Several viral and bacterial illnesses may be accompanied by localized or generalized skin eruptions called exanthems . These eruptions may be the first manifestation of a disorder and often are the reason for parents and patients to pursue medical evaluation. Prompt recognition and diagnosis of exanthems is desirable and may dictate that other examinations be performed to assess for systemic associations. Although identification of the exact infectious agent is not always practical or possible, knowledge of the most common causes of the various exanthems is important from an epidemiologic perspective. For instance, when diagnosing a child with a parvovirus B19-related exanthematous disease, consideration of at-risk contacts (i.e., gravid females, individuals with hemolysis or conditions resulting in decreased red blood cell production) is vital.
The majority of childhood exanthems are caused by viruses and less often by bacterial or rickettsial agents. Most childhood exanthems are diagnosed and managed by primary care providers, and thus a thorough familiarity with both classic and atypical exanthems is desirable for the pediatric primary care practitioner. When evaluating the patient with an exanthem, several features should be considered, including the morphology of individual lesions, the distribution pattern, prodromal and concurrent symptoms, known exposures, associated enanthem (eruption of the mucous membranes), local epidemiology, and the findings of a thorough review of systems and physical examination. Whereas some exanthematous processes present with a characteristic patterning of lesion morphology and distribution, others may reveal no pathognomonic features, and the cutaneous findings must be taken into consideration along with the overall presentation of the patient. Exanthems may be divided into erythematous, vesicular, and papular forms and have also been invariably described as morbilliform (“measles-like”), rubelliform (“rubella-like”), scarlatiniform (“scarlet fever-like”), or urticarial. Pustular and petechial changes may occasionally be noted.
The classic childhood exanthems are listed in Table 16-1 . These disorders were originally classified with a numeric designation in the early 1900s. Since then much has been learned about the etiologic agents of the classic exanthems, and several newer exanthematous disorders have been described. This chapter includes a discussion of the classic exanthems: varicella, infectious mononucleosis (IM), and exanthems caused by enteroviruses, Mycoplasma , and rickettsial agents. In addition, nonspecific viral exanthems are discussed, as are some atypical exanthems including papular acrodermatitis of childhood, unilateral laterothoracic exanthem (ULE), and papular-purpuric gloves-and-socks syndrome (PPGSS). Kawasaki disease, which may have clinical overlap with multiple exanthematous disorders, is discussed in Chapter 21 . Acute generalized exanthematous pustulosis (AGEP), another exanthematous disorder, is discussed in Chapter 20 .
| Numerical Designation | Name(s) | Agent(s) | Comment |
|---|---|---|---|
| 1 | Measles, rubeola | Measles virus (paramyxovirus) | Declining incidence with vaccination; occasional epidemics of imported cases and cases in unimmunized individuals in United States |
| 2 | Scarlet fever, scarlatina | GABHS | Toxin-mediated |
| 3 | German measles, rubella | Rubella virus (togavirus) | Rare with vaccination |
| 4 | Filatow-Dukes disease | ?SA | No longer considered a distinct entity |
| 5 | Erythema infectiosum, fifth disease, slapped cheek disease | Parvovirus B19 | Patients not contagious once rash is present |
| 6 | Exanthem subitum, roseola infantum, sixth disease | HHV-6 and HHV-7 | Diffuse rash appears after abrupt defervescence |
Varicella (Chickenpox)
Varicella-zoster virus (VZV) is a member of the herpesvirus family, and the causative agent of both varicella (chickenpox) and herpes zoster (shingles, see Chapter 15 ). Varicella results from primary infection with VZV and is a highly communicable human disease. Once acquired, VZV becomes permanently established in the sensory ganglia in a latent form with intermittent reactivation in a dermatomal distribution, resulting in herpes zoster. Although acute varicella is usually a self-limited infection, before extensive use of the varicella vaccine most children had upward of 250 to 500 skin lesions, approximately 9000 to 11,000 children were hospitalized annually, and up to 100 individuals per year died of the disease or its complications.
VZV is found worldwide, and annual epidemics occur most often during late winter and spring. Varicella is highly contagious and is usually spread between the prodrome and the first 3 days of the skin eruption. VZV is transmitted via respiratory droplets, then enters regional lymph nodes, and eventually a viremia ensues with more widespread dissemination. Primary infection elicits a humoral immune response with production of immunoglobulin (Ig)A, IgM, and IgG anti-VZV antibodies, the latter of which help protect against reinfection. Patients are considered contagious until at least 5 days after onset of the rash or until all existing lesions are dry and crusted.
A live attenuated varicella vaccine (Oka strain) was introduced in 1995, and in 1996 the Advisory Committee on Immunization Practices (ACIP) recommended universal one-dose vaccination of all children at age 12 to 18 months, with catchup vaccination of all susceptible children before age 13 years. The ACIP and the American Academy of Pediatrics (AAP) have expanded their recommendations to include use of the vaccine for postexposure prophylaxis and for some immunocompromised children. The vaccine is also indicated in adults who are susceptible to varicella. The vaccine has been quite effective in decreasing the overall incidence of varicella, and in vaccinees who develop breakthrough disease, the disease tends to be milder with fewer lesions that may remain papular rather than becoming vesicular. In a 6-year case-control study of the effectiveness of the varicella vaccine over time, the effectiveness in the year after vaccination (97%) was greater than in years 2 through 8 after vaccination (84%), although most cases of breakthrough disease in vaccinees were mild. Another study of the effectiveness of a one- versus two-vaccination regimen revealed high vaccine efficacy over 10 years for both groups (94.4% and 98.3%, respectively), and again, breakthrough disease was mild. A decline in varicella-related hospitalizations for invasive group A β-hemolytic streptococcus (GABHS) infections has been documented between the period of the prevaccine era and that of widespread vaccine use. Although some studies have demonstrated lower effectiveness of the varicella vaccine than expected, overall the data appear to support its universal use. In 2006, because of insufficient population immunity to prevent community transmission, the ACIP and AAP changed the varicella vaccine policy, recommending a universal two-dose vaccination program.
The varicella vaccine is available as a monovalent vaccine (Varivax, Merck & Co, Kenilworth, NJ) and as part of a quadrivalent measles-mumps-rubella-varicella vaccine (MMRV; ProQuad, Merck & Co). Postlicensure studies have suggested a slightly increased risk of febrile seizures among children aged 12 to 23 months who received MMRV for their initial dose (versus separate MMR and varicella vaccines). A personal or family history of seizures is considered a precaution for use of MMRV, and unless a parent expresses strong preference for this vaccine, it is recommended that the first dose be administered as separate measles-mumps-rubella (MMR) and varicella vaccines. For the second dose at any age or the first dose at ages 48 months and older, the combination MMRV vaccine is recommended.
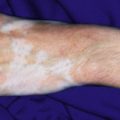
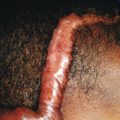
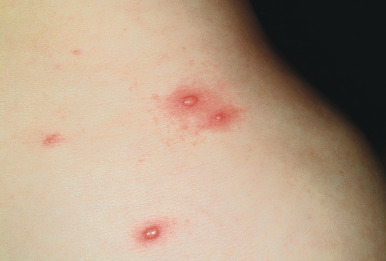
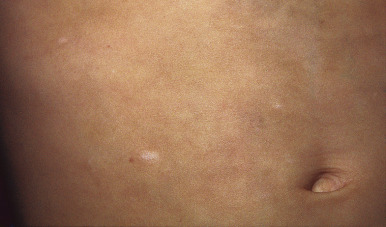
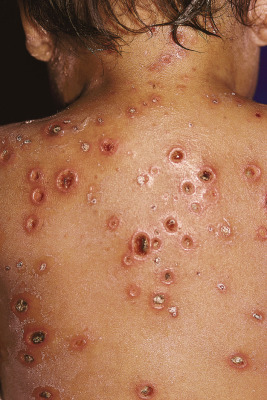
Primary varicella begins with a prodrome of fever, chills, malaise, headache, arthralgia, and myalgia. After 24 to 48 hours, the earliest skin lesions become evident, initially as red macules or papules ( Fig. 16-1 ) that progresses rapidly to a vesicular phase. The fully developed lesion has been likened to a “dewdrop on a rose petal” ( Fig. 16-2 ). Varicella lesions present initially on the scalp, face, or trunk and then spread to the extremities. Older lesions crust over, and new lesions continue to develop, resulting in the pathognomonic finding of lesions in various stages being present at the same time ( Fig. 16-3 ). New lesions continue to develop for 3 to 4 days, and by day 6, most lesions have crusted over. Patients with sunburn or dermatitis may develop a more severe exanthem. An associated enanthem may be present and consists of painful erosions in the oropharynx, conjunctivae, or vaginal mucosae. The lesions of varicella heal with hypopigmentation ( Fig. 16-4 ) and scarring ( Fig. 16-5 ), especially at sites of the initial lesions.
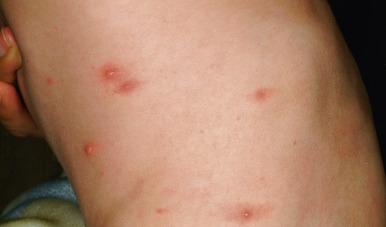
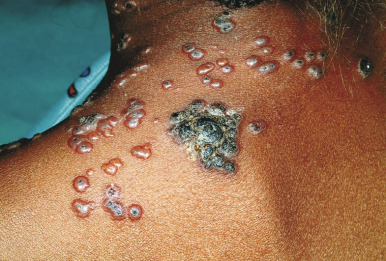
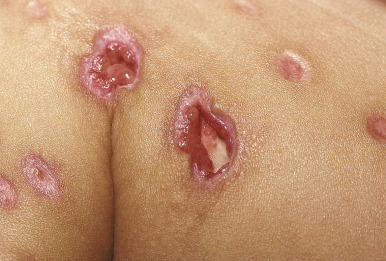
Varicella tends to be a mild, self-limited disease in most immunocompetent hosts. Complications, however, may occur. The most common complication is secondary bacterial superinfection, usually the result of Staphylococcus aureus or GABHS. Invasive GABHS infections, including streptococcal toxic shock syndrome and necrotizing fasciitis, may rarely occur. Secondary bacterial infection usually presents as isolated secondary fever or localized symptoms, such as bulla (large blister) formation or cellulitis. Varicella gangrenosa is diagnosed when an area reveals rapidly progressing erythema, induration, and pain. Peripheral gangrene may occur in the distal extremities. Other skin-related complications include deep ulcerative lesions ( Fig. 16-6 ), subcutaneous abscesses, and regional lymphadenitis. Patients under the age of 5 years who live in a household with older children seem to be at greatest risk for bacterial complications. In patients who develop secondary bacterial infection, bacteremia may occur, as may pneumonia, arthritis, or osteomyelitis.
Second to bacterial superinfection, the most common complication in patients with primary varicella is neurologic involvement. This may present as encephalitis, meningoencephalitis, cerebellar ataxia, transverse myelitis, or Guillain–Barré syndrome. Reye syndrome was a fairly common complication of varicella before the association with salicylates was identified. Reye syndrome (acute encephalopathy and fatty degeneration of the viscera) presents with decreased level of consciousness, vomiting, and abnormalities in liver function. The incidence of this complication has fallen dramatically over recent years. Varicella pneumonia is the most common serious complication in adults but is rare in healthy children. When it occurs during the course of childhood varicella, it is usually bacterial in origin. Severe pulmonary deterioration may occur, and it may occasionally be fatal. Thrombocytopenia, arthritis, uveitis, nephritis, myocarditis, pancreatitis, and hepatitis may also occasionally occur. When arthritis occurs in this setting, it may be bacterial ( Streptococcus pyogenes or S. aureus ) or aseptic (“varicella arthritis”) in origin. The latter is typically confined to a single joint, most commonly the knee, ankle, shoulder, or foot. Purpura fulminans is a rare and life-threatening complication of varicella, and disseminated intravascular coagulopathy may occasionally occur.
Immunocompromised hosts with varicella, including those with leukemia, lymphoma, or human immunodeficiency virus (HIV) infection; those receiving corticosteroids; and those with a history of organ or bone marrow transplantation may have more serious disease and a higher incidence of complications. These individuals have an increased risk of disseminated varicella, lung disease, thrombocytopenia, and other organ involvement. Bacterial superinfection may also occur with increased incidence, and hemorrhagic complications of the disease are more common. Varicella during pregnancy poses an increased risk of serious disease to both the mother and the fetus (see Chapter 2 ). Pregnant females have a higher incidence of varicella pneumonia, and mortality from the disease is increased.
The diagnosis of primary varicella is usually based on the history and clinical findings. Laboratory confirmation, when desired, can be performed by either virologic or serologic methods. Virologic methods include direct and indirect immunofluorescence studies, which are performed on tissue (i.e., cells scraped from the base of a fresh vesicle or organ biopsy tissue in patients with disseminated disease). These studies have the advantages of being both rapid and sensitive. Tzanck smears on skin scrapings identify multinucleated giant cells and confirm the diagnosis of herpesvirus infection but are not specific for VZV. Cell culture is unequivocally confirmatory but not ideal for VZV given the prolonged time necessary for cytopathic effects to appear. Other virologic methods include hybridization and polymerase chain reaction (PCR) assays. Serologic studies for diagnosing VZV infection include acute and convalescent IgM and IgG antibody titers.
The treatment of varicella in otherwise-healthy children is usually symptomatic, with the goals being control of pain and pruritus and prevention of secondary superinfection. Supportive therapies include oral antihistamines for pruritus, acetaminophen for fever or pain, and topical care. Useful topical regimens may include cool compresses, oatmeal baths, and application of topical products such as bacitracin ointment, calamine lotion, pramoxine-containing preparations, or menthol-camphor (Sarna) lotion. Antibiotics should be given if secondary bacterial infection is present.
The decision regarding the use of antiviral therapy for primary varicella depends on several factors, including host immune status, the extent of the infection, and timing of the diagnosis. In general, routine use of antiviral agents for varicella in otherwise-healthy children is not recommended. Acyclovir, famciclovir, and valacyclovir are antiviral agents licensed for treatment of VZV infections, although famciclovir is not approved for primary varicella. Oral acyclovir has been suggested in some studies to decrease the severity of primary VZV infection, including a decrease in the days of fever, number of days of new lesion formation, total number of lesions, and pruritus. In a review of published studies on acyclovir treatment of varicella in otherwise-healthy children and adolescents, a reduction in the number of days with fever was noted, but the results were inconsistent with respect to the number of days to no new lesions, maximum number of lesions, and number of days to relief of itching. In addition, no differences in the incidence of varicella complications between acyclovir and placebo groups were consistently noted. Oral antiviral therapy should be considered, however, for healthy individuals who are at risk for moderate or severe disease, such as those over 12 years of age (especially unvaccinated), those with chronic skin or lung disorders, and those receiving therapy with salicylates (long-term courses) or corticosteroids (short, intermittent, or aerosolized courses). When used in the treatment of varicella in otherwise-healthy patients, antiviral therapy should be started within 24 hours of the appearance of the rash. The use of oral antivirals for pregnant females with uncomplicated varicella remains controversial, although some experts recommend its use for those who develop it during the second or third trimesters. Intravenous acyclovir is recommended for pregnant females with serious complications of varicella.
In immunocompromised children, intravenous antiviral therapy with acyclovir should be administered and is most effective when initiated early in the course of the disease (preferably within 24 hours of the onset of the rash). Some experts endorse high-dose oral acyclovir in selected immunocompromised individuals who are perceived to be at a lower risk for severe disseminated varicella. In 2008 valacyclovir was also approved for treatment of primary varicella in children 2 to under 18 years of age; some experts have used this agent for treating selected immunocompromised patients. In susceptible individuals with a known exposure to VZV, options include administration of varicella-zoster immune globulin (VariZIG) or varicella vaccine. VariZIG is a high-titer preparation of VZV IgG that is given via the intramuscular route. It is recommended for susceptible individuals at high risk for infection and pregnant females who have been exposed to VZV ( Box 16-1 ). VariZIG is best given within 48 hours of the exposure, but the United States Food and Drug Association (FDA) recently extended the approved period for which it can be given to up to 10 days.
Types of exposure to varicella-zoster:
Residing in same household as infected patient
Playmate: face-to-face indoor play
Hospital: varicella in roommate; face-to-face contact with infected staff member or patient; visit by a person deemed contagious with varicella; intimate contact with person deemed contagious with zoster
Newborn: onset of varicella (not herpes zoster) in mother from 5 days or less before delivery or within 48 hours after delivery
Candidates for VariZIG:
Immunocompromised child without history of varicella or varicella immunization
Susceptible pregnant female (no evidence of immunity)
Newborn infant whose mother develops varicella (see above)
Hospitalized premature infant:
≥28 weeks’ gestation, if mother lacks reliable history of varicella or serologic evidence of antibodies
<28 weeks’ gestation or birthweight 1000 g or less, regardless of maternal history of varicella or serologic status
Rubeola (Measles)
In the prevaccine era, more than 500,000 cases of measles, or rubeola, were reported annually in the United States. After introduction of a live-virus vaccine in 1963, a significant reduction in the incidence of infection was noted. The MMR vaccine has been distributed in excess of 575 million doses and is one of the most widely used combination viral vaccines in the world. However, a dramatic resurgence occurred from 1989 to 1990, and during this period the highest mortality since 1977 was noted. Those affected most during the measles resurgence were preschool-age children from the inner-city areas, especially those were were unvaccinated and from low-income families. A majority of the deaths occurring from measles infection during this period occurred in children aged 5 years or younger. Other target populations during the measles resurgence included vaccinated school-age children and college students, probably owing to the insufficiency of a single vaccine dose and waning immunity.
In response to the measles resurgence of the 1980s, the Committee on Infectious Diseases of the AAP and the ACIP recommended an amendment to the previous vaccination schedule and suggested two doses of measles vaccination rather than one. The current recommendation for measles vaccination calls for a two-dose schedule, with the first given at 12 to 15 months and the second at 4 to 6 years of age, and revaccination of children 11 to 12 years of age or older who received only one previous dose of measles vaccine. Changes in the measles vaccination strategy resulted in a markedly diminished incidence of the disease in the United States, with 100 cases reported in 1999. Although the incidence in the United States declined to extremely low levels compared with the prevaccine era, measles continues to be a major health problem worldwide, with the World Health Organization estimating around 700,000 deaths per year from the disease. Of the 100 cases reported in the United States in 1999, 33% were imported from other countries. In developing countries, infants under 9 months of age who are too young to have received vaccination have a high incidence of measles with more multisystem involvement and a greater risk of death. Importantly, sporadic epidemics of primarily importation-associated measles in the United States continue to occur. During January through July 2008, 131 measles cases were reported to the Centers for Disease Control and Prevention (CDC), and these occurred primarily in unvaccinated school-aged children. The majority of these children were eligible for vaccination but had parents who chose not to have them vaccinated. In 2011, 222 measles cases were reported to the CDC, the majority of which were associated with importations from other countries and more than 85% of cases occurring in individuals who were unvaccinated or had unknown vaccination status. In 2014, measles cases in the United States had risen to 644. In early 2015 the United States experienced a large, multistate outbreak of measles infection linked to an amusement park in California. Among 110 of the earliest patients in this outbreak, 45% were unvaccinated, 5% had received only one dose of the vaccine, and 43% had unknown or undocumented vaccination status. Of the unvaccinated patients, 67% were intentionally unvaccinated because of personal beliefs (65% of these were children under the age of 18 years). Given such observations, maintaining high vaccination coverage and index of suspicion for measles and ensuring a rapid public-health response at times of outbreaks are critical in the attempt to maintain measles elimination in the United States.
Measles is caused by a single-stranded ribonucleic acid (RNA) virus in the family Paramyxoviridae. Infection begins in the nasopharyngeal epithelium and less commonly through the conjunctivae. Transmission of measles is primarily via respiratory droplets and less commonly by small particle aerosols. The incubation period is around 10 to 14 days. From the initial site of infection, the virus enters the lymph nodes and lymphatics and multiplies within the reticuloendothelial system with a subsequent viremia. The virus is then disseminated to multiple lymphoid tissues and other organs, including the skin, liver, and gastrointestinal tract. Measles immunity includes cell-mediated, humoral, and mucosal responses. Measles antibodies are responsible for protection from future infection or reinfection.
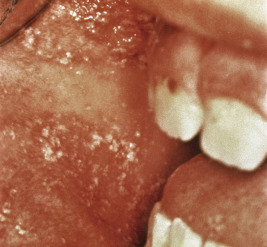
Measles classically presents with fever and the three C s: cough, coryza, and conjunctivitis. The pathognomonic enanthem, Koplik spots, usually occurs during this prodromal period and presents with punctuate, gray-white to erythematous papules distributed on the buccal mucosa ( Fig. 16-7 ). These lesions were recently found to be highly predictive of confirmed measles. The skin eruption of measles usually begins 2 to 4 days after the prodrome, which may intensify several days before the onset of rash. It begins on the face ( Fig. 16-8 ), especially the forehead, hairline, and behind the ears, and spreads downward onto the trunk ( Fig. 16-9 ) and extremities. The lesions are erythematous to purple-red macules and papules that may become confluent and fade in the same order as their appearance, leaving behind coppery macules and desquamation.
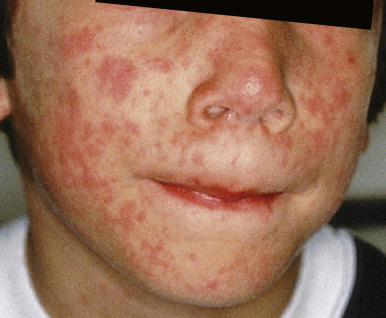
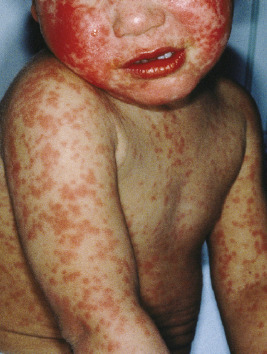
Complications of measles include pneumonia, laryngotracheobronchitis, otitis, gastroenteritis, myocarditis, and encephalitis. Pneumonia may be caused by the measles virus itself or be related to secondary bacterial or viral infection. Keratoconjunctivitis is common, especially in children with vitamin A deficiency, in which case it may result in blindness.
Modified measles is measles occurring in a previously vaccinated individual. In this situation the prodrome is milder and of shorter duration, the exanthem is less prominent, and Koplik spots may be absent. This presentation of measles may pose a diagnostic challenge given the nonpathognomonic presentation. Atypical measles occurs rarely in contemporary times and is seen in individuals exposed to natural measles after vaccination with the killed measles vaccines. These vaccines were utilized for a short period in the 1960s in the United States and for more variable periods of time in other countries including Canada and Sweden. It has occasionally been reported in children who received the live attenuated vaccine. Atypical measles presents with high fever, headaches, and myalgias and eventually the measles rash and often pneumonia. Hemorrhagic features may be present, and the exanthem may be confused with Rocky Mountain spotted fever (RMSF). Subacute sclerosing panencephalitis (SSPE) is a delayed neurodegenerative disease characterized by seizures, personality changes, coma, and death within 5 to 15 years of the measles diagnosis. It classically occurs many years later in 1 in 100,000 patients with measles but fortunately has been dramatically decreased in incidence with widespread vaccination strategies.
The diagnosis of measles is traditionally made based upon clinical presentation with laboratory confirmation. Acute and convalescent serologic studies documenting a fourfold increase in titer confirm the diagnosis of measles. PCR and semiquantitative real-time PCR assays have been developed, the latter being quite sensitive and specific and potentially useful in situations where early and rapid diagnosis is vital. The treatment of uncomplicated measles is largely supportive, because specific antiviral therapies do not exist. Ribavirin, a synthetic nucleoside analog useful in the treatment of other paramyxovirus (i.e., respiratory syncytial virus) infections, has been utilized in severely ill or immunocompromised patients. Antibiotics are useful in patients with secondary bacterial infections such as otitis media or bacterial pneumonia. Vitamin A supplementation has been recommended by the World Health Organization and the United Nations International Children’s Emergency Fund for children with measles who reside in areas where vitamin A deficiency is a recognized problem or where the measles case fatality rate is 1% or greater. In these instances, vitamin A appears to reduce mortality and ameliorate the severity of diarrhea, possibly through a protective action on the epithelial lining of the gastrointestinal tract, increased mucus secretion, and enhanced local barriers to infection. Hospitalized patients with measles should be kept in respiratory isolation for 4 days from the onset of the rash. Most patients will recover completely without sequelae and have lifelong immune protection against reinfection.
Scarlet Fever
Scarlet fever (scarlatina) is a bacterial exanthem that may at times be confused with a variety of viral exanthematous diseases. It is caused by GABHS and is primarily a disease of children between the ages of 1 and 10 years. It is very rarely diagnosed in infants. Transmission of GABHS is usually via respiratory secretions. Epidemiologic changes over time in the types of streptococcal pyrogenic exotoxin (SPE) produced by the organism have been associated with changes in the severity of this disease. The shift from SPE-A- to SPE-B- and SPE-C-producing strains, which was seen early in the twentieth century, has paralleled the decrease in morbidity and mortality from scarlet fever, as well as the decline in incidence and severity of rheumatic fever (RF) in the United States. However, RF and rheumatic heart disease (RHD; progressive valve damage) continue to burden many low- and middle-income countries, as well as some populations in higher-income countries. Immigration from countries where RF and RHD are endemic to higher-income areas have resulted in an increased prevalence in some regions where they were once believed to be eliminated. In the United States, the incidence of RF is generally lower than in developing countries, but there have been some regional outbreaks, including an outbreak in Utah in the 1980s whereby the incidence in children aged 3 to 17 years approached 12 per 100,000.
Scarlet fever presents with fever, throat pain, headache, and chills along with cutaneous findings. It is less commonly associated with streptococcal skin infection rather than pharyngitis. The primary distinction between streptococcal pharyngitis and scarlet fever is the accompanying exanthem present in the latter. Oropharyngeal inspection reveals tonsillopharyngeal erythema, exudates, and petechial macules of the palate. During the first few days of illness, the tongue may reveal a white coating with red and edematous papillae projecting through (white strawberry tongue). By the fourth or fifth day, the coating peels off, leaving behind a red, glistening tongue studded with prominent papillae (red strawberry tongue).
The differential diagnosis of streptococcal pharyngitis includes infection with viral agents (especially mononucleosis), Mycoplasma , Chlamydia , and Arcanobacterium haemolyticum . In adolescents the differential diagnosis should include groups C and G Streptococcus species and Neisseria gonorrhoeae , and in developing countries Corynebacterium diphtheriae may be implicated. A. haemolyticum , formerly known as Corynebacterium haemolyticum , is a Gram-positive bacillus that most often infects adolescents and young adults and may result in a syndrome of pharyngitis and a scarlatiniform exanthem. The pharyngitis in these patients is usually severe (occasionally mistaken for diphtheria), and the exanthem may also occasionally mimic toxic shock syndrome, measles, urticaria, or erythema multiforme. Infection with A. haemolyticum can occasionally be associated with bacteremia, severe sepsis, and soft-tissue infections (especially in patients with concomitant diabetes mellitus). Lemierre syndrome, a condition characterized by pharyngitis, bacteremia (usually with anaerobic organisms including Bacillus and Fusobacterium ), and thrombophlebitis of the internal jugular vein, has also been reported in association with this organism.
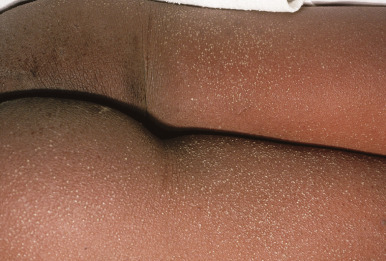
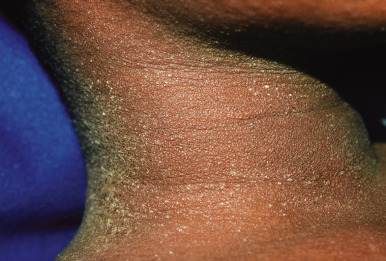
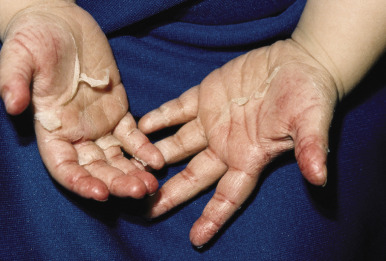
Tender anterior cervical adenopathy is commonly present in patients with scarlet fever, and rhinorrhea and cough are usually absent. Some experts argue that presence of the latter two findings is a negative factor for the diagnosis of GABHS infection. The exanthem of scarlet fever presents as a fine, erythematous, macular and papular eruption ( Fig. 16-10 ) that has been described as “sandpapery.” It involves the trunk and extremities and may be accentuated in flexural areas with a petechial component (Pastia lines). In darker-skinned individuals, the exanthem of scarlet fever may be more difficult to recognize and may consist only of punctate papules resembling cutis anserina (“goose flesh”) ( Fig. 16-11 ). Circumoral pallor may be a useful clinical sign and is visualized as a rim of pallor encircling the perioral area. The exanthem generally resolves over 4 to 5 days and may heal with thick sheets of desquamation, especially over the hands, feet, toes, and fingers ( Fig. 16-12 ). An interesting observation is that the SPEs produced by GABHS contribute to the exanthem by their ability to stimulate a delayed-type hypersensitivity response that requires prior exposure of the host to the organism.
Scarlet fever is diagnosed based on the clinical presentation in conjunction with results of laboratory testing. The gold standard laboratory examination is throat culture with growth of GABHS. Rapid antigen detection (rapid strep testing), when properly performed, has a high sensitivity and specificity. In addition, antistreptococcal serologies may occasionally be useful. Complications of scarlet fever include pneumonia, pericarditis, meningitis, hepatitis, glomerulonephritis, and RF. Although the symptoms of streptococcal pharyngitis and scarlet fever will often improve spontaneously, treatment more rapidly alleviates symptoms and more importantly is the primary mode of prevention for the subsequent occurrence of RF. The prevailing theory regarding RF prevention is that antimicrobial therapy should be started within 9 days from the onset of symptoms of GABHS pharyngitis.
The drug of choice for treatment of scarlet fever is penicillin V. Although amoxicillin and ampicillin are commonly used, they have no microbiologic advantage over penicillin. In patients allergic to penicillin, erythromycin or another macrolide (i.e., clarithromycin or azithromycin) or clindamycin may be utilized. Other options include a first-generation cephalosporin (although the possibility of cross-reaction in patients with penicillin allergy must be considered) or intramuscular penicillin G, which has the advantage of not being compliance-dependent but the disadvantage of being painful during administration.
Rubella (German Measles)
Rubella, or German measles, is a viral exanthematous disease with a worldwide distribution. The incidence of rubella has greatly decreased since widespread rubella vaccination began in the United States in 1969. Before that time, epidemics occurred every 6 to 9 years, and major epidemics or pandemics occurred every 10 to 20 years. The primary goal of the rubella vaccination program was to prevent fetal infection, which may have various effects including miscarriage, stillbirth, and congenital rubella syndrome (CRS) (see Chapter 2 ). Although reported rubella cases in the United States are now quite low, outbreaks still occur, primarily among Hispanic adults, most of whom were born outside of the country and are unvaccinated. As of 2013, goals for rubella elimination had been established in two World Health Organization regions, the Americas (by 2010) and the European region (by 2015), and accelerated control of rubella and prevention of CRS have been established for the Western Pacific region. In Japan, rubella outbreaks significantly increased in 2012 and 2013 with a corresponding increase in CRS. The effects of such outbreaks can be far-reaching given the possibility of disease importation into other countries and serves as a reminder of the need to maintain high levels of vaccination coverage and infectious-disease surveillance.
Rubella is caused by an RNA virus in the Togaviridae family. Humans are the only source of infection, and postnatal disease is spread through direct or droplet contact from nasopharyngeal secretions. Up to 50% of cases of rubella are asymptomatic, and mild, self-limited disease is common. The incubation period ranges from 14 to 23 days.
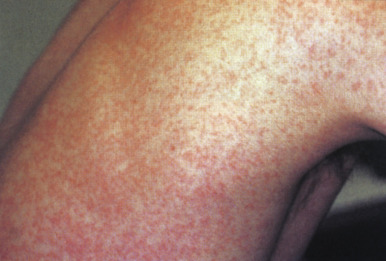
Prodromal symptoms may occur, especially in adolescents and adults, and include low-grade fever, headache, malaise, eye pain, myalgias, sore throat, rhinorrhea, and cough. The prodrome usually presents 2 to 5 days before the exanthem appears. The skin eruption consists of erythematous to rose-pink macules and papules ( Fig. 16-13 ) that tend to become confluent and most commonly involve the face and trunk. The eruption spreads in a cephalocaudad manner and begins to involute after 1 to 3 days, fading in the same order in which it appeared. After severe rubella eruptions, a fine flaky desquamation may be observed in areas of maximum involvement.
Generalized lymphadenopathy often occurs, especially in the suboccipital, postauricular, and cervical regions. Although this pattern of lymph node enlargement is highly characteristic of rubella, it is not pathognomonic and may occur in other disorders including measles, varicella, adenovirus infection, and mononucleosis. Arthralgias and arthritis are common, especially in females in whom they occur up to 52% of the time. The most common joints affected are those of the fingers, wrists, and knees. Complete resolution of the joint symptoms may take up to several weeks, and chronic arthritis occasionally develops. A characteristic enanthem, Forschheimer spots, may be present in patients with rubella and presents with erythematous and petechial macules on the soft palate. Other complications of rubella include encephalitis (which occurs in 1 in 6000 patients), myocarditis, pericarditis, and hepatitis. Anemia, neutropenia, and thrombocytopenia may also occur, as may hemolytic uremic syndrome.
A clinical diagnosis of rubella is difficult to make given the potential overlap with multiple other exanthematous diseases. Serologic tests are most useful in confirming the diagnosis of rubella. The presence of rubella-specific IgM antibody indicates recent infection, as does a fourfold or greater increase in titer between acute and convalescent serum taken 1 to 2 weeks apart. The virus can be isolated from nasal specimens plated onto appropriate cell-culture media. The treatment of postnatal rubella is generally supportive. Hospitalized patients require contact isolation (and nonhospitalized children should be excused from school or day care) for 7 days after onset of the rash.
The ongoing efforts of global rubella eradication campaigns will hopefully result in eventual worldwide elimination of the disease. Continued efforts to ensure the immunity of women of child-bearing age will help to decrease the incidence of vertical transmission and CRS.
Filatow–Dukes Disease
In 1900, Dukes described what was believed to be a unique exanthem and contrasted it from rubella and scarlet fever. This disorder was termed fourth disease and subsequently became known as Filatow–Dukes disease given a similar description by Filatow 15 years earlier. The exanthem was described as bright red papules with a diffuse distribution, skin tenderness, and fever, and there was significant overlap with other exanthematous diseases, including staphylococcal scalded skin syndrome, scarlet fever, and rubella. Significant controversy followed the initial descriptions of fourth disease, and some authors propose that a distinct disorder never existed. The possibility that epidermolytic toxin-producing staphylococci were the etiologic agent for the disorder described in the original reports has been suggested. In general, researchers and practitioners alike have abandoned the idea of Filatow–Dukes disease representing a distinct entity.
Erythema Infectiosum
Erythema infectiosum (EI; fifth disease) is a common childhood exanthematous illness caused by parvovirus B19 (referred to as B19 ). Although parvoviruses are ubiquitous in nature and may cause significant disease in a wide range of animals, B19 is the only one resulting in human disease. Several disorders have been linked to B19 infection in humans ( Box 16-2 ), including EI, arthritis, aplastic crises, and fetal hydrops. B19 infection is common in most countries, and in the United States 60% of adults are seropositive for the virus. Infection is most common in school-age children. In immunocompromised hosts, persistent bone marrow suppression may occur, resulting in severe cytopenias that may be misinterpreted as being related to malignancy relapse or drug reactions.
Asymptomatic infection
Exanthematous disorders
Erythema infectiosum (fifth disease)
Papular-purpuric gloves-and-socks syndrome
Asymmetric periflexural exanthem
“Bathing trunk” exanthem
Petechial exanthems
Other disorders
Arthritis
Transient aplastic crises
Chronic anemia
Refractory anemia after solid-organ or stem-cell transplantation
Hemophagocytic lymphohistiocytosis
Myelodysplastic syndrome
Fetal hydrops
Vasculitis
Neurologic disease
Rheumatologic disease
In patients with classic EI, B19 infection is transmitted via the respiratory tract and is followed by a viremia that ends after 5 to 7 days with the production of IgM anti-B19 antibody. Prodromal symptoms such as headaches, fever, and chills are common during the viremic phase. Respiratory symptoms may also be present. IgG antibody appears during the third week of illness and coincides with the appearance of the rash and arthralgias, and hence patients with the cutaneous findings of EI are not considered infectious. Outbreaks of EI occur primarily during the winter and spring.
EI is the most recognizable B19-associated manifestation. It is also known as “slapped cheek” disease given the characteristic fiery-red facial erythema ( Fig. 16-14 ) that occurs 2 to 3 days after the prodromal symptoms. The cheeks are most prominently affected, with the nasal bridge and perioral areas usually being spared. This phase of the illness has been termed the first stage, and is subsequently followed by stages two and three. The differential diagnosis of the initial eruption of EI includes phototoxic reaction and systemic lupus erythematosus.
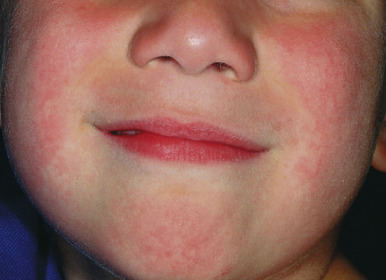
During stage two, the patient develops a lacy, reticulated eruption on the extremities and trunk 1 to 4 days after the facial rash. This eruption may be pruritic and is often evanescent. It often begins with a confluent pattern followed by central clearing, which results in the lacy and reticulated appearance ( Figs. 16-15 and 16-16 ). The palms and soles are usually spared. The rash of the second stage of EI tends to fade over 2 to 3 weeks but may intermittently recur in response to environmental stimuli including sunlight, warm temperatures (i.e., a hot bath), or physical activity. This intermittent waxing and waning represents the third stage of EI, and the duration is variable, usually 1 to 3 weeks.
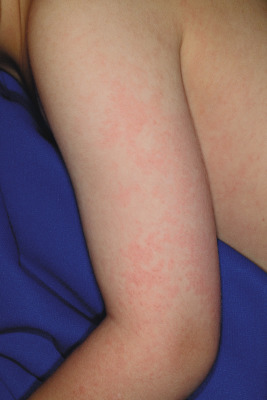
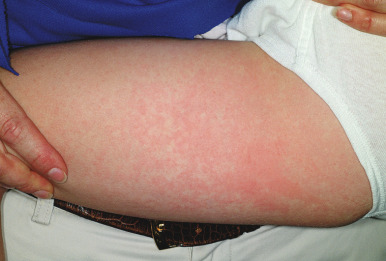
Joint symptoms occur in 8% to 10% of children with EI but in up to 60% of adults with primary B19 infection, especially females. The most commonly involved joints include the metacarpophalangeal joints, proximal interphalangeal joints, knees, wrists, and ankles. For most patients, the joint symptoms are transient and self-limited. Occasionally, affected children develop a chronic arthritis. Some experts have suggested a potential association between B19 infection and rheumatic arthritis or other connective tissue diseases, although the strength of this association remains unclear.
B19 has a remarkable affinity for erythroid precursors, binding to a receptor known as P antigen ( globoside ). Direct infection of the red blood cell precursors results in a transient arrest in red blood cell production and resultant transient anemia. Transient aplastic crises may occur in patients suffering from disorders of decreased red blood cell production or increased red blood cell destruction or loss. Predisposing disorders include iron-deficiency anemia, spherocytosis, sickle cell disease, thalassemia, glucose-6-phosphate dehydrogenase deficiency, and pyruvate kinase deficiency. Although these episodes may be asymptomatic with spontaneous recovery, severe involvement may result in chills, pallor, weakness, fatigue, vasoocclusive crises, or congestive heart failure. Treatment with red blood cell transfusions may be indicated and for some patients is life-saving.
Because B19 can cross the placenta, fetal infection is possible in nonimmune females with acute infection. With time it has become clear that B19 may result in a variety of fetal effects, occasionally even death. This epidemiologic consideration is important in caring for pediatric patients with B19-associated disorders. Fetal effects of B19 infection may include anemia (ranging from mild and self-limited to severe), high-output congestive heart failure, hydrops fetalis (generalized edema with ascites, pleural effusions, and polyhydramnios), and intrauterine fetal demise. Neonatal encephalopathy, encephalitis, cerebral migratory abnormalities, and meningitis have rarely been reported. The majority of pregnant women with B19 infection are asymptomatic, which makes the true incidence of fetal involvement difficult to determine. It is estimated that 30% to 66% of adult females are immune to B19 infection, and therefore their fetuses are not at risk. In the majority of fetuses who acquire acute infection in utero , the infection is self-limited and the infants are delivered asymptomatic and at term. The greatest risk appears to be when infection is acquired before 20 weeks’ gestation, and the majority of fetal losses occur between 9 and 28 weeks’ gestation, mostly between 20 and 24 weeks. The overall risk of B19-related fetal loss in pregnancies complicated by B19 infection is estimated around 1% to 9%. Surviving infants tend to be healthy with normal development and neurologic outcome. B19-related teratogenicity has only rarely been reported. The diagnosis of B19 infection during pregnancy can be confirmed by maternal B19 IgM and IgG antibodies or PCR assay (see below). These studies are usually corroborated with findings on prenatal ultrasonography, most often evidence of fetal anemia and hydrops. PCR assays on fetal cord blood or amniotic fluid samples have also been utilized for diagnosis. Management of severely afflicted fetuses with B19 infection includes fetal digitalization and in utero blood transfusions.
B19 may be associated with a variety of purpuric or petechial exanthems, including PPGSS (see below) and more generalized presentations. In one outbreak of petechial rashes, B19 was confirmed in 76% of 17 children, and the petechiae were widely distributed, often with accentuation in the axillary and inguinal regions as well as the distal extremities. Some authors have suggested the terminology parvovirus B19-associated purpuric-petechial eruption for these polymorphous presentations that appear to correspond to the viremic phase of primary infection.
Papular-Purpuric Gloves-and-Socks Syndrome
Papular-purpuric gloves-and-socks syndrome (PPGSS) is another viral exanthematous illness that in many (but not all) cases has been documented to be caused by B19. This rare disorder presents most often in young adults and less often in children and is usually diagnosed during the spring and summer months. Patients with PPGSS present with the acute onset of rapidly progressive, symmetric swelling and erythema of the hands and feet, often with a petechial or purpuric component ( Figs. 16-17 and 16-18 ). Bullous lesions are rarely present. The eruption has a sharp demarcation at the wrists and ankles and is usually quite pruritic. A more diffuse, papular exanthem may occur elsewhere on the body. An associated enanthem consisting of hyperemia, petechiae, and erosions is often present and affects the soft and hard palate, pharynx, tongue, and inner lips. The symptoms of PPGSS tend to be milder in children and adolescents when compared with adults.
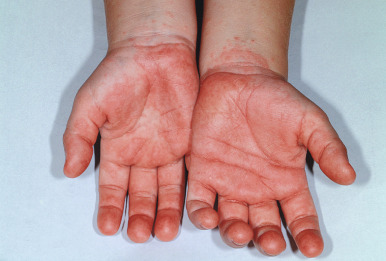
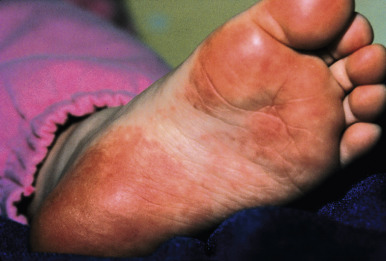
Associated symptoms include fever, arthralgias, malaise, and respiratory or gastrointestinal complaints. Hematologic complications including leukopenia and thrombocytopenia are rarely observed. Mononeuritis multiplex has been described in association with perineuritis noted on skin biopsy. PPGSS resolves spontaneously over 1 to 2 weeks, and recurrences are rare. Importantly, the antibody response to B19 seen in PPGSS may differ from that observed in patients with EI, such that patients with the exanthem of PPGSS may still be viremic and therefore infectious. The B19 VP2 structural protein has been demonstrated in the endothelial lining of dermal blood vessels in skin biopsies from patients with PPGSS.
The diagnosis of B19 infections is often made clinically in patients presenting with classic EI. In immunocompetent children who are otherwise well, laboratory confirmation of B19 infection is usually unnecessary. Instances in which such confirmation may be indicated, however, include atypical presentations, immunocompromised hosts, individuals with hematologic diseases, and those who have been exposed to gravid females. Serologic studies including enzyme immunoassays and radioimmunoassay are useful in detecting anti-B19 IgM and IgG antibodies, and other useful tests include direct hybridization with deoxyribonucleic acid (DNA) probes, in situ hybridization, and PCR studies. In a pregnant female who has been exposed to B19, serologic testing for IgM and IgG antibodies should be performed, and if acute infection is documented, serial fetal ultrasonography is usually indicated. As mentioned, maternal and fetal serum PCR techniques may be useful, especially when serologic study results are unclear, and B19 antigen detection in amniotic fluid samples has also been described.
Roseola Infantum
Roseola infantum (exanthem subitum, sixth disease) is a common childhood disease caused by human herpesvirus (HHV) type 6 or 7. HHV-6 and HHV-7 are ubiquitous members of the Herpesviridae family. These DNA viruses preferentially infect activated T cells, resulting in enhancement of natural killer cell activity and induction of numerous cytokines. As with other herpesviruses, they become latent after primary infection and may reactivate during times of altered immunity. Serologic studies have demonstrated that most children have been infected with HHV-6 before 3 years of age and with HHV-7 by 6 to 10 years of age.
Transmission of HHV-6 and HHV-7 is believed to be primarily via saliva, and horizontal transmission between mother and child is well documented. Persistent or intermittent excretion of HHV-6 in saliva and stool has been documented in parents of children who had documented primary infection. Although most newborns have transplacentally acquired antibodies, by 6 months nearly all have become seronegative and are therefore susceptible to infection. HHV-6 has been divided into two variants, HHV-6A and HHV-6B. Most childhood infections are ascribed to the HHV-6B variant, and HHV-6A may be more commonly implicated in immunocompromised hosts. The diagnosis of these infections can be made via virus isolation in peripheral blood mononuclear cell (PBMC) cultures, serologic studies, qualitative PCR, reverse transcriptase-PCR (RT-PCR), and real-time quantitative PCR. PCR-based diagnosis has been applied to a variety of body fluids and tissues including plasma, whole blood, saliva, urine, cervical swabs, placenta and PBMCs. RT-PCR can detect messenger RNA (mRNA), which is present only during active replication and hence may be useful in distinguishing between primary infection and past or latent infection.
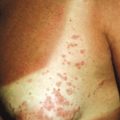
A broad range of conditions has been potentially linked to infection with HHV-6 and HHV-7 ( Table 16-2 ). By far, however, the best-recognized association for both agents is that of roseola infantum. Roseola is a mild exanthematous illness that most often occurs in children under 3 years of age. The classic presentation is that of high fever (38 to 41° C [101 to 106° F]) that lasts for 3 to 5 days in an otherwise-well infant. With normalization of the temperature, the classic exanthem appears, initially on the trunk and eventually spreading to involve the extremities, neck, and face. The skin eruption is composed of fairly nondescript, erythematous, blanchable macules and papules ( Fig. 16-19 ) that occasionally display a peripheral halo of vasoconstriction. The exanthem usually resolves over 1 to 3 days.
| Condition | HHV-6, HHV-7, or Both | Comment |
|---|---|---|
| Roseola infantum | Both | HHV-6 more common |
| Fever | Both | Infants mainly |
| Febrile seizures | Both | In young infants |
| Otitis media | 6 | |
| Meningitis | 6 | |
| Encephalitis | Both | |
| Encephalopathy | 6 | Basal ganglia/white matter abnormalities on brain MRI |
| Hepatitis | Both | |
| Lymphadenopathy | Both | |
| Lymphoproliferative disease | 6 | Proposed but unproven |
| Infections in patients who have had transplants | 6 | Viral reactivation |
| Hemophagocytic syndrome (macrophage activation syndrome) | 6 | Especially in patients who have had transplants |
| Drug hypersensitivity syndrome | Both, but mainly 6 | Proposed as complex interplay between drug reaction and HHV infection |
| HIV-1 cofactor | Both | Proposed but unproven |
| Pityriasis rosea | 7 | Controversial |
| Mononucleosis | Both | In adults |
| Multiple sclerosis | 6 | In adults; controversial |
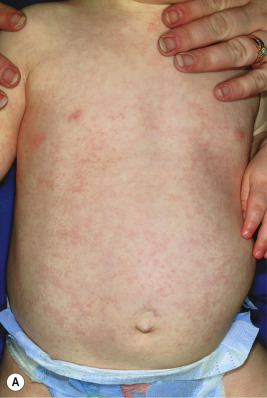
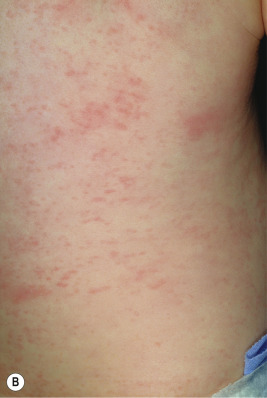
Associated signs and symptoms may include irritability, diarrhea, bulging fontanel, cough, cervical lymphadenopathy, and edematous eyelids. Periorbital edema is quite common and when present in a febrile but otherwise well-appearing child may be a useful clue to the diagnosis during the preexanthematous stage. Nagayama spots are erythematous papules involving the mucosae of the soft palate and uvula and represent the enanthem that occurs in up to two-thirds of patients. The differential diagnosis of the skin eruption in roseola infantum may include measles and rubella, although the temporal characteristics (rash after abrupt defervescence) are usually suggestive of roseola.
Primary HHV-6 infection may also be asymptomatic or may present in a manner distinct from classic roseola infantum. Nonspecific fever in infants with or without otitis media may often be the result of HHV-6 infection. First febrile seizures in young children may also commonly be associated with primary HHV-6 infection. Other potential central nervous system complications include encephalopathy and encephalitis, which tend to occur during the preexanthematous stage and may be associated with long-term neurologic morbidity. HHV-6-associated encephalitis has occurred in both immunocompetent and immunocompromised individuals. The prognosis is variable in these patients and ranges from complete recovery to severe neurologic sequelae or death. Solid-organ and bone marrow transplant recipients are at increased risk of reactivation disease, which may be asymptomatic or present with rash, fever, encephalitis, pneumonitis, hepatitis, or bone marrow suppression. In some patients, the clinical presentation of HHV-6 reactivation may mimic that of acute graft-versus-host disease.
Treatment for roseola infantum is unnecessary, and the illness usually spontaneously resolves without long-term sequelae. Although in vitro studies or clinical observations have suggested anti-HHV activity of ganciclovir, foscarnet, and cidofovir, these agents are rarely used clinically for patients with HHV-6 or HHV-7 infection. However, further research to identify effective treatment options is desirable given the wide array of complications that may be associated with severe infection or infection in the immunocompromised host.
Other Viral Exanthems
Nonspecific Viral Exanthems
Although several exanthematous eruptions may present with characteristic features such as lesion morphology or distribution, the majority are somewhat nonspecific and may be difficult to distinctly categorize. These eruptions may be hard to distinguish from drug reactions or in some younger patients, miliaria rubra (prickly heat). Unique defining characteristics such as an associated enanthem or symptom complex may be absent. Most nonspecific exanthems fall into the erythematous and/or papular categories, presenting with blanchable red macules and papules with a diffuse distribution ( Fig. 16-20 ). Associated symptoms, which are generally nonspecific as well, may include fever, headache, myalgias, fatigue, and respiratory or gastrointestinal complaints. Most nonspecific exanthems resolve without treatment over 1 week without long-term sequelae.
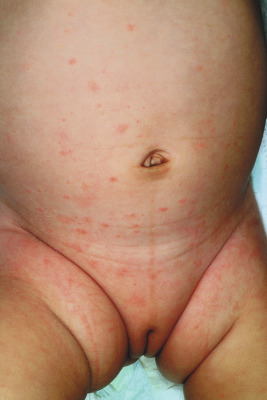
Common causes of nonspecific exanthems include nonpolio enteroviruses (see below) and respiratory viruses (i.e., adenovirus, rhinovirus, parainfluenza virus, respiratory syncytial virus, influenza virus). Other potential agents include Epstein–Barr virus (EBV), HHV-6 and HHV-7, and parvovirus B19, although these agents more often result in distinct exanthematous illnesses as discussed elsewhere in this chapter. In general, the majority of nonspecific exanthems occurring in the winter months are caused by the respiratory viruses and those occurring during the summer months are most often caused by the enteroviruses.
Petechial exanthems, in particular, may pose a diagnostic dilemma to clinicians (given their potential association with life-threatening infections) and are associated with increased rates of hospitalization and diagnostic testing. Multiple viral organisms may be associated with a petechial exanthem, including parvovirus, respiratory syncytial virus, influenza and parainfluenza viruses, rhinovirus, EBV, cytomegalovirus (CMV), adenovirus, and enteroviruses. In a prospective study of children (0 to 18 years of age) who had petechiae and signs or symptoms of infection, real-time PCR was utilized to identify a variety of viruses in nasopharyngeal aspirates. Of 58 patients with a petechial rash, 67% were positive for a viral pathogen, and 41% were notable for viral coinfections (two or more pathogens). The most common pathogens in this study were CMV and EBV (each 18% of the patients), followed by enteroviruses and rhinovirus (each 14% of the patients). Influenza A H1N1 and human bocavirus were also noted fairly often (9% each of the patients). Distinguishing clinically between viral and bacterial processes may be challenging. The ability to rapidly diagnose viral infection via quantitative PCR, however, may help minimize unnecessary antibiotic therapy, invasive diagnostics, and hospitalizations.
Gianotti–Crosti Syndrome
Gianotti–Crosti syndrome (GCS; papular acrodermatitis of childhood) was initially described in 1955 by Gianotti as an erythematous papular eruption symmetrically distributed on the face, buttocks, and extremities of children. A subsequent description by Crosti and Gianotti was made in 1956, and since then the disorder has been commonly known as Gianotti–Crosti syndrome. A viral etiology was suspected early on after the description of the disorder, and subsequently the notion of hepatitis B virus as a cause was hypothesized. It is currently well accepted that GCS is a distinct viral exanthem that may occur after infection with any of several viral agents, with hepatitis B being one possible (but uncommon) cause in certain parts of the world, especially Italy and Japan.
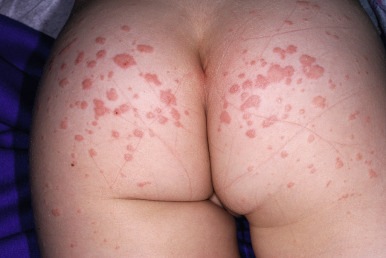
GCS occurs predominantly in children between the ages of 1 and 6 years. Before the appearance of the exanthem, upper respiratory symptoms, fever, and lymphadenopathy may be present. The eruption is characterized by edematous, erythematous, monomorphous papules and occasionally papulovesicles distributed symmetrically over the face, buttocks ( Fig. 16-21 ), and extensor surfaces of the upper ( Fig. 16-22 ) and lower ( Fig. 16-23 ) extremities. Occasionally the papules coalesce into larger, erythematous plaques ( Fig. 16-24 ). Hemorrhagic changes or localized purpura ( Fig. 16-25 ) may occasionally be present. The trunk is usually (but not always) spared in patients with GCS. In most patients the skin eruption is asymptomatic, although mild pruritus may be present.