and Frank Hölzle2
(1)
Department of Oral and Maxillofacial Surgery, Klinikum rechts der Isar, Technische Universität Munich, Munich, Germany
(2)
Department of Oral and Maxillofacial Surgery, University Hospital of RWTH Aachen University, Aachen, Germany
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-3-319-53670-5_11) contains supplementary material, which is available to authorized users.
11.1 General Introduction
With the increasing knowledge about the vascular anatomy and the blood supply of the skin and the refinement of surgical skills and instruments, sophistication of free tissue transfer could further develop. The first precise descriptions about the structure of the integument and its vessels were given by Spateholz in 1893 [515], who even then made a distinction between direct and indirect cutaneous vessels (perforators), branching off from a source vessel. A detailed analysis of the fine blood vessels of the skin was already published some years before by Manchot in 1889 [332]. He introduced vascular territories of the individual skin vessels, which later were further objectified by radiographic studies using lead oxide tincture injections [49]. On the basis of this knowledge, Esser was the first to build island flaps in which only the fine cutaneous perforators were preserved [49].Whereas in times when cutaneous flaps were raised without the knowledge of the specific anatomy of the skin as random pattern flaps, the vessel course at the skin was taken into consideration by Mc Gregor, who described the first axial pattern skin flaps [49, 360]. Moreover, the significance of the muscles as carriers for cutaneous vessels was pointed out by Orticochea [49, 409], and the blood supply of the skeletal muscles of the human body was classified into five types by Mathes and Nahai [346]. They already noticed that in muscles having a dominant proximal pedicle (type I), a cutaneous side branch often can be found close to the muscle hilum. A further important contribution for the understanding of skin perfusion was made by Ponten [49]. In his paper, he emphasized the relevance of the deep fascia for skin perfusion and introduced the definition of fasciocutaneous flaps. A first classification of this type of flaps was proposed by Cormack and Lamberty, who differentiated four types of fasciocutaneous flaps according to the number and course of the perforators, including osteomyofascial flaps (type D). An even more detailed definition of six different patterns of perforating vessels was provided by Nakajima, giving a description of the different vessel courses through muscle, septa, and subcutaneous fatty tissue [396]. Based on detailed anatomical dissections, dye injections, and radiographic investigations, Taylor and Palmer introduced the angiosome concept, in which the blood supply of three-dimensional blocks of tissue is correlated with specific vessels. They described an angiosome as a composite block of tissue that is supplied anatomically by source vessels that span between the skin and bone. In addition to supplying the deep tissues, the source vessel of these angiosomes supplies branches to the overlying skin, which pass either between the deep tissues or through the deep tissues, usually muscle, to pierce the outer layer of the deep fascia. Hence, perforator flaps, when dissected to the underlying source vessels, involve tracing vessels either between the deep tissues, whether muscle, tendon, or bone, or through the deep tissues, usually muscle [535]. Thus, these three-dimensional tissue blocks can consist of all possible tissue components but are perfused by one single source vessel. Like vascular territories of the skin, adjacent angiosomes are connected by fine vascular anastomoses (choke anastomotic vessels).
The venous architecture was described as a continuous network of arcades that follow the connective-tissue framework of the body. It mirrors the arterial supply in the deep tissues, so that angiosomes consist of matching arteriosomes and venosomes [536].
A simplified classification of perforating vessels which was proposed by Hallock distinguishes direct from indirect perforators. Whereas the first travel directly from the source vessel through the subcutaneous fatty tissue to the skin and only penetrate the deep fascia, indirect perforators follow fascial spaces, intermuscular septa, or have a course through muscle tissue until they penetrate the deep fascia to reach the skin [399]. This simple classification finds general acceptance among surgeons [73]. Niranjan pointed out that indirect perforators also can travel through periosteum or tendons instead of muscle before they reach the skin, but this is only seldom the case [49].
Meanwhile, perforator flaps are harvested from many donor sites such as the anterolateral thigh (ALT) [73, 281, 282, 308, 425], medial or lateral thigh [180, 289], the tensor fasciae latae [116, 280, 298], latissimus dorsi [270, 274, 294], rectus abdominis [8, 195, 293], and gluteal region [7, 571], from the forearm [37, 458], the lower leg [72, 239, 292, 297, 300, 551], and many other regions of the body [141, 345]. Nevertheless, the potential of these flaps which offer new donor sites and a high precision of flap designs has by far not yet been reached [592].
11.2 Classification and Definition of Perforator Flaps
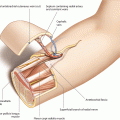
Soft tissue flaps which are perfused by defined singular vessels which penetrate the deep fascia can be named as perforator flaps. They consist of skin and/or subcutaneous fatty tissue. The first description for a perforator flap without the source vessel was given by Koshima for the thinned paraumbilical perforator flap [293], and many others followed describing numerous donor sites throughout the body. Following the simplified classification of Hallock, perforator flaps are named according to the course of the vessel penetrating the deep fascia. Thus, direct perforator flaps are perfused by a vessel traveling directly from the source artery to the deep fascia which they penetrate to enter the skin. Indirect perforator flaps are subdivided into myocutaneous or septocutaneous flaps, depending on the course of the vessel through muscle tissue or along intermuscular septa. Thus, for the raising of a perforator flap, the deep fascia must always be opened to follow the perforating vessel down to the source artery. Whereas in direct perforator flaps the dissection is performed only through fatty tissue, in indirect perforator flaps, muscle fibers must be transected or separated (myocutaneous perforator flap), or at least one intermuscular septum must be opened (septocutaneous perforator flap). The individual perforator flap is then specified according to the name of the source vessel (deep inferior epigastric artery perforator flap) or according to the muscle which has to be separated (vastus lateralis perforator flap). Since the anastomosis of a perforator flap can either be performed without sacrifice of the source vessel at the perforating vessel itself (short pedicle with small caliber) or at the source vessel (long pedicle with large caliber), the structure of the vascular pedicle should be mentioned as a further characteristic of the flap (soleus perforator only flap, vastus lateralis long pedicle perforator flap). According to a definition by Kim, a perforator flap based on a musculocutaneous perforator should be named according to the name of the muscle perforated, and perforator flaps based on other types of perforators should be named according to the name of the proximal vessel. The term “perforator based” should further define those flaps harvested without sacrificing the proximal vessels [273].
Therefore, the main difference to conventional flaps is that for elevation of a perforator flap, a specific skin vessel the location of which can be variable must be exposed and followed to the source vessel by incision of the deep fascia and depending on its course, septa, or muscle tissue. This technique allows for the preservation of structures not needed for defect coverage, particularly muscles including their motor innervation. Apart from the individual design of the skin flap, which can be exactly tailored to fit the defect, the major advantage of perforator flaps is their minimal donor site morbidity. However, for a successful reconstruction, mastering of the subtle dissection which is required for flap raising, exact knowledge of the vascular anatomy and its possible variations, and reliable suturing of vessels with a diameter of 1 mm or less is mandatory.
11.3 Development and Indications
When Song et al. in 1984 published their paper about the thigh as a donor site for three new flaps with the anterolateral thigh, they closely fulfilled all characteristics of a perforator flap [509].
To raise the septocutaneous flap, opening of the deep fascia and safe inclusion of the skin vessel located between the rectus and vastus lateralis muscle were significant components of the operating procedure. But other than elevating a true perforator flap according to the above-mentioned definitions, the conventional anterolateral thigh flap mostly includes a significant amount of subcutaneous fatty tissue and often a substantial portion of muscle tissue from the vastus lateralis in order to protect the perforating skin vessels and to guarantee for a safe blood supply of the skin. Whereas initially the perforators were only visualized or partially exposed at the level of the deep fascia, they are now completely skeletonized along their course from the skin to the source vessel, which mostly is the descending branch of the lateral circumflex femoral artery. Doing this, any muscle tissue is discarded, and the subcutaneous fat is shaped or thinned according to the requirements of the defect. Moreover, different vascular pedicles arising from different source vessels can be used in the anterolateral thigh perforator flap, allowing to raise the flap in a “freestyle” approach as long a safe anastomosis is provided by the pedicle which was selected.
After the first description by Song, it took several years until this flap became popular for different reconstructive purposes. With different focuses on the variety and shapes which are possible when using the conventional technique, a great number of clinical series with many different soft tissue reconstructions have been reported since the early 1990s. The flap has been used as a fasciocutaneous [162, 299, 497, 509, 584, 635], fascial [215], thinned skin flap [281, 282, 584, 606, 607], chimeric flap in combination with bone flaps [299, 584], muscle-only flap [605], myocutaneous flap [81, 117, 162, 279, 419, 584, 591, 595, 605], dermofat flap [81, 175, 234, 254, 603], or flow-through flap [16, 291] for reconstructions of the head and neck including scalp, oral cavity, lips and esophagus, upper and lower extremities, foot and hand, trunk, female breast, abdominal wall, and other regions of the body.
With this tremendous amount of indications and clinical experience, the anterolateral thigh made an evolution to become a donor site for flaps which are primarily defined by the location, length, and course of its individual perforating vessel instead by the gross anatomical location of the skin island [336, 552].
11.4 Anatomy
Since the anatomy of the anterolateral thigh is already described in ► Chap. 5, only a short review of the vascular anatomy with special regard to the perforating vessels is given here.
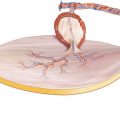
Perforator flaps from the anterolateral thigh receive their blood supply from perforating vessels that arise from the descending branch of the circumflex femoral artery. Nevertheless, perforators can also spring off from the transverse or the ascending branch of the lateral circumflex femoral artery, or as a seldom variation from an oblique branch or from the profunda or the superficial femoral artery itself [16, 94, 277, 336, 497, 531, 611]. In 89 consecutive patients, Wong et al. found a mean of 1.9 sizeable cutaneous perforators that were identified with musculocutaneous perforators in 85% and septocutaneous vessels in 15% of all cases. Those perforators located close to the septum have a short, direct intramuscular course [611]. The branching pattern of the skin vessels in a series of 74 clinical cases could be classified into eight categories, but no variation was found making flap raising impossible. In this series, 2.3 perforators per case were found, 82% of them having a myocutaneous course, branching off at different levels from the descending branch [17, 277]. An oblique branch of the lateral circumflex femoral artery was noted to be present in 35% of cases, being the source vessel for the dominant perforator in 14% of cases [611]. A near-total thigh flap, nourished by three independent perforators originating from the superficial and profunda femoris artery as well as from the descending branch, was transferred to an extended facial defect by anastomosing all pedicles separately, hereby supercharging the flap perfusion [505]. Similarly, an auto flow-through technique was described, allowing for the capture of perforators from two different sources and maximizing the reach of the pedicle [182]. Besides these variations of the source vessel, the absence of any cutaneous branches is possible in seldom cases [94, 288, 308, 584, 591, 611] and was described to occur in up to 5.4% [277]. In the middle third of the thigh, the descending branch divides in about 30% of the patients into a medial and a lateral branch. Whereas the medial branch gives off feeders to the rectus femoris muscle and the skin at the medial aspect of the thigh, the lateral branch is the source vessel for septo- or myocutaneous perforators to the skin of the anterolateral thigh. In a clinical study of 115 flap-raising procedures at the anterolateral thigh, the descending branch was found to be absent in 22.6%, being replaced by the medial descending branch or other strong muscle branches [17]. According to the results of anatomical investigations, the dominant cutaneous vessel of the anterolateral thigh was found to have a myocutaneous course in 60–80% [117, 277, 584, 591, 611], showing a septocutaneous pattern more frequently in the proximal part of the thigh [335]. In these cases, the dominant cutaneous vessel is having a direct course to the skin, running along the lateral intermuscular septum between the rectus femoris and vastus lateralis muscle and piercing the fascia lata without traversing through the vastus lateralis muscle. These flaps are raised without any muscle tissue and thus are offering thin and pliable skin paddles, being well suited for reconstructions in the head and neck area including the oral cavity. The dominant cutaneous vessel can be found within a 4 cm radius at the midpoint of a line between the anterior superior iliac spine and the lateral border of the patella in nearly all cases [346, 598].
Apart from this main perforator, the descending branch is giving off one to three additional cutaneous branches, reaching the skin more distally to the main perforator. Whereas the most distally located of these additional vessels are not reliable for skin perfusion, a second perforator can be found in about 90% of all cases 4–9 cm distal to the main perforator, making it possible to build a second independent skin paddle. Like the dominant perforator, this additional cutaneous vessel is having a myocutaneous course in 80–90%, piercing the muscle in a distance of 2–5 cm from its medial rim [591, 598]; other authors describe a range of 0.1–7 cm, with a mean distance of 1.8 cm [330]. Because most of the perforators showing a myocutaneous course enter the muscle within 2–3 cm, only a small muscle cuff is necessary if the tedious dissection of these musculocutaneous perforators is not desired. Wong et al. investigated the course of the perforators in a series of 89 flaps and found perforators with a more lateral and distal localization having a tortuous intramuscular course through the vastus lateralis [611]. A detailed description of the course of the perforators was given by Shieh et al., who classified them into four types according to their derivation and the direction in which they traversed the vastus lateralis muscle. In type I, vertical musculocutaneous perforators from the descending branch were found in 56.8%, being 4.8 cm long in average. In type II, horizontal myocutaneous perforators from the transverse branch were found in 27%, having a length of 6.7 cm. Type III was found in 10.8% containing perforators having a vertical septocutaneous course from the descending branch with a length of 3.6 cm in average. In type IV (5.4%), horizontal septocutaneous perforators were found from the transverse branch, being about 8 cm long [497].
11.5 Advantages and Disadvantages
Compared to the conventional ALT flap which can include a significant amount of subcutaneous fatty tissue and muscle bulk, the meticulous dissection of the perforator allows the raising of thin and pliable flaps individually designed according to the needs of the defect. Kimura and Satoh were the first to describe that vascular anatomy of the cutaneous perforators of the lateral thigh gives a suitable basis for primary flap thinning procedures [281]. In their first five cases, they removed the subcutaneous fatty tissue uniformly from the whole flap except for the region around the perforator, obtaining a flap thickness of only 3–4 mm. Consequently, other authors performed flap thinning also if the perforator had a myocutaneous course by using the technique of perforator flap elevation and discarding the superfluous muscle tissue [65, 162, 584, 606]. Further experience with primary thinning has shown that the radical removal of fatty tissue does not impair flap perfusion, if the subdermal vascular plexus is preserved and attention is paid to the vascular territory of the corresponding flap vessels [290]. Although Ross and coworkers found a higher complication rate in their clinical series [447] and Alkureishi et al. could experimentally find a reduced dye perfusion of the thinned flaps [6], the literature generally reports low complication rates [6, 290, 584, 606, 621]. In an anatomical study by Nojima et al., the vascular territories of unthinned and thinned perforator flaps were compared by selective dye injection into the largest perforator of the descending branch, the diameter of which was 1 mm in average. The mean vascular territory of the unthinned flaps was 351 cm2 and 256 cm2 after thinning to a thickness to 6–8 cm. Hence, flap thinning led to a reduction to about 80% of the safe vascular territory [403]. In fresh cadavers, three- and four-dimensional computed tomographic angiography and venography showed changes in flap perfusion after thinning. The authors found that thinning reduces the size of the vascular territory by transecting recurrent vessels at the level of the suprafascial plexus [472]. Using three-dimensional imaging and latex dissections, these large-diameter linking vessels in the suprafascial level were also described by Saint-Cyr and coworkers. They pointed out that these vascular links enable perfusion of adjacent vascular territories and thus make it possible to raise extended flaps which are reliably perfused by a single dominant perforator [460]. All authors agree, however, that flap thinning must be performed with a high degree of technical skill and exact knowledge of the vascular anatomy. If the subfascial vascular plexus is completely preserved, the size of the vascular territory of a thinned flap corresponds to conventional flaps [289, 393, 584].
One of the most important reasons for flap failure is the inadvertent division of the perforator at the fascial plane [73]. Thus, to reduce the uncertainty in predicting the anatomy of the perforators and to facilitate their exposure, preoperative mapping using an audible Doppler is generally recommended. Although the definite course of this dominant cutaneous vessel can only be explored during flap raising, a myocutaneous pattern can be expected if the Doppler signal is detected not directly over the palpable groove between the rectus and vastus lateralis muscle, but 2–4 cm lateral to the septum above the medial portion of the muscle. The use of the preoperative color Doppler assessment was investigated by comparing their intraoperative findings with the number and location marked by preoperative color Doppler flowmetry. Here, a high predictive value of this method was found in prospective studies [245, 553]. On the other hand, sensitivity and specificity were found to be less reliable in audible Dopplers and to be dependent on the type of the handheld Doppler used [267, 553, 628]. Rozen et al. proposed preoperative CT angiography, which provided better and more reliable information about the descending branch and its perforators compared to Doppler sonography. With this method, limbs with unsuitable perforators can be identified before surgery and a better done area can be selected [451], and this method was also found to modify the reconstructive strategy by predicting the most suitable vessels [159]. A laser-assisted indocyanine green imaging was applied by Sacks et al., who with this method were able to choose optimal perforators according to the best perfusion values [454]. Moreover, a video-assisted flap harvesting was proposed after successfully identifying the perforator positions endoscopically [382].
The dissection of the perforators gives the opportunity to perform small-vessel anastomoses, if adequate small recipient vessels are present at the defect site. This can be helpful in patients with a paucity of conventional recipient vessels having had previous surgery. In these cases, only arteries with a visible pulsation may be selected since otherwise no adequate flow will be established within the flap. If also an unconventional recipient vein is used, it must be evident that it provides reliable drainage to the flap. It is mandatory that all such small vessels must be handled with a high degree of sensitivity and great care to prevent vascular spasm and any kind of damage to the vessel wall. Because the sacrifice of the descending branch does not cause any disadvantage to the patient, it is generally recommended to raise a long pedicle ALT perforator flap and to make use of the long and high-caliber vascular pedicle it offers.
11.6 Patient Positioning
Despite the anatomical variations described for the vascular pedicle of the anterolateral thigh/vastus lateralis flap, angiography is not helpful in locating the variable positions of the septo- or myocutaneous branches of the descending branch. Preoperative evaluation of the perforators should be performed using a Doppler probe by carefully auscultating the skin in the region of the lateral intermuscular septum and over the medial parts of the vastus lateralis muscle. The patient is placed in a supine position, and the whole leg is included into the operating field to allow for free positioning of the extremity and for modifying the flap design, if necessary. Circular disinfection is performed from the hip down to the lower leg.
11.7 Flap Design
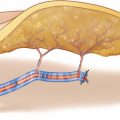
As the skin paddle of the ALT perforator flap is completely dependent on the location of the perforator selected for flap raising, the definite design of the flap cannot be determined before a useful perforator showing a strong pulsation has been exposed. This normally can be found a few centimeters proximal to the midpoint of the interconnection between the anterior iliac spine and the lateral rim of the patella. The longitudinal axis of the flap is designed parallel and 2–4 cm lateral to the septum between the rectus and vastus lateralis muscle. The length of the flap can reach up to 25 cm, if the subfascial vascular network is preserved; flap width is limited to about 8 cm, which is the limit for tension-free primary closure. When thinning the flap radically in the suprafascial plane, it is recommended to reduce the size of the flap moderately for safe perfusion. The skin incision is marked medial to the tensor muscle at the proximal thigh and straight above the rectus muscle toward the knee, keeping a safe distance to the septum of about 3 cm (◘ Figs. 11.1 and 11.2).
Skin Incision
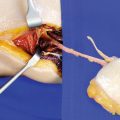
After palpation of the rectus muscle and the lateral intermuscular septum, the skin incision is performed slightly curved medially above the rectus femoris muscle, starting in the proximal third opposite to the tensor fasciae latae muscle. A safety distance of 2–3 cm is kept from the lateral intermuscular septum. So, all perforators to the anterolateral thigh emerging from the descending branch are safely included. Moreover, if the main perforator arises from the transverse, ascending, or horizontal branch, it also can be exposed using this incision in most cases. Since the perforator is exposed subfascially, the fascia lata is incised so that the fibers of the rectus muscle clearly become visible (◘ Figs. 11.3 and 11.4).
Reflecting the Fascia Lata
At the incision margin, the fascia is held with fine hooks or a forceps and gently reflected. Using meticulous and blunt dissection, the intermuscular septum is approached and carefully scanned for perforators arising between the rectus and vastus lateralis muscle. Before the fascia lata is incised at the proximal or distal incision margin to allow for more flexibility, any perforator that might cross here must be identified (◘ Fig. 11.5).
Exposure of the Perforator
With further blunt dissection, a dominant perforator is identified as a myocutaneous vessel in a distance of about 3 cm from the anterior rim of the vastus muscle, a few centimeters cranial to the midpoint of the interconnection line between SIAS and patella. Before this perforator is selected, its pulse should be clearly visible indicating that this vessel is reliable for perfusion of the skin flap. No tension, pressure, or torque must be applied on this vessel during the whole flap-raising procedure (◘ Fig. 11.6).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree