Introduction
Oncoplastic breast conservation surgery combines oncologic principles with plastic surgical techniques. But it is much more than a combination of two disciplines – it is a philosophy that requires vision, passion, a knowledge of anatomy, and an appreciation and understanding of aesthetics, symmetry and breast function. The oncoplastic surgeon must be constantly thinking ‘How can I remove this cancer with large margins of normal tissue while at the same time making the patient look as good or better than she looks now?’ ( Box 5.1 ).
The goals of oncoplastic breast conservation surgery include:
- 1.
Complete removal of the lesion
- 2.
Clear margins – the larger the better
- 3.
Good to excellent cosmetic result
- 4.
Going to the operating room one time to perform the definitive procedure
The ultimate oncoplastic achievement would be to convert what would normally be an oncologic and/or cosmetic failure using standard techniques into both an oncologic and cosmetic success. Avoiding mastectomy when it seems inevitable and ending with an excellent cosmetic result is one way to achieve that goal. The following case demonstrates just such a scenario.
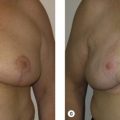
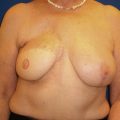
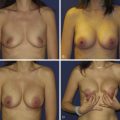
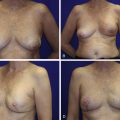
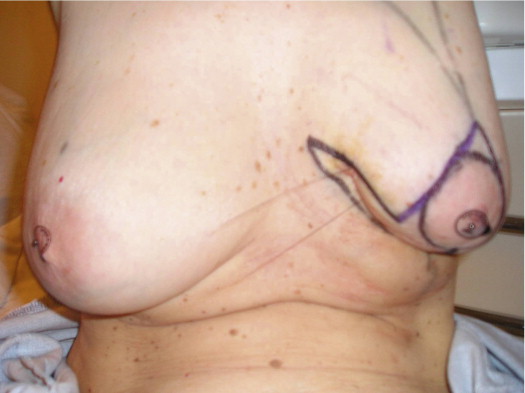
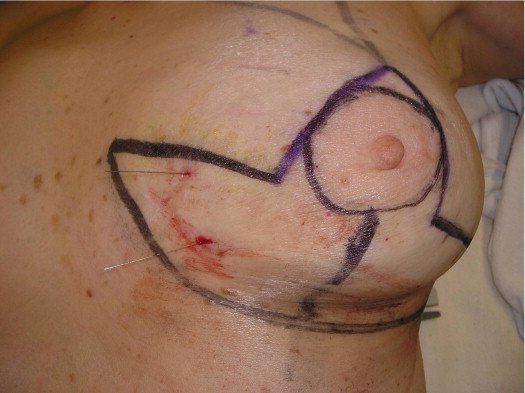
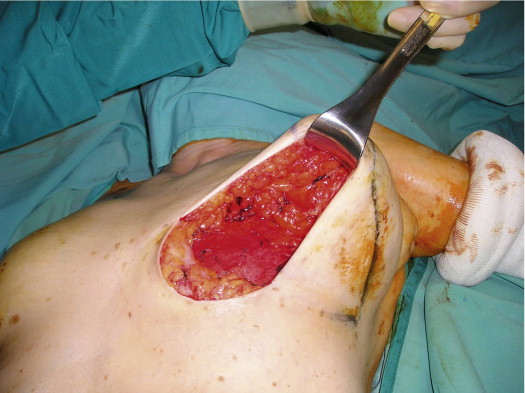
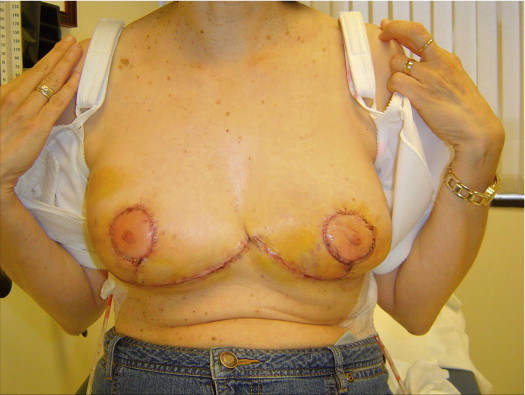
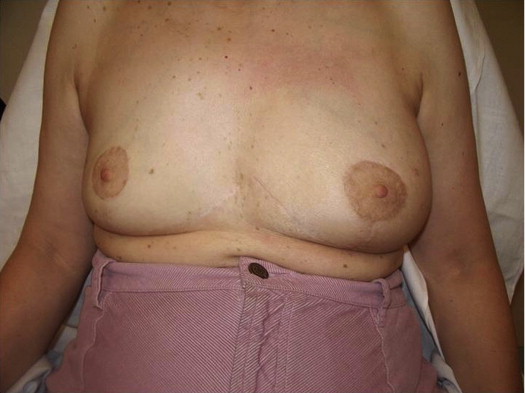
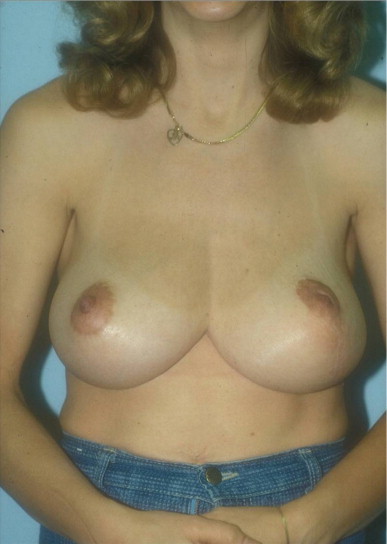
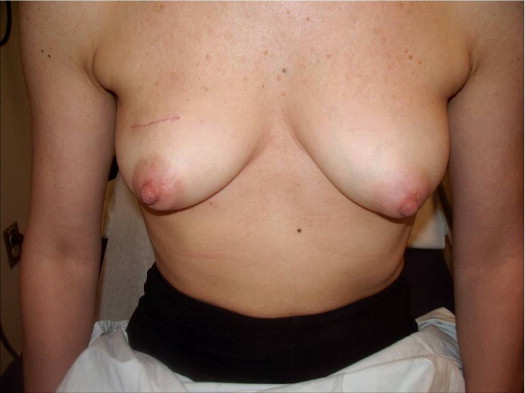
A 58-year-old female presented with recurrent ductal carcinoma in situ (DCIS) in the left lower inner quadrant of the breast. A previous excision had been performed 2 years earlier obtaining a minimal margin of clearance. The left breast was severely deformed following this procedure ( Fig. 5.1 ). Following the local recurrence, she was told by multiple surgeons that mastectomy was the only option. After re-evaluation, including digital mammography, ultrasound, and magnetic resonance imaging (MRI), it was determined that the only disease present was at the edge of the previous excision. A left wire-directed segmental resection using a reduction incision with a contralateral (right) reduction was performed ( Figs 5.2–5.5 ). This procedure excised the residual DCIS with a margin of excision that exceeded 10 mm in all directions. The cosmetic appearance was much improved and was persistent 4 years later ( Fig. 5.6 ).
The history of oncoplastic breast conservation surgery in the United States
There is little written about the origins of oncoplastic breast conservation surgery in the United States. We began developing oncoplastic techniques in the early 1980s by accident.
The first free-standing Breast Center in the United States was founded in Van Nuys, California, in 1979. Our group consisted of plastic surgeons, oncologic surgeons, medical oncologists, radiologists, radiation oncologists, and a psychiatrist. In the early years, due to staffing issues it was not uncommon for an oncologic surgeon to assist a plastic surgeon with a reduction and for a plastic surgeon to assist an oncologic surgeon with a mastectomy or axillary dissection. Because of this, both disciplines (plastic and oncologic surgery) became quite familiar with what the other discipline could accomplish.
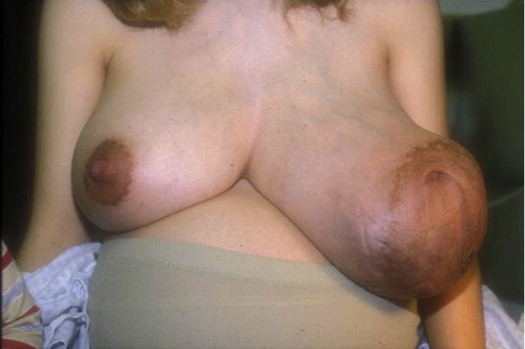
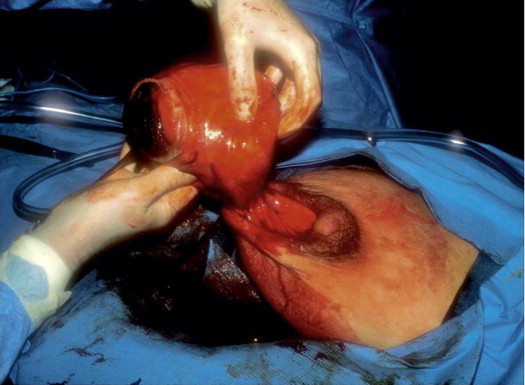
Our first true oncoplastic case occurred in 1982. A young pregnant woman was referred with a biopsy-proven giant fibroadenoma of pregnancy ( Fig. 5.7 ). We waited until she delivered her baby and then a few weeks later a team of one oncologic and one plastic surgeon removed the benign tumor using a reduction approach and simultaneously reduced the opposite side ( Fig. 5.8 ). Owing to the large size of the tumor (about 20 cm), the nipples ended up a bit too high, but overall it was an outstanding oncoplastic result ( Fig. 5.9 ).
Shortly thereafter, one of our plastic surgeons did a reduction by removing a large segment of superior breast tissue in an older woman who did not want a standard reduction. When asked what he called this strange incision, he answered ‘Batwing … because it looks like the Batman symbol.’ At the same time, we were learning more about the importance of widely clear margins, something that had not been appreciated prior to the mid 1980s. We quickly adopted the ‘batwing’ for women with breast cancer in the upper half of a larger breast that could benefit from lifting of the nipple–areola complex (NAC). The rest, as they say, is history. We rapidly added more and varied excisions to our ‘oncoplastic’ armamentarium, many of which will be illustrated below.
Oncoplastic resection
When treating a patient with biopsy-proven breast cancer, the non-oncoplastic approach would be to make a small cosmetically placed curvilinear incision over the area to be removed ( Fig. 5.10 ). This would typically include no skin and a relatively small piece of breast tissue. The definition of a clear margin was based on non-transection of the tumor. Complete and sequential tissue processing is not usually performed and postoperative radiation therapy is the usual protocol.
But the trend is changing. During the last 25 years, my colleagues and I have developed a comprehensive multidisciplinary oncoplastic approach for the excision of breast cancer. It requires surgical coordination with a pathologist, a radiologist and, often, a plastic surgeon. Oncoplastic surgery combines sound surgical oncologic principles with plastic surgical techniques. Coordination of the two surgical disciplines may help to avoid poor cosmetic results after wide excision and may increase the number of women who can be treated with breast-conserving surgery by allowing larger breast excisions with more acceptable cosmetic results. These techniques are applicable to patients with both noninvasive (DCIS) and invasive breast cancer.
Oncoplastic resection is a therapeutic procedure, not a breast biopsy. It is performed on patients with a proven diagnosis of breast cancer. This approach was strongly supported by the 2005 Consensus Conference on Image-Detected Breast Cancer. An important goal in caring for a woman with breast cancer is to go to the operating room a single time and to perform a definitive procedure that does not require re-operation. Whenever possible, the initial breast biopsy should be made using a minimally invasive percutaneous technique. This usually provides ample tissue for diagnosis.
When excising breast cancer, the surgeon faces two opposing goals: clear margins versus an acceptable cosmetic result. From an oncologic point of view, the largest specimen possible should be removed in an attempt to achieve the widest possible margins. From a cosmetic point of view, a much smaller amount of tissue should be removed in order to achieve the best possible cosmetic result. The surgeon must tread a fine line as he or she tries to satisfy two masters. The first attempt to remove a cancerous lesion is critical. The first excision offers the best chance to remove the entire lesion in one piece, evaluate its extent and margin status, and to achieve the best possible cosmetic result.
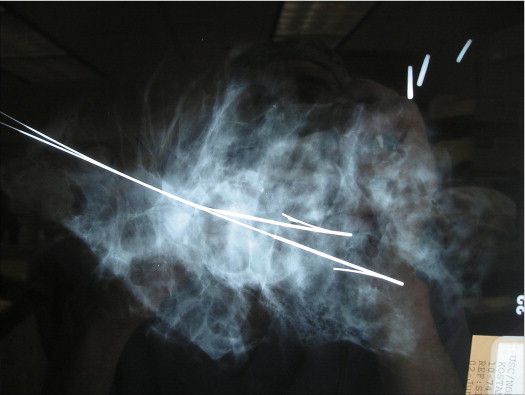
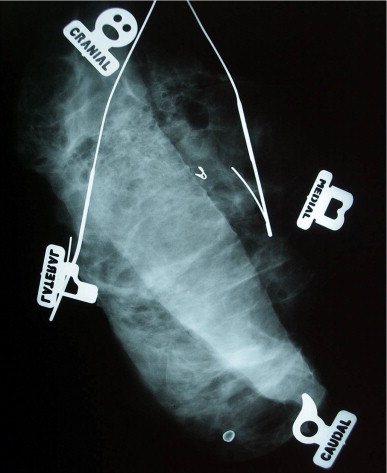
Currently, as many as 40–50% of new breast cancer cases are discovered by modern state-of-the-art imaging (mostly mammography) and, intraoperatively, are grossly both non-palpable and non-visualizable. Under these circumstances, the surgeon essentially operates blindly. Multiple hooked wires can help define the extent of the lesion radiographically. Using bracketing wires, the surgeon should make an attempt to excise the entire lesion within a single piece of tissue. This often will include overlying skin as well as pectoral fascia ( Figs 5.11 and 5.12 ). The tissue should be precisely oriented for the pathologist.
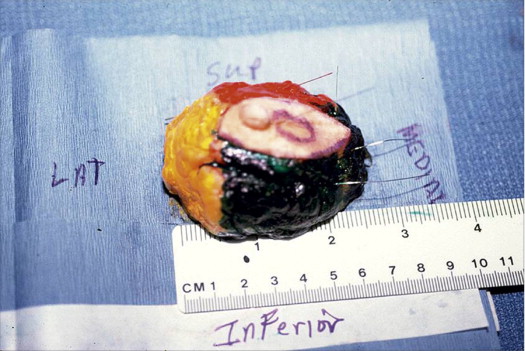
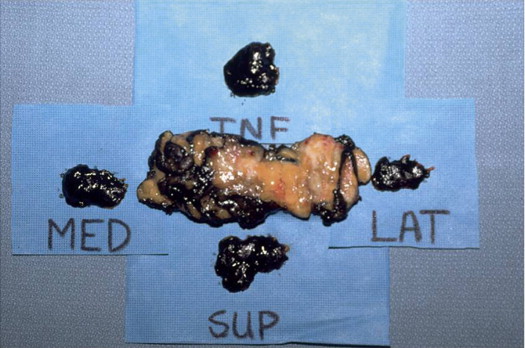
If the specimen is removed in multiple pieces rather than a single piece, there is little likelihood of evaluating margins and size accurately. Figure 5.13 shows an excision specimen with four additional pieces that allegedly represent the new margins. The additional pieces are too small and do not reflect the true margins of the original specimen. If one makes a judgment on margin clearance based on these small additional pieces, that judgment might very well be incorrect.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree