Introduction
Breast conservation therapy, or partial mastectomy, continues to be a popular treatment option for breast cancer patients. As a result, the demand for suitable oncoplastic reconstructive options is imperative. This trend has been supported by the increased use of neoadjuvant chemotherapy, rise of mammographic screening leading to earlier detection of breast cancers, and proliferation of breast cancer reconstruction awareness. Advancements in partial breast radiation therapy and intraoperative radiation technique support breast conservation therapy displaying effective local control with potentially less soft tissue damage and long-term soft tissue sequelae.
Most defects created from breast conservation therapy can be closed primarily; however, the aesthetic outcomes are often unpredictable. Adjuvant radiation therapy may distort the breast, and the ensuing scarring and fibrosis can make it difficult to correct. Some of the clinical changes to the breast seen with breast conservation therapy include asymmetry, skin dimpling, contour irregularity, and nipple retraction. These obstacles were considered by Werner Audretsch who provided the German experience of repairing lumpectomy defects before radiation to American plastic surgeons in the late 1990s. This was considered a revolutionary concept at the time. Although breast conservation and mastectomy therapies result in equivalent long-term survival, oncoplastic reconstruction following breast conservation was historically slow to catch on in the United States. Although advancements in technology and surgical technique have emerged with time, the primary consideration in breast conservation reconstruction has always remained consistent: tumor-to-breast size ratio. A large tumor in a small breast presents reconstructive challenges due to minimal remaining breast parenchyma following excision. Conversely, small to moderate tumors in a large breast allow for many reconstructive approaches. These fundamental considerations create the basis for the volume displacement and replacement techniques to be discussed in this chapter.
Volume loss is conceivably the most important change with breast conservation therapy. With oncoplastic reconstruction, volume loss can be corrected. Small to moderate losses of volume allow for the use of volume displacement techniques including parenchymal remodeling and plication practices, local tissue rearrangement, and mastopexy or reduction ( Table 7.1 ). Larger losses of breast volume call for volume replacement techniques like adjacent or distant tissue transfers, fat transfers, and even implant placement. When anticipated volume loss exceeds 50% and the reconstructive plan entails using a distant tissue transfer, the option of mastectomy should be given strong consideration. In this setting, the potential advantages of breast conservation therapy including preservation of breast tissue and sensation, less surgical invasiveness, and shorter recovery are lost.
| Volume Displacement Techniques | Volume Replacement Techniques |
|---|---|
|
|
Timing of Repair
The timing of oncoplastic reconstruction can be at the time of the lumpectomy or partial mastectomy (immediate), after the tumor excision but before radiation (staged immediate), or at an unspecified time after radiation therapy (delayed). Whenever possible, immediate reconstruction should be favored by the oncologic team. This involves proper planning and communication between the resecting surgeon, reconstructive surgeon, and radiation oncologist. Many studies have shown higher success rates in immediate reconstruction than those reconstructions performed in the delayed setting. More importantly, immediate reconstruction does not pose a problem with postoperative cancer surveillance. Local recurrence rates have been shown to be as low as 5%. There are, however, potential limitations of the immediate approach, which include delayed wound healing, positive tumor margins, longer surgery time, and poor result. All of these factors may also delay the onset of radiation therapy.
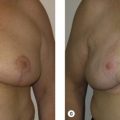
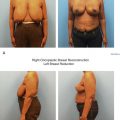
When there is concern for positive tumor margins such as with large or multicentric tumors, or multifocal disease, then a staged immediate approach should be considered. This allows for confirmation of negative margins and, when necessary, re-excision of positive margins before radiation therapy ( Fig. 7.1 ).
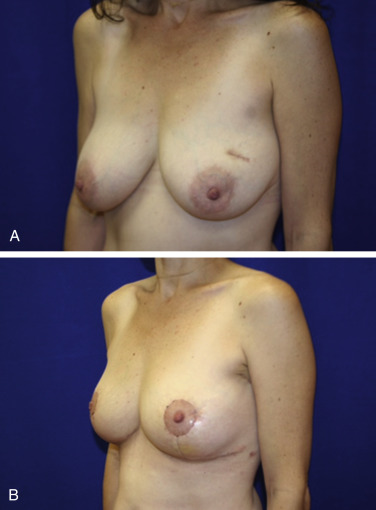
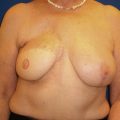
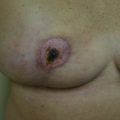
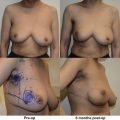
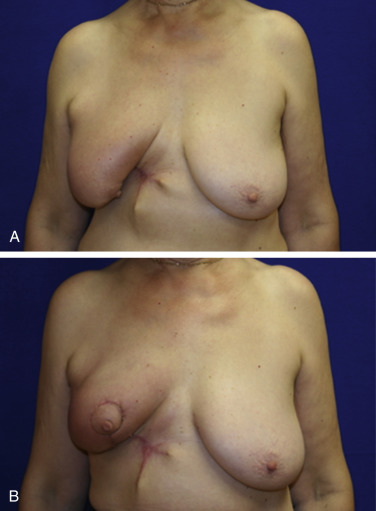
Once scarring and fibrosis has set in following radiation therapy, manipulation of the breast parenchyma is limited. Higher complication rates have been seen in delayed reconstruction groups with difficulties including wound breakdown, scar contracture, cellulitis, and seroma. Even the most optimal result in the delayed setting fares poorly when compared with immediate reconstructions ( Figs. 7.2 and 7.3 ).
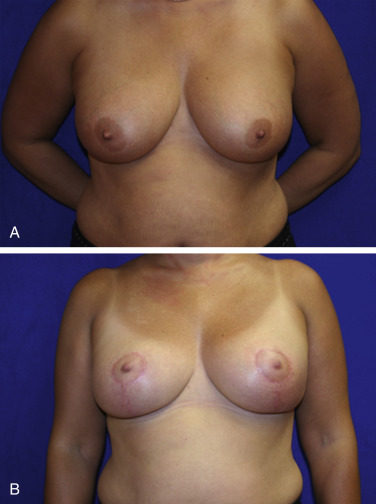
The timing of the symmetry procedure is up to the reconstructive surgeon; there is no existing standard. It is my practice to delay the symmetry procedure until the radiation therapy of the affected breast has been completed. This approach optimizes breast symmetry, reduces surgical time, and eliminates the possibility of delaying radiation treatment due to healing issues in an otherwise healthy breast. The obvious limitation of this approach is the need for a second surgery. The potential changes in the radiated breast may be an increase in size from chronic edema related to impaired lymphatic drainage or a decrease in size due to atrophy from radiation-induced fat necrosis. Allowing time for observation of these potential changes ensures the most accurate symmetry. Consideration must also be given to the patient’s emotional state and quality of life. For patients with severe macromastia, a simultaneous symmetry procedure is offered, and the possibility of postoperative asymmetry due to radiation changes is explained. Some have also reported leaving the ipsilateral radiated breast roughly 10% larger during simultaneous reductions to allow for radiation fibrosis correction. Otherwise, the symmetry procedure is offered 2–3 months after the completion of radiation.
Classification of Partial Breast Defects
The variability in partial mastectomy defects range from small to large. Therefore, their respective reconstructive needs have led to numerous classification systems and algorithms for repair. The various classifications systems share in common the evaluation of initial breast volume, the extent and location of the anticipated defect, and the remaining available breast tissue ( Table 7.2 ). In addition to the extent of the defect, the classification system should reflect how much skin is removed and where the defect is in relation to the nipple–areolar complex.
| Description | Potential Outcome | Treatment | |
|---|---|---|---|
| Type 1 (favorable) | Small, peripheral defect Large or small breasts | Breast shape is preserved with acceptable symmetry | Primary closure, breast tissue advancement flap |
| Type 2 (unfavorable) | Medium to large defectCentral defects | Size and shape deficiency with noticeable distortion and asymmetry | |
| Type 2a | Small breasts, with or without ptosis | Volume replacement procedures | |
| Type 2b | Medium to large breasts, with or without ptosis | Quadrantectomy defect outside standard Wise pattern | Volume replacement technique |
| Any lumpectomy defect | |||
| Quadrantectomy defect within standard Wise pattern | Volume displacement techniques | ||
| Type 3 (unfavorable) | Extensive resection with little remaining breast tissue | Major deficiency | Mastectomy and reconstruction |
| Large or small breasts |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree