The management of recurrent breast cancer is a multidisciplinary challenge. Clinical and radiological follow-up of patients following breast cancer treatment aims to detect early locoregional and distant metastasis. Local breast recurrence represents the reappearance of cancer and can occur at or near the site of excision following breast-conserving surgery (BCS) or in the soft tissues of the anterior chest wall or skin flap following mastectomy. In the case of oncoplastic surgery, the majority of local recurrence appears at the initial site where the resection was performed.
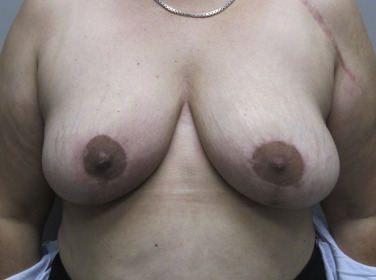
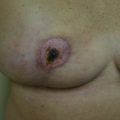
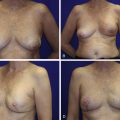
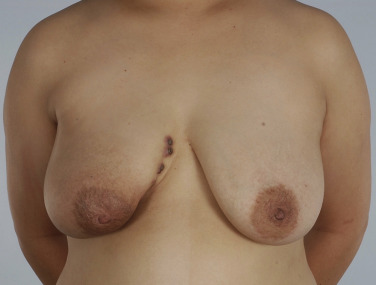
The clinical presentation of a patient with a local recurrence may vary and includes a new lump or firmness within the operated breast, nipple inversion or discharge, as well as skin changes, for example, erythema, rash, firm nodules, oedema, tethering, or thickening of scar tissue ( Figs. 18.1 and 18.2 ). Differential diagnoses include postoperative fibrosis, fat necrosis, suture granulomas, benign breast disease, and post-radiotherapy skin changes. Persistent edema and erythematous skin overlying the operated breast should raise the suspicion of inflammatory recurrent breast cancer and warrants punch biopsy; however, this can be difficult to differentiate from mastitis and post-radiotherapy skin changes. Even rarer is cutaneous metastasis “en cuirasse” located on thoracic and abdominal walls characterized by infiltrated, hard, and sclerodermiform plaques.
Mammography is the mainstay of surveillance imaging following BCS detecting 8–50% of ipsilateral recurrences, whereas ultrasound and magnetic resonance imaging remain supplementary surveillance modalities. Mammographic appearances of ipsilateral recurrence include new nondystrophic microcalcifications, a new mass compared with baseline mammogram, increased architectural distortion or opacity, and an increase in skin thickening after posttreatment changes have subsided. Not all local recurrences will be associated with mammographic change, and a normal mammogram does not exclude local recurrence. Stereotactic and core biopsies histologically confirm the diagnosis of local recurrence.
Local recurrence rates following surgery vary widely in the literature due to differences in patient cohort selection, extent of surgery, and use of adjuvant therapies. The incidence of local recurrence after BCS and radiotherapy ranges from 10–22% at 10 years and, after mastectomy, ranges from 5–15% at 10 years. The median time to recurrence is 3–4 years for BCS and 2–3 years for mastectomy. Breast recurrence is a predictor of distant metastasis and decreased survival. A meta-analysis by the Early Breast Cancer Trialists’ Collaborative Group demonstrated a negative effect of a local recurrence on survival. In 5–15% of cases, local recurrence is associated with concomitant regional and distant metastasis; therefore, computed tomography and positron emission tomography staging is considered for all patients with confirmed local recurrence.
The a etiology of local recurrence remains unclear; however, theories include incomplete excision of the primary tumor, unrecognized multifocal disease, entrapment of tumor cells within obstructive lymphatics, and local implantation of systemic circulating cells. In the context of local recurrence alone, excision surgery and adjuvant therapies have curative intent. For patients presenting with local recurrence and distant metastasis, a multidisciplinary approach will be required to assess the need for palliative resection of the local recurrence as an adjunct to systemic treatment.
Determinants of Local Recurrence
The importance of negative margins in BCS cannot be overstated, and positive margins are a risk factor for increased local recurrence. Positive margins according to the joint guidelines of the American Society of Breast Surgeons/Society of Surgical Oncology/American Society of Radiation Oncology constitute “ink on tumor” for invasive breast cancer and less than 2 mm margins for ductal carcinoma in situ. Current UK guidelines from the Association of Breast Surgery consensus define positive margins as 1 mm for both invasive and ductal carcinoma in situ. A systematic review on oncologic reporting in BCS highlighted variation in the frequency of margin involvement (0–36%) and local recurrence rates of 0–10.8%. Positive margins (ink on invasive or ductal carcinoma in situ) are associated with a twofold increase in the risk of ipsilateral recurrence compared with negative margins. More widely clear margins than no ink on tumor do not significantly decrease the rate of recurrence compared with no ink on tumor even in high-risk groups with unfavorable biology. Local recurrence is influenced more by tumor biology and therapy than surgical margin. Young age at diagnosis (less than 40 years) is a significant risk factor for local recurrence at 5 years with a relative risk of 2.21 (95% CI 1.62–3.02). Body mass index gain following BCS is also significantly associated with higher rates of recurrence.
Pan et al performed a meta-analysis of the results of 88 trials involving 62,923 women with estrogen receptor (ER)-positive breast cancer who were disease-free after 5 years of scheduled endocrine therapy. The risk of disease recurrence, both locally and distant, was strongly correlated with the original TMN classification (tumour, node, metastasis) status and tumor grade. During the study period from 5–20 years, the absolute risk of local recurrence with T1N0 ER positive breast cancer or a contralateral breast cancer were low grade 17%, moderate grade 22%, and high grade 26%. Tumor grade and size are significant predictors of recurrence after adjustment to other variables.
Breast tumors can be divided into subtypes based on molecular profiling, particularly those differing in proliferation. Measurement of the level of activation of the proliferation pathway is via Ki-67 expression. Ki-67 is a nuclear protein associated with cellular proliferation. Immuno-staining can assess the percentage of breast cancer cells expressing Ki67 (<14%; >14%). Molecular subtypes of breast cancer include luminal A (ER-positive or progesterone receptor [PR]-positive and Ki-67 < 14%); luminal B (ER- or PR-positive and Ki-67 ≥ 14%); luminal human epidermal growth factor receptor 2 (HER2) (ER- or PR-positive and HER2-positive); HER2-enriched (ER-negative, PR-negative, and HER2-positive); and basal-like (ER-, PR-, and HER2-negative, triple negative). Luminal B, HER2 positive, and triple negative subtypes all show significant increased risk for both local recurrence and distant recurrence following BCS. Five-year local recurrence rates following BCS and radiotherapy for each subtype have been reported: 0.8% for luminal A, 2.3% for luminal B, 1.1% for luminal HER2, 10.8% for HER2-enriched, and 6.7% for triple-negative disease. Similarly, after mastectomy, patients with luminal A tumors had the lowest rates of local recurrence, 8% at 10 years. Gene expression profiling by microarray analysis is being used to identify gene expression profiles that can predict local recurrence. Although still being refined, researchers have been able to identify subgroups of patients at increased risk of developing local recurrence following BCS.
Indications and Techniques for Second Oncoplastic Breast Surgery
The standard treatment recommended for ipsilateral recurrence following BCS is salvage mastectomy with radiotherapy to the chest wall if not already administered previously. The management plan following salvage surgery is formulated in a multidisciplinary approach to determine the benefits of radiotherapy, endocrine, and chemotherapy on a case-by-case basis with the aim to reduce secondary recurrence and distant metastasis. The adjuvant treatment strategy will take into account the biology of the original tumor, time to recurrence, patient TN status, age, previous adjuvant treatments, and comorbidities.
Patients presenting with ipsilateral recurrence in the tumor bed may decline salvage mastectomy and seek alternative treatment options. For this small subgroup of patients, repeat breast-conserving surgery (rBCS) with or without additional radiotherapy may be considered. Patients with inflammatory breast cancer, small volume breasts, multicentric or multifocal recurrence, skin involvement, or poor tumor biology (e.g., high grade, triple negative, HER2-positive) are not candidates for this approach. Patient choice would be the main determining factor to consider a second conservative surgery and is not to be offered routinely. The evidence base supporting rBCS is limited and therefore cannot currently be recommended over mastectomy. Small unifocal recurrence in a large volume breast, a long interval to recurrence, and amenable tumor location would be factors enabling rBCS to be technically feasible. Cosmesis in the re-operated breast may be compromised in rBCS due to reduced pliability of the breast parenchyma, significant volume disparity between breasts, and post-radiotherapy changes to the skin envelope. There are no published level 1 studies comparing salvage mastectomy versus rBCS ± radiotherapy for patients with ipsilateral breast recurrence. Low incidence of recurrence and heterogeneity of the recurrence population, for example, timing to recurrence, tumor staging and biology, means recruitment, and design of a large-scale study comparing the two treatments, proves difficult. Two phase II prospective trials investigating the use of partial breast re-irradiation following rBCS are in progress ( https://clinicaltrials.gov ).
The use of secondary repeat breast conservation surgery for local recurrence was first described by Kurtz et al in a retrospective analysis of 52 patients. Wide local excision alone was performed for stage I–II breast cancer patients for small, slow-growing, ipsilateral recurrence. They reported no significant differences between mastectomy and rBCS for 10-year overall survival (54% and 64%, respectively). A higher rate of secondary recurrence was however observed in the rBCS without re-irradiation (38% vs 25%). Since then, the majority of published studies on this topic are small retrospective, non-randomized cohort studies. They are subject to selection bias and poor study quality. Patients receiving rBCS are more often to have better prognostic characteristics compared with those treated with mastectomy (e.g., size, multifocality, aggressive tumor biology). A recent retrospective cohort study by Kolben et al has reported data supporting the use of rBCS in selected patients. Of 170 patients, 34.1% of women underwent secondary BCS, whereas 65.9% were treated by salvage mastectomy. Five-year ipsilateral recurrence-free rate after secondary BCS was 77.6% (Standard Deviation (SD) ± 6.1%) and 75.0% (SD ± 4.5%) for patients after mastectomy. Five-year disease-free survival was 57.3% (SD ± 8.2%), and 61.9% (SD ± 5.5%); 5-year overall survival was 84.7% (SD ± 5.8%), and 72.6% (SD ± 5.1%), respectively. Ishitobi et al reported a 20% local recurrence rate following rBCS alone. Survival rates following rBCS compared with salvage mastectomy are conflicting. Alpert et al reported a 64.5% survival rate at 10 years with no significant difference between salvage mastectomy and rBCS observed in 271 patients undergoing rBCS or mastectomy for recurrence (55% vs 45%). Yoshida et al reported no significant difference in overall survival between the two surgical approaches after adjustment for clinical and tumor characteristics. In contrast, Chen et al observed significantly better survival rates at 5 years following salvage mastectomy when compared with rBCS (78% vs 67%, p = 0.003).
Kurtz et al reported risk factors associated with secondary recurrence following rBCS alone. Through multivariate analysis, the group reported that the disease-free interval to recurrence and resection margins significantly influence local control following rBCS. Five-year local control was 92% for recurrences occurring after 5 years versus 49% for shorter intervals, and 73% for negative margins versus 36% for positive or indeterminate margins. The authors concluded that wide excision represents an alternative to mastectomy in salvage treatment for selected patients with mobile tumors, 2 cm or smaller in diameter, and no signs of rapid growth. Prior adjuvant systemic therapy, skin or muscle infiltration, and lymphovascular infiltration in the breast recurrence are additional risk factors for shorter disease-free survival following rBCS.
Re-irradiation following rBCS is an emerging concept in the treatment of recurrence, although it remains in an experimental phase. Repeat irradiation to the resected breast cavity following rBCS is a technique applied to reduce secondary local recurrences first reported in the 1990s. Ishitobi et al reported 5-year second recurrence survival rates are improved with post-rBCS radiotherapy; radiotherapy after initial surgery, radiotherapy after salvage surgery, and no radiotherapy were 78.0%, 93.5%, and 52.7%, respectively. There is no consensus regarding the optimal treatment for patients who have previously had BCS with irradiation. The normal tissue tolerance does not permit a second full-dose course of radiotherapy to the entire breast after a second BCS. Re-irradiation to the entire breast is poorly tolerated due to poor cosmetic outcome and high toxicity rates. New modes of delivering focal radiotherapy to the breast have raised the possibility of localized radiation therapy for women post-rBCS. Accelerated partial breast irradiation (APBI) is proposed as an alternative to whole breast re-irradiation. APBI limits irradiation to the target area post-wide local excision plus 1–2 cm margin of tissue in a shorter time period while delivering equivalent doses to whole breast irradiation. A number of techniques are reported including multicatheter interstitial brachytherapy (IB), balloon catheter brachytherapy, intraoperative radiation therapy (IORT), and external beam irradiation (EBI). A recent Cochrane review summarizing evidence for partial breast irradiation (PBI) and APBI for treatment of primary breast cancer versus whole breast radiotherapy highlighted the limitations of the data currently available in this field. The authors reported cosmetic outcomes and late effects were worse with PBI/APBI, but less skin toxicity was noted. Local recurrence was increased with PBI/APBI (the difference was small) with no evidence of detriment in other oncologic outcomes.
Studies applying this technology to recurrent breast cancer treatment following rBCS are limited to small case series, varying follow-up durations, and subject to selection bias. The majority of studies report results for rBCS with brachytherapy for patients with small T0-2 recurrent lesions, no metastasis, and negative resection margins with late onset of recurrence (mean 70 months). Authors report EBI with 45 Gy (1.8 Gy per fraction) to rBCS is generally well tolerated with low acute and late skin toxicities and good local disease control. The prognostic factors for favorable overall survival rates were younger age ( p = 0.045), lower T category ( p = 0.019), and N0 category ( p = 0.005).
Hannoun-Levi et al have reported promising results for 217 women treated with multicatheter brachytherapy following rBCS. Secondary local recurrence rates were 5.6% (1.5–9.5) at 5 years and 7.2% (2.1–12.1) at 10 years. Overall survival rates for 5 and 10 years were 88.7% (83.1–94.8) and 76.4% (66.9–87.3%), respectively, and report comparable results to salvage mastectomy. Similar overall survival and disease-free survival rates have been reported in other small case series. Grade 1–2 toxicities are observed in the majority of patients (skin atrophy, pigmentation, telangiectasia, slight induration, and loss of subcutaneous fat). Approximately 11% of women experience grade 3 or 4 toxicities (marked skin atrophy, gross telangiectasia, ulceration, severe fibrosis, field contracture, necrosis). The most common side effect reported from IB is cutaneous and subcutaneous fibrosis (up to 60%) as a function of dose and volume, which may considerably affect overall cosmetic outcome following secondary oncoplastic surgery due to parenchymal tethering. Factors affecting cosmesis include the distance of the catheter or balloon from the skin, the type of device used, the volume of residual breast tissue after rBCS, and degree of initial breast asymmetry. Complications related to device include persistent breast pain, a chronic abscess in the sinus tract of the balloon catheter, and infections of the catheter tract requiring mastectomy and antibiotics, respectively. Proponents of the technique report stable cosmetic scores (from excellent to good) and patient satisfaction in 60–80% of patients; however, most studies did not use standardized evaluation schemes. In 2007, Kraus-Tiefenbacher et al reported on the use of IORT following rBCS. Fifteen patients were treated after previous EB radiotherapy after ipsilateral breast recurrence. At median follow-up, short, acute toxicity 26 months after IORT was reported as mild with no grade 3–4 toxicities in the group. Further high quality research is required to support rBCS with or without re-irradiation in routine clinical practice.
Reconstructive Options Following Salvage Mastectomy for Recurrence
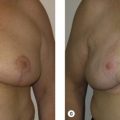
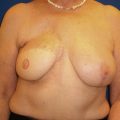
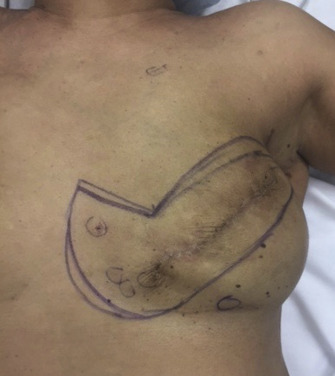
Despite international guidelines recommending salvage mastectomy following recurrence, there are no level 1 studies to guide surgeons’ choice for reconstruction following BCS and radiotherapy, and data are limited to observational studies. Decisions regarding reconstruction in this cohort should be made within a multidisciplinary team with a tailored individual approach. Options regarding the timing and types of reconstruction vary considerably among surgeons, particularly in this context. Authors emphasize the need for careful detailed assessment and discussion with patients regarding their risk factors, disease extent, reconstructive options, and personal preferences. Figs. 18.3 and 18.4 illustrate a patient diagnosed with left breast cancer managed with oncoplasty who early on developed a recurrence managed with mastectomy and device-based reconstruction.