Vascular Treatments of the Lower Extremity
Dillon Clarey, MD
Ashley Wysong, MD, MS
Chapter Highlights
Chronic venous disease secondary to valve reflux is a common and increasing problem.
Numerous risk factors are present, including increasing age, family history, multiparity, and body mass.
The disease has a significant impact on the quality of life for those affected in addition to increased economic burden to society.
A thorough history (aching/tiredness, pain, tenderness, edema) and physical exam (varicose veins, reticular veins, telangiectasias, edema) are critical prior to choosing management modalities.
Sclerotherapy is the mainstay of treatment for asymptomatic telangiectasias.
Endovenous ablation has predominantly replaced high ligation and vein stripping in the management of saphenous vein varices.
Ambulatory phlebectomy (mini-vein stripping) has been broadly replaced by minimally invasive techniques but is still used in select patients and on areas overlying bony prominences.
Broadly speaking, chronic venous disease (CVD) refers to the inability of blood to be returned from the lower extremities to the heart.1,2 In a properly functioning vein, blood is propelled from the legs to the heart by calf contractions (muscular pump) and prevented from refluxing by bicuspid venous valves.3 This combination enables a unidirectional flow.4 Venous flow is influenced by both intrinsic (venous contractions, arterial inflow, thoracic/abdominal pressure, valve integrity, vein wall recoil) and extrinsic (gravity, atmospheric pressure, centrifugal force, compression) factors.4
The inability of a vein to return blood most commonly arises due to malfunctioning venous valves.5 This is most often secondary to primary valve incompetence.5 Incompetence can also result secondarily from trauma, deep venous thrombosis (DVT), lack of a muscular pump, or a congenital anomaly (May-Thurner, Ehlers-Danlos, Von Hippel-Lindau).5 Risk factors for CVD include increasing age (decreased calf muscle mass leads to decreased muscle contraction, weakened vein walls, progressive inflammatory degradation of valves),1
family history (risk as high as 90% if both parents are affected),6 and multiparity (due to effects of hormones causing smooth muscle relaxation and increased abdominal pressure during pregnancy).6,7,8,9,10 Refluxed blood travels from the deep venous system through the perforating venous system and ultimately into the superficial venous system.5
family history (risk as high as 90% if both parents are affected),6 and multiparity (due to effects of hormones causing smooth muscle relaxation and increased abdominal pressure during pregnancy).6,7,8,9,10 Refluxed blood travels from the deep venous system through the perforating venous system and ultimately into the superficial venous system.5
Nearly 60% to 70% (females > males) of the population has been estimated to have some degree of CVD, with the incidence of varicose veins at roughly 2% per year.11 The incidence is typically higher in areas that are more industrialized, such as the Western countries.2 Worldwide, CVD of the lower extremities costs billions of dollars a year.12 In addition to the monetary impact, the psychological impact of CVD on the quality of life is significant, particularly in the case of venous ulceration.12
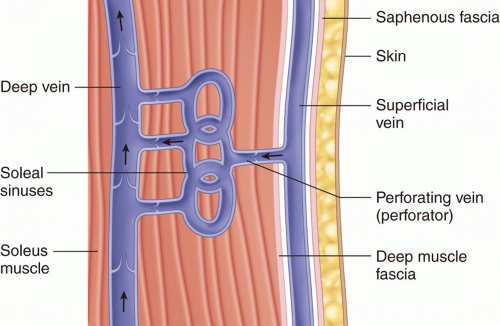
The vein walls of the lower extremity are much thinner than arteries. They contain intimal, medial, and adventitial layers.13 Each of these layers plays important roles in vein function. The primary role of the intimal layer is to serve an antithrombogenic function; it does this through a number of mechanisms (prostaglandin I2 production, tissue-type plasminogen activator [t-PA] production, others).3 This antithrombogenic activity can be disturbed with damage to the intimal layer, leading to increased thrombogenicity.3 Smooth muscle, collagen, and elastin compose the medial layer.3,14 The adventitial layer, primarily composed of collagen, forms the outer layer and functions to provide veins with a degree of stiffness that enables the calf muscle pump to propel blood.3 The lower extremity venous system is divided into three different compartments: deep, superficial (saphenous), and perforating (Figure 8.1).15,16
Deep Venous Compartment
The veins of the deep venous system lie beneath a muscular fascia and run with their associated arteries (Figure 8.1).3 Due to this location, they function to drain the muscles of the lower extremity.3 The deep compartment relies on calf muscle contraction and relaxation for propulsion of blood back toward the heart.3 Functioning bicuspid valves of the deep and perforating system prevent reflux.3
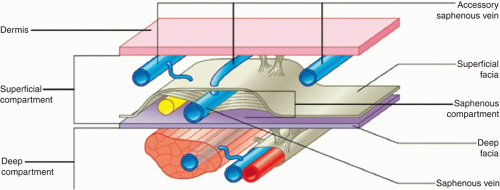 FIGURE 8.1 Diagrammatic representation of the three different compartments comprising the lower extremity. (Reprinted from Bergan J, Pascarella L. Chapter 4: Venous anatomy, physiology, and pathophysiology. In: The Vein Book. Elsevier; 2007:39-45. Copyright © 2007 Elsevier. With permission.) |
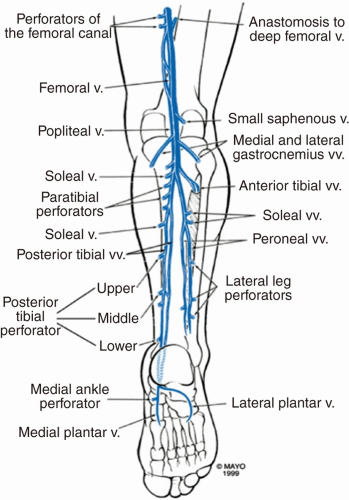 FIGURE 8.2 Lower extremity deep venous system. (Reprinted from Mozes G, Gloviczhi P. Chapter 2: Venous embryology and anatomy. In: The Vein Book. Elsevier; 2007:15-25. Copyright © 2007 Elsevier. With permission.) |
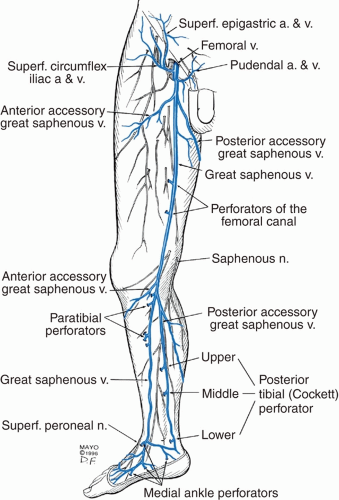
Starting in the foot, the digital and metatarsal veins drain to form the deep plantar venous arch.17 The deep plantar venous arch runs proximally over the foot to form the medial and lateral plantar veins (Figure 8.2).17 At the ankle, the medial and lateral plantar veins drain into the posterior tibial vein. The anterior tibial vein is formed from the dorsal pedal vein.3 The posterior tibial vein is responsible for draining blood from the posterior leg and medial and plantar foot, while the anterior tibial vein is responsible for the anterior lower leg and dorsal foot.3 The peroneal vein drains blood from the lateral foot.18 The posterior tibial vein receives drainage from the peroneal vein near the posteromedial fibula.18 The anterior and posterior tibial vein join at the lower aspect of the posterior knee to form the tibioperoneal trunk and popliteal vein.3 The popliteal vein ascends through the popliteal fossa and becomes the femoral vein upon entrance into the adductor hiatus.3 The femoral vein joins the deep femoral vein to form the common femoral vein.3 The saphenofemoral junction (SFJ), located 4 cm inferolateral to the pubic tubercle, is formed from the drainage of the great saphenous vein (GSV) into the common femoral vein.18 The common femoral vein becomes the external iliac vein at the inguinal ligament.18
Superficial Venous Compartment
The location of the superficial venous system above the deep fascia allows these veins to drain the cutaneous circulation.19 CVD is most prevalent in the superficial veins of the lower extremities secondary to their decreased muscular support compared to deeper veins.3 The main vessels of this compartment include the truncal veins (e.g., GSV and small saphenous vein) as well as the named tributaries, or branches, off the saphenous system (e.g., anterior and posterior accessory of the GSV).3,20 Reticular veins are a group
of veins that are located between the dermis and saphenous fascia and function to drain the skin and soft tissue (Figure 8.3).3,20 The perforating venous system allows for communication of reticular veins to both the deep venous system and saphenous tributaries.3,20
of veins that are located between the dermis and saphenous fascia and function to drain the skin and soft tissue (Figure 8.3).3,20 The perforating venous system allows for communication of reticular veins to both the deep venous system and saphenous tributaries.3,20
The GSV lies in the saphenous compartment, an area in the superficial compartment bordered superficially and deep by saphenous fascia and muscular fascia, respectively (Figure 8.1).3,20 The saphenous compartment also contains nerves and arteries associated with the saphenous vein but does not contain the reticular veins (more superficial).3,21 The GSV originates on the dorsomedial foot from the dorsal pedal venous arch.3,20,21,22 From there, the vein traverses superiorly anterior to the medial malleolus.3 At the intersection of the middle and distal calf, the vein crosses and courses over to the posteromedial knee.3 It then continues to ascend medially up the thigh to a point 3 to 4 cm inferolateral to the pubic tubercle. Here, it penetrates the deep fascia to join the common femoral vein at the SFJ.3 At the SFJ, the superficial circumflex iliac vein (drains groin), superficial epigastric vein (drains abdominal wall) (Figure 8.4), and external pudendal vein (drains pelvis) are present.
As the saphenous vein courses up the mid-distal thigh, it may pierce the saphenous fascia to become more superficial.22 A lack of fascial support has been proposed as a possible explanation for the more frequent presentation of varicose veins in these areas.3,20 The GSV has also been shown to have duplication in the thigh (8%) and in the calf (25%).3,21 These duplicated vessels lie within the saphenous compartment and later rejoin.22 Of note, the saphenous nerve is present anterior to the GSV in the calf and must be monitored for in cases where GSV incompetence management ranges into the calf.3
The small saphenous vein arises from the dorsal pedal arch (Figure 8.5).3,23 It ascends posterolateral to the lateral malleolus and has a variable termination into the popliteal vein.3 Sixty percent of short saphenous veins (SSVs) join the popliteal vein within 8 cm of the knee, 20% join the GSV, and 20% join another deep vein (deep femoral, femoral, internal iliac).3 The SSV can also continue superiorly as the vein of Giacomini and drain into the GSV via the posterior thigh circumflex vein.3,24 The sural nerve, responsible for cutaneous sensation to the lateral foot and posterolateral leg, is located lateral to the SSV and Achilles tendon in the saphenous compartment. It pierces the muscular fascia prior to termination.3
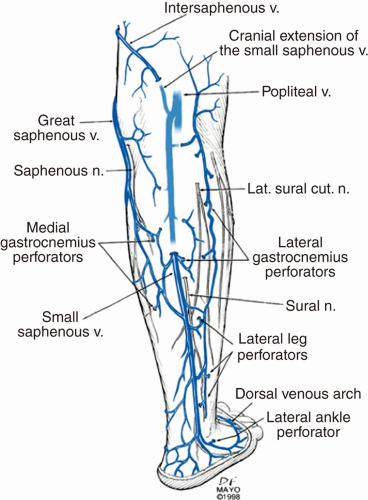 FIGURE 8.5 Lower extremity superficial venous system. (Reprinted from Mozes G, Gloviczhi P. Chapter 2: Venous embryology and anatomy. In: The Vein Book. Elsevier; 2007:15-25. Copyright © 2007 Elsevier. With permission.) |
Perforating Venous Compartment
The veins of the perforating venous system (average of 64 from the ankle to the groin) course through the muscular fascia and function to connect the superficial and deep venous compartments (Figure 8.3).3 This allows blood to flow unidirectionally from superficial to deep.21 There are four clinically significant groups of perforators based on location: foot, medial and lateral calf, and thigh.3 These veins prevent reverse flow by closing their valves during the calf muscle pump, a time when there is increased deep venous pressure.3 Conversely, the valves open up with relaxation of calf muscles to allow blood to flow superficially to deep along pressure gradients.3 The only perforating system that does not direct blood in a superficial to deep direction is that found in the foot.3
A thorough history should be obtained upon initial evaluation.1 This should include a description and duration of symptoms, precipitating and alleviating factors, number of pregnancies, pelvic symptoms (worse with menses, intercourse, standing), history of venous thromboembolism (VTE), family history of varicose veins, ulcers, VTE, coagulopathy,25 peripheral vascular disease,26 coronary artery disease,26 and prior treatments.1 CVD has a wide array of presentations.27,28 The most common presenting symptom is aching/tired legs, although feelings of pain (throbbing, burning, pulling, stretching), swelling (most specific symptom), tenderness over a vein, and restless legs are also experienced.3 Symptoms are worse with prolonged sitting or standing and improve with exercise, leg elevation, nonsteroidal anti-inflammatory drugs (NSAIDs), and compression therapy.29,30
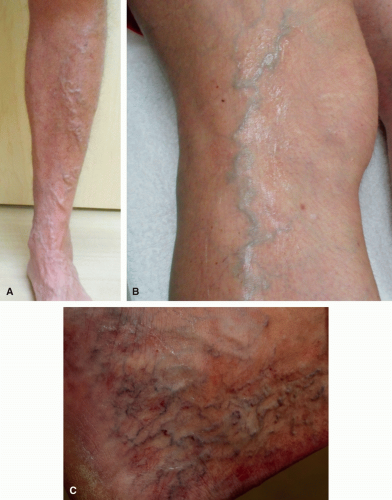
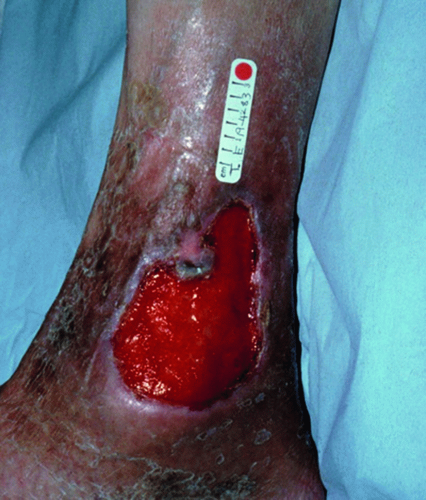
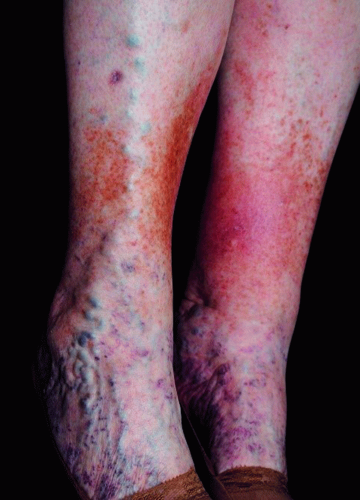
Following a thorough history, a focused physical exam should then be performed.1 On examination, it is imperative to check arterial pulses.31 Confirmation of adequate arterial flow is required prior to usage of any compression therapy.31 Examination findings include varicose veins (3-8 mm, Figure 8.6A), reticular veins (2-4 mm, blue, Figure 8.6B), telangiectasias (0.2-1 mm, red, “spider veins,” Figure 8.6C), edema, ulceration (Figure 8.7), and skin color changes (Figure 8.8).1 Noting the specific locations of varicosities and/or ulcerations can be beneficial in identifying the underlying pattern of insufficiency (Table 8.1). Importantly, abdominal, suprapubic, vulvar, inner thigh, or gluteal varicose veins are concerning for potential ilio-femoral obstruction/thrombosis and may necessitate further workup and imaging.37 Engorgement of pelvic (Figure 8.9) and intrabdominal veins in these areas may present with pain of the low back, pelvis, vulva, and upper thigh areas. This syndrome, known as pelvic congestion syndrome, is characterized by chronic pelvic pain lasting for more than 6 months.36 A history of multiple pregnancies and a family history of lower extremity varicose veins are common.32
Following completion of a history and physical examination, the algorithm in Figure 8.10 can be used to help guide work-up decisions. Any symptomatic patient should receive a bilateral duplex ultrasound performed by a registered vascular technologist. Gray scale ultrasound is used in identification of vein size and vein anatomy (mapping, congenital anomalies, ruling out of obstruction). Color doppler ultrasound is used for identification of flow direction and frequency shifts, and color flow with compression is helpful in ruling out DVT or other obstruction. Pulsed wave doppler can identify abnormal reflux: >0.5 seconds in the superficial system,33 >1 second in the deep system,33 and >0.35 seconds in perforators.34 Other imaging modalities include conventional venography, computed tomography, and magnetic resonance venography/arteriography (MRV/MRA). If a patient is not symptomatic, however, and worrisome findings are noted on examination (lower extremity swelling, extensive varicosities, skin changes), a complete venous mapping and duplex ultrasound is performed. If symptoms and signs are not present, sclerotherapy can be utilized for reticular veins and telangiectasias.
The Clinical-Etiology-Anatomy-Pathophysiology (CEAP) classification (Table 8.2) is a system that standardizes analysis of CVD and management alternatives.35 It is not used as a measure of venous disease clinical severity or as a measure of response to therapy.35 The clinical signs (C) of CEAP are used to categorize the observable signs of CVD. Those with C0-C3 or C4 disease are typically asymptomatic.35
Medical Management
As with many diseases, management of CVD is initially conservative, starting with external compression stockings, elevation of the legs, and exercise therapy, sometimes referred to as the “EEE.”1 In addition, obese or overweight patients are encouraged to also focus on weight loss strategies. These are all efforts to decrease the pooling of blood in the legs, which will help mitigate the symptoms of CVD.1 Additional systemic medications can be utilized, including NSAIDs (decrease pain/inflammation), rutosides (diminish aching/swelling),36 horse chestnut seed extract (increases venous tone, decreases filtration, alters prostaglandins/histamine),37 and pentoxifylline (improves blood flow).38 For symptoms related to stasis dermatitis, topical management can be added (emollients, corticosteroids).39
Despite these efforts, conservative medical management will be unlikely to cure the underlying valvular dysfunction and venous insufficiency present in a large truncal vein. With technological advances, definitive management of CVD has progressed from ligation and vein stripping to more minimally invasive and endovenous approaches (Table 8.3).40 The overall approach to minimally invasive vein treatments can be seen in Figure 8.10. The most commonly used procedures, endovenous thermal ablation (EVTA), sclerotherapy (visual and ultrasound guided), and ambulatory phlebectomy, will be discussed below.40
Endovenous Ablation: Thermal and Nonthermal
Endovenous ablation, a relatively new way of managing CVD, has replaced high ligation and stripping (HL and S) as the management gold standard for saphenous vein varices.40 Endovenous ablation affords numerous benefits over HL and S, including its minimally invasive technique, ability to be performed on an outpatient basis, equivalent management success, decreased side effects (pain, wound infection, hematoma formation), decreased
recurrence rates, and its ability to allow for a rapid return to activity.40 It is often used in combination with sclerotherapy or phlebectomy, as addressing saphenous vein incompetence is imperative prior to management of lower extremity telangiectasias or reticular veins in order to decrease the recurrence risk.41 Two main approaches to energy delivery comprise endovenous ablation: thermal (radiofrequency ablation (RFA) and endovenous laser therapy [EVLT]) and nonthermal (cyanoacrylate glue, VenaSeal, mechanochemical ablation [MOCA]).42
recurrence rates, and its ability to allow for a rapid return to activity.40 It is often used in combination with sclerotherapy or phlebectomy, as addressing saphenous vein incompetence is imperative prior to management of lower extremity telangiectasias or reticular veins in order to decrease the recurrence risk.41 Two main approaches to energy delivery comprise endovenous ablation: thermal (radiofrequency ablation (RFA) and endovenous laser therapy [EVLT]) and nonthermal (cyanoacrylate glue, VenaSeal, mechanochemical ablation [MOCA]).42
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree