The major groups of animals responsible for bites, stings, and parasitic infections in humans belong to the phyla Arthropoda, Chordata, Cnidaria (formerly Coelenterata), Nemathelminthes, Platyhelminthes, Annelida, and Protozoa. This chapter reviews parasitic diseases and the major causes of bites and stings, as well as strategies for prevention.
Phylum Protozoa
The protozoa are one-celled organisms, divided into classes according to the nature of their locomotion. Class Sarcodina organisms move by temporary projections of cytoplasm (pseudopods); class Mastigophora by means of one or more flagella; and class Ciliata by short, hairlike projections of cytoplasm (cilia). Class Sporozoa have no special organs of locomotion.
Class Sarcodina
Amebiasis Cutis
Entamoeba histolytica –induced cutaneous ulcers usually result from extension of an underlying amebic abscess; the most common sites are the trunk, abdomen, buttocks, genitalia, and perineum. Those on the abdomen may result from hepatic abscesses. Penile lesions are usually sexually acquired and associated with anal intercourse. Most lesions begin as deep abscesses that rupture and form ulcerations with distinct, raised, cordlike edges, and an erythematous halo approximately 2 cm wide. The base is covered with necrotic tissue and hemopurulent pus containing amebas. These lesions are from a few centimeters to 20 cm wide. Without treatment, slow progression of the ulcer occurs in an increasingly debilitated patient until death ensues. Patients may also present with fistulas, fissures, polypoid warty lesions, or nodules. Deep lesions are more likely to be associated with visceral lesions.
The sole manifestation of early amebiasis may be chronic urticaria. An estimated 10 million invasive cases occur annually, most of them in the tropics. Infection may be asymptomatic, or bloody diarrhea and hepatic abscesses may be present. In the United States the disease occurs chiefly in institutionalized patients, world travelers, recent immigrants, migrant workers, and men who have sex with men. Penile ulcers are associated with insertive anal intercourse.
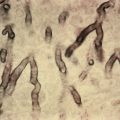
The histologic findings are those of a necrotic ulceration with many lymphocytes, neutrophils, plasma cells, and eosinophils. E. histolytica is found in the tissue, within blood and lymph vessels. The organism measures 50–60 µm in diameter and has basophilic cytoplasm and a single, eccentric nucleus with a central karyosome. The organism is frequently demonstrable in fresh material from the base of the ulcer by direct smear. Culture of the protozoa confirms the diagnosis. Indirect hemagglutination test results remain elevated for years after the initial onset of invasive disease, whereas the results of gel diffusion precipitation tests and counterimmunoelectrophoresis become negative at 6 months. This property can be used to test for recurrent or active disease in persons coming from endemic areas.
When the perianal or perineal areas are involved, granuloma inguinale, lymphogranuloma venereum, deep mycosis, and syphilis must be considered. In chronic urticaria, fresh stool examinations by a trained technician are necessary.
The treatment of choice is metronidazole (Flagyl), 750 mg orally three times daily for 10 days. Abscesses may require surgical drainage.
Other Amebas
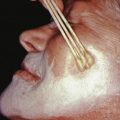
Amebas of the genera Acanthamoeba and Balamuthia may also cause skin lesions in infected hosts. These organisms are ubiquitous in the environment and are found in soil, water, and air. Granulomatous amebic encephalitis is the most common manifestation of infection with these amebas. In the case of Acanthamoeba, invasive infections are almost always in immunocompromised individuals, including those with acquired immunodeficiency syndrome (AIDS) and organ transplant patients, although Acanthamoeba can also involve the cornea in those who use homemade contact lens solution. Disseminated lesions present as pink or violaceous nodules that then enlarge, suppurate, and form ulcers with a necrotic eschar ( Fig. 20.1 ). Other findings include fever, nasal congestion or discharge, epistaxis, cough, headaches, lethargy, altered mental status, and seizures. In patients infected with Acanthamoeba who have disease of the central nervous system (CNS), death usually occurs within days to weeks. The organisms are visible on skin biopsy, and culture is definitive. In patients without CNS involvement, mortality rate is 75%, with successfully treated cases often managed with a combination of 5-fluorocytosine and sulfadiazine. In patients infected with Balamuthia mandrillaris, involvement of the central face is typical. Treatment paradigms are changing, and in vitro evidence suggests that diminazene aceturate is more active than miltefosine or pentamidine, and artemether shows promise ( Fig. 20.2 ). Chlorhexidine topically and surgical debridement are local adjunctive measures that may prove beneficial.
Belizario V Jr, et al: Cutaneous manifestations of selected parasitic infections in western Pacific and Southeast Asian regions. Curr Infect Dis Rep 2016; 18: 30.
Deng Y, et al: Artemether exhibits amoebicidal activity against Acanthamoeba castellanii through inhibition of the serine biosynthesis pathway. Antimicrob Agents Chemother 2015; 59: 4680.
Eichelmann K, et al: Tropical dermatology. Semin Cutan Med Surg 2014; 33: 133.
Khan NA, et al: Targeting brain-eating amoebae infections. ACS Chem Neurosci 2017; 8: 687.

Class Mastigophora
Organisms belonging to this class, the mastigophorans, are also known as flagellates. Many have an undulating membrane with flagella along their crest.
Trichomoniasis
Trichomonas vulvovaginitis is a common cause of vaginal pruritus, with burning and a frothy leukorrhea. The vaginal mucosa appears bright red from inflammation and may be mottled with pseudomembranous patches. The male urethra may also harbor the organism; in the male it causes urethritis and prostatitis. Occasionally, men may develop balanoposthitis. Erosive lesions on the glans and penis or abscesses of the median raphe may occur. Neonates may acquire the infection during passage through the birth canal, but they require treatment only if symptomatic or if colonization lasts more than 4 weeks. Because this is otherwise almost exclusively a sexually transmitted disease (STD), Trichomonas vulvovaginitis in a child should prompt suspicion of sexual abuse.
Trichomoniasis is caused by Trichomonas vaginalis, a colorless piriform flagellate 5–15 µm long. T. vaginalis is demonstrated in smears from affected areas. Testing by direct immunofluorescence is sensitive and specific, and polymerase chain reaction (PCR) analysis is now available.
Metronidazole, 2 g in a single oral dose, is the treatment of choice. Alternatively, 500 mg twice daily for 7 days may be given, and intravaginal metronidazole/miconazole is also effective. Patients should be warned not to drink alcohol for 24 hours after or dosing because of the disulfiram-type effects of this medication. Male sex partners should also be treated. The use of metronidazole is contraindicated in pregnant women, and clotrimazole, applied intravaginally at 100 mg a night for 2 weeks, may be used instead. Disulfiram and nithiamide show in vitro evidence of activity and could prove useful for resistant organisms.
Bouchemal K, et al: Strategies for prevention and treatment of Trichomonas vaginalis infections. Clin Microbiol Rev 2017; 30: 811.
de Brum Vieira P, et al: Challenges and persistent questions in the treatment of trichomoniasis. Curr Top Med Chem 2017; 17: 1249.
Leishmaniasis
All forms of leishmaniasis are caused by morphologically indistinguishable protozoa of the family Trypanosomidae, called Leishmania (pronounced leesh-may-nea). The clinical features of the leishmaniases differ, and in general, these diseases have different geographic distribution. The variable clinical manifestations may result from the diversity of the organism and the person’s immune status and genetic ability to initiate an effective cell-mediated immune response to the specific infecting organism. It is known that the antigen-specific T-cell responses, which lead to the production of interferon (IFN) and interleukin-12 (IL-12), are important for healing of the lesions and the induction of lifelong, species-specific immunity to reinfection that results after natural infection. Both CD4+ and CD8+ lymphocytes appear to be active in the immune response. IL-10–producing natural regulatory T cells may play a role in the downregulation of infection-induced immunity.
Cutaneous Leishmaniasis
There are several types of lesion. All tend to occur on exposed parts because all are transmitted by the sandfly. Old World leishmaniasis manifests mainly in the skin and has also been called Baghdad boil, Oriental sore, leishmaniasis tropica, Biskra button, Delhi boil, Aleppo boil, Kandahar sore, and Lahore sore. Mild visceral disease may occur. Skin lesions of New World infection have been termed uta, pian bois, and bay sore or chiclero ulcer.
Clinical Features.
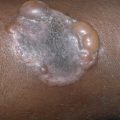
In Old World leishmaniasis, lesions may present in two distinct types. One is the moist or rural type, a slowly growing, indurated, livid, indolent papule ( Fig. 20.3 ), which enlarges in a few months to form a nodule that may ulcerate in a few weeks to form an ulcer as large as 5 cm in diameter. Spontaneous healing usually takes place within 6 months, leaving a characteristic scar. This type is contracted from rodent reservoirs such as gerbils via the sandfly vector. The incubation period is relatively short (1–4 weeks). The dry or urban type has a longer incubation period (2–8 months or longer), develops much more slowly, and heals more slowly than the rural type. In both types, the ulcer or crust forms on a bed of edematous tissue.

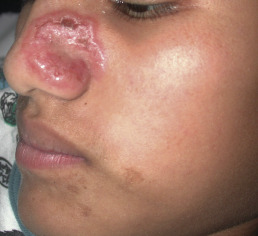
Rarely, after the initial or “mother” lesion is healed, at the borders of the healed area a few soft red papules may appear that are covered with whitish scales and have the “apple jelly” characteristics of granulomatous diseases such as lupus vulgaris. These spread peripherally on a common erythematous base and are the lupoid type. This is also known as leishmaniasis recidivans and occurs most often with the urban type of disease, caused by Leishmania tropica. New World disease may also induce purely cutaneous lesions, of varied morphology. The primary papule may become nodular, verrucous, furuncular, or ulcerated, with an infiltrated red border ( Fig. 20.4 ). Subcutaneous peripheral nodules, which eventually ulcerate, may signal extension of the disease. A linear or radial lymphangitic (sporotrichoid) pattern may occur with lymphadenopathy, and the nodes may rarely yield organisms. Facial lesions may coalesce and resemble erysipelas. Recidivans lesions are unusual in the New World form of disease. In Yucatan and Guatemala, a subtype of New World disease exists: the chiclero ulcer. The most frequent site of infection is the ear ( Fig. 20.5 ). The lesions ulcerate and occur most frequently in workers who harvest chicle for chewing gum in forests, where there is high humidity. This form is a more chronic ulcer that may persist for years, destroying the ear cartilage and leading to deformity. The etiologic agent is Leishmania mexicana and the sandfly vector, Lutzomyia flaviscutellata.



Uta is a term used by Peruvians for leishmaniasis occurring in mountainous territory at 1200–1800 m above sea level. The ulcerating lesions are found on exposed sites and mucosal lesions do not occur.
Disseminated cutaneous leishmaniasis may be seen in both New and Old World disease. Multiple nonulcerated papules and plaques, chiefly on exposed surfaces, characterize this type. The disease begins with a single ulcer, nodule, or plaque from which satellite lesions may develop and disseminate to cover the entire body. The disease is progressive, and treatment is usually ineffective. It is characterized by anergy to the organism. This type of leishmaniasis must be differentiated from lepromatous leprosy, xanthoma tuberosum, paracoccidioidal granuloma, Lobo disease, and malignant lymphoma.
Etiologic Factors
Leishmania tropica, L. major, L. aethiopica, and L. infantum, the cause of Mediterranean visceral leishmaniasis, may cause cutaneous leishmaniasis. Purely cutaneous leishmaniasis is also caused by several species present in the New World. L. mexicana does not induce mucosal disease. Leishmania braziliensis guyanensis produces cutaneous disease, as does L. b. braziliensis and L. b. panamensis; however, the latter two may also result in mucocutaneous disease.
Epidemiology.
Cutaneous leishmaniasis is endemic in Asia Minor and to a lesser extent in many countries around the Mediterranean. Iran and Saudi Arabia have a high occurrence rate. In endemic areas, deliberate inoculation on the thigh is sometimes practiced so that scarring on the face—a frequent site for Oriental sore—may be avoided. Purely cutaneous lesions may also be found in the Americas. In the United States leishmaniasis is largely restricted to southern Texas, although rare reports of human cutaneous disease have occurred as far north as Pennsylvania, and visceral leishmaniasis in immunosuppressed humans is being recognized as an emerging infection in areas not previously thought to be endemic for the disease.
Pathogenesis.
The leishmania protozoan has an alternate life in vertebrates and in insect hosts. Humans and other mammals, such as dogs and rodents, are the natural reservoir hosts. The vector hosts are Phlebotomus sandflies for the Old World type and Phlebotomus perniciosus and Lutzomyia sandflies for New World cutaneous leishmaniasis. After the insect has fed on blood, the flagellates (leptomonad, promastigote) develop in the gut in 8–20 days, after which migration occurs into the mouth parts; from here, transmission into humans occurs by a bite. In humans, the flagella are lost, and a leishmanial form (amastigote) is assumed.
Histopathology.
An ulcer with a heavy infiltrate of histiocytes, lymphocytes, plasma cells, and polymorphonuclear leukocytes is seen. The parasitized histiocytes form tuberculoid granulomas in the dermis. Pseudoepitheliomatous hyperplasia may occur in the edges of the ulcer. Leishmanias are nonencapsulated and contain a nucleus and a paranucleus. Wright, Giemsa, and monoclonal antibody staining may be helpful in identifying the organisms within histiocytes, where they often line up at the periphery of a vacuole. PCR primers are available for a variety of species. PCR is more sensitive than microscopy but less sensitive than culture.
Diagnosis.
In endemic areas, the diagnosis is not difficult. In other localities, cutaneous leishmaniasis may be confused with syphilis, yaws, lupus vulgaris, and pyogenic granulomas. The diagnosis is established by demonstration of the organism in smears. A punch biopsy specimen from the active edge of the ulcer is ideal for culture. It can be placed in Nicolle-Novy-MacNeal (NNN) medium and shipped at room temperature. Parasites can also be cultured from tissue fluid. A hypodermic needle is inserted into the normal skin and to the edge of the ulcer base. The needle is rotated to work loose some material and serum, which is then aspirated. A culture on NNN medium at 22°C–35°C (71.6°F–95°F) is recommended to demonstrate the leptomonads. PCR is now available and is the most sensitive diagnostic test for cutaneous leishmaniasis.
Treatment.
Spontaneous healing of primary cutaneous lesions occurs, usually within 12–18 months, shorter for Old World disease. Reasons to treat a self-limited infection include avoiding disfiguring scars in exposed areas, avoiding secondary infection, controlling disease in the population, and failure of spontaneous healing. In the diffuse cutaneous and recidivans types, leishmaniasis may persist for 20–40 years if not treated.
In areas where localized cutaneous leishmaniasis is not complicated by recidivans or sporotrichoid forms or by mucocutaneous disease, treatment with such topical modalities as paromomycin sulfate 15% plus methylbenzethonium chloride 12%, ketoconazole cream under occlusion, cryotherapy, local heat, 5-aminolevulinic acid hydrochloride (10%) plus visible red light (633 nm), and laser ablation, or with intralesional sodium stibogluconate antimony or emetine hydrochloride may be effective and safe.
In the setting of Old World cutaneous leishmaniasis, some data suggest that intramuscular meglumine antimoniate in combination with intralesional meglumine antimoniate may be superior to intralesional therapy alone. A meta-analysis of studies of Old World cutaneous leishmaniasis concluded that pentamidine was similar in efficacy to pentavalent antimonials, and that both were superior to the other agents studied. Since then, a Pakistani study concluded that itraconazole was more effective and more economical and had fewer side effects than meglumine antimoniate in both wet and dry types of cutaneous leishmaniasis. The number of patients studied was relatively small, and other studies have been disappointing. Oral fluconazole and zinc sulfate have been used to treat Leishmania major infection. A similar meta-analysis of studies of New World cutaneous leishmaniasis concluded that meglumine might be the best agent in its class. Intralesional therapy may be acceptable for small, solitary lesions in areas with a low risk of mucosal disease. Azithromycin has been used in New World disease but is inferior to antimonials. Perilesional injections of IFN-γ have also been reported to be effective but are expensive.
In immunosuppressed patients or those who acquire infection in areas where mucocutaneous disease may occur, systemic therapy is recommended. As with topical treatment, many alternatives have been reported to be effective. Sodium antimony gluconate (sodium stibogluconate) solution is given intramuscularly or intravenously, 20 mg/kg/day in two divided doses for 28 days. It can be obtained from the U.S. Centers for Disease Control and Prevention (CDC) Drug Service (Atlanta, GA 30333). Repeated courses may be given. Antimony n -methyl glutamine (Glucantime) is used more often in Central and South America because of its local availability.
Other systemic medications reported to be effective include fluconazole (200 mg/day for 6 weeks), ketoconazole, dapsone, rifampicin, and allopurinol. Some of these have not been subjected to controlled clinical trials, as is true of most topical treatments. The recidivans and disseminated cutaneous types may require prolonged courses or adjuvant IFN therapy. Amphotericin B may be used in antimony-resistant disease. Lipid formulations of amphotericin B are highly effective in short courses but are expensive. Liposomal amphotericin B may be especially helpful for Leishmania braziliensis and L. guyanensis infections. Intramuscular pentamidine is also used for L. guyanensis cutaneous leishmaniasis, because this infection is resistant to systemic antimony. Miltefosine is being used for cutaneous disease in some areas of the world and may prove to be the treatment of choice for diffuse cutaneous leishmaniasis and post–kala-azar dermal leishmaniasis. However, some studies have shown miltefosine to be ineffective in L. major and L. braziliensis infections. Posaconazole has been used in Old World disease. Control depends chiefly on the success of antifly measures taken by health authorities and personal protection with protective clothing, screening, and repellents. Vaccines are being investigated but are not available.
Mucocutaneous Leishmaniasis (Leishmaniasis Americana, Espundia)
Clinical Features.
The initial leishmanial infection, which occurs at the site of the fly bite, is a cutaneous ulcer. Secondary lesions on the mucosa usually occur at some time during the next 5 years. The earliest mucosal lesion is usually hyperemia of the nasal septum with subsequent ulceration ( Fig. 20.6 ), which progresses to invade the septum and later the paranasal fossae. Perforation of the septum eventually takes place. For some time, the nose remains unchanged externally, despite the internal destruction. At first, only a dry crust is observed, or a bright-red infiltration or vegetation on the nasal septum, with symptoms of obstruction and small hemorrhages. Despite the mutilating and destructive character of leishmaniasis, it never involves the nasal bones. When the septum is destroyed, the nasal bridge and tip of the nose collapse, giving the appearance of a parrot beak, camel nose, or tapir nose.

It is important to recall that the four great chronic infections—syphilis, tuberculosis, Hansen disease, and leishmaniasis—have a predilection for the nose. The ulcer may extend to the lips ( Fig. 20.7 ) and continue to advance to the pharynx, attacking the soft palate, uvula, tonsils, gingiva, and tongue. The eventual mutilation is called espundia. Two perpendicular grooves at the union of the osseous palate and soft tissues, in the midst of the vegetative infiltration of the entire pharynx, are called the palate cross of espundia.

Only in exceptional cases does American leishmaniasis invade the genital or ocular mucous membranes. The frequency of mucous membrane involvement is variable. In Yucatan and Guatemala, it is an exception; in other countries, such as Brazil, it may occur in 80% of cases.
Etiologic Factors.
Mucocutaneous leishmaniasis is mainly caused by Leishmania (Viannia) braziliensis braziliensis and L. b. panamensis, although some Old World organisms, including L. infantum, L. major, and L. tropica, can cause mucosal ulceration. Leishmania has two forms: the nonflagellated form or leishmania, which is found in the tissues of humans and animals susceptible to the inoculation of the parasite; and the flagellated form or leptomonad, which is found in the digestive tract of the vector insect ( Lutzomyia in mucocutaneous disease) and in cultures. The typical morphology of leishmania, as found in vertebrates, is round or oval, usually with one extremity more rounded than the other, measuring 2–4 µm × 1.5–2.5 µm, with cytoplasm, nucleus, and blepharoplast or kinetoplast.
Epidemiology.
Mucocutaneous leishmaniasis is predominantly a rural disease. It most often occurs in damp and forested regions. The disease can be contracted at any time of the year, but the risk is highest just after the rainy season. All ages and races and both genders are equally affected. Epidemics parallel the El Niño cycle.
Histopathology.
In ulcerous leishmaniasis, marked irregular acanthosis and sometimes pseudoepitheliomatous hyperplasia can be found. The dermis shows a dense infiltration of histiocytes, lymphocytes, and plasma cells. In new lesions, some neutrophils are observed. Large Langhans giant cells or typical tubercles are occasionally seen. Numerous organisms are present (mostly in histiocytes), which are nonencapsulated and contain a nucleus and a paranucleus. Wright, Giemsa, and monoclonal antibody staining may be helpful in identifying the organisms. In patients with granulomatous infiltrates containing intracellular parasites within histiocytes, leishmaniasis is one of several diseases to be considered, including rhinoscleroma, histoplasmosis, granuloma inguinale, Chagas disease, Penicillium marneffei infection, and toxoplasmosis. Touch smears stained with Giemsa are helpful in many cases of cutaneous and mucocutaneous leishmaniasis.
Laboratory Findings.
Leishmania is demonstrated in the cutaneous and mucous membrane lesions by direct smears or cultures. In Wright-stained biopsy material, intracellular and extracellular organisms are seen with typical morphology of two chromatic structures: nucleus and parabasal body. In later mucosal lesions, the scarcity of parasites makes identification difficult. The culture is done on NNN medium for leptomonads. PCR is now widely used, and specimens obtained from lesion scarification and blood sample–enriched leukocytes compare favorably with indirect immunofluorescence reaction and culture techniques.
Prophylaxis.
Although it is impractical to eliminate the insect vector, it is still the only valid measure for the control of this prevalent disease. Effective vaccines are not available for mucocutaneous leishmaniasis.
Treatment.
Treatment is the same as described for cutaneous leishmaniasis, except that antimony resistance is common in mucocutaneous disease. Combination therapy using antimonials with drugs such as rifampin or azithromycin, or adding immunomodulators such as IFN-γ, IL-2, or imiquimod may result in cure. Amphotericin B treatment may be necessary.
Visceral Leishmaniasis (Kala-Azar, Dumdum Fever)
Clinical Features.
The earliest lesion is the cutaneous nodule or leishmanioma, which occurs at the site of the initial sandfly inoculation. Kala-azar, meaning “black fever,” acquired its name because of the patchy macular darkening of the skin caused by deposits of melanin that develop in the later course of the disease. These patches are most marked over the forehead and temples, periorally, and on the midabdomen.
The primary target for the parasites is the reticuloendothelial system; the spleen, liver, bone marrow, and lymph nodes are attacked. The incubation period is 1–4 months. An intermittent fever, with temperatures ranging from 39°C–40°C (102°F–104°F), ushers in the disease. Hepatosplenomegaly, agranulocytosis, anemia, and thrombocytopenia occur. Chills, fever, emaciation, weight loss, weakness, epistaxis, and purpura develop as the disease progresses. Susceptibility to secondary infection may produce pulmonary and gastrointestinal (GI) infection, ulcerations in the mouth (cancrum oris), and noma. Death occurs about 2 years from onset in untreated individuals.
Most infections are subclinical or asymptomatic. In patients with AIDS, papular and nodular skin lesions may occur. Dermatofibroma-type or Kaposi sarcoma–like, brown to purple nodules are most frequently reported, although random biopsies of normal skin will reveal organisms. Therefore clinical correlation is necessary to attribute skin findings to Leishmania specifically.
Etiologic Factors.
Leishmania donovani spp. donovani, infantum, and chagasi cause visceral leishmaniasis and are parasites of rodents, canines, and humans. They are nonflagellate oval organisms about 3 mm in diameter, known as Leishman-Donovan bodies. In the sandfly, it is a leptomonad form with flagella.
Epidemiology.
Leishmania donovani donovani causes visceral leishmaniasis in India, with the major reservoir being humans and the vector being Phlebotomus argentipes. L .donovani infantum occurs in China, Africa, the Near East and Middle East, and the Mediterranean littoral, where the major reservoirs are dogs; Phlebotomus perniciosus and P. ariasi are the vectors of the Mediterranean type. American visceral leishmaniasis is caused by L. donovani chagasi and is transmitted by the sandfly Lutzomyia longipalpis. American visceral leishmaniasis principally affects domestic dogs, although explosive outbreaks of the human infection occur sporadically, when the number of Lutzomyia longipalpis builds up to a high level in the presence of infected dogs. Canine visceral infections with Leishmania infantum have been reported in foxhounds in various parts of the United States and Canada.
Diagnosis.
Leishman-Donovan bodies may be present in the blood in individuals with kala-azar of India. Specimens for examination, in descending order of utility, include spleen pulp, sternal marrow, liver tissue, and exudate from lymph nodes. Culturing on NNN medium may also reveal the organisms.
Treatment.
General supportive measures are essential. Pentavalent antimony has long been the drug of choice. In areas of drug resistance, amphotericin B is usually effective, but it is expensive and toxic, and requires intravenous administration. Miltefosine, an oral alkyl-phosphocholine analog, has proved as effective as amphotericin B in some trials. It is often used to treat visceral disease in India and Ethiopia. Mixed infections involving both Leishmania and Trypanosoma cruzi are becoming increasingly common in Central and South America because of overlapping endemic areas. Amiodarone has been used as an unconventional antiparasitic drug in this setting in addition to standard therapy.
Post–Kala-Azar Dermal Leishmaniasis
In kala-azar, the leishmanoid (amastigote) forms may be widely distributed throughout apparently normal skin. During and after recovery from the disease, a special form of dermal disease known as post–kala-azar dermal leishmaniasis appears. This condition appears during or shortly after treatment in the African form, but its appearance may be delayed up to 10 years after treatment in the Indian form. It follows the treatment of visceral leishmaniasis in 50% of Sudanese patients and 5%–10% of those seen in India. There are two constituents of the eruption: a macular, depigmented eruption found mainly on the face, arms, and upper part of the trunk and a warty, papular eruption in which amastigotes can be found. Because it may persist for up to 20 years, these patients may act as a chronic reservoir of infection. This condition closely resembles Hansen disease. High concentrations of IL-10 in the blood of visceral leishmaniasis patients predict those who will be affected by post–kala-azar dermal leishmaniasis. Miltefosine may become the drug of choice.
Viscerotropic Leishmaniasis
Twelve U.S. soldiers developed systemic infection with Leishmania tropica while fighting in Operation Desert Storm in Iraq and Kuwait. None had symptoms of kala-azar, but most had fever, fatigue, malaise, cough, diarrhea, or abdominal pain, and none had cutaneous disease. Diagnostic tests yielded positive results on bone marrow aspiration; lymph node involvement was also documented. Treatment with sodium stibogluconate led to improvement.
Burza S, et al: Leishmaniasis. Lancet. 2018 Sep 15;392(10151):951-970. doi: 10.1016/S0140-6736(18)31204-2. Epub 2018 Aug 17. Review. PubMed PMID: 30126638.
Maxfield L, et al: Leishmaniasis. 2018 Oct 1. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018 Jan-. Available from http://www-ncbi-nlm-nih-gov.easyaccess2.lib.cuhk.edu.hk/books/NBK531456/ PubMed PMID: 30285351.
Meireles CB, et al: Atypical presentations of cutaneous leishmaniasis. Acta Trop 2017; 172: 240.
Oryan A, et al: Worldwide risk factors in leishmaniasis. Asian Pac J Trop Med 2016; 9: 925.
Scorza BM, et al: Cutaneous manifestations of human and murine leishmaniasis. Int J Mol Sc 2017; 18.
Steverding D: The history of leishmaniasis. Parasit Vectors 2017; 10: 82.
Sundar S, et al: Recent developments and future prospects in the treatment of visceral leishmaniasis. Ther Adv Infect Dis 2016; 3: 98.
Wolf Nassif P, et al: Safety and efficacy of current alternatives in the topical treatment of cutaneous leishmaniasis. Parasitology. 2017; 144: 995.
Human Trypanosomiasis
Three species of trypanosome are pathogenic to humans: Trypanosoma gambiense and T. rhodesiense in Africa and T. cruzi in America. The skin manifestations are usually observed in the earlier stages of trypanosomiasis as evanescent erythema, erythema multiforme, and edema, especially angioedema.
In the early stage of African trypanosomiasis, a trypanosome chancre may occur at the site of a tsetse fly bite. Erythema with circumscribed swellings of angioedema then occurs, with enlargement of the lymph nodes, fever, malaise, headache, and joint pains. In the West African (Gambian) form, the illness is chronic, lasting several years, with progressive deterioration, whereas the East African (Rhodesian) form is an acute illness, with a stormy, fatal course of weeks to months. The Rhodesian form is more often associated with cutaneous signs. Annular or deep erythema nodosum–like lesions are frequent manifestations ( Fig. 20.8 ). Lymphadenopathy is generalized, but frequently there is a pronounced enlargement of the posterior cervical group (Winterbottom sign).

In American trypanosomiasis (Chagas disease), similar changes take place in the skin. The reduviid bug (kissing bug, assassin bug) usually bites at night, frequently at mucocutaneous junctions, where the bug’s infected feces are deposited when it feeds ( Fig. 20.9 ). The unsuspecting sleeping person rubs the feces into the bite and becomes infected. If the bite of the infected bug occurs near the eye, Romana sign develops, consisting of unilateral conjunctivitis and edema of the eyelids, with an ulceration or chagoma in the area. The bite of a “kissing bug” becomes extremely swollen and red, whether or not trypanosomes are involved. Acute Chagas disease is usually a mild illness of fever, malaise, edema of the face and lower extremities, and generalized lymphadenopathy. Skin lesions occurring in this phase include nodules at the site of inoculation, disseminated nodules, or morbilliform and urticarial lesions. In chronic Chagas disease, which occurs in 10%–30% of infected persons years to decades later, the heart (myocarditis, arrhythmias, thromboembolism, cardiac failure) and GI system (megaesophagus, megacolon) are most often involved. During the remaining infected but asymptomatic indeterminate phase, patients may transmit the disease through transfusion. When such patients become immunosuppressed (with AIDS or organ transplantation), reactivation skin lesions may occur with a wide range of morphologies, including panniculitis.

Rhodesian trypanosomiasis is endemic among the cattle-raising tribes of East Africa, with the savannah habitat of the vectors determining its geographic distribution. Wild game and livestock are reservoir hosts, in addition to humans. The tsetse fly, Glossina morsitans, is the principal vector.
For Gambian trypanosomiasis, humans are the only vertebrate host, and the palpalis group of tsetse flies is the invertebrate host. These flies are found close to the water, and their fastidious biologic requirements restrict their distribution and thus that of the disease. Incidence is seasonal, with humidity and temperature being determining factors. The highest incidence is in men age 20–40 in tropical areas of West and Central Africa.
Chagas disease is prevalent in Central and South America from the United States to Argentina and Chile; the highest incidence is in Venezuela, Brazil, Uruguay, Paraguay, and Argentina. Approximately 29% of all male deaths in the 29–44 age-group in Brazil are attributed to Chagas disease.
Before CNS involvement has occurred in the Rhodesian form, suramin, a complex, non–metal-containing, organic compound, is the treatment of choice. When the CNS is involved, melarsoprol is the drug of choice. Pentamidine isethionate is the drug of choice for the Gambian disease. Eflornithine appears to be a good alternative to melarsoprol for second-stage West African trypanosomiasis. For American trypanosomiasis, treatment is of limited efficacy. The nitroaromatic compounds nifurtimox and benznidazole clear the parasitemia and reduce the severity of the acute illness, but there is a high incidence of adverse effects. Although benznidazole reduces parasite load during the acute phase, it does not prevent chronic cardiac lesions. Ruthenium complexation improves bioavailability of benznidazole and has the potential to improve outcomes. Conservative treatment is the typical approach to the patient with congestive heart failure from Chagas myocarditis, but recent data suggest that clomipramine, a tricyclic antidepressant that inhibits Trypanosoma cruzi ‘s trypanothione reductase, improves the course of cardiac disease in animal models. GI complications may be treated surgically.
Aksoy S, et al: Human African trypanosomiasis control. PLoS Negl Trop Dis 2017; 11: e0005454.
Chatelain E: Chagas disease research and development. Comput Struct Biotechnol J 2016; 15: 98.
Cullen DR, et al: A brief review of drug discovery research for human African trypanosomiasis. Curr Med Chem 2017; 24: 701.
Duschak VG: Targets and patented drugs for chemotherapy of Chagas disease in the last 15 years-period. Recent Pat Antiinfect Drug Discov 2016; 11: 74.
Class Sporozoa
Toxoplasmosis
Toxoplasmosis is a zoonosis caused by a parasitic protozoan, Toxoplasma gondii. Infection may be either congenital or acquired. Cerebral disease has been reported in the setting of rituximab therapy and widespread lesions can mimic melanoma metastases on positron emission tomography (PET) scans. Congenital infection occurs from placental transmission. Abortion or stillbirth may result. However, a full-term child delivered to an infected mother may have a triad of hydrocephalus, chorioretinitis, and cerebral calcification. In addition, there may be hepatosplenomegaly and jaundice. Skin changes in toxoplasmosis are rare and clinically nonspecific.
In congenital toxoplasmosis, macular and hemorrhagic eruptions predominate. Blueberry muffin lesions, reflecting dermatoerythropoiesis, may be seen. Occasionally, abnormal hair growth and exfoliative dermatitis have also been observed. The differential diagnosis of congenital toxoplasmosis is the TORCH syndrome (toxoplasmosis, other agents, rubella, cytomegalovirus, and herpes simplex). In acquired toxoplasmosis, early skin manifestations consist of cutaneous and subcutaneous nodules and macular, papular, and hemorrhagic eruptions. These may be followed by scarlatiniform desquamation, eruptions mimicking roseola, erythema multiforme, and dermatomyositis or lichen planus, as well as exfoliative dermatitis. As a rule, the exanthem is accompanied by high fever and general malaise.
Diagnosis of acquired toxoplasmosis is of special importance to three groups of adults: healthy pregnant women concerned about recent exposure; adults with lymphadenopathy, fever, and myalgia, who might have some other serious disease (e.g., lymphoma); and immunocompromised persons, such as patients with AIDS, in whom toxoplasmosis might be fatal. It is the most common cause of focal encephalitis in AIDS patients, and this may be accompanied by a widespread papular eruption.
Toxoplasma gondii is a crescent-shaped, oval, or round protozoan that can infect any mammalian or avian cell. Toxoplasmosis is often acquired through contact with animals, particularly cats. Reservoirs of infection have been reported in dogs, cats, cattle, sheep, pigs, rabbits, rats, pigeons, and chickens. The two major routes of transmission of T. gondii in humans are oral and congenital. Meats consumed by humans may contain tissue cysts, thus serving as a source of infection when eaten raw or undercooked. There is no evidence of direct human-to-human transmission, other than from mother to fetus.
The diagnosis cannot be made on clinical grounds alone. It may be established by isolation of T. gondii; demonstration of the protozoa in tissue sections, smears, or body fluids by Wright or Giemsa stain; characteristic lymph node histology; and serologic methods. In the patient with bone marrow transplantation, the organism has caused interface dermatitis, creating the potential for misdiagnosis as graft-versus-host disease.
A combination of pyrimethamine (Daraprim) and sulfadiazine acts synergistically and forms an effective treatment, but toxicity is substantial. Dosages and total treatment time vary according to the age and immunologic competence of the infected patient.
Assolini JP, et al: Nanomedicine advances in toxoplasmosis. Parasitol Res 2017; 116: 1603.
Chellan P, et al: Recent developments in drug discovery against the protozoal parasites Cryptosporidium and Toxoplasma. Bioorg Med Chem Lett 2017; 27: 1491.
Phylum Cnidaria
The cnidarians include the jellyfish, hydroids, Portuguese men-of-war, corals, and sea anemones. These are all radial marine animals, living mostly in ocean water.
Portuguese Man-of-War Dermatitis
Stings by the Portuguese man-of-war ( Physalia physalis in Atlantic or much smaller Physalia utriculus or “bluebottle” in Pacific Ocean) are characterized by linear lesions that are erythematous, urticarial, and even hemorrhagic. The forearms, sides of the trunk, thighs, and feet are common sites of involvement. The usual local manifestation is sharp, stinging, and intense pain. Internally, there may be severe dyspnea, prostration, nausea, abdominal cramps, lacrimation, and muscular pains. Death may occur if the areas stung are large in relation to the patient’s size.
The fluid of the nematocysts contains toxin that is carried into the victim through barbs along the tentacle. The venom is a neurotoxic poison that can produce marked cardiac changes. Each Portuguese man-of-war is a colony of symbiotic organisms consisting of a blue to red float or pneumatophore with a gas gland, several gastrozooids measuring 1–20 mm, reproductive polyps, and the fishing tentacles bearing the nematocysts from which the barbs are ejected. The hydroid is found most frequently along the southeastern Florida coastline and in the Gulf of Mexico as well as on windward coasts throughout the mid-Pacific and South Pacific. Safe Sea, a barrier cream, has been reported as being effective at preventing jellyfish stings off the coast of Florida, but studies of barrier creams in general have been mixed.
Jellyfish Dermatitis
Jellyfish dermatitis produces lesions similar to those of the Portuguese man-of-war, except that the lesions are not so linear. Immediate allergic reactions occur infrequently as urticaria, angioedema, or anaphylaxis. Delayed and persistent lesions also rarely occur.
The Australian sea wasp, Chironex fleckeri, which is colorless and transparent, is the most dangerous of all jellyfish, with a sting that is often fatal ( Fig. 20.10 ). Another sea wasp, Carybdea marsupialis, is much less dangerous and occurs in the Caribbean Sea. Rhopilema nomadica, common in the Mediterranean Sea, has been reported to cause severe delayed dermatitis.

Seabather’s eruption is an acute dermatitis that begins a few hours after bathing in the waters along the Atlantic coast. It affects covered areas of the body as cnidarian larvae become entrapped under the bathing suit and the nematocyst releases its toxin because of external pressure. Thus the buttocks and waist are affected primarily, with the breast also involved in women ( Fig. 20.11 ). Erythematous macules and papules appear and may develop into pustules or vesicles. Urticarial plaques are also present in a smaller number of patients. Crops of new lesions may occur for up to 72 hours, and the eruption persists for 10–14 days on average. It is quite pruritic.

Outbreaks in Florida are usually caused by larvae of the thimble jellyfish, Linuche unguiculata, which patients report as “black dots” in the water or their bathing suits. The larvae of the sea anemone Edwardstella lineata caused one epidemic of seabather’s eruption in Long Island, New York. This organism also has nematocysts; thus the mechanism of the eruption is the same as with the jellyfish-induced eruption. It is likely that different cnidarian envenomations in different waters produce a similar clinical picture. Other reports focus on spring plants, dinoflagellates, protozoans, or crustaceans as potential causes. Because the eruption results from trapping of cnidarian larvae with their nematocysts or other toxic or irritant substances under the bathing suit, it may be limited by seabathers who remove their suit and shower soon after leaving the water.
Hydroid, Sea Anemone, and Coral Dermatitis
Patients contacting the small marine hydroid Halecium may develop a dermatitis. The organism grows as a 1-cm–thick coat of moss on the submerged portions of vessels or pilings. Sea anemones ( Fig. 20.12 ) produce reactions similar to those from jellyfish and hydroids. Coral cuts are injuries caused by the exoskeleton of the corals Milleporina. They have a reputation for becoming inflamed and infected and for delayed healing. The combination of implantation of fragments of coral skeleton and infection (because cuts occur most often on feet) probably accounts almost entirely for these symptoms. Detoxification as soon as possible after the injury is recommended for all these types of sting or cut.

Treatment of Stings and Cuts
Hot water immersion may be an effective remedy for many stings, but scald injuries must be avoided. In the case of box jellies, ice has been shown to be equally effective, but ice can worsen stings of some other jellies. Undischarged nematocytes should be removed. Fresh water, and even sea water, may cause them to discharge. Pacific Chironex (box jellyfish) nematocytes should always be inactivated with 5% acetic acid (vinegar) when it is available, but Pacific Physalia (bluebottle) nematocytes may discharge on contact with vinegar. Large, visible tentacles may be removed with forceps in a double-gloved hand. Remaining nematocysts may be removed by applying a layer of shaving cream and shaving the area gently. Meat tenderizer may cause tissue damage and has been shown to be no better than placebo in some studies.
Pressure dressings and abrasion will worsen the envenomation. Topical anesthetics or steroids may be applied after decontamination. Systemic reactions may occur through either large amounts of venom or a previously sensitizing exposure from which anaphylaxis may result, and systemic treatment with epinephrine, antihistamines, or corticosteroids may be needed. Specific antivenin is available for the box jellyfish, Chironex fleckeri. This should be administered intravenously to limit myonecrosis. Magnesium sulfate (MgSO 4 ) may also be of value in the setting of box jellyfish envenomation. Recurrent jellyfish reactions have shown partial responses to tacrolimus ointment 0.1%.
Sponges and Bristleworms
Sponges have horny spicules of silicon dioxide and calcium carbonate. Some sponges produce dermal irritants, such as halitoxin and okadaic acid, and others may be colonized by Cnidaria. Allergic or irritant reactions may result. Bristleworms may also produce stinging. All these may be treated by first using adhesive tape to remove the spicules, then applying vinegar soaks, as previously described, and lastly, topical corticosteroids.
Sea Urchin Injuries
Puncture wounds inflicted by the brittle, fragile spines of sea urchins, mainly of genus Diadema or Echinothrix, are stained blue-black by the black spines and may contain fragments of the spines. The spines consist of calcium carbonate crystals, which most frequently induce an irritant reaction with pain and inflammation of several days’ duration. Foreign body or sarcoid-like granulomas may develop ( Fig. 20.13 ), as may a vesicular hypersensitivity reaction, 10 days after exposure. Injuries by spines of the genus Tripneustes have been reported to cause fatal envenomation, but this genus is not found on U.S. coasts.
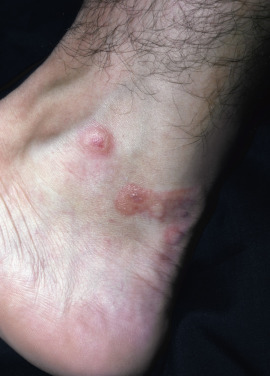
Starfish also have thorny spines that can sting and burn if they are stepped on or handled. Several different types of stinging fish also produce puncture wounds. Stingrays, scorpionfish, stonefish, catfish, and weaverfish may cause such envenomations.
These wounds should be immersed in nonscalding water (45°C [113°F]) for 30–90 minutes or until the pain subsides. Calcified fragments may be visible on x-ray evaluation, with fluoroscopy guiding extraction of spines, especially on the hands and feet. Sea urchin spines have been effectively removed using the erbium:yttrium-aluminum-garnet (YAG) laser. Debridement and possibly antibiotic therapy for deep puncture wounds of the hands and feet are recommended. There is a specific antivenin for stonefish stings.
Seaweed Dermatitis
Although caused by a marine alga and not by an animal, seaweed dermatitis deserves mention with other problems associated with swimming or wading. The dermatitis occurs 3–8 hours after the individual emerges from the ocean. The distribution is in parts covered by a bathing suit: scrotum, penis, perineum, and perianal area. The dermatitis is caused by a marine plant, Lyngbya majuscula Gomont. It has been observed only in bathers swimming off the windward shore of Oahu, Hawaii. Seabather’s eruption, clam digger’s itch, and swimmer’s itch must be differentiated from seaweed dermatitis caused by marine algae. Prophylaxis is achieved by refraining from swimming in waters that are turbid with such algae. Swimmers should shower within 5 minutes of swimming. Active treatment in severe cases is the same as for acute burns.
Dogger Bank Itch
Dogger Bank itch is an eczematous dermatitis caused by the sea chervil Alcyonidrium hirsutum, a seaweed-like animal colony. These sea mosses or sea mats are found on the Dogger Bank, an immense shelflike elevation under the North Sea between Scotland and Denmark.
Isbister GK, et al: Hot water immersion v icepacks for treating the pain of Chironex fleckeri stings. Med J Aust 2017; 206: 258.
Jefferson J, et al: Coral contact dermatitis. Dermatol Online J 2015; 21.
Little M, et al: Successful use of heat as first aid for tropical Australian jellyfish stings. Toxicon 2016; 122: 142.
Yanagihara AA, et al: Cubozoan sting-site seawater rinse, scraping, and ice can increase venom load. Toxins (Basel) 2017; 9.
Phylum Platyhelminthes
Phylum Platyhelminthes includes the flatworms, of which two classes, trematodes and cestodes, are parasitic to humans. The trematodes, or blood flukes, parasitize human skin or internal organs. The cestodes are segmented, ribbon-shaped flatworms that inhabit the intestinal tract as adults and involve the subcutaneous tissue, heart, muscle, and eye in the larval form. This is encased in a sac that eventually becomes calcified.
Class Trematoda
Schistosome Cercarial Dermatitis
Cercarial dermatitis is a severely pruritic, widespread, papular dermatitis caused by cercariae of schistosomes for which humans are not hosts; the usual animal hosts are waterfowl and rodents, such as muskrats. The eggs in the excreta of these animals, when deposited in water, hatch into swimming miracidia. These enter a snail, where further development occurs. From the snail, the free-swimming cercariae emerge to invade human skin on accidental contact. The swimming, colorless, multicellular organisms are slightly less than 1 mm long. Exposure to cercariae occurs when a person swims or, more often, wades in water containing them. They attack by burrowing into the skin, where they die. The species that causes this eruption cannot enter the bloodstream or deeper tissues.
After coming out of the water, the bather begins to itch, and a transient erythematous eruption appears, but after a few hours, the eruption subsides, together with the itching. After a quiescent period of 10–15 hours, the symptoms then recur, and erythematous macules and papules develop throughout the exposed parts that were in the water ( Fig. 20.14 ). After several days, the dermatitis heals spontaneously. There are two types: the freshwater swimmer’s itch and the saltwater marine dermatitis, or clam digger’s itch. Cercarial dermatitis is not communicable.
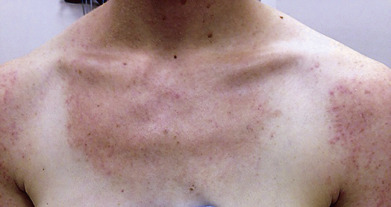
Various genera and species of organism have been reported from various locations worldwide. An outbreak of cercarial dermatitis was reported from Delaware in 1991 in which the avian schistosome Microbilharzia variglandis was implicated as the causative organism. Schistosoma spindale cercaria caused a recent epidemic in southern Thailand.
Thoroughly washing, then drying with a towel after exposure, can prevent the disease. Some advocate rubbing with alcohol as an additional preventive measure. Snail populations can be controlled, or waterfowl may be treated with medicated feedcorn to destroy the adult schistosomes and prevent outbreaks of swimmer’s itch.
Visceral Schistosomiasis (Bilharziasis)
The cutaneous manifestation of schistosomiasis may begin with mild itching and a papular dermatitis of the feet and other parts after swimming in polluted streams containing cercariae. The types of schistosome causing this disease can penetrate into the bloodstream and eventually inhabit the venous system, draining the urinary bladder (Schistosoma haematobium) or the intestines ( Schistosoma mansoni or Schistosoma japonicum ). After an asymptomatic incubation period, the person may develop a sudden illness with fever and chills, pneumonitis, and eosinophilia. Petechial hemorrhages may occur.
Cutaneous schistosomal granulomas most frequently involve the genitalia, perineum, and buttocks. The eggs of S. haematobium or S. mansoni usually cause these bilharziomas ( Fig. 20.15 ). Vegetating, soft, cauliflower-shaped masses, fistulous tracts, and extensive hard masses occur; these are riddled by sinuses that exude a seropurulent discharge with a characteristic odor. Phagedenic ulcerations and pseudoelephantiasis of the scrotum, penis, or labia are sometimes encountered. Histologically, the nodules contain bilharzial ova undergoing degeneration, with calcification and a surrounding cellular reaction of histiocytes, eosinophils, and occasional giant cells. In some cases, eventual malignant changes have been noted in chronic lesions. Animal studies have shown a moderate helper T-cell type 1 (Th1) response to parasite antigens in most tissues, but a strong Th2 response that propagates fibrogenesis within the liver. Infrequently, ectopic or extragenital lesions may occur, mainly on the trunk. This is a papular eruption tending to group in plaques and become darkly pigmented and scaly. A severe urticarial eruption known as urticarial fever or Katayama fever is frequently present along with S. japonicum infection; it occurs with the beginning of oviposition, 4–8 weeks after infection. This condition is seen mainly in China, Japan, and the Philippines. In addition to the urticaria, fever, malaise, abdominal cramps, arthritis, and liver/spleen involvement are seen. This is thought to be a serum sickness–like reaction.
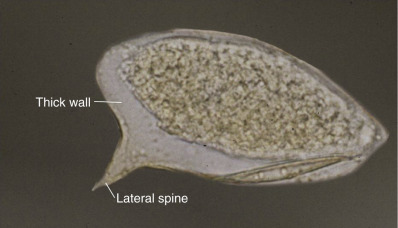
Preventive measures include reducing infection sources, preventing contamination by human excreta of snail-bearing waters, control of snail hosts, and avoiding exposure to cercaria-infested waters. Prophylactic measures are constantly sought to control one of the world’s worst parasitic diseases, but as yet, none has been found to be practical. For both S. haematobium and S. mansoni, praziquantel (Biltricide), 40 mg/kg orally for each of two treatments in 1 day, is the therapy of choice. S. japonicum treatment requires 60 mg/kg in three doses in 1 day. Schistosomicides exhibit toxicity for the host as well as for the parasite, and the risk of undesirable side effects may be enhanced by concomitant cardiac, renal, or hepatosplenic disease.
Kollipara R, et al: Emerging infectious diseases with cutaneous manifestations. J Am Acad Dermatol. 2016; 75: 19.
Patel TA, et al: Treatment of schistosomiasis in a patient allergic to praziquantel. Am J Trop Med Hyg 2016; 95: 1041.
Cysticercosis Cutis
The natural intermediate host of the pork tapeworm, Taenia solium, is the pig, but under some circumstances, humans act in this role. The larval stage of T. solium is Cysticercus cellulosae. Infection takes place by the ingestion of food contaminated with the eggs or by reverse peristalsis of eggs or proglottides from the intestine to the stomach. Here the eggs hatch, freeing the oncospheres. These enter the general circulation and form cysts in various parts of the body, such as striated muscles, brain, eye, heart, and lung.
In the subcutaneous tissues, the lesions are usually painless nodules that contain cysticerci. They are more or less stationary, usually numerous, and often calcified and are therefore demonstrable radiographically. Pain and ulceration may accompany the lesions. The disease is most prevalent in countries where pigs feed on human feces. It may be confused with gumma, lipoma, and epithelioma. A positive diagnosis is established solely by incision and examination of the interior of the calcified tumor, where the parasite will be found. Fine-needle aspiration has also been used to establish the diagnosis.
Albendazole or praziquantel is effective; however, the status of the CNS, spinal, and ocular involvement needs to be thoroughly assessed before treatment. The length of therapy and use of concomitant corticosteroids depend on the location of the cysts. However, none of the regimens clears the calcified parasites, which need to be surgically removed.
Sparganosis
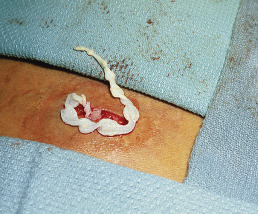
Sparganosis is caused by the larva of the tapeworm Spirometra. The adult tapeworm lives in the intestines of dogs and cats. This is a rare tissue infection occurring in two forms. Application sparganosis occurs when an ulcer or infected eye is poulticed with the flesh of an infected intermediate host (such poultices are frequently used in the Orient). The larvae become encased in small nodules in the infected tissue. Ingestion sparganosis occurs when humans ingest inadequately cooked meat, such as snake or frog, or when a person drinks water that is contaminated with Cyclops, which is infected with plerocercoid larvae. One or two slightly pruritic or painful nodules may form in the subcutaneous tissue or on the trunk, breast, genitalia, or extremities. Cerebral disease may also occur. Diagnosis is usually made by excision of the nodule, although noninvasive imaging has also been used.
Humans are the accidental intermediate host of the sparganum, which is the alternative name for the plerocercoid larva. Treatment is surgical removal or ethanol injection of the infected nodules ( Fig. 20.16 ). This may be difficult because of the swelling and extensive vascularity.
Echinococcosis
Echinococcosis is also known as hydatid disease. In humans, infection is produced by the ova reaching the mouth from the hands, in food, or from containers soiled by ova-contaminated feces from an infected dog. This leads to Echinococcus granulosus infestation of the liver and the lungs. Soft, fluctuating, semitranslucent, cystic tumors may occur in the skin, sometimes in the supraumbilical area as fistulas from underlying liver involvement. These tumors become fibrotic or calcified after the death of the larva. Eosinophilia, intractable urticaria and pruritus, and even acute generalized exanthematous pustulosis may be present. Such reactive findings may be present as skin manifestations of many of the helminthic infections, including other types of tapeworm. The treatment is excision, with care being taken to avoid rupturing the cyst. Albendazole combined with percutaneous drainage may also be used. Hymenolepis nana is a cosmopolitan dwarf tapeworm endemic in the tropics that may cause a treatment-resistant pruritic papular eruption associated with eosinophilia. Stool specimens for ova and parasites are definitive, and praziquantel is curative.
Moosazadeh M, et al: Epidemiological and clinical aspects of patients with hydatid cyst in Iran. J Parasit Dis 2017; 41: 356.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree