and Frank Hölzle2
(1)
Department of Oral and Maxillofacial Surgery, Klinikum rechts der Isar, Technische Universität Munich, Munich, Germany
(2)
Department of Oral and Maxillofacial Surgery, University Hospital of RWTH Aachen University, Aachen, Germany
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-319-53670-5_14) contains supplementary material, which is available to authorized users.
14.1 Development and Indications
Based on the concept of myocutaneous flaps which was introduced in the late 1970s, the posterior aspect of the calf has been used as a donor site for local musculocutaneous flaps from the gastrocnemius muscle for defect coverage at the lower leg and around the knee joint [120, 143, 355, 356]. Before that, a successful local defect coverage with the lateral gastrocnemius muscle head, which was supplied by a direct branch of the popliteal artery, was reported by Pers and Medgyesi in 1973. In these first descriptions, the authors highlighted the excellent blood supply to both heads of the gastrocnemius muscle from either the medial or lateral sural artery, allowing for wide rotation of the muscle including skin to reach defects from above the patella down to the upper portion of the lower tibia and anterior lower leg. Because a substantial amount of tissue could safely be transposed into defects from a well-perfused donor site of the same leg, this technique immediately started to replace the formerly used crossleg flaps in lower-leg reconstructions. Consecutively, the muscle was transferred to the distal third of the lower leg as a free flap by Salibian et al. and Keller et al., thereby using the lesser saphenous vein as an interposition graft to elongate the sural artery [265, 461]. According to Matthes and Nahai, the muscle has a type I pattern of circulation [350].
Before providing evidence that myocutaneous vessels allowed for raising of perforator flaps from the deep vascular system, several anatomical studies and case series focused on the possibility to elevate fasciocutaneous flaps at the posterior lower leg based on a direct cutaneous branch of the medial or lateral sural artery. One of the first studies was performed by Haertsch, who found a dominant superficial median artery using intra-arterial dye injections [181]. At the same time, Ponten described a cutaneous artery running along the sural nerve which then was known as the superficial sural artery [417]. Although this vessel had the potential to perfuse fasciocutaneous flaps at the posterior lower leg, it showed great variations with regard to its location, origin, and size. Further anatomical investigations confirmed that the superficial arteries in this region can have diameters of 0.5 mm only or be missing completely [137, 467, 594]. Whereas pedicled cutaneous flaps from the posterior calf have been described as reliable for local defect coverage, there were only very few reports with small numbers of patients about microvascular transplantations on the superficial vascular system [467, 575, 594]. Therefore, the posterior lower leg was considered unreliable for harvesting free cutaneous flaps until the principle of perforator flap raising was applied to this donor site.
Although it was evident from the early reports that the skin overlying the gastrocnemius muscle was perfused via myocutaneous vessels, it took more than 20 years until the sural artery perforator flap was described, first as a local flap [380], then as a free flap [72]. This long time period was even more surprising, because, based on cadaver dissections, a detailed description of a gastrocnemius musculocutaneous perforator was given by Taylor and Daniel as early as in 1975 [538]. In this publication, the authors proposed raising a skin flap from the posterior calf on a single vessel perforating the gastrocnemius muscle. By intramuscular dissection, the authors could follow this perforator to its origin from the sural vessels, representing the flap pedicle. With this procedure, the raising of a perforator flap had already been described even before the term “perforator flap” became established during the 1990s. In their series, Cavadas et al. used this flap as a free flap in five cases for lower-limb reconstruction. Subsequently, detailed anatomic investigations were carried out to further define number, localization, and reliability of the perforators originating from the deep sural artery system, and it was shown that a dominant myocutaneous perforator was present at the medial head of the gastrocnemius muscle in more than 90% of the cases [191, 194, 258, 272]. Consequently, the medial sural artery perforator flap was used for many reconstructive purposes. Chen [86] and Kao [258, 259] were the first to use the medial sural perforator flap for defect coverage in the oral cavity. In a prospective study, this flap was found to have the same success rate and reconstructive quality as the radial forearm flap by leaving lesser donor-site morbidity [259]. Other authors confirmed the excellent suitability of this flap for many applications in the head and neck [75, 232, 404, 508] including reconstruction of the tongue [211, 632] and for defect coverage in the extremities [193, 232, 253, 633].
14.2 Anatomy
Similar to other donor sites in the human body, the skin at the posterior calf is perfused via a superficial and deep vascular system. Apart from this, various superficial sural vessels acting as the vascular pedicle for fasciocutaneous flaps and myocutaneous perforators from the deep medial or lateral sural vessels provide an important and reliable source for the cutaneous blood supply of the posterior lower leg. According to Hallock, an inverse relationship exists between these two sources, so that in case of very small-caliber or even missing perforators, fasciocutaneous flaps from the superficial sural system have to be raised instead [193].
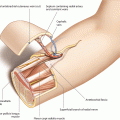
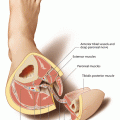
The medial sural artery perforator flap is a relatively thin cutaneous flap which is designed over the medial head of the gastrocnemius muscle. Its blood supply comes from at least one, preferably the dominant myocutaneous perforator, which develops from the medial sural artery. This vessel is usually accompanied by two venae comitantes (2–4 mm) and has its origin either from a common sural trunk, which also gives rise to the lateral sural artery, or directly from a popliteal artery [50, 108]; additionally, the greater saphenous vein can also be used as a drainage [191]. In many cases, more than one medial sural artery can be found [50]. During its course along medial gastrocnemius head, one or more perforating vessels are given off which pierce the deep fascia to enter the subcutaneous vascular plexus of the posterior medial calf. Although according to injection studies the width of the flap can be more than 8 cm in average, the size of the medial sural artery skin paddle is predominantly limited by the ability to close the donor site directly; this is usually possible if the flap width does not exceed 6–7 cm. The detailed anatomy of the vascular pedicle with the exact location, size, and number of perforators to the skin was the subject of several anatomical studies. The first detailed description was published by Cavadas et al., who found a mean of 2.2 perforators over the medial gastrocnemius muscle, clustered about 12 (proximal perforator) and 17 cm from the popliteal crease. With intramuscular dissections, Wong et al. found that the medial sural artery divides into two branches, each of them giving rise to myocutaneous perforators along the medial head of the gastrocnemius muscle. They found between two and six perforators being located between 6 and 22.5 cm distal from the popliteal crease. Perforators with origin from the medial of the two branches were generally larger. In most cases, a large, proximal perforator with a straight intramuscular course could be identified 10 cm distal to the flexor crease and 2 cm medial to the posterior midline of the calf on average, and a distal one about 16 cm away from the flexor crease [614]. Whereas the intramuscular course of the perforator is mostly short, it tends to have a long extramuscular and subfascial course before penetrating the deep fascia. Therefore, Hallock suggests to expose the perforator in the subfascial plane and to accordingly adapt the position of the skin paddle [194]. In anatomic dissections, Okamoto et al. confirmed that the flap also can be raised safely in Asians. They found no perforator higher than 5 cm or lower than 17.5 cm from the popliteal crease. In 36%, two perforators were found, the proximal one with a mean distance of 9.6 and 12.8 cm for the distal one, respectively. All perforators were in an area between 0.5 and 4.5 cm from the midline. Another anatomic study was carried out by Kosutic and coworkers who pointed out that raising a perforator flap from the lateral sural artery is less reliable. They only found perforators in 31% of the lateral sural arteries, of which only in 9.4% had a dominant vessel. On the other hand, medial sural artery perforators were present in 94% of all legs, 37% of them being dominant vessels. To estimate the possible size of the skin paddle, Altaf et al. performed injection studies and investigated the stained skin area and found that the possible size of the medial sural artery perforator flap can be 8 × 13 cm on average. According to their studies, the medial sural artery has an external diameter of 3 mm at its origin and gives off two perforators to the skin, the larger one with a diameter of 0.9 mm, giving the possibility to raise a pedicle of up to 18 cm. Dye injections were also performed by Tione et al. on 20 fresh lower limbs. According to their results, flaps can be harvested with a size of 13 × 8 cm on average, nourished by a mean of 1.9 perforators per flap. This group also investigated the intramuscular course of the perforators and found that about two third of the perforators originated from the lateral branch of the medial sural artery and one third from the medial one [542]. Based on cadaver studies, angiographies and 3D CT scans, Kim et al. found that the main perforators of the medial sural artery were located on a line drawn from the midpoint of the popliteal crease to the midpoint of the medial malleolus. On this line, the first perforator was located within the distal half circle drawn around a point approximately 8 cm, and the second one 15 cm away from the midpoint of the popliteal crease [272]. Similar results were obtained by the group of Kao, who evaluated number, location, and topographic patterns of the perforators. They found 2.7 perforators on average with a mean distance of a sizable perforator perpendicular to the flexion crease of about 11 cm; most sizable perforators entered the medial gastrocnemius muscle at one-fifth to one-third of the lower leg length [258]. All authors conclude that at least one sizeable perforator with a diameter of at least 1 mm could be found arising from either the medial or lateral branch of the medial sural artery, most of them being located about 10 and 16 cm distal to the popliteal crease, respectively [614]. Their distance from the posterior midline was between 0.5 and 4 cm. Thus, pedicle lengths with an average of 10–12 cm (range 5–18 cm) can be achieved, consisting of sizeable vessels (artery 1–2 mm, veins 2–4 mm), which allow for reliable anastomoses.
14.3 Advantages and Disadvantages
For many reasons, the MSAP flap has become one of the most frequently used perforator flaps for flat and moderate-sized defects. After its first descriptions as a pedicled [380] or free flap for defect coverage in the lower limb [72], the flap has been used for many other applications due to its thinness and its long and high-caliber vascular pedicle. The flap thickness was reported to range from 4 [549] to 10 mm [93, 404] only, and, depending on the location of the perforators, the flap pedicles varied between 5 [493] and 14 cm [271, 493], or even 18 cm [10] mostly with an average length of 10–12 cm [75, 211, 404, 549, 632]. Owing to these favorable anatomical properties, except from defect coverage at the distal limb [576], hand [613, 633], and foot [85, 271], the MSAP flap has also become popular in head and neck [93, 258, 404, 493, 508] and intraoral [75, 211] reconstruction. In a prospective clinical study, Kao et al. compared the use of the MSAP flap with the radial forearm flap and found it to be a good alternative in smaller defects with a lower donor-site morbidity than the RFF [258]. A similar study was performed by Song and coworkers, with the same conclusion: they also prefer the MSAP flap to the RFF due to its better appearance and lower morbidity of the donor site [508], as long as direct closure can be achieved. According to literature reports, this is possible up to a flap width of about 6–7 cm. An even larger comparative study was carried out by Zhao et al., who additionally compared the MSAP flap to the ALT flap; they found that the thickness and function of the MSAP flap were similar to those of the RFF but better than those of the ALT flaps; in appearance and function of the donor site, the medial sural perforator flaps were similar to the ALT but better than the RFF [632]. Although the donor-site morbidity was generally reported to be low by nearly all authors, hypertrophic scars, itching, pigmentation, numbness, paresthesia or muscle weakness can occur after raising the flap [194, 614]. Moreover, the donor site can be inacceptable for women, and for aesthetic reasons, skin grafts have to be avoided in any case [193].
As with any other perforator flap, one disadvantage of the MSAP flap compared to conventional flaps is its dependence on at least one perforating vessel, the location of which can vary considerably. Despite the anatomical reports describing most perforators to be located in the area between 10 and 16 cm distal to the popliteal crease [614], their individual location cannot be predicted exactly. Therefore, measures to prepare for flap raising are generally emphasized. Although this method may be imprecise [267], the most common instrument for localizing the perforators is a hand-held Doppler, which has proven to be of good reliability for preoperative mapping [428, 632, 633], and even better results can be achieved with the two-dimensional color duplex ultrasound [301]. Using preoperative CT angiography, Higueraz et al. were able to visualize perforators in the lower limb with high specificity and reliability [220]. They reported to have hardly any flap failures after angio-CT, and operative findings correlated perfectly with preoperative imaging. A similar result was reported by He et al., who could not reveal significant differences in the location of perforators between preoperative CT angiograms and intraoperative findings [211]. To decide about the better suitability of either the legs for flap raising, Dusseldorp et al. suggested a routine use of CT angiography to help determine the branching pattern and intramuscular course of the pedicle [129]. An even more sophisticated approach to localize the perforator was proposed by Shen et al., who used endoscopy to confirm the localization of the MSA perforator [493].
Although localizing the perforator can be mastered by careful preoperative mapping and forward-looking dissection according to the above-described anatomy, the MSAP flap can have a critical or even insufficient venous drainage if anastomoses are routinely performed to one of the deep veins only. Because the skin of the lower leg is additionally drained by various superficial veins, mostly the lesser saphenous vein or another unnamed venous branch of the superficial system can be the main venous drainage of the flap, especially if the perforator is located close to the posterior midline [194, 428, 549]. In these cases, one of the two veins which accompany the perforator travels toward a vein of the flap’s superficial system. Therefore, when circumscribing the skin paddle and dissecting the flap, any bigger vein running across the flap’s longitudinal axis should be preserved and checked for its venous outflow. If there is substantial venous backflow from the superficial vein, it is recommended to use this vein for an additional venous anastomosis. This procedure of augmentation of the flap’s venous outflow has already been established in DIEAP flaps because of its proven efficacy and reduction of flap morbidity [407, 615]. Finally, direct manipulation, especially to small perforators during flap raising, can easily cause vasospasm [319], making topical application of vasodilators like papaverine or lidocaine necessary.
14.4 Flap Raising
14.4.1 Patient Positioning
Because of its location at the posterior calf, the MSAP flap can easily be raised having the patient in prone position, but flap elevation is also possible if the patient is in supine position without problems. The leg is then abduced in the hip and bent in the knee joint, and rotated outwards, if possible. Preoperative mapping of the perforators using a Doppler is highly recommended in this situation which best marks the location of the vessels at the skin, which is always a little dependent on skin laxity and sagging. Careful auscultation should be performed in an area between about 5 and 18 cm distal to flexion crease of the knee joint along a line drawn between midpoint of the joint and the medial ankle. Because perforators can arise from a lateral or medial branch of the medial sural artery, the area of auscultation should cover at least 2–3 cm at either side of this line. It is most likely that two perforator signals are obtained, the proximal about 10 cm, and the distal about 16 cm away from the flexion crease. After exact mapping of the perforator(s), circular disinfection is performed from above the knee joint down to the toes. No tourniquet is used to allow for visualization of the perforator’s pulsation and to check the backflow of a superficial vein, which might be included. Although flap raising is possible from both sides of the donor leg, the surgeon has a better view to the operating field from the contralateral position.
14.4.2 Flap Design
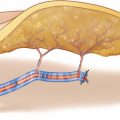
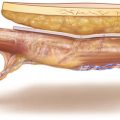
As the perfusion of the skin paddle is dependent on the chosen perforator(s), position and design of the flap cannot be determined before a sizeable perforator of at least 1 mm with good pulsation has been exposed. The longitudinal axis of the flap is designed parallel to the line connecting midpoint of the knee joint and medial ankle, bringing the perforator in a central or slightly proximal eccentric position when outlining the flap borders. The length of the flap can be at least 10 cm when using one or up to 15 cm, if a second perforator with a more distal location is used. Although the vascular territory allows for raising flaps more than 8 cm wide, the width of the MSAP flap should not exceed 6 cm for primary closure. The posterior midline of the calf is marked, and the skin incision is outlined along the posterior midline, keeping a distance of at least 2 cm to the marked Doppler signal (◘ Figs. 14.1, 14.2 and 14.3).
Incision of Skin and Deep Fascia
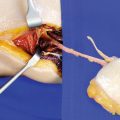
The skin is incised along the posterior midline, keeping a safety distance of 2 cm to presumed position of the perforator according to the Doppler signal. In most cases, the perforator pierces the deep fascia close to the connecting line between midpoint of the knee joint and medial ankle. During transection of the fatty tissue, care should be taken not to injure the perforator which might run across the dissection plane. If present, subcutaneous veins with diameters of more than about 2 mm should be preserved for a possible later use as additional drainage. The deep fascia is incised longitudinally like the skin, and the medial head of the gastrocnemius muscle is exposed. The deep fascia is elevated by a hook, and the space between the fascia and the muscle is scrutinized for the presence of a sizeable myocutaneous perforator. A perforator and two venae comitantes become visible at the surface of the muscle, traveling toward the deep fascia within a thin gliding layer of fatty tissue. Note how easily the perforator can be overseen, neglected, or even destroyed during the dissection. Owing to their fine structure and small caliber, these perforators must be handled with great care at every step to follow (◘ Fig. 14.4).
Incision of Muscle
The deep fascia is carefully reflected, and the muscle is incised distal to the perforator, keeping a safety distance of 1–2 cm to the point where the perforator penetrates the muscle surface. The perforator is not exposed along its course through the muscle at this time (◘ Fig. 14.5).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree