and Frank Hölzle2
(1)
Department of Oral and Maxillofacial Surgery, Klinikum rechts der Isar, Technische Universität Munich, Munich, Germany
(2)
Department of Oral and Maxillofacial Surgery, University Hospital of RWTH Aachen University, Aachen, Germany
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-319-53670-5_13) contains supplementary material, which is available to authorized users.
13.1 Development and Indications
As a consequence of the well-reported morbidity which can be associated with harvesting the conventional rectus abdominis flap, strategies to reduce the amount of muscle have been developed, first resulting in the muscle-sparing flap-raising technique and finally evolving to the perforator flap principle. Very early, the significance of the perforating vessels of the rectus muscle to perfuse the abdominal skin was emphasized by numerous authors [225, 441, 524], but it took until 1989 that Koshima and Soeda reported the first clinical application of the inferior epigastric artery perforator flap [296]. With the introduction of this important modification, complications like weakness of the abdominal wall with herniation or bulging were largely avoided. This made the several previously described techniques for closure of the muscle defect, like direct approximation [204, 560], using turnover flaps from the contralateral rectus sheath [364], or inserting synthetic meshwork material into the created muscle defect [126, 315] unnecessary. After identifying a strong perforating vessel, Koshima left all the rectus muscle tissue intact when he followed this perforator to the deep epigastric system by gently spreading the muscle fibers. This procedure first met all criteria of raising perforator flaps according to the current definition [296]. The attractiveness of this procedure was further increased when the same author introduced the technique of primary flap thinning, which became possible with the targeted dissection and preservation of the perforator, allowing for safe removal of all superfluous fatty tissue [293]. Like with the conventional transverse rectus abdominis muscle (TRAM) or rectus abdominis flaps, these deep inferior epigastric artery perforator (DIEAP) flaps were still anastomosed to the deep inferior epigastric vessels [9, 161]. Allen and Treece were among the first to use this flap for breast reconstruction and confirmed its advantages of decreasing the possibility of ventral hernia or muscle weakness [9]. If there is a need to extend the flap to the contralateral side of the abdomen, which is regularly the case in breast reconstruction, a perforator from the contralateral side can additionally be anastomosed to the end of the ipsilateral deep inferior epigastric vessels, thus increasing the safety of blood supply (supercharging technique) [161, 414]. This technique is also useful in the presence of vertical scars which can reduce blood circulation contralateral to the side of anastomoses [478]. A similar technique was also described by Blondeel and Boeckx, who additionally used contralateral perforators, which they anastomosed to the internal thoracic vessels instead of using the ipsilateral main vessels of the flap alone [47]. The first anastomosis of a deep inferior epigastric perforator flap exclusively to its perforating vessels was again performed by Koshima et al., describing this technique as supramicrosurgery [290]. Moreover, they recognized another significant advantage of perforator flaps, namely, the possibility of primary radical flap thinning. Indications of those super-thin perforator flaps with a short pedicle were superficial skin defects of the lower extremities, scalp, abdomen, or head and neck [290, 487, 527, 622]. Meanwhile, many comparative studies have shown that DIEAP flaps are associated with higher patient satisfaction, reduced abdominal bulge, but comparable aesthetic results like TRAM flaps [44, 379, 395].
13.2 Anatomy
Anatomy and blood flow of the DIEAP were investigated in detail by Blondeel and co-workers [47, 51, 161]. They could show that in about 75%, the DIEAP divides into two branches, the lateral of which being the dominant vessel (54%). The medial branch has a larger caliber in 18%, but blood flow through the perforators is lowest in these cases. According to their results, in 28%, the DIEAP runs as a singular vessel with small, multiple side branches centrally at the posterior surface of the muscle. Other clinical and anatomical findings have shown that the pedicle can also divide into three branches, which happens in 14% of all cases, making it possible to split the muscle without endangering the blood supply of each segment [455, 559, 564]. Thus, perforators can penetrate the muscle laterally, medially, or at its center. The highest density of cutaneous perforators from the DIEAP can be found around the umbilicus [60]. A detailed anatomical study by Blondeel et al. revealed between two and eight perforators on each side of the muscle, all having a diameter of at least 0.5 mm. Most of these vessels were located in an area of 1–6 cm lateral and between 2 cm cranial and 6 cm caudal to the umbilicus [46]. Vandevoort et al. described the topography of the perforators and their course through the rectus muscle in a series of 100 clinical cases. In 65%, they found a short direct course through the muscle, whereas in 16%, the perforators pierced through a tendinous inscription. A long intramuscular course of the perforators was found only in 9%, but flap raising was most difficult in these cases [569]. Similar results were reported by Munholz et al., who found 34% of the perforators located in a lateral row along the rectus muscle, having a rectilinear course through the muscle in 79%. In the medial row, only 18.2% showed this configuration [391]. In their anatomic investigation on 329 DIEAP perforators, Kikuchi et al. differentiated between “large” perforators having a diameter of at least 1 mm [201], suitable perforators that run parallel to the muscle fibers [367], and “ideal” perforators which unify both features [111]. These ideal perforators were located at the intermediate third of the rectus muscle, 10–30 mm lateral to the umbilicus [269]. With a theoretical model, Patel and Keller described the arterial flow in the DIEAP flap perforators and concluded that the flap is best perfused by one single but large-diameter vessel. Although inclusion of additional perforators will increase the flow, the authors did not consider this additional trauma of dissection and increased operating time as beneficial to the patient [413]. By comparing flap outcomes by the number of perforators, Grover and coworkers could not find a significant influence of this parameter on flap survival; however, the rate of flap necrosis was significantly higher in DIEAP flaps based on a single perforator only [176].
Using a fluorescent perfusion technique, Holm and coworkers demonstrated that blood flow from the pedicle to TRAM flaps, covering all four perfusion zones as defined by Hartrampf, first travels to the ipsilateral side before crossing the midline. The authors also observed a different pattern of perfusion depending on the existence of lateral or medial row perforators [223]. This was confirmed by Wong et al., who used three- and four-dimensional computed tomographic angiography to reappraise the zones of vascularity. They found a mean vascular territory for a medial row perforator DIEAP flap of nearly 300 cm2, whereas from the lateral row, less than 200 cm2 of skin was perfused. Moreover, they found that medial row perforators conform to the Hartrampf zones and that lateral perforators follow the observations made by Holm, who according to their results proposed to reverse zones II and III. These perforators only rarely provide perfusion crossing the midline [609].
The importance to select a large perforator with a palpable pulse and a diameter of at least 1 mm was stressed by Kroll, who found a high rate of fat necrosis in his first DIEAP flaps performed in unselected cases, often having small perforating vessels [303]. On the other hand, it could be shown that blood flow in the perforator is higher than in the source artery and increases with time [148]. When choosing a long-pedicle perforator flap, the deep superior epigastric artery (DSEA) can also be selected as the vascular pedicle instead of the DIEA. This was confirmed in an anatomical and clinical study of Mah et al., who used a deep superior epigastric artery perforator flap with a long pedicle for sternal reconstruction [329].
13.3 Advantages and Disadvantages
Since its first descriptions, the DIEAP flap has proved to be a valuable and safe alternative to the conventional TRAM flap, as long as the dissection and anastomosis of the perforators can technically be mastered by the surgeon. Thus, the decision on whether to raise a conventional rectus abdominis or TRAM flap or to choose a “true” perforator flap with a short pedicle is mainly dependent on microsurgical skills. But apart from this, also other factors influence the type of flap to be raised: Since the main advantage of the DIEAP is the targeted dissection of the perforator and the possibility to leave the rectus muscle completely intact in patients needing a guaranteed integrity of the muscular abdominal wall, the little higher risk of vascular complications which is associated with a short and small-caliber pedicle seems to be justified. Blondeel et al. reported about a higher number of venous congestion in zone IV when using the DIEAP flap compared to the conventional TRAM flap, which they explained with a dominant venous drainage of the superficial venous system within this area [45]. Therefore, if a strong superficial epigastric vein is present, they proposed to perform an additional venous anastomosis. This additional venous drainage also was performed by Tran et al. in five of their 100 consecutive DIEAP flaps with favorable results [550]. Venous drainage of zones I–III is always guaranteed by the deep inferior epigastric vessels alone [45, 68]. If the comitant veins of the primary anastomosis are found to be widely thrombosed, a venous bypass using the ipsilateral basilica vein to the superficial inferior epigastric vein is possible [179]. A similar method as a solution for venous congestions can be a venous bypass to any chest wall vein, even if the primary venous anastomosis is still patent [550]. Also, a reverse-flow venous anastomosis can be established between the superficial and the deep inferior epigastric veins [321]. On the other hand, Cheng et al. did only in one out of 74 flaps find necrosis of zone IV, but fat necrosis in 13.5% [90]. Using computed tomographic angiography on DIEAP flaps from fresh cadavers, Schaverien et al. could show that zone IV was not perfused following injection of the lateral row perforators, so that a medial row perforator should be selected if zone IV perfusion is required [473]. Like with the vastus lateralis perforator flap, measures for preoperative imaging were proposed for the planning of perforator flaps. Whereas Rozen et al. used CT angiography with which they found a good correlation of imaging and the operative findings [451], Blondeel and coworkers prefer using the color duplex scanning method [46].
As an alternative to the commonly used horizontal design of the skin paddle, a vertical orientation of the DIEAP flap was proposed, especially in patients with midline abdominal scars which he extended up to 13 cm in length [465, 527]. To speed up flap raising and to reduce the difficulty in perforator identification, an antegrade pedicle dissection is proposed instead of the commonly used retrograde dissection, which often leads to prolonged operating time [142].
By including the sensate branch of the segmental nerve which runs along with the perforating vessel, sensory nerve repair in perforator flaps for autologous breast reconstruction is possible. When performing a nerve anastomosis, a better response was given to pressure and thermal stimuli compared to flaps without nerve repair [48].
A critical review of perioperative complications was given by Hofer et al., who initially found a high complication rate in the first 30 DIEAP flaps of 40%. As a consequence of continuous learning, perioperative complications in the next 144 flaps were reduced to 13% with only one complete flap loss [222].
Donor-site complications of the DIEAP flap are reported to be lower compared to the TRAM flap [44, 169, 195], but can be higher in women with preexisting scars [412]. Also in overweight or obese women undergoing breast reconstruction with the DIEAP flap, the occurrence of abdominal wall laxity, herniation, or bulging was uncommon and not statistically different from normal-weight patients [158]. On the other hand, Vyas et al. found obesity to be a significant risk factor for donor-site complications, whereas in their study, no significant increase in donor-site complications was associated with prior abdominal operations [574]. Bajaj et al. compared donor-site-related complications between free DIEAP and muscle-sparing TRAM flaps and found no differences. Thus, they advocated using the most expeditious and reliable flap based on the vascular anatomy of the DIEAP system [28]. In a direct comparison of unilateral DIEAP versus TRAM flaps, Schaverien et al. found no differences in terms of subjective functional limitations of daily activities between both groups [474]. Abdominal strength was objectively determined after the raising of muscle-sparing TRAM- or DIEAP flaps by Bonde and coworkers. They found a clinical small, but significant advantage in the DIEAP group when measuring the eccentric muscle strength [53]. Despite the obvious advantages of this further evolution in raising flaps from the anterior abdominal wall, studies comparing conventional TRAM with perforator-based DIEAP flaps have evaluated no significant differences except for higher costs for hospitalization in patients having obtained DIEAP flaps [528].
To minimize abdominal donor-site morbidity, flaps based at the superficial vascular system (superficial inferior epigastric artery [SIEA]) were proposed instead of DIEAP flaps or muscle-sparing TRAM flaps, since harvesting flaps at the superficial inferior epigastric artery proved to be the least invasive method with no muscle injury at all [612]. Muscle injury can also be reduced if damage to motor nerves is avoided during perforator dissection. In an anatomical study, Rozen et al. found four to seven nerve branches entering the rectus muscle from the lateral border or posterior surface, running with the most lateral branch of the DIEA and its perforators. Thus, the medial row perforators which are not related to those motor nerves are ideal for inclusion in the flap. Using intraoperative stimulation, they also found small nerves innervating small longitudinal stripes of rectus muscle, which may be sacrificed without functional detriment due to overlapping innervation from adjacent nerves. However, large nerves at the level of the arcuate line innervate the entire width of the rectus muscle and may contribute to donor-site morbidity if sacrificed [452]. The appearance of the donor site after DIEAP flap harvesting can be improved if musculofascial plication techniques and abdominoplasties are carried out immediately after flap harvesting [392].
13.4 Flap Raising
13.4.1 Patient Positioning
The patient is positioned as described above. As it is proposed for other perforator flaps, localization of the perforators around the umbilicus using Doppler can be carried out before surgery to facilitate identification of a suitable perforator. This is especially the case if a small flap is planned with the perforator in the center of the skin island.
13.4.2 Flap Design
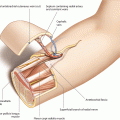
Because the localization of the skin paddle of the DIEAP flap is completely dependent on the perforator selected for flap raising, the definite design of the flap cannot be determined before a useful perforator showing a strong pulsation has been exposed. The most suitable perforators normally can be found at the intermediate third of the rectus muscle, 10–30 mm lateral to the umbilicus. Since the perforators feed a dense subcutaneous network of vessels, which communicate through a system of choke arteries, the skin paddle can be oriented in any direction, as long as a strong periumbilical perforator is included. To increase the safety of the flap, it should not be extended to zone IV, which means lateral to the opposite linea semilunaris. On the ipsilateral side, the skin paddle can reach up to the costal margin, if an oblique flap design is selected. This design corresponds to the dominant orientation of the subcutaneous network. Depending on the laxity of the abdominal skin and the orientation of the flap axis, the width of the flap can be 10 cm or more (◘ Figs. 13.1, 13.2, 13.3, and 13.4).
Incision of Skin Until Muscle Fascia
The skin is incised down to the anterior rectus sheath, including at least one strong perforator according to the preoperative mapping using the Doppler. The medial edge of the flap is located at the midline, and the lateral flap pole can reach the costal arch (◘ Fig. 13.5).
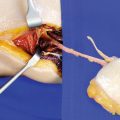
A Perforator Is Visualized Close to the Umbilicus
Starting laterally, the skin paddle is elevated at the level above the fascia, and dissection is carried out toward the semilunaris line. A few centimeters medially, the first perforator becomes visible, and its strength and pulse are observed (◘ Fig. 13.6).
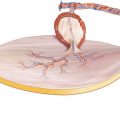
Two Perforators Piercing Through the Anterior Rectus Sheath Close to the Umbilicus Are Exposed
The dissection of the skin paddle is continued superficial to the anterior rectus sheath, and a second strong perforator is exposed close to the umbilicus. The skin paddle is completely mobilized around these two perforators (◘ Fig. 13.7).
Deep Inferior Epigastric Artery Becomes Indirectly Visible by Elevating the Flap
The skin paddle is carefully elevated, and the anterior rectus sheath is completely exposed to prepare for further dissection of the vessels. In slim patients, the DIEA becomes indirectly visible by slightly lifting the flap upward (◘ Fig. 13.8).
Dissection of Perforators Through the Tendinous Fibers of the Rectus Muscle
The anterior rectus sheath is carefully incised around the first perforator, and the accompanying sensible nerve, if present, is transected (◘ Fig. 13.9).
Exposure of the Deep Inferior Epigastric Artery
Side Branches to the Muscle Become Visible When Uplifting the Looped Deep Inferior Epigastric Artery
The DIEA is circumferentially dissected, and a loop is placed around the vessel. Side branches to the rectus muscle are clipped or cauterized (◘ Fig. 13.12).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree