BASIC SCIENCE
Seven serotypes of botulinum toxin termed A through G have been isolated from various strains of
C. botulinum.
1,3,6,7 While each serotype produces chemodenervation, there are some differences in their cellular structure and mechanisms of action.
6,7 The most potent types, A and B, are predominantly used in clinical practice.
7Structurally, botulinum toxins are all 150-kDa polypeptides composed of a 100-kDa heavy chain and a unique 50-kDa light chain which are linked by heat-labile disulfide bonds. The toxin is then complexed with nontoxic proteins, mainly hemagglutinins, by covalent bonds and dimerized to form the larger final compound.
3,6,7 In the United States, there are four commercially available BoNT-A: onabotulinumtoxinA (Botox
®) weighing 900-kDa, prabotulinumtoxinA (Jeuveau
®) weighing 900-kDa, abobotulinumtoxinA (Dysport
®) weighing 500-kDa, and incobotulinumtoxinA (Xeomin
®) weighing 150-kDa as it is a monomeric protein free of any complexing proteins.
4,5,8,9,10 Herein these products will be referred to as onabotA, prabotA, abobotA, and incobotA, respectively.
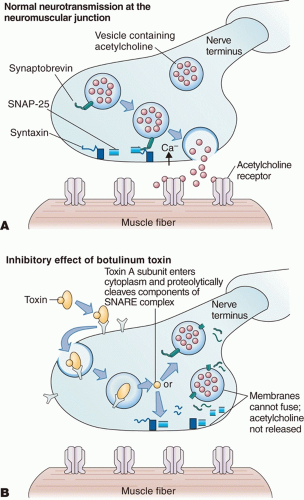
The mechanism by which botulinum toxin inhibits skeletal muscle contraction is due to the inhibition of acetylcholine release from the neuromuscular junction (NMJ).
1,3,6,7 After injection, the complex rapidly and irreversibly binds to synaptotagmin, a docking receptor on the presynaptic terminal of the NMJ. The toxin is then internalized via endocytosis, and the disulfide bonds are cleaved allowing the free light chain to translocate into the cytoplasm. Inhibition of acetylcholine release occurs when the free light chain fuses with proteins in the synaptic fusion complex and deactivates it by a proteolytic zinc-dependent endopeptidase. Several key proteins make up the synaptic fusion complex, known as SNARE (soluble
N-ethylmaleimide-sensitive factor attachment protein receptors). Specifically, botulinum toxin types A, C, and E cleave the 25-kDa synaptosomal-associated protein (SNAP25), while toxin types B, D, F, and G cleave synaptobrevin, also known as vesicle-associated membrane protein.
6,7 Ultimately, the docking, fusion, and release of acetylcholine vesicles are permanently disrupted inhibiting downstream muscular contraction (
Figure 2.1).
Clinically, muscle weakness is often apparent 2 to 3 days after injection with full response seen by 8 to 10 days, although timing can vary based on dose, treatment area, and the individual.
3 Despite the fact that chemical denervation is considered permanent, muscular activity typically returns 3 to 6 months after injection due to neurogenesis.
3,7 Peripheral axonal sprouts form, making new connections with the muscle, and to a lesser degree SNAP25 proteins regenerate in the original NMJ, allowing regained muscular function.
7 For this reason, injections of botulinum toxin are performed at 3 to 6 month intervals for facial rhytides.
3 Some data suggest that with repeated treatments, there is a tendency toward greater interval’s of time between future treatments.
11Immunogenicity is a controversial topic in the literature, but generally speaking, although neutralizing antibody formation against BoNT-A has been reported, the phenomenon is rarely, if ever, witnessed with doses used for cosmetic purposes.
3,7 However, patients treated with 150 to 300 units for other neuromuscular diseases have developed resistance to the toxin’s effects.
7 In these cases, switching to botulinum toxin type B (BoNT-B), available in the United States as rimabotulinumtoxinB (Myobloc
®), may be helpful.
3,6,7 Using the lowest effective doses and longer intervals between injections may minimize the possibility of antibody formation.
3,7

COMMERCIALLY AVAILABLE TOXINS IN THE UNITED STATES
There are four BoNT-A products and one BoNT-B approved by the US FDA to date (
Table 2.1).
4,5,8,9,10,12 Several similarities exist, but each formulation has unique chemical properties and therefore are not interchangeable. Dosing among products cannot be standardized or calculated with any exact conversion ratio as one unit of toxin corresponds
to the calculated median lethal intraperitoneal dose (LD50) in mice which varies across manufacturers.
7 It is critical that physicians understand these differences in order to achieve safe and clinically effective results.
OnabotA (Botox
®), initially approved in 1989, is a sterile lyophilized botulinum A exotoxin distributed as a vacuum-dried single-use vial without preservative. Each vial contains 0.5 mg of human albumin and 0.9 mg of sodium chloride. OnabotA has the most FDA indications including the treatment of glabellar lines, lateral canthal lines, forehead lines, and axillary hyperhidrosis.
4,5AbobotA (Dysport
®), initially approved in 2009, is also a sterile lyophilized botulinum A exotoxin but manufactured with different purification techniques. Each vial contains 0.125 mg of human albumin and 2.5 mg of lactose.
8 Most studies suggest similar efficacy using a conversion ratio of 2-3:1 (abobotA:onabotA).
13 The only aesthetic FDA indication is the treatment of glabellar lines.
8IncobotA (Xeomin
®), approved in 2011, is a sterile lyophilized botulinum A exotoxin that is free of any complexing proteins making it theoretically less immunogenic. Each vial contains 1 mg of human albumin and 4.7 mg of sucrose.
9 Clinical and preclinical data suggest similar potencies between incobotA and onabotA using a conversion ratio of 1:1 or 1.2:1.
13 Like abobotA, the only cosmetic FDA indication is the treatment of glabellar lines.
9PrabotA (Jeuveau
®), approved in 2019, is another sterile lyophilized botulinum A exotoxin distributed as a vacuum-dried single-use vial without preservative. Similar to onabotA, each vial contains 0.5 mg of human albumin and 0.9 mg of sodium chloride.
10 Limited data exist comparing its potency to onabotA, but noninferiority has been established using a 1:1 conversion ratio in phase 3 clinical trials for glabellar and lateral canthal lines.
14,15 The only FDA indication is for the treatment of glabellar lines.
10RimabotulinumtoxinB (Myobloc
®), approved in 2000, is the sole BoNT-B available and the only nonlyophilized stable liquid formulation. Depending on the vial size, each vial contains concentrations of human albumin, sodium chloride, and sodium succinate.
12 Dosing is variable with conversion ratios as high as 1:100 (onabotA:rimabotB) being reported for muscular diseases. RimabotB injections are typically more painful, offer a shorter duration of action, and have more autonomic side effects.
16 There are no cosmetic indications.
12,16
Diffusion
Neurotoxin diffusion refers to the slow dispersion of toxin beyond the original site of injection.
17 In some cases, higher diffusion is desirable such as with the treatment of hyperhidrosis. In comparison, minimal diffusion may be preferred for injections of facial muscles to minimize unwanted adverse effects. Several factors contribute to the rate of diffusion including dose, type of skin, anatomical location, and the density of botulinum toxin receptors.
17 Definitive data are lacking, although some studies using forehead anhidrosis as the end point have suggested innately higher diffusion with abobotA compared to onabotA with equivalent injection volumes.
18,19
Reconstitution
All products available in the United States, except rimabotB, require reconstitution prior to injection.
4,5,8,9,10,12 Reconstitution with preservative-free saline is recommended by the FDA; however, most injectors prefer bacteriostatic saline due to the preservative benzyl alcohol and its anesthetic properties.
20 The amount of diluent added varies greatly among injectors. The most common reconstitution volumes for onabotA, prabotA, and incobotA
ranges from 1 to 5 mL per 100 unit vial compared to abobotA wherein volumes range from 1.5 to 6 mL per 300 unit vial (
Table 2.2). Several studies have evaluated the association between reconstitution volume and diffusion, but the results are rarely clinically significant or reproducible in patients.
17 Since clinical efficacy and result duration do not seem to be correlated with dilution, it may be easiest to pick one dilution initially.
To add the diluent to the botulinum toxin, first remove the plastic cap covering and wipe the top of the vial with alcohol, allowing it time to thoroughly dry. Then, use an 18 to 23 gauge needle to pierce through the bottle top. As the bottle is vacuum sealed, the diluent should be easily pulled into the vial.
4,5,8,9,10 After the diluent is added, gently swirl the vial in a circular motion until all particulate matter is well mixed. Avoid vigorous shaking or foaming of the product.
3,17 Record the date and time of reconstitution as well as the final concentration on the label. All of these data in addition to the lot number and vial expiration date should be included in the patient’s record during treatment.
Safe Handling
Unopened vials of neurotoxin should be kept frozen or refrigerated in temperatures ranging from 2°C to 8°C and used prior to the label’s expiration date.
4,5,8,10,12 IncobotA is the exception which can be stored at room temperatures (20°C-25°C) for up to 36 months or until the expiration date.
9 After reconstitution, the package insert for all neurotoxins suggests storage in a refrigerator (2°C-8°C) and administration within 24 hours.
4,5,8,9,10,12 These narrow shelf life guidelines have been viewed as impractical and are rarely followed. Recent clinical studies have proven maintained safety and product efficacy for up to 6 weeks post reconstitution with appropriate storage techniques.
21 Although this reduces product waste and consequently costs, it is still necessary to utilize safe handling and storage techniques to prevent contamination.

INJECTION TECHNIQUES
Pretreatment preparation is overall minimal both for the patient and for the physician. Traditionally, patients are advised to avoid nonessential use of aspirin, nonsteroidal anti-inflammatory drugs, vitamin E, and other supplements for one week prior to treatment to minimize the risks of bruising.
23 Regardless, botulinum toxin can still be utilized in patients who are taking these medications. If neuromodulator injections are the only procedure planned for the visit then anesthetics are ordinarily not required. Ice packs or topical anesthetics may be applied prior to injections to minimize discomfort in more sensitive individuals.
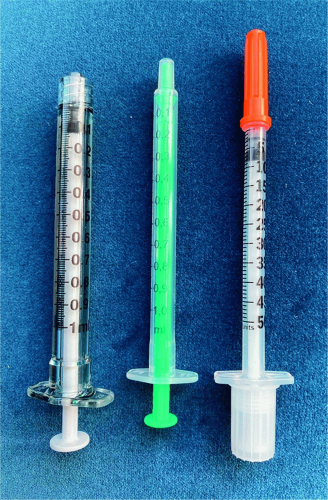
After appropriate consultation with the patient, all treatment sites should be identified with intended doses outlined per treatment area. Use of a marking pen or a white eyebrow pencil to pinpoint each injection site may be considered, especially for novice injectors. The reconstituted botulinum toxin is then drawn up using an 18 to 23 gauge needle on a 1-mL Luer-Lok syringe. The product may either be kept in this syringe for injection or transferred to an ultrafine BD insulin syringe, both of which have clear markings to allow for easy observation of smaller injection volumes. Syringes are available that have a plunger designed to reduce wasting and are used by the authors (
Figure 2.2). Withdrawing all product from the needle bevel will help minimize product waste before transitioning to a 30 to 32 gauge half-inch needle for the actual injection. An additional pearl to minimize product waste is to remove the vial’s cap and stopper and tilt the vial 45° to draw up any remaining droplets (
Figure 2.3). Separating syringes by treatment site can allow for more efficient and accurate injections and decreases the possibility of needle dulling which ultimately leads to improved patient satisfaction.
The areas to be treated should be cleaned with an alcohol wipe or other antiseptic and allowed to dry completely before proceeding with injections. Patients are typically seated upright with their head rested against the procedure chair.
3 It is essential for both the patient and the injector to be in an ergonomically comfortable position throughout the treatment. Typically, syringes are held perpendicular to the skin surface, although technique certainly varies based on the treatment area and injector preference (
Figure 2.4). Injections are delivered slowly into the intramuscular plane or in some areas intradermally or subcutaneously.
3,22 Gauze or cotton tip applicators should be easily accessible in the advent of bleeding at which time gentle pressure can be applied to minimize bruising.


 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access