In tumescent anesthesia, the concentration of lidocaine ranges from 0.05% to 0.15%. The lowest, 0.05%, results in the greatest tumescence and is the dose recommended in Klein’s seminal study.
4 However, some authors note that higher concentrations of 0.1% or 0.15% provide better anesthesia for sensitive sites such as the inner thighs, stomach, flanks and breasts. One strategy to maximize the efficiency of each treatment is to infiltrate with 0.05% lidocaine solution and keep 0.1% solution on hand during the session for any areas that require additional anesthetic for patient comfort. The recipe for tumescent lidocaine solution, sample dosing volumes, and a maximum dose calculation is shown in
Tables 7.3,
7.4, and
7.5, respectively.
Maximum anesthetic dosing limits the amount of liposuction that can be done in a given tumescent treatment session. When more than one treatment session is needed to maintain safe dosing levels, we recommended 1 month interval between treatments. However, some surgeons treat additional sites within several days. In order to maximize the efficiency of each treatment, it is important to aspirate within 10 to 30 minutes after infiltration. While the lidocaine’s onset is almost instantaneous, epinephrine takes longer to reach the therapeutic threshold. Blanching of the overlying skin is a visible clue that vasoconstriction has occurred. Waiting too long after infiltration diminishes the efficacy of the lidocaine and increases the chance that more anesthetic will be needed.
Prilocaine does not have an indication for tumescent anesthesia in the United States, but it is used in Germany and other European countries. Limited clinical data show it to be effective and well tolerated.
14,15,16 In comparison to lidocaine, prilocaine has less cardiotoxicity and more rapid excretion. Some surgeons advocate for use of a combination of lidocaine and prilocaine when large volumes of tumescent anesthesia are needed in order to reduce the risk of side effects.
14 One disadvantage of prilocaine is that it results in the dose-dependent formation of methemoglobin. However, this has not proven to be clinically significant in causing any adverse events while used during tumescent liposuction.
16 There are no data for other anesthetics, including bupivacaine, for use in tumescent anesthesia during liposuction.
Patient Selection and Preoperative Counseling
Appropriate surgical candidates for liposuction are generally in good health and at or near ideal body weight. Localized areas of excessive fat deposits can be targeted. Patients should understand that liposuction is not an appropriate treatment for generalized obesity.
A thorough medical history is obtained with particular attention given to history of clotting disorders, liver disease, and allergy to lidocaine. Medications are reviewed and adjustments made to those that inhibit cytochrome P450.
A physical exam is performed. During this time, the provider will formulate a plan with the patient based on priority areas to treat and anticipated volume of tumescent anesthesia. Preoperative photographs are obtained. If indicated by history, review of systems, or extent of planned treatment, preoperative laboratory studies can be performed including complete blood count with differential, basic metabolic panel, liver function tests, prothrombin time, and partial thromboplastin time. Hepatitis and HIV screening may also be done.
Once the treatment plan has been agreed upon with the patient, expectations for the treatment and postoperative period are reviewed. Patients are instructed to wear dark, loose fitting clothing. They can eat breakfast and medications normally on the day of surgery. Supplements that cause increased risk for bleeding or those that inhibit lidocaine metabolism are stopped. While there is no data to support this recommendation, most surgeons prescribe antibiotics beginning the evening before the procedure and extending for 5 to 7 days after. We prefer a first-generation cephalosporin. Trimethoprim/sulfamethoxazole or doxycycline are appropriate substitutes if there is a contraindication or a patient history of methicillin-resistant Staphylococcus aureus. To help with pain control and patient comfort, lorazepam can be given before the procedure.
Procedural Approach
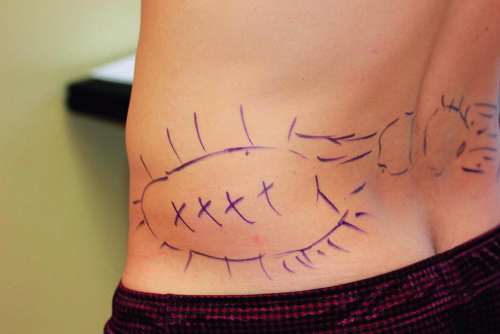
Careful marking with the patient in standing position using a permanent ink is helpful to delineate treatment areas and sections of peripheral feathering (
Figure 7.5). This is important to do in order to set clear patient expectations of treatment areas and to mark any asymmetries and the exact desired areas of adipose tissue prior to swelling from the tumescent anesthesia occurs.
In order to access the subcutaneous adipose tissue, incisions through the skin are made with a 15 blade, 11 blade, or punch biopsy tool. They should be sized to the cannula. For example, if using a 2 mm cannula, a 2 mm punch tool can be used. Entry points can be anesthetized with the same tumescent solution used for the fat compartment, but some use injection of 0.5 to 1 mL of 1% lidocaine with 1:100,000 epinephrine. Some undermining can be helpful to facilitate cannula entry. The number of incisions depends on the area treated and the reach of the cannula. They should be oriented to allow the cannula to travel along the long axis of the site and to enable overlapping treatment in a triangular criss-cross pattern. After surgery, some surgeons suture the entry points, but often these wounds are left open to heal by second intention and facilitate drainage.
Once the entry sites have been made, anesthetic solution is infused into the fat compartment. One hand is placed on the target area to feel tissue tumescence. The end point is achieved when the maximum volume of anesthesia has been infused and/or the tissue is noted to be appropriately tumesced with blanching and a peau d’orange pebbled appearance of the skin is visible. To maximize patient comfort, the infusion is started slowly (i.e., 1 mL/min) and is increased as tolerated. The entire area to be treated is anesthetized in a pattern that minimizes needle repositioning. Anesthesia should be infused a few centimeters beyond the edge of the treated area.
After infiltration is complete, wait at least 10 minutes before aspiration, or start with the first area anesthetized. This allows for full onset of the vasoconstrictive effect of epinephrine. Aspiration of fat is patterned similar to the anesthesia, often in an overlapping fanned pattern. To ensure even results, make crossing tunnels at different depths in the fat. This often requires multiple passes over the treatment area from each access point with progressively larger cannulas.
When handling the cannula, the near dominant hand is used to move it back and forth and the far “safe” hand is positioned overlying the treatment area to gauge position and depth. Grasping the fat with the far hand can help move the cannula into different areas. The cannula is continuously repositioned at slightly different angles in both radial and axial planes such that small tunnels are made throughout the treatment area. The cannula is feathered into adjacent tissues to ensure sufficient blending. The end point is reached once the goal volume of aspirate is obtained. Gently pinching the skin between the thumb and the second finger can be used to measure adequacy of fat removal. These authors recommend carefully standing the patient once to twice during the treatment session to assess symmetry with the aid of the original placed markings.
Postprocedural Care and Recovery
After the procedure is complete, the wounds are dressed with a sterile compression dressing. This is composed of an absorbent pad, netting, and a compression wrap or garment. The patient can expect active drainage during the first day, decreasing over the next several days. They will need to clean the entry points with antimicrobial soap and replace the dressings as they become saturated, likely several times per day the first 48 hours after treatment. Compression can be used for several days and is beneficial for up to 2 weeks.
The most commonly reported side effects of liposuction include swelling, bruising, itching, and numbness in the treated area. These usually subside over 1 to 2 weeks. Some patients will have persistent focal areas of numbness as well as palpable bumpiness or nodularity in the fat that can be massaged and treated with warm compresses. There are no reports of death among patients receiving liposuction from dermatologists using tumescent local anesthesia. The overall serious adverse event rate (per case) ranges from 0% to 0.16%.
2,3,7,17,18,19,20 Reported events include infection, venous thromboembolism, hematoma/seroma formation, and allergic reactions to antibiotics or garments. A meta-analysis of 24 liposuction studies showed that tumescent anesthesia alone had the lowest serious adverse event rate when compared to other methods that incorporated systemic anesthesia.
21
Laser-Assisted Liposuction
Laser-assisted liposuction (LAL) augments manual aspiration by using selective photothermolysis to target fat and promote collagen contraction.
22 This can be performed during liposuction or before it as a separate procedure. Lasers used include the 980 nm diode, 1064 nm neodymium-doped yttrium aluminum garnet (Nd:YAG), 1064/1320 nm Nd:YAG, and 1440 nm. There are a variety of devices available commercially. Many use a combination of wavelengths to both target vasculature within the adipose tissue and stimulate collagen formation and skin tightening. Laser cannulas are small and minimally invasive. These “microcannulas” are ˜1 mm in diameter and contain a smaller laser fiber within the cannula.
A recent meta-analysis comparing LAL to traditional liposuction found LAL to have better fat reduction, skin tightening, and patient satisfaction.
23 However, many of the included studies had a high risk of bias. Complication rate and severity from LAL do not differ significantly from traditional liposuction.
19