and Frank Hölzle2
(1)
Department of Oral and Maxillofacial Surgery, Klinikum rechts der Isar, Technische Universität Munich, Munich, Germany
(2)
Department of Oral and Maxillofacial Surgery, University Hospital of RWTH Aachen University, Aachen, Germany
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-319-53670-5_7) contains supplementary material, which is available to authorized users.
7.1 Development and Indications
In 1974, an upper transverse abdominal flap for breast reconstruction was described by Tai and coworkers, who gave the first description of this myocutaneous flap based on the deep superior epigastric vessels and their perforating vessels through the rectus muscle [524]. Another early report of flap raising procedures at the anterior abdominal wall including parts of the rectus muscle was given by Brown et al., who described the myocutaneous rectus abdominis flap used as a pedicled flap for covering an extensive ipsilateral defect of the arm after a shotgun injury [62]. Two years later, Drever described a superiorly based rectus abdominis flap with a vertical skin island which he used as a pedicled flap to treat a scar contracture at the chest. In this publication, he also proposed using this flap for many reconstructive purposes, especially in the head and neck region, as a free flap. Like Brown and Tai, he recognized the dependence of skin perfusion from perforators through the rectus muscle, and they pointed out that long transverse skin island of the abdominal skin could be based on these perforators. In the years to follow, the superiorly based rectus abdominis flap, nourished via the internal mammary vessels, was primarily taken with a horizontal skin paddle and used for breast reconstruction [368, 381]. These reports were followed by the first microvascular transfer with anastomoses to the deep inferior epigastric vessels performed by Pennington and Pelly [415]. In their report, they present two free flaps based on the deep inferior epigastric artery and highlight the long, wide lumen pedicle and the ease of flap raising. Nevertheless, the authors also mentioned the need to reconstruct the anterior rectus sheath and the possible bulk of the flap as disadvantages. Before that, free flaps from the lower abdominal wall have already been raised, but using the superficial epigastric vascular system. Antia and Buch [15] are credited to be the first having successfully performed a microsurgical free-flap transfer by transferring a dermis-fat flap from the lower abdomen for facial contour augmentation, thereby using the superficial inferior epigastric vessels and including a cuff of the femoral artery for safe anastomosis [15]. A few years later, Taylor and Daniel [538] gave an anatomic description on this superficial epigastric vascular system, which, compared to the deep epigastric vessel, was highly variable and consisted of small caliber vessels. Both, the deep and the superficial system, have partially overlapping vascular territories and are connected by choke arteries. Of these, the paraumbilical perforators originating from the deep system are the strongest and therefore contribute most to skin perfusion of the anterior abdominal wall [51, 60, 161]. Very soon after the first reports, a number of different flap designs and orientations of the skin paddle were described, all confirming the unique usefulness of myocutaneous flaps from the deep inferior vascular system. Intraoperatively, Harris and coworkers studied the blood flow of the deep and superficial epigastric vessels and showed that most vascular connections between both systems are concentrated centrally. Nevertheless, they emphasized elevating the entire muscle to preserve all vascular connections to the skin [202]. The main use of this conventional myocutaneous flap, which includes significant parts of the rectus abdominis muscle, is in breast reconstruction, but it is also used for other regions of the body like the skull base, face, head and neck, trunk, and extremities [64, 126, 130, 204, 372, 537, 558, 560, 618]. Due to the dense anastomotic network between the deep inferior and superior epigastric vessels, which was extensively investigated using cadaver dissections and angiograms [368, 381], the perfusion of the flap is also reliable via the internal mammary vessels, making the pedicled transfer to defects of the breast or thorax possible. Undoubtedly, the most common design when using the rectus abdominis flap is the TRAM (transverse rectus abdominis muscle) flap with a horizontal orientation, using the deep inferior epigastric vessels as the pedicle and incorporating the skin from the entire lower abdomen, but many other variations of the skin paddle have been described, such as the standard longitudinal skin paddle positioned over the entire length of the rectus muscle, the oblique design, or the raising of the muscle without any skin. It has been estimated that the deep inferior epigastric artery may provide the largest territory of vascularized skin available in the human body [60]. The skin territory can be extended even more using the “supercharging” technique, if the most distal flap zone is additionally anastomosed to an artery or vein from the superficial system, thereby enhancing the blood supply to the flap provided by the deep inferior epigastric vessels [526].
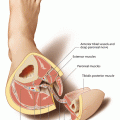
7.2 Anatomy
The rectus muscle, which originates from the cartilagineous parts of the sixth, seventh, and eighth ribs and the xiphoid process, is subdivided into four components by tendinous inscriptions. In a study of 54 cadavers, it was shown that almost always three tendinous intersections are present, and only in 2% of the cases four or even five intersections can be found [13].Together with the oblique muscles, the rectus abdominis muscle forms the anterior abdominal wall. At its insertion at the pubic bone, it is covered by the pyramidalis muscle in about 80% of the cases. The muscle is enveloped in the rectus sheath, which forms the muscle-free linea alba at the midline. This muscle fascia is anteriorly built by the tendinous fibers of the external oblique and the anterior parts of the internal oblique abdominis muscle. Posteriorly, the internal oblique together with the transversalis muscle forms the posterior rectus sheath cranial to the arcuate line. The arcuate line is located on each side with a cranial convex orientation between both anterior superior iliac spines and the midline. Caudal to this line, there is no posterior component of the rectus sheath. Therefore, it is of great importance to preserve the anterior rectus sheath below the arcuate line during flap raising to enable for direct and tension-free closure of the abdominal wall in this region. Also on both sides, the linea semilunaris marks the lateral border of the rectus muscle, reaching from the lower ribs to the pelvis. Here, the segmental nerves coming from the terminal branches of the lower six intercostal nerves penetrate the posterior rectus sheath about 3 cm medial to the linea semilunaris. They provide motor innervation to the rectus muscle as well as sensory innervation to the abdominal skin. These mixed nerves travel between the transverse and internal oblique muscles and then along the posterior surface of the rectus [128]. Because of their segmental configuration, stimulating one of these nerves will provoke a local contraction of the rectus muscle, giving the anatomic basis for facial reanimation [206, 561].
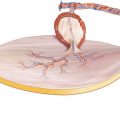
The blood supply to the abdominal skin and the rectus muscle, which according to the classification of Mathes and Nahai has a type III perfusion, derives from the deep inferior epigastric artery (DIEA) and two venae comitantes, running at the posterior surface of the muscle within the rectus sheath. Very reliably, the vessels anastomose with the deep superior epigastric vessels, which originally served as the flap pedicle; thus, the deep superior epigastric artery (DSEA) can also be used as the source vessel [192, 401]. According to the results of cadaver dissections and angiograms, a variety of branching patterns exist between the superior and inferior vessels, forming one (29%), two (57%), or three and even more (14%) anastomoses, all above the level of the umbilicus [387]. Whereas the deep superior epigastric vessels develop directly from the internal mammary vessels, the DIEA branches off from the external iliac artery opposite to the deep circumflex iliac artery (DCIA). It runs extraperitoneally in a medial and cranial direction, and after reaching the lateral rim of the rectus muscle, the pedicle penetrates the transversalis fascia about 3–4 cm caudal to the arcuate line [559]. The vessels enter the rectus muscle at its undersurface mostly at its middle (78%), seldom at its distal (17%) or proximal (5%) third [369]. Nevertheless, the approach to the pedicle has been discussed in the literature, because it can travel a long way up the lateral aspect of the muscle before arborizing. In these cases, an incision of the anterior rectus sheath medial to the linea semilunaris can potentially destroy the pedicle [561]; therefore, a transverse suprapubic incision has been proposed for exposing the deep inferior epigastric vessels [185, 537]. The inferior and the superior deep epigastric arteries can either not divide when traveling along the posterior surface of the muscle (type I), or they can arborize into two (type II) or even three branches (type III), giving the anatomic basis for a split muscle transfer [455, 561]. With a diameter of 2–4.5 mm, both artery and veins have a large caliber, and the extramuscular length of the pedicle is 10 cm in average [49]. In most cases, the veins unify before joining the external iliac vein. Boyd and coworkers have shown that the DIEA dominantly provides blood flow to the anterior abdominal wall. Apart from the continuation to the deep superior epigastric artery (DSEA), the vessel anastomoses cranially with branches of the intercostal arteries, and laterally and caudally, anastomoses exist with the superficial inferior epigastric artery (SIEA) and the DCIA via multiple choke arteries. It is well documented that the venous drainage is provided by the superficial and the deep system, communicating by way of veins accompanying the arterial musculocutaneous peforators [68]. The branches of the periumbilical perforators radiate toward any direction of the abdominal skin, thus giving the possibility to harvest flaps with virtually any orientation [559]. However, according to the anatomical work by Taylor, the dominant orientation of the subdermal vascular network is 45° from the horizontal [538, 559], which makes an oblique design of the skin paddle most reliable.
Based on the clinical experience in his first 16 unipedicled TRAM flaps, Hartrampf described four perfusion zones for the lower abdomen, which have been adopted for the free TRAM as well as for the free DIEAP flap. When using a transverse skin paddle extending to both sides of the abdomen, four flap zones are defined Zone I lies ipsilateral over the rectus muscle, zone III ipsilateral and lateral to the linea semilunaris; according to this, the location of zones II and IV are contralateral to the side of the pedicle. Contrast injection studies have confirmed the clinical experience that the blood supply of zone IV is the most tenuous [263]. Using laser-induced fluorescence of indocyanine green, Holm et al. found decreased or no perfusion in zone IV and best perfusion in zones I and III [223]. Moreover, selective dye injections have shown that zones I–III regularly are supplied by a single perforating vessel, whereas zone IV was only partially or not stained [212]. To improve blood supply in zone IV in large flaps with a horizontal design (TRAM), blood flow was augmented by anastomosing the distal end of the ipsilateral deep inferior epigastric vessels to a contralateral periumbilical perforator [414].
Only few anatomic variations have been described, nearly none of them affecting the possibility to harvest the flap. Boyd et al. reported about a possible variation of the deep inferior epigastric artery, originating from the external iliac artery in the form of a double trunk in about one third of the cases [60]. Whereas in an earlier study the deep superficial epigastric artery was reported to be missing in 3 out of 115 cadaver dissections, but in none of the cases affecting the deep inferior epigastric vessels [369], a complete bilateral absence of the deep inferior epigastric vessels was found in a clinical case, necessitating a change in the planned reconstruction [434]. Another variation concerned the origin and course of the perforator, which in seldom cases can run directly through the anterior rectus sheath, if the deep inferior epigastric pedicle travels along the superficial surface of the rectus muscle [170].
7.3 Advantages and Disadvantages
The rectus abdominis muscle is a very reliable and largely proven myocutaneous flap, which due to numerous different possible designs can be used for an extremely wide scope of reconstructive purposes all over the body. In large clinical series, the utility of this flap became evident very early particularly for the reconstruction of the female breast, and only marginal rates of flap losses have been reported [134, 144, 205, 373, 480, 481, 501]. Moreover, the pedicled transfer of the deep superior epigastric flap to the female breast or defects of the upper trunk enables reconstructions without performing microvascular anastomoses. Many authors also emphasize the usefulness of this flap in head and neck reconstruction, particularly in the region of the midface and skull base [256, 372, 558, 618]. However, there are potential disadvantages with the flap, all of them related to its donor site at the abdomen. Here, the often abundantly present fatty tissue can limit the use of the flap, if no primary thinning procedures are carried out. Moreover, the fatty tissue can develop necrosis due to critical blood supply and increased flap weight [390, 579]; this is especially the case in flaps weighing more than 1000 g, but having only one suitable perforator [390]; in these cases, fat necrosis can occur in up to 45% of the cases. A similar observation was also made by Baumann et al., who showed that the lowest risk of fat necrosis occurred in flaps with 3–5 perforators, whereas flaps having only 1–2 perforators originating from the deep inferior epigastric artery can develop fat necrosis in up to 25% [38]. Moreover, the selection of recipient vessels was shown to influence the frequency of fat necrosis with superior results when using the internal mammary artery instead of the thoracodorsal vessels [305]. Generally, when selecting the transverse design of the flap, the inclusion of zone IV should be avoided.
Besides these drawbacks concerning flap perfusion, flap raising can be associated with herniation of the anterior abdominal wall, if the rectus sheath has not been closed properly, independent of the amount of muscle that has been harvested. Already in the first years of its use, managing the donor-site defect has been an important issue, because despite careful direct closure, more than 40% developed weakness, bulging, or hernia [126, 304]; on the other hand, other authors report about less than 10% or even no herniation [561, 579]. Nevertheless, strategies have been developed to overcome this complication using a mesh for reconstructing the anterior rectus sheath [110, 125, 315, 514], leading to significant reduction or even complete avoidance of herniation. In a large series of patients with a mean follow-up of 4 years, Bucky and May [63] found a hernia rate of only 1.5% and the same incidence for mesh-related infection. Thus, the authors believed the use of the mesh allows for harvesting the whole rectus muscle, including all perforators to the skin. Another solution with only 8% of herniation was proposed by Kroll and Marchi [304], who preserved one fifth of the lateral muscle rim, allowing for a two-layer closure of the rectus sheath. Hartrampf and coworkers approximated the residual anterior fascial margins directly and could achieve less than 1% of abdominal wall laxity in their series of 300 patients; these results were confirmed by Urken et al. [561], who was following the same principle. Apart from avoiding bulging and herniation, also the motor function and strength of the abdominal wall can at least partially be maintained by reducing the amount of muscle which is included in the flap. Using myosonography, Seidenstücker et al. could prove a significantly better muscle function after raising the DIEAP instead of the TRAM flap [485], and this was confirmed by a prospective clinical evaluation of muscle strength 1 year postoperatively. Selber and coworkers found significantly worse muscle function after raising bilateral TRAM flaps compared to DIEAP flaps, and they concluded that reduction of abdominal wall strength depends on the degree of surgical muscle sacrifice [489]. However, in a systemic review, Atisha and Alderman could identify 20 studies on abdominal function after either pedicled or free TRAM or DIEAP flap reconstructions and found that the objectively measured deficits in flexion abilities, which were up to 53% in the TRAM group, do not subjectively translate to detriments in the performance of activities of daily living [20].
7.4 Flap Raising
7.4.1 Patient Positioning
Patients with a history of major abdominal surgery or having scars across the operation field must be excluded from flap raising. Flap raising is only possible in supine position, and the whole abdomen is prepped across the midline, including both lower rib arches and both sides of the upper thigh as well as the pubic area. Because of their constant anatomy, no preoperative diagnostic measures concerning the deep inferior epigastric vessels are necessary. Raising of the flap with a vertical design will include more than one perforator, which therefore do not have to be mapped preoperatively.
7.4.2 Flap Design
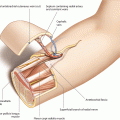
A vertical flap design is outlined, with the skin paddle covering the rectus muscle unilaterally. The vertical borders are positioned above the linea alba medially and the linea semilunaris laterally. The linea semilunaris can be expected 7–10 cm lateral to the linea alba. The proximal and distal flap poles can reach up to the ribs and down to the pubic area, respectively. The center of the skin paddle must always be positioned at the level of the umbilicus. Whereas the upper and lower poles can be separated from the muscle without endangering the blood supply, the central portion may never be detached from the anterior rectus sheath, because here, the myocutaneous perforators of the deep epigastric vessels travel to the skin. The maximal width of the flap for primary closure should be estimated according to the individual body shape and skin laxity but is normally possible up to 12 cm (◘ Figs. 7.1 and 7.2).
Definition of the Superior Pole
Forming the superior pole as a triangle, skin and fatty tissue are incised down to the anterior rectus sheath. Keeping a distance from the central flap area around the umbilicus, the lipocutaneous portion of the upper pole is uplifted and peeled away from the deep fascia. The deep fascia, representing the anterior rectus sheath, is incised 5–7 cm cranial to the umbilicus across the whole width of the flap without incising the muscle, which readily becomes visible (◘ Fig. 7.3).
Identification of the Deep Epigastric Vessels
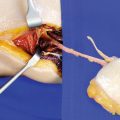
Carefully, the vertically oriented muscle fibers of the superior pole are horizontally incised with a scalpel, but also using blunt dissection with scissors, so that the deep inferior epigastric vessels which normally run along the deep surface of the muscle can be identified. In this case, only a lateral branch is present, but a medial or even a third branch can often be found (◘ Fig. 7.4).
Definition of the Inferior Pole
A line is drawn connecting both anterior superior iliac spines which defines the caudal incision of the anterior rectus sheath. Below this line, there is no posterior rectus sheath. Therefore, the anterior rectus sheath caudal to the arcuate line may never be included into the flap. If it is preserved, it guarantees for a tight linear closure of the abdominal wall.
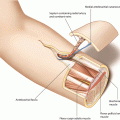
Caudal to this line, a triangular incision of skin and fat is carried out down to the deep fascia, and the lipocutaneous inferior pole of the flap is detached from the fascia until the level of the arcuate line has been reached. The position of the arcuate line is represented by the interconnection of both iliac spines (◘ Fig. 7.5).
Opening of the Anterior Rectus Sheath
Identification of the Inferior Muscle Segment
After folding away both sides of the split anterior rectus sheath, the caudal portion of the rectus muscle becomes visible. Because the deep inferior epigastric vessels can enter the muscle at different levels, no incision of the muscle is done at this step as it might damage the pedicle (◘ Fig. 7.8).
Circumscribing the Skin Island
Using a scalpel, the whole skin paddle is now circumscribed without opening the deep fascia. By doing this, the central portion of the flap, containing the myocutaneous perforators, must be left firmly attached to the fascia (◘ Fig. 7.9).