Introduction
The surgical management of breast cancer has seen a paradigm shift from radical ablation to maximum conservation. The Halstedian radical mastectomy of the late 1800s gradually evolved by 1975 to spare muscle, lymph nodes, skin, and ultimately manifested in the modern-day nipple–areola sparing mastectomy. The 1970s and 1980s brought forth prospective randomized trials supporting breast conserving therapy for tumors ≤5 cm as a viable alternative to mastectomy. Breast conserving therapy was widely adopted and evolved from quadrantectomies requiring wide margins to localized wire-directed excisions requiring merely no ink on tumor. These advances in breast conservation allowed a cosmetic benefit to the patient without sacrificing oncologic care. Unfortunately, many patients were left with post-lumpectomy deformities that compromised the aesthetic goal of the operation. The advent of oncoplastic surgery was led by the awareness of these post-lumpectomy deformities and gradual acceptance of the concept that optimal oncologic care and breast cosmesis are not mutually exclusive. As a result, post-lumpectomy deformities were not deemed an inevitable or unavoidable outcome and proactive measures to reduce their incidence were encouraged. Oncoplastic surgery further advanced breast conservation to allow larger and larger resections, while still maintaining the aesthetic appearance of the breast.
Modern medical therapies, with improved hormonal and chemotherapy, new techniques and protocols in radiation oncology, and a better understanding of the biology of breast cancer, have improved overall and breast cancer specific survival. Naturally, local recurrence rates have improved after both mastectomy and breast conservation. Recent prospective randomized trials have demonstrated local recurrence rates less than 1.5% at 5 years for patients randomized to excision plus whole breast radiotherapy. However, prospective randomized trials studying breast conservation were only conducted for tumors ≤5 cm. Consequently, women with tumors >5 cm, or those with multicentric or multifocal disease, are often denied breast conservation and are relegated to mastectomy with adjuvant radiotherapy.
Oncoplastic surgery presents an opportunity to re-examine this paradigm. A successful aesthetic result after oncoplastic surgery largely depends on a favorable tumor-to-breast volume ratio and the application of appropriate tissue rearranging maneuvers. One of the simplest scenarios is a small tumor in the lower pole of a large breast that is easily addressed with a standard Wise pattern or vertical pattern mammaplasty. However, when tumors are in unfavorable locations, are close to the overlying skin, or involve the skin, these standard techniques can be modified to still allow an oncoplastic reconstruction, albeit with nontraditional scar patterns. Gradually, we have evolved to push the limits of these techniques and apply to them to patients with less favorable tumor:breast volume ratios. With experience, it becomes apparent that even patients with >5 cm tumors, or those with multifocal or multicentric disease, can potentially have breast conservation while still achieving excellent cosmetic outcomes. It is particularly tempting when comparing this reconstructive approach to a post-mastectomy reconstruction. Patients with large or locally advanced tumors typically require post-mastectomy radiotherapy. Implant-based reconstruction with radiotherapy is fraught with poor results and higher complication rates. Autologous reconstruction avoids this issue but often requires multiple procedures and long recovery periods. With current data reporting low local recurrence rates, the impetus for defaulting to mastectomy is fading. Although it is unlikely that randomized prospective data will be gathered to support breast conservation for >5 cm tumors, there is clear interest in this concept, as evidenced by a growing body of literature dedicated to this topic.
At our institution, oncoplastic reconstruction is offered to all patients whenever technically feasible. Extreme oncoplasty is the term applied to a specialized concept for a breast conserving operation, using oncoplastic techniques, in a patient who, in most physicians’ opinions, requires a mastectomy. Extreme oncoplasty applies to breast conservation using a reduction mammaplasty technique in patients with larger lesions, with the goal of clear surgical margins, while maintaining or improving the cosmetic outcome. These lesions are generally large, >5 cm, multifocal, or multicentric tumors. They may be locally advanced, and many will have positive lymph nodes. Most of these patients will require radiation therapy, even if they are treated with mastectomy.
Oncoplastic reconstruction generally yields a cosmetic result superior to a mastectomy with immediate reconstruction and radiation therapy. There is less operative and postoperative morbidity with extreme oncoplasty, fewer surgeries required, and finally, radiation therapy is far kinder to breast conservation than to mastectomy with reconstruction.
Patient Selection
The ideal patient for extreme oncoplasty is a woman with a large breast, who desires a smaller breast, who desires breast conservation, and has disease limited to a single quadrant, with limited comorbidities. Naturally, not all patients meet every one of the criteria, and many decisions are made on a case-by-case basis. Neoadjuvant chemotherapy is often utilized to downstage a patient and convert from obligatory mastectomy to potential breast conservation. The overriding theme is that if a method of reconstruction with a volume-displacing technique is possible, regardless of the aforementioned criteria, then the patient is a candidate. The value of extreme oncoplasty is in its ability to condense surgical therapy. The patient will undergo the ablative surgery, immediate reconstruction, and immediate contralateral breast surgery within a 3-hour outpatient operation. The recovery is usually rapid, similar to the recovery process of a reduction mammaplasty, and there are no additional reconstructive procedures necessary. Although most patients are treated with volume displacement oncoplasty, occasionally a volume replacement technique involving local fasciocutaneous flaps (e.g., LICAP) may be utilized. Regional or distant tissue transfer techniques for oncoplastic reconstruction are usually not utilized, given the uncertainty of margin status at the time of reconstruction. Delayed partial breast reconstruction with autologous methods is another option; however, these patients may be better suited for mastectomy if they are considering this significant investment in reconstruction.
Preoperative History and Consideration
Oncoplastic surgery requires a multidisciplinary approach and thorough preoperative planning. Multidisciplinary planning requires discussion, at minimum, with the oncologic surgeon, radiologist, and plastic surgeon; however, other team members should be included as well. Oncoplastic surgery requires a philosophy that the appearance of the breast after tumor excision is important. All preoperative imaging tests must be carefully evaluated and integrated with information about the pathological subtype, tumor size, location in relation to the nipple, skin quality, and patient preferences. Other concerns include invasive lobular cancers that may be larger on the microscopic level than expected based on initial imaging, extensive in situ components with similar risk for understaging on imaging. Also, radiation effects on the size and shape of the ipsilateral breast compared with the contralateral side undergoing a symmetry procedure must be considered.
A common misconception is that the goal of breast reconstruction is to create the “perfect breast.” The goal should be to achieve an outcome that best suits the patient’s goals for reconstruction and desires for final breast appearance, while still considering the primary goal of treating the cancer. The reconstructive plan can be formulated only after analysis of the tumor size and location; the preoperative breast shape, size, and degree of ptosis; and understanding the patient’s oncologic and reconstructive desires. The ideal is to minimize the amount of surgery, recovery period, risk of complications, and surgical failure rates, while maximizing the desired aesthetic and oncologic outcome.
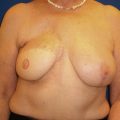
At our institution, the oncologic breast surgeon assumes the role of “leader” to guide the team and ensure excellent communication among all team members. During the first visit we generate a surgical plan, called the “Flight Plan,” that summarizes the diagnosis, includes photos of the patient’s chest and relevant imaging, and lists the plan of action leading up to and including the planned operation ( Fig. 15.1 ). The surgical plan is given to the patient, as well as distributed to all team members, and updated as the patient moves through the consultation process.
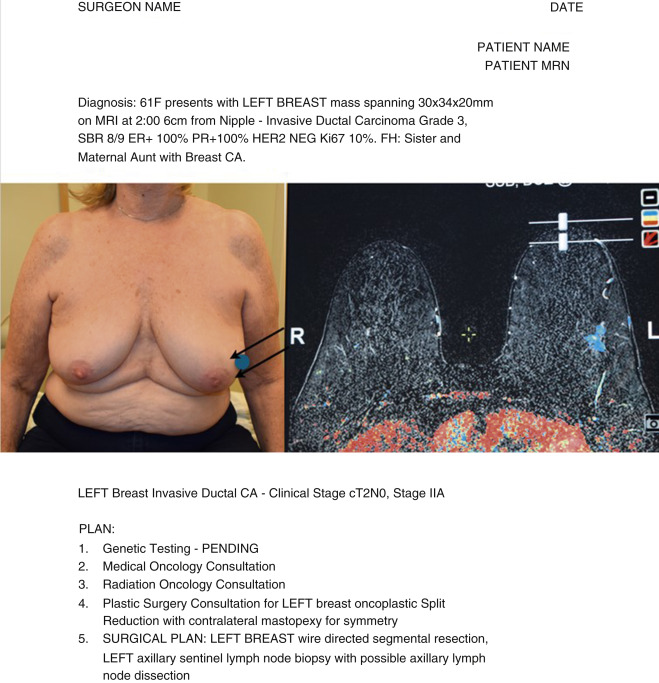
Preoperative work-up should include a full history and physical, paying close attention to prior breast surgery and location of prior surgical incisions. Breast imaging should include a combination of mammogram, ultrasound, and breast magnetic resonance imaging (MRI). MRI can often better determine extent of disease in mammographically subtle findings and has improved sensitivity for invasive lobular carcinoma.
Indications and Contraindications
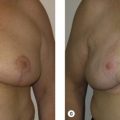
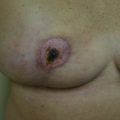
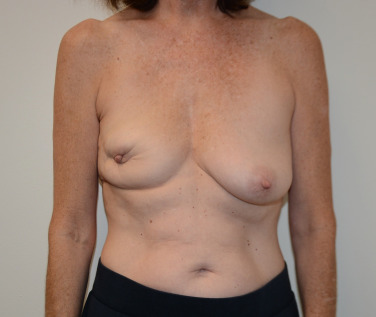
Historically, women were commonly left feeling maimed after breast cancer surgery when a lumpectomy cavity was left to form a seroma that later resorbs, leading to deformation after radiation therapy ( Fig. 15.2 ). In a typical lumpectomy, an incision is made over the tumor, the tumor is removed, and no specific effort is made to fill the defect left with remaining breast tissue. Even when surrounding breast tissue is used to fill with a small local tissue flap advancement, this can still lead to puckering or dimpling when the patient is out of the supine position and sitting or standing upright. Unfortunately, as many as 36% of simple excisions fail to achieve adequate margins in a single operation, leading to re-excision, worsening cosmesis, and conversions to mastectomy. Oncoplastic surgery, using a reduction mammaplasty, allows removal of the tumor but also prevents undue distortion of the breast by allowing the defect to be filled with remaining breast tissue.
Oncoplastic surgery can be performed in tandem with a lumpectomy in most cases where the tumor is ≤5 cm. Extreme oncoplasty can be considered when women with tumors >5 cm are seeking an alternative to mastectomy, when oncologically feasible. In either case, oncoplastic surgery allows for breast conserving therapy with a better cosmetic result. Oncoplastic surgery should be considered in all patients who are candidates for surgical treatment of breast cancer .
Operative Approach
The workhorse of oncoplastic surgery at our institution is performed with the inverted T or Wise pattern as the preferred incisional approach for the mammaplasty. This versatile technique is the ideal option for women with preoperative macromastia. Based on tumor location, a skin pattern and nipple–areolar complex (NAC) pedicle are designed preoperatively to allow for resection of the tumor and filling of the tumor cavity defect with the remaining breast tissue. This technique can also be applied to tumors that fall outside the Wise pattern by shifting tissue and rotating the reduction pattern. The wide amount of skin excised allows for correction of ptosis, exposure to the entire breast, the ability to widely resect tissue from any quadrant, and significantly reduce overall breast volume to aid radiation dose homogeneity. Once the amount of required tissue resection is determined on the ipsilateral side, the contralateral breast is reduced to match as a symmetrizing procedure.
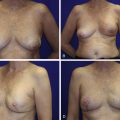
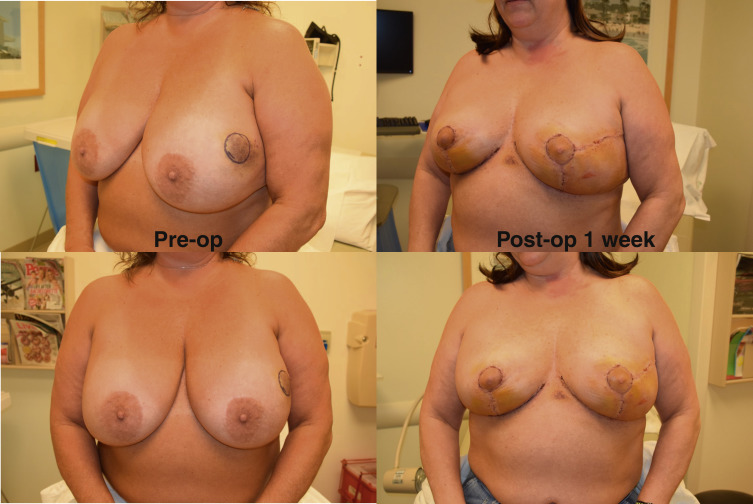
The need to be certain of the anterior tumor margin led our group to develop the Split Reduction. The main strength of the Wise pattern is the independence of the skin resection and the parenchymal resection. The parenchymal reduction does not need to follow the skin reduction pattern; the end goal is creation of a breast mound over which the skin can be re-draped. For an aesthetic breast reduction, it is desirable to place the scars in the least visible areas. Thus, the Wise pattern is designed to limit the scars to the circumareolar border, the vertical midline of the breast, and the inframammary crease. For oncoplastic breast surgery, we do not need to limit ourselves to this ideal skin pattern. Because the need for tumor clearance trumps this aesthetic ideal, we may modify the traditional Wise pattern to displace a hidden scar from the medial or lateral inframammary fold onto the visible breast, directly over the tumor, in favor of clearing the anterior margin in lesions close to the skin. This modification, that we term a Split Reduction, allows definitive clearance of the anterior (skin) margin or access to a lesion that falls out of the standard Wise pattern. The end result is resection of a comparable amount of skin as a traditional Wise pattern with maintenance of a similar, cone-shaped skin envelope but higher visibility of the scar. In our opinion, this trade-off is acceptable, because the alternative of a close or involved anterior margin leading to mastectomy is avoided ( Fig. 15.3 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree