Introduction
In general, Stage I or II breast cancer can be comparably treated with either a partial mastectomy (lumpectomy, quandrantectomy, segmentectomy, etc.) followed by adjuvant radiation therapy – together referred to as breast-conserving therapy (BCT) – or mastectomy. Six prospective randomized trials have convincingly demonstrated that overall and disease-free survivals are similar between BCT and mastectomy ( Table 4.1 ). Historically, the limitation of BCT centered on concerns of a higher risk of tumor recurrence after BCT than following mastectomy for equivalent disease. While three of the previously noted six prospective trials demonstrated higher local recurrence rates with BCT, two of these studies showing higher recurrence rates with BCT did not have well-established negative margins, an imperative to avert recurrence ( Table 4.2 ). Recurrence of breast cancer is complex and multi-factorial. Factors that contribute to recurrence after BCT (such as young age, lack of chemotherapy and aggressive disease) also predispose to recurrence after mastectomy. Recurrence relates not simply to the operative technique but to intrinsic tumor characteristics and the adjuvant and neoadjuvant therapies utilized. The Early Breast Cancer Trialists’ Collaborative Group demonstrated quite convincingly that the avoidance of local recurrence translates into a survival advantage and that, properly executed, BCT is oncologically equivalent to mastectomy.
| Trial | Endpoint | Number of patients | Overall survival (%) | Disease-free survival (%) | ||||
|---|---|---|---|---|---|---|---|---|
| BCS + XRT | Mastectomy | P -value | BCS + XRT | Mastectomy | P -value | |||
| Milan | 18 years | 701 | 65 | 65 | NS | |||
| Institut Gustave-Roussy | 15 years | 179 | 73 | 65 | 0.19 | |||
| NSABP B-06 | 12 years | 1219 | 63 | 59 | 0.12 | 50 | 49 | 0.21 |
| National Cancer Institute | 10 years | 237 | 77 | 75 | 0.89 | 72 | 69 | 0.93 |
| EORTC | 10 years | 874 | 65 | 66 | NS | |||
| Danish Breast Cancer Group | 6 years | 904 | 79 | 82 | NS | 70 | 66 | NS |
| Trial | Endpoint | BCS + XRT | Mastectomy | P -value |
|---|---|---|---|---|
| Milan | Cumulative incidence at 18 years | 7 | 4 | NS |
| Institut Gustave-Roussy | Cumulative incidence at 15 years | 9 | 14 | NS |
| NSABP B-06 | Cumulative incidence | 10 | 8 | |
| National Cancer Institute | Crude incidence median follow-up of 10.1 years | 19 | 6 | 0.01 |
| EORTC | Actuarial at 10 years | 20 | 12 | 0.01 |
| Danish Breast Cancer Group | Crude incidence median follow-up of 3.3 years | 3 | 4 | NS |
Preservation of the breast is appealing to a number of women. The increased application of BCT in lieu of mastectomy reflects not only the demonstration of the oncologic parity of the technique in early breast cancer, but also a number of advances in complementary, parallel imaging and treatment modalities. Improved screening has resulted in increased detection of early-stage breast cancers. The use of adjuvant chemotherapy and endocrine therapies has lowered the risk of ipsilateral breast disease. Neoadjuvant chemotherapy has enabled some tumors initially too large to be treated with BCT to be downsized and rendered amenable to this treatment. These factors all have implications for determining the optimal oncologic result for women with breast cancer considering their treatment.
The interest in averting aesthetic deformity after BCT has increased as well, evidenced by the emergence of the discipline of Oncoplastic Surgery. The term oncoplastic is derived from the Greek onco meaning tumor, and plastic , meaning to mold, and combines oncologic principles with some form of local tissue rearrangement to avoid deformity with BCT.
Optimizing outcomes
Assessing outcomes in breast reconstruction: BCT vs mastectomy?
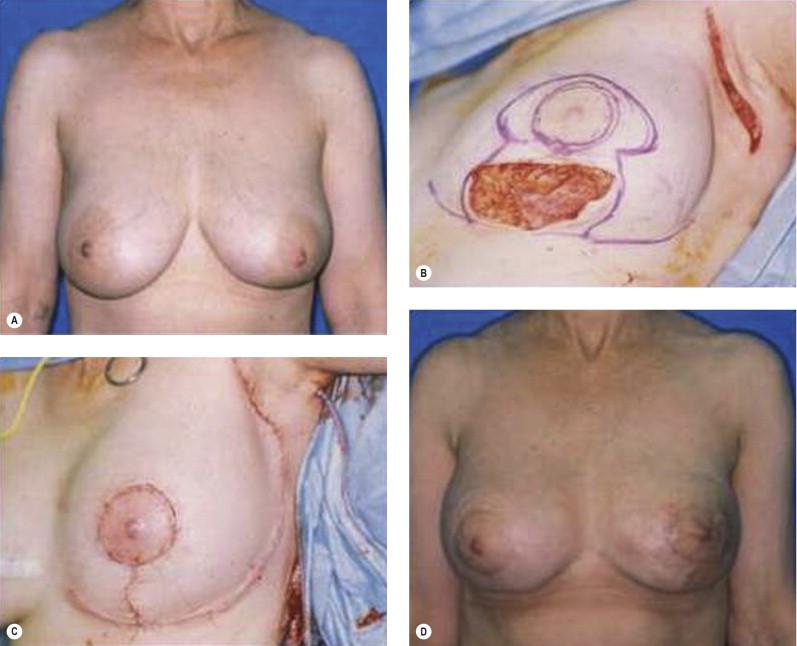
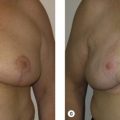
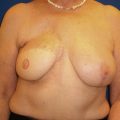
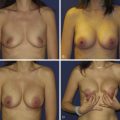
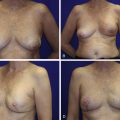
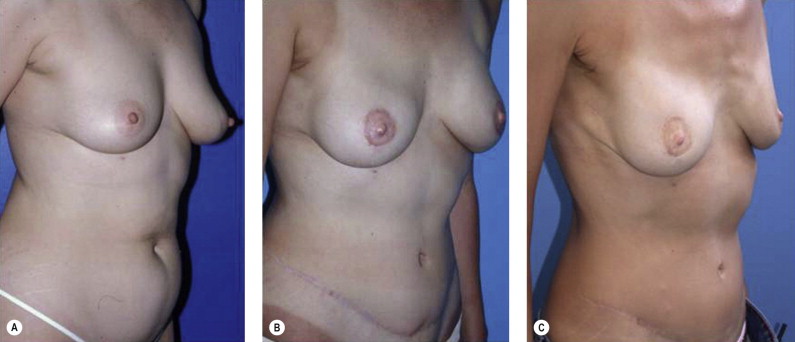
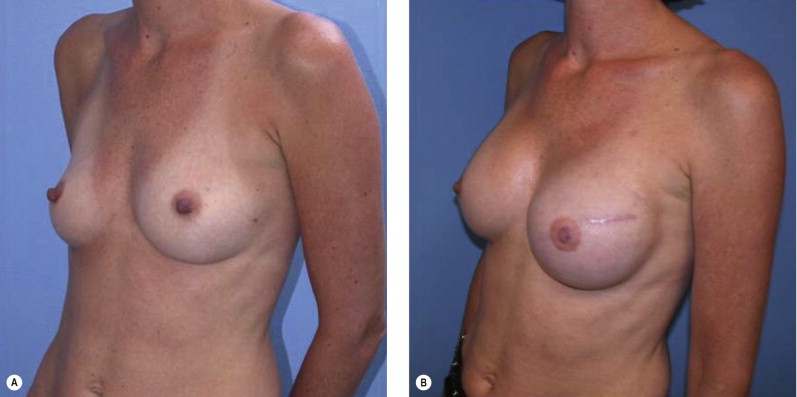
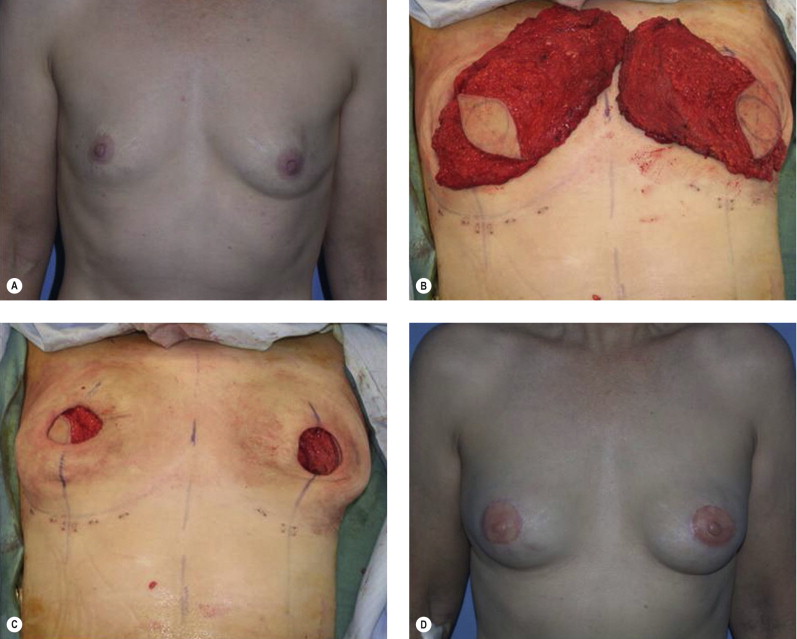
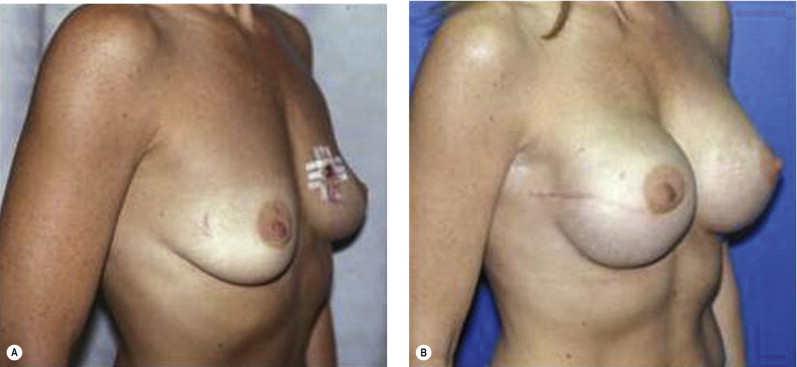
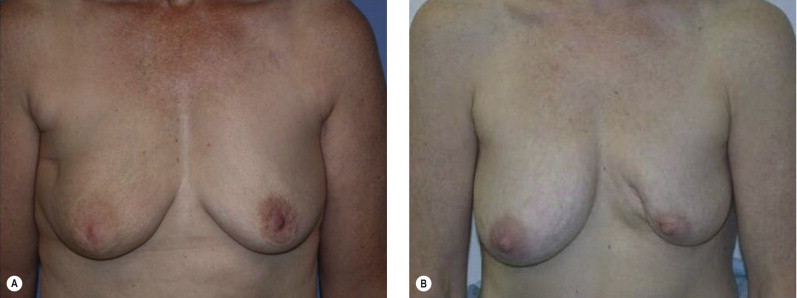
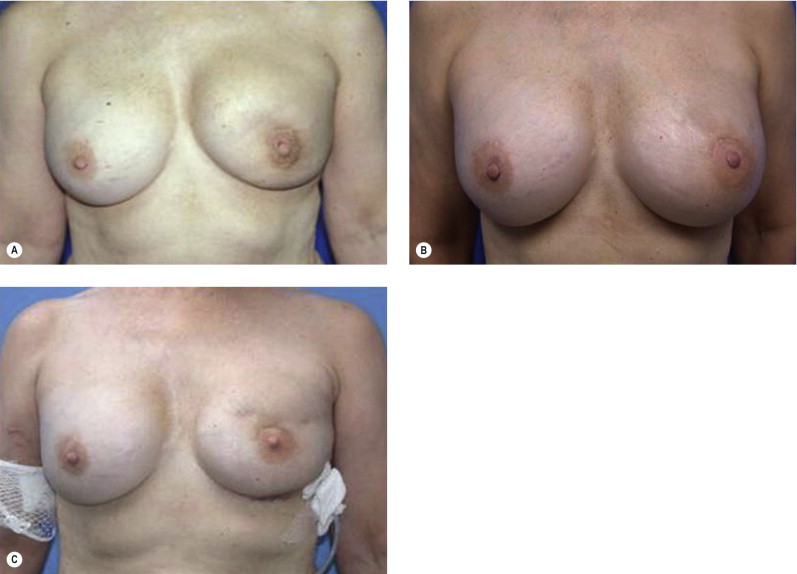
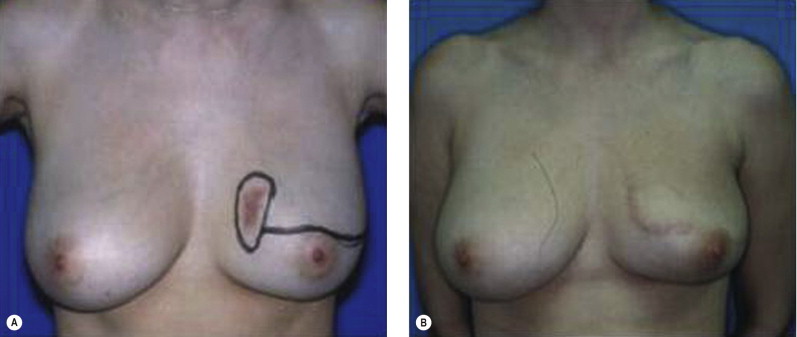
Once the oncologic imperative is met, it then becomes incumbent upon surgeons to determine the best means to address reconstruction in breast cancer, incorporating aspects of patient satisfaction and aesthetic outcome. What factors will necessitate reconstruction in BCT? What are the most accurate determinants of whether local tissues or distant flaps will be the best reconstructive strategy? What is the appropriate timing of reconstruction in BCT: immediate or delayed? If delayed at what time frame is reconstruction most suitable? Can we pinpoint specific patient, tumor or treatment-related variables to guide clinical decision making toward the most favorable outcome? In this context, what factors should compel us to recommend a skin-sparing mastectomy (SSM) with reconstruction over BCT? While numerous studies have attempted to establish guidelines for optimal management in aesthetic outcomes in breast cancer, there is a lack of consensus. In general, proponents of BCT and those of SSM with reconstruction have polarizing and opposing views. Preservation of the breast mound, skin and nipple–areolar complex (NAC) has been cited to afford a psychological advantage to BCT over mastectomy. BCT additionally should avoid ‘major’ reconstructive surgery and the attendant potential for donor site morbidity in autologous reconstruction or implant-related complications such as fibrous capsular contracture, infection, and extrusion. In contrast, proponents of SSM with reconstruction would point out the excellent results currently achieved ( Fig. 4.1 ) Free tissue transfer is considered the gold standard in terms of aesthetic outcome. It is durable and reliable (loss of free flaps for breast reconstruction is generally less than 2–5%). Implant-based reconstruction, while less natural in appearance and feel than autologous approaches, can provide reasonable results in suitable patients ( Figs 4.2 and 4.3 ). Mastectomy for early-stage disease will generally obviate the need for radiation therapy and its untoward sequelae. The toxicity (both cutaneous and cardiopulmonary) associated with radiation in BCT is far less than that incurred with treatment protocols for advanced disease, yet it will limit reconstructive options, and the longitudinal effect on both native breast and flap tissues is difficult to predict. Preservation of the NAC is considered an advantage in BCT, but the supportive data of the efficacy of this aspect of the technique are not clear. There are no well-defined outcomes studies on the retention of sensibility with BCT. The increased popularity of the NAC-sparing mastectomy, currently under prospective investigation at our institution, also may alter patient perspective on total mastectomy versus BCT ( Fig. 4.4 ). Investigations comparing the aesthetic outcome of BCT versus total mastectomy with reconstruction have netted uniformly conflicting results. Rendering ‘objectivity’ to aesthetic outcomes is highly problematic and dependent on a number of factors, including the patient’s perception of outcome as well as the background and bias of the observer doing the ranking. It is well established that there is poor concordance amongst physicians’ assessments (plastic surgeons, radiation oncologists, surgical oncologists) and patients’ judgments in terms of aesthetic outcomes. Resource allocation has taken on an increasingly high priority in the current medical marketplace. Cost analysis suggests that implant-based breast reconstructions are generally less expensive than autologous-based reconstructions due to the increased operative commitment of the latter. The cost of radiation therapy renders the expenditure for BCT comparable to that of mastectomy with reconstruction. In that vein, mastectomy alone with no reconstruction is the least expensive option, but few would argue that cost alone should be a deciding factor in determining such matters. Patient quality of life and satisfaction after breast surgery are difficult to assess accurately in terms of the superiority of one treatment approach over another. Outcome studies have shown comparable satisfaction amongst patients undergoing BCT, mastectomy with reconstruction and even those with mastectomy alone without reconstruction. There appears to be a complex interplay of variables, including age, education, cancer fear, and premorbid psychological status, that contribute to patient satisfaction, making assessments problematic. It is apparent from a number of studies that the perceived level of control that the patient has in the decision process of her treatment – both for the therapeutic and reconstructive approach in her breast cancer – is one of the most universal and significant factors dictating patient satisfaction. While patients’ decision making may be motivated by different factors to a variable degree, including preservation of the breast, cosmetic results, operative morbidity, treatment duration, and convenience, patients are more satisfied if they received the treatments that they wanted.
Complications and side effects
Reconstruction in BCT: limitations of delayed repair
While patients may be happier with the treatment strategy they choose, this satisfaction is predicated on a favorable outcome. Poor outcomes should be anticipated and avoided as far possible. It is estimated that 20–35% of patients treated with BCT have unfavorable outcomes, and a significant proportion of these patients are dissatisfied enough to seek additional corrective surgery. Repair of a deformity after BCT is not straightforward. The effects of radiation therapy figure heavily in the complexity of effecting an adequate repair. Prosthetic breast implants for repair after partial mastectomy are generally best avoided due to prohibitive rates of fibrous capsular contracture. Local tissue rearrangement of the index breast is limited by radiation. Additionally, the patient may be asked to wait a significant amount of time (often 2–3 years after the end of radiation therapy) for the tissues to stabilize and the full effect of the deformity to be manifested prior to intervention. Patients often find this longitudinal period of delay frustrating. Patients who initially desired ‘less surgery’ in their treatment choice for extirpation of their cancer are not eager for a significant operative venture to fix the problem.
The approach to repair of delayed defects after BCT, and the success of this operative venture, will vary by the extent of the deformity. A classification of the deformities associated with BCT has been outlined by a number of authors, to help guide management and predict treatment outcome. A straightforward system based on Clough delineates deformity after BCT as: Type I (Mild) defects demonstrating volumetric deformity with asymmetry; Type II (Moderate) defects demonstrating contour deformity: Type III (Severe) defects with significant contour and volumetric deformity. As the index breast is not deformed but smaller and usually ‘perkier’ than the contralateral breast, Type I defects are most commonly managed with a reduction mammaplasty of the non-treated breast, without manipulation of the index breast. This approach is the most straightforward but is predicated on the willingness of the patient to accept a smaller-volume breast and scars. The non-irradiated breast will invariably descend or drape over time, and a secondary ‘re-pexy’ may be required. Fortunately, patient satisfaction with this approach is high (80%) and complications are limited ( Fig. 4.5 ). The more severe the defect, the higher the complication rate and the lower the level of patient satisfaction. Type II and III deformities usually require flap transfer for correction. The effects of radiation will compromise the aesthetic result and complicate the technical transfer and inset of the flap, and this must be stressed to the patients. Attention to both the skin and parenchymal deformity must be addressed, and due to the contracture in a delayed setting the defect will initially appear smaller. In order to avoid undercorrection, the original defect must be re-created and then the flap designed. The irradiated tissue will invariably contract over the flap and ‘shrink wrap’ around it, such that well-vascularized tissue needs to be introduced to all areas of the breast and chest wall and axilla, requiring correction to achieve the best possible result. The juncture of skin paddles between flap and native breast will usually have a color and texture mismatch and a contracted or biscuit/pincushion deformity ( Fig. 4.6 ). The later contour deformity may be minimized with a more linear (as opposed to circular) flap design, but will be especially notable and essentially inevitable if there has been chronic inflammation or infection in the breast being repaired ( Fig. 4.7 ).
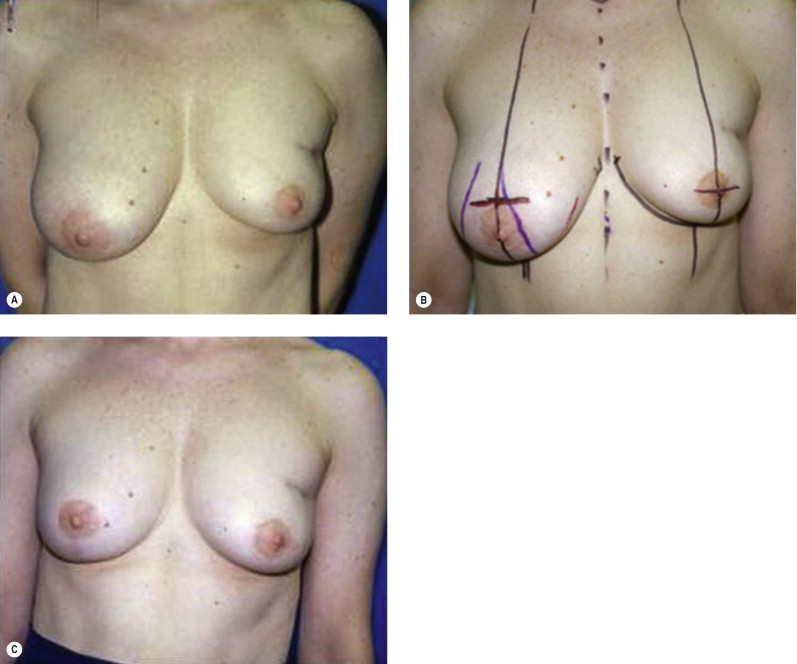
Type II deformities may require a contralateral balancing procedure in addition to the flap transfer. Patient satisfaction with this repair at 3 years is approximately 45%. ( Fig. 4.6 ). Type III deformities will require a flap for correction, as well as a contralateral balancing procedure ( Fig. 4.7 ). Patient satisfaction may be as low as 20% in this setting. A number of these patients may be better served by a total mastectomy and reconstruction, depending on the severity of the deformity.
Avoiding deformity in BCT: key factors
As delayed repair appears largely unsatisfactory, it becomes clear that we must identify those patients who will likely experience a significant deformity after BCT and attempt to prevent this occurrence. The three factors that will reliably motivate a patient to seek reconstruction after partial mastectomy are: (1) volume discrepancy of greater than 20%; (2) contour deformity; (3) nipple malposition. While it is impossible to fully anticipate all unfavorable results, the aforementioned sequelae are the predictable result of several operative interventions in BCT. The most common causes of an unfavorable result in BCT include: (1) Removal of more than 15–20% of the breast parenchyma in a small-volume (A, B cup) breast or 30% or more of the breast in a larger breast. This volumetric discrepancy will be noted by and concern the patient. (2) Resection of tissue in an aesthetically sensitive area. Patients will often tolerate some contour deformity but not one in the critical cleavage area. Accordingly, a tumor extirpation that results in removal of a significant amount of parenchyma and/or skin, especially in an area in which there is a paucity of redundant tissue, such as the medial/superior pole of the breast, will likely be problematic in the long term. (3) A contour deformity will be made more severe if the focal defect results in adherence of the skin to the underlying pectoralis muscle or fascia. These defects result when less than 1–2 cm of subcutaneous tissue is left on the skin flap and/or the parenchyma/fatty tissue of the breast is removed, exposing the pectoralis muscle/fascia. This defect is often manifest later in the postoperative course as the inevitable seroma (the result of dead space, and/or the intraoperative placement of fluid by the oncologic surgeon) resorbs over time. These deformities will be exacerbated by the longitudinal effects of radiation therapy. (4) Nipple–areolar malposition is difficult to camouflage. Removal of a relatively small amount of skin adjacent the NAC may result in nipple malposition. Even if little skin is removed, loss of breast parenchyma under the NAC will cause an angulation deformity or ‘tip’ the nipple. Excision of skin will both ‘tip’ and change the nipple position significantly. Excisions medial, lateral, superior but most commonly inferior to the nipple may result in a highly unfavorable deformity. This deformity is classically manifest after a transverse or ‘Langer line’ excision in the lower pole of the breast (between the NAC and inframammary fold (IMF)) and will result in severe nipple malposition unless a radial incision and/or some tissue rearrangement is employed and minimal parenchyma is excised. These contour and shape deformities are not only visible unclothed, but often make it difficult for a patient to wear undergarments and certain types of clothing, prompting patients to request repair.
The parameters which appear to impact most significantly on the aesthetic outcome in BCT, and suggest the need for reconstruction with BCT or mastectomy with reconstruction, include the extent and location of the tumor and volume of resection, the ptosis and volume of preoperative breast size, and the ratio of tumor volume excised to breast size. The most appropriate reconstructive strategy for management of these challenges depends not only on the characteristics of the defect, but also the willingness of the patient to accept a significant volume/shape change in the index as well as the contralateral breast to achieve symmetry, as well as their willingness to undergo a flap reconstruction. One of the greatest challenges in the assessment for the need for reconstruction in BCT is the accurate estimation of the impact of radiotherapy on the surgical defect. The balance of both treatment and patient-related factors will determine the reconstructive plan and timing, and ultimately which patients will be best served by partial mastectomy alone and those that will require reconstruction in conjunction with BCT. The approach must be further refined to determine those patients best managed with an ipsilateral tissue rearrangement, those that will necessitate both ipsilateral and contralateral surgical intervention for symmetry, and those patients in whom a regional flap may be needed and ultimately those in whom SSM and immediate breast reconstruction are preferable to BCT on aesthetic grounds.
Indications and contraindications
Indications for immediate reconstruction in BCT
Extent of resection and tumor variables
The absolute volume of the surgical specimen in BCT appears to be of less import than the ratio of the defect to the remaining breast parenchyma. Defects in large breasts are usually better tolerated than those in smaller breasts. In patients with a small breast, even a small defect can create a big problem. This is especially important in tumors involving areas critical to the appearance of the breast, such as the medial superior pole, in which resection often results in a highly visible contour deformity or cases in which significant volume of breast parenchyma or skin between the NAC and IMF is resected, which may not only lead to a contour deformity but also a marked distortion of nipple position ( Fig. 4.8 ). Unless there is a compensatory adjustment of both skin and parenchyma to correct for removal of breast tissue in these areas, contour deformities will develop and will be exacerbated by radiation. Immediate reconstruction is imperative in these cases as attempts at scar release in a delayed fashion are usually unsuccessful.
The greater the extent of surgery, the more likely is deformity after BCT. In the breast, the defect incurred from resection is more accurately assessed as volume rather than weight, as the breast is conical in nature. The total breast volume can be estimated from a mammogram using the formula for a cone: V = 1/3 π r 2 h (where h is the height of the breast off the chest wall and 2 r is the diameter of the breast). Resection of less than 100 cm 3 results in better cosmesis than when larger volumes are resected. Larger resections (120 cm 3 ) will usually require concurrent reconstruction to avoid deformity.
Additional factors found to contribute to a poor aesthetic outcome after BCT and necessitating consideration for reconstruction include inner as opposed to outer quadrant tumor location, need for re-excision for margin control, need for axillary lymph node dissection, greater extent of surgery, and existing significant volume discrepancy between the breasts when a tumor involves the smaller breast. In these circumstances the surgeon must be critical as to whether the best result may be achieved by immediate reconstruction in BCT or conversion to SSM and reconstruction.
Patient-related indications for reconstruction in BCT
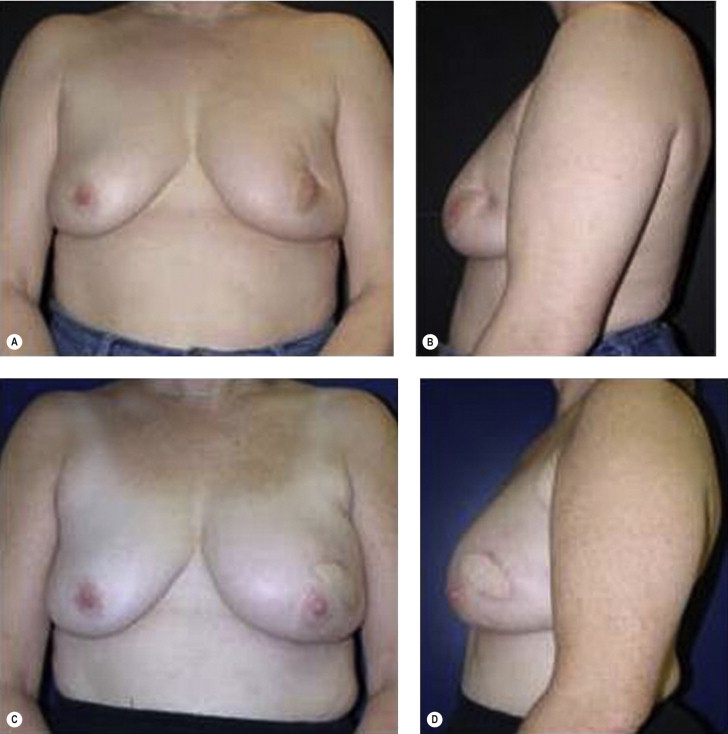
Older patients with more ptotic breasts have less favorable results after BCT, suggesting the need for immediate reconstruction or SSM and mastectomy. Obese patients and patients with large breasts are often excellent candidates for tumor resection in concert with bilateral reduction mammaplasty. Obese patients present a reconstructive challenge after total mastectomy, as in the higher weight ranges (body mass index (BMI) > 35) patients may have significant morbidity with autologous breast reconstruction. Implants alone as a reconstructive strategy are rarely large enough to recreate a proportional breast for their body size and carry a high complication rate in this population. Breast reduction strategies can permit good aesthetic outcomes after resection of large volumes at any location in the breast. This approach is best undertaken at the time of tumor resection as local tissue rearrangement/reduction of a radiated breast is fraught with complications and not generally recommended. Additionally, reduction of the breast will often limit some of the skin toxicity and potential inhomogeneous dosing of radiotherapy associated with large, ptotic breasts.
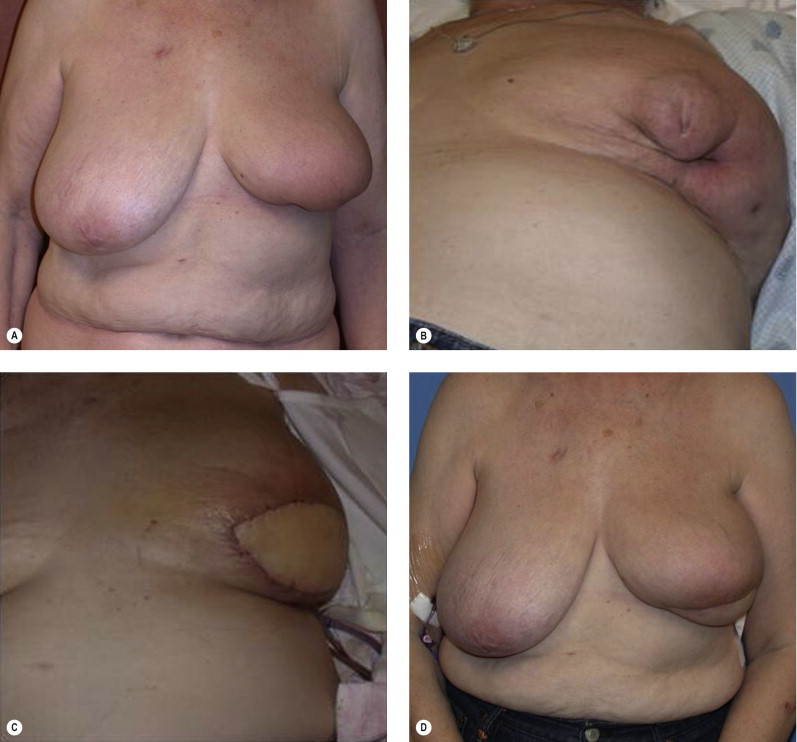
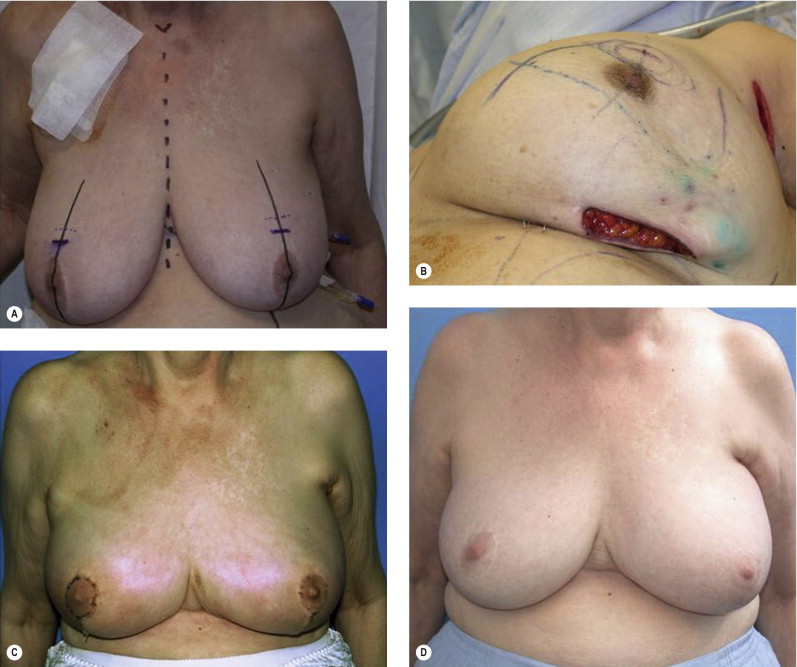
The prior augmentation mammaplasty patient presents a difficult management challenge in all types of breast reconstruction. These patients often have a high aesthetic standard and wish to minimize scarring and maintain their implants. The long-term sequela of implants that have been preserved in BCT is generally unfavorable. These patients may be best served by mastectomy and reconstruction in order to optimize aesthetic outcome, but may be resistant to this approach. The larger the implant relative to the remaining breast parenchyma, the worse the result is after radiotherapy. Patients must be prepared for the inevitable fibrous capsular contracture and distortion of the breast, as well as the potential for rib erosion and fractures and implant loss and infection ( Fig. 4.9 ). Repair of these deformities will often require significant operative intervention with autologous tissue and patients must be made aware of the potential for these outcomes.
Radiotherapy: impact on reconstruction in BCT?
While the impact of radiation therapy on overall aesthetic outcome is, not unexpectedly, debated by surgeons and radiation oncologists, it is generally considered to be the single most significant obstacle in the achievement of an aesthetic outcome. Irradiation of the breast usually results in shrinkage and fibrosis, with an average 10–20% decrease in breast volume. Unfortunately, the degree to which radiation therapy impacts on the final result does not appear entirely predictable, and this makes anticipation of the need for reconstruction in BCT and/or the necessary degree of overcorrection of the deformity problematic. Patients respond to radiation differently, and patients with similar volumes of resection in breasts of similar size may have very different outcomes, depending on their idiosyncratic response to radiation. Certain factors have been more closely implicated in a poor result after radiation for BCT. Classically, application of radiotherapy to smaller breasts or more ptotic breast has been generally considered less favorable, and avoided, but this is far from universal. The use of a boost in radiation therapy, and/or therapy incorporating iridium as opposed to electrons, appears to be implicated in poorer results in BCT. Radiation following greater extent of surgery, such as significant undermining of the breast and/or devascularization of the breast as manifested by poor healing, skin flap compromise or fat necrosis also appears to be implicated in poorer results in BCT. Mature breasts in which there is a lower volume of breast tissue and a greater degree of fat replacement tend to have a worse cosmetic outcome after radiation than dense breasts. These criteria suggest the need for immediate reconstruction in BCT as correction of these sequelae is usually unsuccessful.
Perioperative history and considerations
Role of perioperative planning and neoadjuvant therapy in BCT outcomes
Limiting the volume of tissue resected will limit or potentially avoid any needed reconstructive effort and improve outcomes after BCT. Careful preoperative review of the imaging studies of the breast facilitates decision making regarding the appropriate localization technique (mammography or sonography), the number of needles to be placed for marking the extent of disease, and placement of the incision and therefore the precise area and volume to be resected. Centering the lesion in the partial mastectomy specimen will minimize the amount of normal tissue that must be resected to obtain a negative margin ( Fig. 4.10 ). Patients with large primary tumors may benefit from neoadjuvant (preoperative) chemotherapy to shrink the tumor. In patients with tumors that are initially too large to permit BCT, neoadjuvant chemotherapy may make BCT possible, and has not compromised survival. In patients who are candidates for BCT at initial presentation, neoadjuvant chemotherapy may reduce the volume of tissue that has to be resected and thereby lead to improved cosmesis.
Intraoperative margin assessment
Positive margins have been shown to be predictors of local recurrence and decreased disease-specific survival after BCT. Aggressive local therapy is necessary to ensure adequate surgical margins and to minimize the risk of ipsilateral breast tumor recurrence. If immediate reconstruction is planned in BCT it is of course imperative that negative margins are verified prior to the reconstructive effort. The specimen must be oriented such that if re-excision is necessary the appropriate region can be easily identified and resection limited to the area of involvement. Accordingly, when immediate reconstruction is undertaken in BCT, extensive margin analysis is necessary prior to any tissue movement for reconstruction. At our institution we expend considerable time to assess the margins accurately. The specimen is inked with different colors to indicate superior, inferior, superficial, deep, medial, and lateral aspects and examined by the pathologist. Specimen radiography is crucial to the intraoperative assessment of margins, especially in nonpalpable tumors. The specimen is serially sectioned, and reviewed by the radiologist and the pathologist. All microcalcifications and any marking clips placed preoperatively have been excised with the specimen. Only once the surgeon, pathologist, and radiologist are comfortable that margins are negative and all abnormalities have been excised should the tissue rearrangement for reconstruction in BCT begin. Surgical clips should be placed to delineate the territory of resection. These clips will guide treatment planning by the radiation oncologist.
Although intraoperative margin assessment can be time consuming and labor intensive, it is worthwhile, especially if tissue rearrangement is planned. Barrios et al showed that intraoperative evaluation of surgical margins by macroscopic, cytological, and histological analysis at the time of initial surgery revealed inadequate margins and led to re-excision in 37.3% of cases. Intense intraoperative processing of the breast specimen as described above can significantly decrease the need for re-excision to achieve negative margins from 40% to 22% and can contribute to enhanced aesthetic results.
Operative approach
Surgical approaches for reconstruction in BCT: considerations for outcome and timing
Repair of defects resulting from partial mastectomy can generally be grouped into three main categories: local tissue rearrangement with composite breast flaps, reduction mammaplasty, and transfer of remote tissue in the form of a vascularized regional or distant flap. The technical nuances of these options will be discussed in subsequent chapters of this book. The following discussion will instead focus on considerations for the relative merits and limitations of each surgical approach and key considerations for the most suitable application of each reconstructive strategy.
Skin incisions
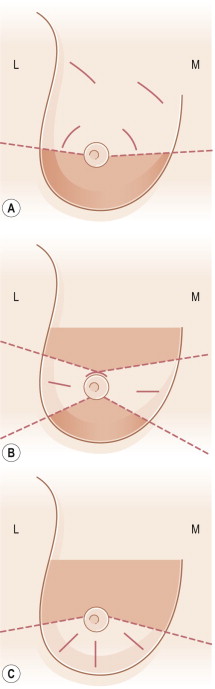
The unfavorable location and/or orientation of skin incisions have been linked to unfavorable outcomes in BCT. In a small-volume resection, the skin incisions used for partial mastectomy have a significant impact on the aesthetic outcome of BCT. Oncologically, the scar should be centered over the tumor. Appropriate scar orientation may make the difference between a poor and an acceptable aesthetic result, and avert the need for significant reconstructive venture ( Fig. 4.11 ). Oncologic surgeons have been taught to follow natural ‘wrinkle lines’ in the breast and to employ curvilinear incisions following Langer’s lines (concentric lines parallel to the edge of the areola) or Kraissl’s lines (natural horizontally oriented skin creases) in the breast. This approach can be problematic, as the majority of patients do not have significant rhytids in the breast in which to ‘hide the scar’ and the scar contracture coupled with underlying parenchymal loss results in noticeable distortion of the breast. Periareolar incisions are well tolerated, but only with small resections, and limit access for distant tumors. The utilization of radial incisions is a far more favorable approach and is a fundamental tenet of reduction mammaplasty, adopted by oncoplastic surgery to avoid contour deformity and/or malposition of the nipple by complementary advancement of parenchyma and skin. It is imperative that skin incisions be planned such that if a mastectomy is ultimately required for margin control, the incision site can be comfortably included within the mastectomy skin island.
Local composite tissue rearrangement
Local tissue rearrangement is the most straightforward option for repair of partial mastectomy defects. This approach obviates donor site issues, preserves subsequent options for reconstruction and should be the first reconstructive strategy considered. Aesthetic outcomes with this approach are optimal when the defect is of limited size and the rearrangement is performed in the immediate setting. Composite breast flaps include full-thickness breast parenchyma plus skin and are best rotated or transposed en bloc. It is imperative to ensure that the tissues to be transferred are well vascularized, to avoid subsequent aesthetic compromise (or oncologic concerns) due to fat necrosis and scarring of the transferred breast parenchyma. These flaps, which were popularized by the late Stephen Kroll and others, are primarily used to effect a subaxillary shift of tissues from lateral to medial ( Fig. 4.12 ). Composite flaps assure a reliable vascular base and breast contour but are not appropriate for large defects, in which composite flaps can result in very noticeable scarring and distortion of the nipple. This limitation and the often extensive scarring attending this approach have likely led to its essential eclipse by reduction mammaplasty techniques for immediate reconstruction in BCT.
Reduction mammaplasty techniques
In women with macromastia, performing breast reduction in conjunction with the oncologic surgery permits BCT and can produce excellent cosmetic results. The reduction surgery permits better dosimetry and reduces the number of ‘hot spots’ and the volume of the lung and thoracic structures in the irradiation field. Reduction improves the homogeneity of radiation treatment in women with large breasts and lessens the risk of moderate to severe late radiation changes such as breast fibrosis. Reduction mammaplasty also relieves symptomatic macromastia (shoulder grooving, cervical and thoracic strain, and mastodynia) and can be considered to improve breast health. Other potential benefits of reduction mammaplasty for oncoplastic breast surgery are reduction in the risk of cancer of the contralateral breast. Although the efficacy of reduction mammaplasty as a risk reduction procedure is controversial, this premise has some support. While the incidence of finding an occult cancer in a routine breast reduction specimen is low (0.16–0.5%), the risk may be higher when a contralateral breast cancer has already been diagnosed. This highlights the need for bilateral diagnostic mammography prior to any surgical interventions.
When a reduction mammaplasty approach is being considered, the expected oncologic and aesthetic outcomes must be carefully considered. If there are concerns that the resection needed to obtain a negative margin will result in significant deformity that cannot be corrected with local options, reconstruction of the defect immediately with remote flaps or endorsement of a total mastectomy and reconstruction must be considered. These concerns are most common in small-breasted patients. Unless the patient has significant macromastia, resection of greater than 30% of the breast will usually result in an unattractive result. In the majority of patients with large breasts, this is not an issue as the volume of the partial mastectomy specimen is most often significantly smaller than the volume of tissue removed for breast reduction.
The effects of radiation on the breast after partial mastectomy are difficult to anticipate, and reduction mammaplasty is no exception. In the early postoperative setting, edema and residual seroma pockets give the breast a quite favorable appearance that may be different from the final outcome at 18–36 months after radiation therapy. Some authors suggest ‘over-reducing’ the contralateral breast to account for the likely fibrosis and shrinkage of the index breast in response to irradiation. However, this approach is not universally endorsed given the highly variable and unpredictable outcome of radiotherapy. The patient should be advised preoperatively that an additional small balancing procedure may be required on the nonirradiated contralateral breast. Breast reduction and adjustment of the irradiated breast have been reported but are associated with a high risk of complications and should be avoided. The reports of successful use of this approach noted poor wound healing, tissue loss, and nipple compromise. Patients should be counseled that the success of intervention on a radiated breast, without the introduction of well vascularized tissue, will most likely not be successful.
In order to achieve the best possible aesthetic results of reduction mammaplasty, it is imperative to address the location and amount of both breast parenchyma and skin that is to be removed. To achieve symmetry, similar reduction techniques and scar placement should be used for the index breast and the contralateral breast. This may be difficult to predict preoperatively. Symmetry is most easily and often best achieved if the contralateral breast is shaped after the oncologic surgery on the index breast has been completed and margins verified. Flexibility in the application of an adequate vascular base for the breast mound in a reduction mammaplasty approach in BCT is critical and consideration for both skin and parenchymal components is important. The ultimate shape of the breast may be considered to result from the parenchyma as well as the skin. Short scar/vertical approaches are used in smaller, less ptotic breasts or a traditional inverted T scar in patients with significant skin redundancy ( Figs 4.10 and 4.13 ) The vascular pedicle for the breast mound can be adjusted to the tumor location, as nicely delineated by Losken ( Fig. 4.14 ). As tumors more commonly occur in the lateral quadrants, medially based pedicles, which derive their vascular supply from the internal mammary perforators, are very useful.