Parasites are a fascinating and important cause of skin disease in children. They produce their effects in various ways: mechanical trauma from bites or stings, injection of pharmacologically active substances that induce local or systemic effects, allergic reactions in a previously sensitized host, persistent granulomatous reactions to retained mouth parts, direct invasion of the epidermis, or transmission of infectious disease by blood-sucking insects.
Arthropods
Arthropods are elongated invertebrate animals with segmented bodies, true appendages, and a chitinous exoskeleton. Those of dermatologic significance are the eight-legged arachnids (mites, ticks, spiders, and scorpions) and the six-legged insects (lice, flies, mosquitoes, fleas, bugs, bees, wasps, ants, caterpillars/moths, and beetles).
Arachnids
The term mite refers to a large number of tiny arachnids, many of which live at least part of their lives as parasites upon animals or plants or in prepared foods. Of greatest clinical significance are itch mites ( Sarcoptes scabiei ), grain mites, and harvest mites (chiggers). Avian mite dermatitis has also been recognized with increased incidence in recent years. Mites attack humans by burrowing under or attaching themselves to the skin, where they inflict trivial bites and cause associated dermatitides.
Scabies
Scabies is a common skin infestation caused by the mite S. scabiei . This mite is an obligate human parasite residing in burrowed tunnels within the human epidermis. Mites of all developmental stages may burrow into skin, depositing feces in their tracks. The female mite also lays eggs in these burrows, which serves to further propagate the infestation. The adult female mite has a lifespan of around 15 to 30 days, measures around 400 μm, and lays 1 to 4 eggs per day, which hatch in 3 to 4 days. After hatching, the larvae mature into adult mites in 10 to 14 days, and the duration of the whole life cycle is 30 to 60 days.
Scabies is transmitted most often by direct contact with an infested individual, although acquisition from fomites such as bedding and clothing is also possible. The survival time for a mite separated from the human host is estimated at several days. It is estimated that 15 to 20 minutes of close contact time is required for the transmission of scabies from one person to another. Scabies disproportionately affects women and children, as well as individuals with certain predisposing conditions, including immunocompromise, severe mental or physical handicap, and human immunodeficiency virus (HIV) infection. Scabies is seen worldwide in infants and children, and in the United States there seems to be a fairly high incidence in foreign-born adoptees, especially those from Asia or Latin America. This latter observation is likely related to the high incidence of infestation seen in orphanages. Scabies continues to be a significant public health issue in the developing world, with a disproportionate burden of disease noted in children who live in overcrowded, lower socioeconomic tropical regions. High endemic rates of scabies are noted in closed communities and institutional environments. In institutions, risk factors for scabies include institution type (with long-term care facilities and larger-sized institutions having higher incidence), extensive physical contact between patients and care workers, movement of patients, delay in diagnosis, and poor implementation of infection control or treatment.
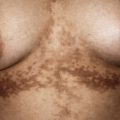
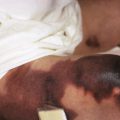
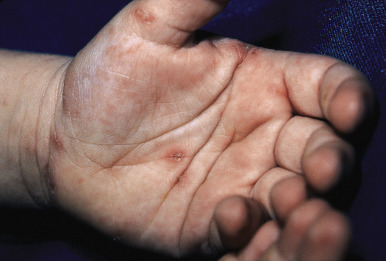
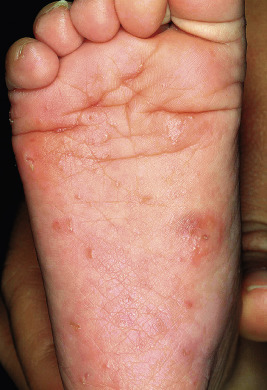
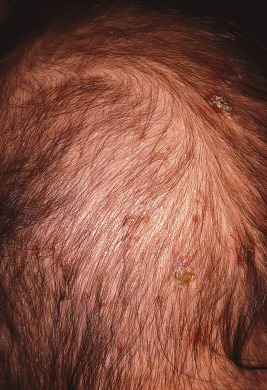
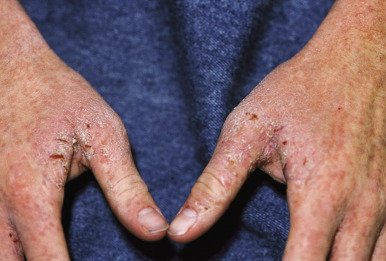
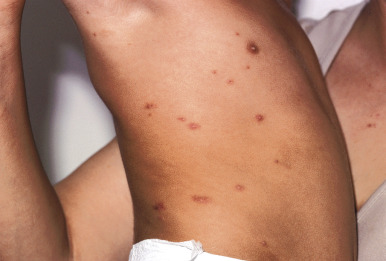
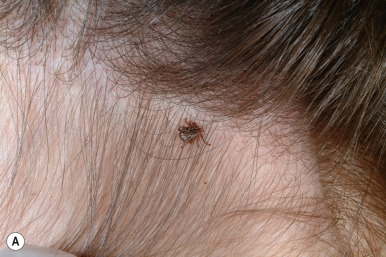
The incubation period for classic scabies is around 3 weeks, with reinfestation resulting in more immediate symptoms. The initial symptom of scabies is usually pruritus, which is often evident well before the clinical signs become apparent. Patients with scabies usually complain of worsening pruritus during nighttime hours. Infants and young children may also experience irritability and poor feeding. The skin findings include papules, nodules, burrows, and vesiculopustules ( Figs. 18-1 through 18-5 ). The most common locations for scabies lesions are the interdigital spaces, wrists, ankles, axillae, waist, groin, palms, and soles. Burrows on the palms may reveal a pattern of scale reminiscent of the wake left on a water surface by a moving boat and has been termed the wake sign. In infants, lesions may also be seen on the head ( Fig. 18-6 ), which is rarely involved in older patients. Infants commonly have involvement of the palms, soles, and axillae. In older children and adolescents, the most common sites of involvement are the wrists, interdigital spaces, and waist. Although bullous lesions are uncommon in scabies, vesicles are often found in infants and young children, owing to the predisposition for blister formation seen in this age group. Occasionally the Darier sign may be positive, and hence the diagnosis of urticaria pigmentosa may be considered. Genital involvement presenting as papules, crusted papules, or nodules ( Figs. 18-7 and 18-8 ) and areolar lesions are other classic presentation patterns. Excoriations are commonly noted.
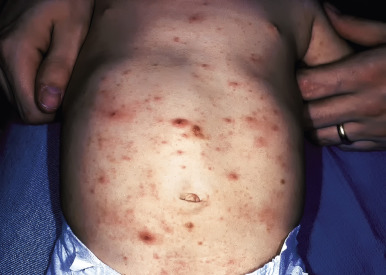
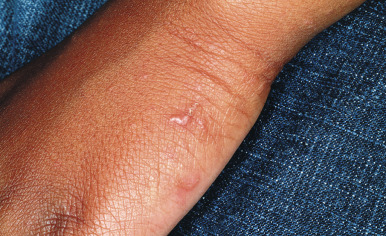
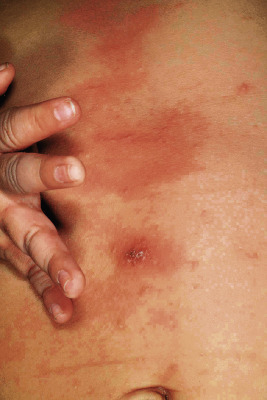
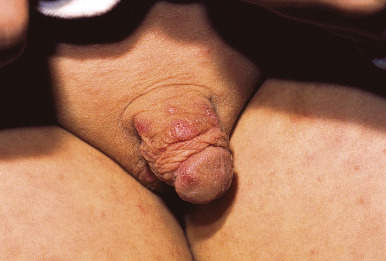
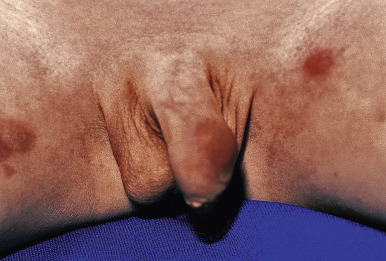
Scabies nodules are red-brown nodules ( Figs. 18-9 and 18-10 ) that represent a vigorous hypersensitivity response of the host. They occur most commonly on the trunk, axillary regions, and genitalia and are seen primarily in infants. Although they eventually resolve, scabies nodules may be present for several months. They are occasionally misdiagnosed (both clinically and histologically) as signs of a neoplastic disorder (i.e., leukemia or lymphoma cutis).
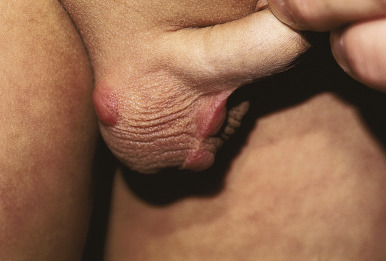
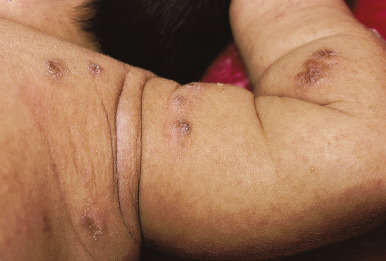
Crusted, or Norwegian, scabies is a form of the disorder that presents as scaly, dermatitic papules or plaques. Crusted scabies may be localized or generalized and occurs primarily in immunocompromised patients (especially those with HIV infection) and in those who are mentally retarded or physically incapacitated. The lesions of this disorder may mimic eczema ( Fig. 18-11 ), psoriasis, warts, or a drug reaction, and nail dystrophy may be present. Crusted scabies is commonly misdiagnosed and mismanaged. A delay in the diagnosis of crusted scabies may lead to the inadvertent exposure of multiple physical contacts. The lesions may become heavily crusted and hyperkeratotic and often are minimally pruritic. Crusted scabies is extremely contagious, given the large numbers of mites (thousands) that may be present. In classic scabies, only around 12 mites on average are present. Patients with crusted scabies are often the source for large epidemics within hospitals, given the lack of recognition and subsequent diagnostic delay.
Complications of scabies are generally mild. They include secondary bacterial infection, impaired skin integrity, pain, and rarely debilitation related to limitations in movement secondary to pain. Secondary infection is most commonly the result of Staphylococcus aureus or group A β-hemolytic streptococci and presents as crusting, oozing pustules or vesiculopustules. Bullous lesions have been described primarily in older adults, which on occasion may simulate (or possibly evolve into) the autoimmune blistering disorder, bullous pemphigoid. Necrotizing vasculitis in the presence of lupus anticoagulant has also been reported.
Animal-transmitted scabies, which is Sarcoptes infestation transmitted from domestic animals, usually results in a short-lived infestation with spontaneous resolution in the human host. The close relationship of humans with dogs makes the canine form of animal scabies ( S. scabiei var. canis ) the most common type transmitted to humans. Canine scabies, or sarcoptic mange, causes patchy loss of hair with scaling in the dog. It is seen most commonly in undernourished, heavily parasitized puppies, and a presumptive diagnosis can be made on the basis of exposure to a pet with a pruritic eruption, alopecia, and the characteristic mouse-like odor of animals with extensive sarcoptic infestation. In children with canine scabies the papular eruption most commonly involves the forearms, lower region of the chest, abdomen, and thighs. The distribution differs from that of human scabies in that it tends to spare the interdigital webs and the genitalia, and burrows are absent. The mite of canine scabies does not reproduce on human skin, and therefore the infestation is usually self-limited, clearing spontaneously over several weeks.
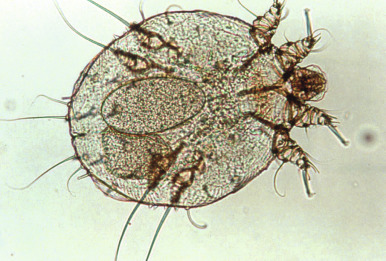
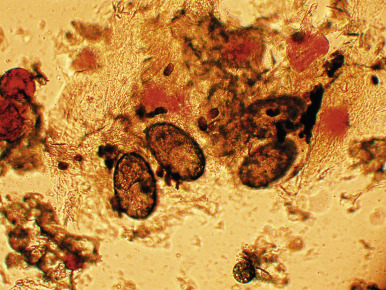
The diagnosis of scabies is suggested in the child with pruritus, a papular or papulovesicular eruption with burrows, and the characteristic distribution pattern. However, the differential diagnosis may be broad and includes atopic dermatitis, contact dermatitis, seborrheic dermatitis, Langerhans cell histiocytosis, impetigo, pyoderma, viral exanthem, papular urticaria, arthropod bites, and acropustulosis of infancy. The latter, which may represent a postscabetic hypersensitivity response, is discussed in detail in Chapter 2 . A definitive diagnosis of scabies is made via mineral-oil examination ( Box 18-1 ) with the microscopic identification of mites ( Fig. 18-12 ) or their eggs or feces (scybala) ( Fig. 18-13 ). Ideal skin lesions for sampling include burrows and fresh papules. In young patients in whom skin scrapings might be difficult, obtaining samples from adult contacts (i.e., parents) with lesions is a consideration. Skin biopsy is rarely necessary. A disposable skin curette (an instrument used for the destructive skin procedure, curettage) may be substituted for the scalpel blade if available and may be less frightening for young children and thereby minimize the risk of injury when performing the procedure in a very young or uncooperative child. High-magnification video dermatoscopy is a noninvasive technique that has been reported for diagnosing scabies.
- 1.
Apply a drop of mineral oil to the lesion(s) to be scraped.
- 2.
Scrape through the lesion with a number 15 scalpel blade (a small amount of bleeding is expected with appropriately deep scrapings).
- 3.
Smear contents of scraping on a clean glass slide.
- 4.
Add a few more drops of mineral oil.
- 5.
Place cover slip over oil and examine under microscope at low power.
Criteria for a positive mineral oil examination:
Scabies mite
or
Ova (eggs)
or
Scybala (feces)
Therapeutic options for scabies are listed in Table 18-1 , and a few are discussed here in more detail. The treatment of choice for scabies is 5% permethrin cream, which is applied from the neck down and left on for 8 to 14 hours and followed by thorough rinsing. Permethrin, a synthetic pyrethroid, is a neurotoxin and an excellent scabicide with low potential for toxicity. There appears to be little, if any, resistance of scabies to permethrin, and overall the response rate to treatment is excellent. A second treatment with permethrin 1 week after the first is often recommended, although studies suggest a relatively high cure rate after a single application. Treatment of all close contacts is also recommended in an effort to minimize ongoing propagation of the infestation. In a systematic review and meta-analysis of scabies therapies, permethrin was demonstrated to be the most effective treatment.
| Name | Instructions for Use | Comment |
|---|---|---|
| Permethrin 5% cream (Elimite, Acticin) | Apply from neck down; rinse in 8–14 hours | Not for use under 2 months of age; may repeat in 1 week if necessary; treat scalp in infants |
| Lindane 1% lotion | Apply from neck down; rinse in 8–12 hours | Not recommended for infants; not first-line therapy; potential CNS toxicity |
| Sulfur 6% ointment | Apply from neck down for 3 consecutive nights; rinse 24 hours after last application | Older therapy; malodorous; compounded in petrolatum; safe in infants, pregnant females |
| Crotamiton cream (Eurax) | Apply from neck down for 2 consecutive nights; rinse 48 hours after last application | High failure rate; may require up to 5 applications |
| Benzyl benzoate | Apply nightly or every other night for 3 applications | Not available in the United States |
| Ivermectin (Stromectol) | 200 μg/kg per dose given orally for 2 doses, 2 weeks apart | Off-label use; consider for severe infestations, crusted scabies, IC patients, scabies epidemics; should not be used under 5 years of age |
Permethrin should be applied in a thin, even coat and rubbed in well to all skin surfaces. In infants, application should include the scalp and face, with care to avoid the regions around the eyes and mouth. Special attention should be paid to applying the medication to web spaces, the umbilicus, the genitals, and the gluteal cleft. Fingernails and toenails should be trimmed short and the medication applied as well as possible to the region under the nail edge. Permethrin is not recommended for infants younger than 2 months of age or for pregnant (pregnancy class B) or nursing women. It is vital for patients and parents to understand that after appropriate therapy, signs and symptoms of scabies may not clear for 2 to 6 weeks after successful treatment, because the hypersensitivity state does not cease immediately after eradication of the infection. The family should have this information before starting therapy, and it may be useful to provide a topical corticosteroid and oral antihistamine for symptomatic relief during this period.
Lindane 1% lotion was, for years, the primary therapy for scabies before the development of permethrin and escalating reports of lindane-related toxicities. Lindane is applied to the skin for 8 to 12 hours with reapplication in 1 week. Although the majority of untoward effects attributed to the use of lindane have been associated with inappropriate, prolonged, or repetitive use, true toxicity potential does exist and becomes more relevant in infants and small children, owing to a relatively greater skin surface and possibly higher blood level accumulations in this age group. When in vitro percutaneous absorption of 5% permethrin was compared to 1% lindane, it was shown that human skin was 20-fold more permeable to lindane. In addition, in vivo guinea pig blood and brain levels of lindane were fourfold greater than permethrin levels after a single application. Reactions attributed to the use of lindane have included eczema, urticaria, aplastic anemia, alopecia, muscular spasms, and central nervous system toxicity manifested by irritability, nausea, vomiting, amblyopia, headache, dizziness, and convulsions. Lindane-resistant scabies has also been an increasing problem in the United States and Central America. This agent, which is recommended as a second-line therapy, is banned in California. Other topical agents utilized in some areas for the treatment of scabies include 10% to 25% benzyl benzoate, 10% crotamiton, 0.5% malathion, and 2% to 10% sulfur ointment.
Ivermectin has been reported with increasing incidence as an off-label treatment option for scabies (it is not approved for this indication in the United States). This agent has been used extensively in veterinary medicine since 1981 for a wide variety of parasitic infestations in farm and domestic animals. Oral ivermectin (Stromectol) is approved for the treatment of Strongyloides and onchocerciasis in adult humans. It is also effective for the treatment of loiasis and bancroftian filariasis. Ivermectin acts by blocking chemical transmission across invertebrate nerve synapses that utilize glutamate or γ-aminobutyric acid (GABA), resulting in paralysis and death. It has selective activity against human parasites because of its high affinity for the channels found in the peripheral nervous system of invertebrates. Several studies have evaluated the role of ivermectin in the treatment of scabies and found it very effective in either a single- or two-dose regimen of 200 μg/kg per dose. Many recommend that this agent be given in two doses when used for classic scabies, separated by 1 to 2 weeks, because it is not ovicidal. Ivermectin is especially useful in immunocompromised patients with scabies and patients with crusted scabies, where it may be used alone or in combination with topical scabicides or keratolytic agents. When it is used in crusted scabies, ivermectin may need to be administered in three to seven doses, depending on the severity of the infection. Topical 1% ivermectin, which is not readily available, has reportedly been effective as well.
Regardless of the treatment used for scabies, environmental decontamination is important given the possibility of spread of scabies from fomites. Clothing, bed linens, and towels should be machine washed in hot water and dried using a high-heat setting. Clothing or other items (i.e., stuffed animals) that cannot be washed may be dry cleaned or stored in bags for 3 days to 1 week, because the mite will die when separated from the human host. In addition, systemic antibiotic therapy should be given if secondary bacterial superinfection is present. Scabies nodules can be treated symptomatically with topical or intralesional corticosteroids.
Other Mites
Mites are small arachnids with mouthparts capable of puncturing and feeding on host tissue fluids. A variety of food and animal mites may result in pruritic dermatoses in exposed humans, and may also be vectors of infectious diseases. Mite bites should be considered in the patient with an unexplained, itchy papular skin eruption.
Harvest mites (chiggers, jiggers, “red bugs”) are a member of the American harvest mite family and are commonly found in the southern United States. Also known as Trombicula alfreddugesi, the chigger is distinct from other mites in that only the larva is parasitic to humans and animals. The eight-legged adult and nymphal stages are spent in a nonparasitic existence. Chiggers live in grain stems, in grasses, or in areas overgrown with briars or blackberry bushes, where they exist and feed on vegetable matter, minute arthropods, and insect eggs. The six-legged larva clings to vegetation awaiting the passage of an unsuspecting host and then attaches to the skin when the host brushes against the foliage. The mite then injects an irritating secretion that causes itching and then drops to the ground to molt (or is scratched off) within a few days. The lesions of chigger bites present as urticarial, erythematous papules ( Fig. 18-14 ). They occasionally reveal a hemorrhagic punctum, and in some patients there may be more diffuse erythema, vesicles, or bullae. Pruritus tends to be intense. The “summer penile syndrome” refers to a seasonal acute hypersensitivity reaction to chigger bites and presents with penile swelling, pruritus, and occasional dysuria. Most patients have a history of recent exposure to woods, parks, or lawns.
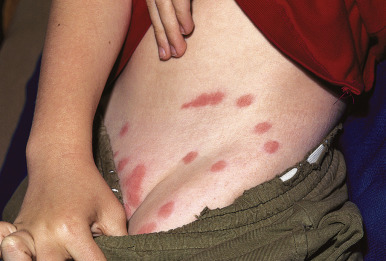
Treatment of chigger bites consists of antihistamines, cool baths or compresses, and topical corticosteroids. Secondary infection should be treated with systemic antibiotics. Household vinegar (5% acetic acid) has been suggested as a useful measure for postexposure prophylaxis and treatment of pruritus. Chiggers may be disease vectors for scrub typhus, hemorrhagic fever with renal syndrome, hantavirus pulmonary syndrome, and ehrlichiosis.
Grain mites (straw itch mite, Pyemotes ventricosus ) feed on the larvae of insects, seeds, grains, and plant stems. Persons coming into contact with infested straw and grain are particularly susceptible to this form of dermatitis. This seasonal eruption presents as severely pruritic, pale pink to bright red macules, papulovesicles, or pustules followed by urticarial wheals. Occasionally, a purpuric eruption develops. In severe cases, constitutional symptoms and fever may be present, and the presentation may be mistaken for acute varicella. The eruption is self-limited and treatment, as with chigger bite reactions, is supportive.
Avian mites (fowl mites, bird mites) are divided into two genera, Dermanyssus and Ornithonyssus . They are known to infest humans accidentally, resulting in a pruritic, widespread papular dermatitis. Gamasoidosis is the term used to describe the human skin disorder resulting from nonburrowing, blood-sucking mites from birds and other animals. The diagnosis of avian mite dermatitis is often overlooked because of lack of awareness and the small size of the mites. Common causes include Dermanyssus gallinae (chicken mite), Dermanyssus americanus (American bird mite), Ornithonyssus sylviarum (northern fowl mite), and Ornithonyssus bursa (tropical fowl mite). D. gallinae , the most common cause, infects various birds including chickens, parakeets, pigeons, canaries, and starlings. Although the mite remains on the bird at night, during the day it may migrate to the nest and can survive without feeding for several months. When birds leave their nest in the spring or early summer, the mites search for other hosts, and human infestation may be the result. Clinical examination reveals papules ( Fig. 18-15 ), vesicles, and urticarial lesions without burrows, because the mite does not burrow into the skin. The diagnosis of avian mite dermatitis is confirmed by identification of the mites found in pillows, bed sheets, clothing, nests, birds, and only rarely humans. Special attention should be given to potential sites for nesting, such as porches, attics, eaves, air conditioning systems, and ventilation ducts. Pet gerbils were the source for O. sylviarum and D. gallinae in one report. Treatment for avian mite dermatitis, as with other mite bites, is symptomatic.
Other mites may result in similar, nonspecific, pruritic papular eruptions in the human host. The rat mite, Ornithonyssus bacoti, is found in rats, and extermination is often necessary for eradication. It has also been reported in pet hamsters. Cheyletiella mites are large, animal-specific nonburrowing mites that include Cheyletiella blakei (found on cats), Cheyletiella yasguri (found on dogs), and Cheyletiella parasitovorax (found on rabbits). When these mites are in close proximity to a human host, they bite quickly and run, returning to their animal host. They are very difficult to visualize with the human eye. Skin lesions include grouped pruritic papules, urticarial wheals, and bullous lesions. Pets infested with Cheyletiella exhibit patches of fine, powdery scale (“walking dandruff”). Other mites of potential human concern include house-dust mites ( Dermatophagoides species), the snake mite ( Ophionyssus natricis ), and the house-mouse mite ( Liponyssoides sanguineus ).
Demodex mites are present normally in adult human hair follicles and sebaceous glands of the face, where they are considered to be “commensal ectoparasites.” They are less commonly present in childhood skin. The two species are Demodex folliculorum and Demodex brevis . “D emodex folliculitis” presents as a very itchy papular and pustular eruption on the face, usually in children with leukemia or HIV infection. There may be an accentuation in the perioral regions. Reported topical therapies have included 5% permethrin, metronidazole, sulfur, and sodium sulfacetamide.
Ticks
Ticks are large, globular arachnids with short legs, hard leathery skin, and mouthparts adaptable for sucking blood from mammals, birds, and reptiles. They are important vectors of diseases such as relapsing fever, rickettsial infections (Rocky Mountain spotted fever, Mediterranean spotted fever, Q fever, and ehrlichiosis; see Chapter 16 ), Lyme disease (see Chapter 14 ), babesiosis, Colorado tick fever, and tularemia. Although tick bites may be painful, the majority are painless and may go unnoticed.
Ticks are classified into three family groups: hard ticks (Ixodidae), soft ticks (Argasidae), and Nuttalliellidae. They are found in grass, shrubs, vines, and bushes, from which they attach themselves to dogs, cattle, deer, and humans. Ticks penetrate the human epidermis with distal mouthparts called chelicerae ( Fig. 18-16 ) and then insert another part called the hypostome, through which they feed on blood and deposit a variety of agents including anticoagulant and anti-inflammatory agents. Both acute and chronic dermatoses may result from tick bites. Acute changes include erythema, papules, nodules, bullae, ulceration, and necrosis. Multiple pruritic papules caused by infestation with tick larvae have also been reported. Chronic changes include granulomas, alopecia, and secondary bacterial infection. Localized foreign-body-type reactions may result from retained mouthparts or improper tick removal and occasionally persist for months to years. T- or B-cell cutaneous lymphoid hyperplasia may also be noted, often with copious numbers of eosinophils when tissue biopsies are analyzed histologically. The B-cell type may be difficult to distinguish from true B-cell lymphoma.

Nondermatologic findings that may result from tick bites include anaphylaxis, fever, headache, flu-like symptoms, abdominal pain, and vomiting. Tick paralysis is a reversible disorder characterized by ascending motor weakness and flaccid paralysis. It is believed to be caused by a salivary neurotoxin injected into the victim while the tick is engorging, and it appears that the tick must feed for several days before paralysis occurs. Tick paralysis occurs more commonly in young girls, possibly owing to their longer hair, which may augment tick attachment and hiding. The classic clinical presentation includes an unsteady, ataxic gait followed by ascending symmetric paralysis, usually starting within 2 to 6 days of tick attachment. Upper extremity weakness and cranial nerve involvement (which may present as dysphagia, drooling, facial weakness, and dysphonia) may occur. If intervention does not occur, involvement may extend to the respiratory musculature, resulting in respiratory failure and subsequent death. Most cases of tick paralysis occur in children, and the mainstay of treatment is tick removal, which usually results in rapid resolution of the signs and symptoms.
Various methods of prevention for tick-borne diseases have been advocated. Avoidance of tick habitats may reduce risks, but this is not always feasible. Recommendations for clothing include wearing light-colored, long-sleeved clothes and footwear, tucking pants into socks and taping exposed edges. Insect repellents containing N,N-diethyl-3-methylbenzamide (DEET) or picaridin applied to exposed areas of skin and permethrin applied to clothing are both effective parts of prevention (see below). Frequent examination of skin for ticks is also important in high-risk areas and during high-risk seasons.
Prompt tick removal is also important in reducing the transmission of disease. The entire tick must be removed completely, including the mouthpart and the cement the tick secretes to secure attachment. A variety of commercial tick-removal devices are available and are quite successful. Manual extraction of a tick is performed with blunt, medium-tipped forceps. The tick is grasped as close to the skin as possible, and perpendicular traction is used to gradually extract the entire tick, taking care not to twist the forceps. Should a portion of the tick be retained, a cutaneous punch biopsy will effectively remove it.
Spiders
Spiders, because of their menacing appearance, are often blamed for more damage than they actually create. Virtually all of the serious spider bites on the North American continent are caused by the black widow spider and the brown recluse spider. Bites of the former tend to have milder outcomes, whereas bites of the latter often result in more severe sequelae. Children are more vulnerable to the effects of spider bites. Patients reporting spider bites are often diagnosed in emergency departments as having skin or soft-tissue infection. The gold standard for diagnosing spider bites is collection and proper identification of the biting spider, although this rarely occurs.
The black widow spider, or Latrodectus mactans , is a potentially dangerous spider found mainly in southern Canada, the United States, Cuba, and Mexico. It is recognized by its coal-black color and globular body (1 cm in diameter) with a red or orange hourglass marking on the underside of its abdomen ( Fig. 18-17 ). A web spinner (in contrast to burrowing spiders), it lives in cool, dark places in buildings and little-used structures, woodpiles, and garages, and often spins its web across outdoor furniture. Consequently, a significant number of bites in the southern United States occur around the genitalia and buttocks.

The black widow spider bites humans only in self-defense. The female spider is more dangerous than the male as a result of her larger size and more potent neurotoxin. α-Latrotoxin, the potent black widow neurotoxin, triggers synaptic vesicle release from presynaptic nerve terminals and results in its effects via exocytosis of neurotransmitters. Two red, punctate marks and local swelling are often seen, and burning or stinging with intense pain develops at the site of the envenomation. This is followed, usually within 10 minutes to an hour, by severe cramping abdominal pain and spasmodic muscular contractions, which peak in around 3 hours. Irritability, sweating, anxiety, and agitation are also common. Priapism may occur. Hypertension within the initial hours after the bite occurs in 20% to 30% of patients, and although it may be severe, it is often asymptomatic in children. Although the bite may be fatal, most children recover spontaneously in 2 to 3 days.
Treatment options for black widow spider bites include analgesics, benzodiazepines, calcium gluconate, and specific antivenin. Diazepam is the most commonly utilized benzodiazepine. Intravenous opioids are also utilized and seem very effective in providing symptomatic relief. Antivenin therapy has been demonstrated useful, even when given as late as 90 hours after the bite. Although specific antivenin may not be available in some parts of the world, redback-spider ( Latrodectus hasselti ) antivenin has also been shown to be effective in neutralizing L. mactans venom in mouse models. Indications for the administration of Latrodectus antivenin are shown in Table 18-2 , although use of this agent remains controversial in the United States. Potential reactions to antivenin include anaphylaxis and serum sickness.
| Indication | Comment |
|---|---|
| Hypertension | If uncontrolled |
| Pregnancy | |
| Comorbid conditions | Coronary artery disease, chronic obstructive pulmonary disease |
| Intractable pain | |
| Respiratory difficulty |
Spiders of the Loxosceles genus cause cutaneous and subcutaneous injury primarily via the enzyme sphingomyelinase D (SMD) (see below). There are roughly 100 species of Loxosceles spiders, with 80% of them being found in the Western hemisphere. The brown recluse spider, or Loxosceles reclusa, is the one of the most dangerous spiders in the United States. This spider is indigenous to the area spanning eastward from southeastern Nebraska and the eastern half of Texas to the western part of Georgia. The northern boundary includes southern Missouri, Illinois, Indiana, and Ohio, and the southern boundary is the Gulf of Mexico. In nonendemic areas, such as the western United States, physician overdiagnosis of brown recluse bites (also known as loxoscelism ) is common.
The brown recluse spider has an oval, tan to dark brown body, about 1 cm in length and 4 to 6 mm in width. A dark-brown violin-shaped marking extends dorsally from the eyes back to the distal cephalothorax ( Fig. 18-18 ), and there are three pairs of eyes, rather than the four seen in other spiders. The brown recluse spider thrives in human-altered environments such as attics, basements, and boxes. When in the house, the spider is often found in storage closets (among clothing); when outdoors, it generally resides in grasses, rocky bluffs, woodpiles, and barns. Because of its normal shyness and predilection for dark recesses, it bites only in self-defense when approached.

The venom of L. reclusa contains numerous enzymes, most notably SMD2, as well as alkaline phosphatase, esterase, adenosinetriphosphatase (ATPase), and hyaluronidase. SMD may result in many effects, including lysis of red blood cells, complement activation, platelet activation, and thrombosis. Activation of neutrophils results in the classic cutaneous necrosis seen and is a result of neutrophil secretion with subsequent endothelial damage, thrombosis, and ischemia. The local effect on tissues seems to be directly related to the amount of venom injected.
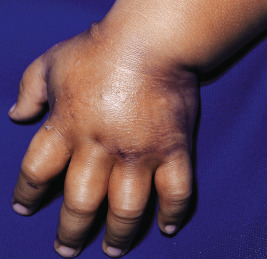
The clinical presentation of loxoscelism begins within 6 hours of the bite, usually with pruritus, pain, and/or erythema at the site. These bites, however, may be relatively painless, and patients may be unaware of them occurring, which accounts for the rarity of spider identification in diagnosing these bites. Within 24 hours, a red ring develops around the bite site and is followed by blue-purple discoloration ( Fig. 18-19 ), occasional hemorrhagic blisters, and an increase in pain. Ultimately, gangrenous ulceration with necrosis and eschar formation occurs ( Fig. 18-20 ), occasionally with bulla formation. Anesthesia is often present in the center of the affected area. A generalized petechial or morbilliform eruption may also be present. Systemic findings may include chills, fever, nausea, and joint pain. Rare findings include severe hemolysis, renal failure, and pulmonary edema. Necrotizing fasciitis has also been reported. Lymphangitis is not uncommon when the bite occurs on an extremity. In mild cases, L. reclusa bites may result only in a mild urticarial reaction. Systemic loxoscelism is uncommon overall (but when it occurs, it appears to be more common in children), and symptoms may include fever, malaise, vomiting, headache, and acute hemolytic anemia with jaundice. Renal failure rarely occurs.
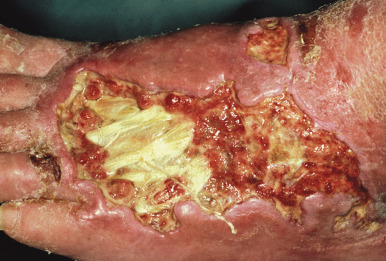
Brown recluse spider bites heal very slowly, and scarring often results. Progression to pyoderma gangrenosum-like ulcers has been reported but is rare. For the majority of small, uncomplicated bite reactions, spontaneous healing is the rule. Conservative treatment measures for these patients include rest, ice compression, and elevation (RICE) of the affected area. Antibiotics should be considered if secondary bacterial infection is suspected. Aspirin, antihistamines (most notably cyproheptadine), and tetanus vaccination may be considered, and dapsone therapy has been utilized but must be closely monitored given the toxicity profile. Dapsone is felt by some to be effective when administered early in the course of the bite reaction and may have its therapeutic effects via suppression of neutrophil chemotaxis. However, side effects including dose-related hemolysis, agranulocytosis, cholestasis, and methemoglobinemia limit its widespread use. When systemic signs and symptoms are present, fluid support should be given and systemic corticosteroids may be considered. Other reported therapies have included hyperbaric oxygen, vasodilators, heparin, nitroglycerin, electric shock, curettage, and surgical excision. Loxosceles antivenin is available in some countries.
Scorpions
Scorpions are tropical photophobic arachnids that hide by day and hunt by night. They differ from other arachnids in that they have an elongated abdomen ending in a stinger ( Fig. 18-21, A ). Scorpions are found worldwide, particularly in the tropics, and in North America they are generally seen in the southwestern United States and Mexico. The Buthidae family are the most toxic of scorpions and are found mainly in India, Spain, the Middle East, and northern Africa. The major scorpion offender in the United States is Centruroides exilicauda (also known as Centruroides sculpturatus or the “bark scorpion”).

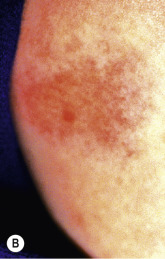
Although the majority of scorpion stings in the United States are not severe and do not warrant significant medical therapy, children seem to be at greater risk of developing severe scorpion envenomation. Hence stings in children should be watched carefully, particularly during the first 4 hours, to assess for a more serious reaction. Most symptoms have their onset within 15 minutes after envenomation. The venom apparatus of the scorpion is carried in its curved stinger at the tip of the tail, which is swung over the scorpion’s head to penetrate the victim’s skin. During the day scorpions hide in shoes, closets, clothing, and crevices. Some species of ground scorpions, however, may burrow and hide in gravel or children’s sandboxes. They rarely attack humans but will do so when accidentally disturbed, brushed against, or stepped upon. The effect of the sting depends upon the amount of envenomation and the age and size of the individual. The majority of published reports of severe or fatal envenomation have been in children younger than 10 years of age.
During a sting, the scorpion releases two noxious agents: a localized hemolytic toxin and a dangerous neurotoxic venom. The hemolytic toxin may cause a painful burning sensation with pronounced redness ( Fig. 18-21, B ), swelling, discoloration, severe necrosis, lymphangitis, and in some patients, disseminated intravascular coagulation or renal failure. The neurotoxic venom, which reaches the systemic circulation primarily via the lymphatics, may produce local numbness and a severe generalized reaction consisting of sweating, salivation, tightness in the throat, abdominal cramps, cyanosis, muscle jerking, tongue fasciculations, convulsions, and particularly in small children, respiratory paralysis and death. Tachycardia and hypertension are common and may persist for several hours. Pulmonary edema, with or without hemoptysis, and shock may develop. Local cutaneous reactions may vary and include erythema, petechiae, purpura, bullae, edema, induration, necrosis, and ulceration. The diagnosis of scorpion sting is a clinical one, and experienced clinicians who practice in endemic regions can often recognize the clinical symptom pattern.
Treatment for scorpion envenomation is difficult and without consensus on one gold standard of care. Immediate measures include application of a tourniquet above the area of the sting and the application of ice or cold water. Serotherapy, which refers to administration of scorpion antivenin, has been advocated by some but has been found ineffective by some investigators, and does not prevent the development of cardiac problems. Antivenin Centruroides (scorpion) equine immune F(ab′) 2 (Anascorp) has been approved by the United States Food and Drug Administration (FDA) as an orphan drug and has been shown to be associated with more rapid resolution of envenomation symptoms, decreased use of sedative-hypnotics, and lower levels of plasma venom levels.
Fluid resuscitation, sedation, and treatment of hypertension may be indicated after scorpion stings. Prazosin therapy has been shown to decrease the development of acute pulmonary edema. A large series of severe scorpion envenomation in children suggested that the beneficial effects of antivenin and/or prazosin is questionable when hospital admission is delayed. Continuous intravenous midazolam infusion has also been utilized to control agitation and involuntary motor activity. Opioid analgesics may be required for pain relief. Although intravenous high-dose corticosteroid therapy was once recommended, it appears to offer no significant benefit in terms or mortality, duration of hospital stay, or cost.
Insects
Insects are the class of arthropods characterized by division into three parts (a head, thorax, and abdomen). Noxious insects are ubiquitous, affecting all humans in some manner at one time or another. The insects of medical significance include lice, mosquitoes, flies, fleas, bed bugs, bees, wasps, ants, caterpillars/moths, and beetles.
Lice (Pediculosis)
Lice have plagued humans since ancient times. Infection is most common during times of stress, such as war, or in crowded environments such as schools, camps, or institutions. After widespread use of dichlorodiphenyltrichloroethane (DDT) after the end of the Second World War, there were relatively few reports of pediculosis in the United States. Subsequent restrictions on the use of DDT in the United States since 1973, however, resulted in an increase in the number of cases, particularly pediculosis capitis and pediculosis pubis. Lice occur wherever there are humans. They spend their entire life as ectoparasites depending on human blood for sustenance. Their existence independent of humans is generally not possible. All lice feed by pressing their mouth against the host’s skin, piercing the surface, and injecting an anticoagulant to facilitate the blood flow during feeding.
Lice are small, six-legged, wingless insects with translucent, gray-white bodies that become red when engorged with blood. They measure 1 to 4 mm in size, and are visible to the naked eye on close inspection. Three species of lice infest humans:
- 1.
Pediculus humanus corporis (the body louse)
- 2.
Pediculus humanus capitis (the head louse)
- 3.
Phthirus pubis (the pubic or “crab” louse).
Body lice and head lice ( Fig. 18-22 ) are very similar in appearance, with elongated bodies and three pairs of claw-like legs. Pubic lice, on the other hand, have short bodies and resemble crabs in appearance. The body louse is the only louse that can carry human disease, including louse-borne relapsing fever, trench fever, and epidemic typhus. Body and head lice are transmitted by close contact or occasionally via fomites including clothing (body louse) and combs, brushes, and hats or other headgear (head louse). Pubic lice are transmitted by intimate physical contact and are considered in most cases to be a sexually transmitted disease. Nonsexual transmission of pubic lice may occur, though, and probably accounts for most cases of infestation involving the eyebrows and/or eyelashes in children, although the possibility of sexual abuse should always be considered in this setting.

Lice egg cases are called nits and are firmly attached to the hair shaft with a cement that makes them difficult to remove. They are gray to yellow-white in color and are seen as tiny pinhead specks measuring 0.3 to 0.8 mm ( Fig. 18-23 ). The nits of head lice are attached to the hair shaft at a more acute angle than those of pubic lice. Head-lice nits are usually laid within 1 to 2 mm of the scalp and, in general, attached nits further from the scalp are more likely to be nonviable. Nits are usually easily distinguished from hair casts or dandruff by the inability to remove them from the hair shaft and, if necessary, microscopic evaluation. The differential diagnosis of nits is shown in Table 18-3 . Nits incubate for about 1 week, and then young lice hatch, passing through several nymph stages and growing into adult lice over 1 to 2 weeks. P. corporis (body lice) most commonly occurs in homeless individuals, refugees, victims of wars and natural disasters, and those living in crowded conditions. The nits of the body louse attach firmly to clothing fibers, where they may remain viable for weeks. The louse itself is rarely observed on the skin. It obtains its nourishment by clinging to the patient’s clothing and intermittently piercing the skin. The primary lesion is a small, red macule, papule, or urticarial wheal with a hemorrhagic central punctum. Primary lesions may be difficult to visualize owing to excoriations related to scratching, and secondary impetiginization may be present. The diagnosis of body lice is confirmed by finding lice or nits in clothing, often in seams. Treatment consists primarily of thorough deinfestation of all clothing and bedding. Treatment of the patient with 5% permethrin cream or lindane 1% lotion may also be useful, because an occasional louse may remain and return to feed.
| Diagnosis | Comment |
|---|---|
| Nits | Firmly adherent to hair shaft; not easily removed with fingers |
| Seborrheic dermatitis (dandruff) | Diffuse scalp scaling; scales occasionally adhere to hair but easy to remove; scalp erythema may be present |
| Hair casts | Keratin protein that encircles hair shaft; easily removed |
| Piedra | Fungal infection of hair; firm nodules attached to hair shafts, white or black in color |
| Psoriasis | Thick silvery scales, often present overlying red plaques on the scalp |
| Hair products | i.e., hairspray, mousse, gel |
Pediculosis pubis (pubic lice, crab lice) involves primarily the pubic area but may also involve the scalp, eyebrows and eyelashes, beard, and other hairy areas. Blepharoconjunctivitis may be the presenting signs when eyelashes are involved, and this pattern has been reported in infants and children. Live lice and nits are usually seen. Itching may be the initial symptom, but in persistent cases eczematization or secondary infection may occur. A characteristic skin finding is maculae ceruleae, which are gray-blue macules on the abdomen and thighs. They are felt to represent hemosiderin deposition in the deep dermis as a result of the bites. Treatment of pubic lice is generally similar to that used for head lice (see below), as well as laundering of all clothing and bed linens. Eyelash involvement is usually treated with an occlusive agent such as petroleum jelly.
P. capitis (head lice) is by far the most common form of lice to affect children and usually affects those between 3 and 12 years of age. As mentioned, it is spread primarily through head-to-head contact and less commonly via fomites including headgear, combs, brushes, towels, and upholstery. The transmission from fomites may contribute to the increasingly challenging cycle of head-lice infestation. However, because most transmission occurs from direct contact with the head of an infested individual, the focus of control activities should be primarily on reducing the number of lice on the head and decreasing head-to-head contact. All socioeconomic groups are affected, although African-Americans are less often infested with head lice, possibly related to the diameter, shape, or twisted nature of their hair shafts (which makes grasping of the shaft more difficult for the louse). The clinical findings consist primarily of pruritus, with secondary excoriation and occasional secondary bacterial infection. Cervical and suboccipital lymphadenopathy may be present. Head lice do not transmit other infectious disease agents. The diagnosis is confirmed by finding live lice ( Fig. 18-24 ) or viable nits on the scalp. In patients in whom the diagnosis is based only on nits, their viability can be confirmed by mounting them on a glass slide and performing microscopic evaluation at low power. Viable nits have an intact operculum (cap) on the nonattached end and a developing louse within the egg ( Fig. 18-25 ).