Ear Reconstruction
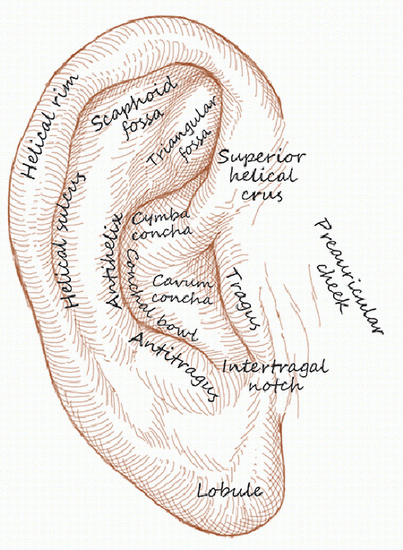 |
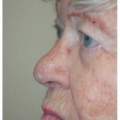
The ear is a complex structure whose lateral surface provides minimal loose tissue for side-to-side or flap repair. The skin on this side is bound down to a convoluted cartilaginous structure with convex and concave surfaces. As such, repair on the lateral surface is usually limited to second intention healing, skin graft, or a flap from the medial pinna or other nearby cosmetic unit (i.e., preauricular cheek or postauricular scalp). The skin on the medial surface of the ear is a little looser and may provide adequate tissue for side-to-side or flap repair of small- to medium-sized defects, although flap repair frequently recruits tissue from the postauricular sulcus area. The helical rim provides the characteristic shape to the pinna, and reconstruction is most successful if the shape, size, and appearance of this contiguous structure is maintained. Fortunately, for many defects on the helical rim, the inferior helical rim and lobule provide an adequate reservoir of loose tissue from which to fashion a local flap repair.
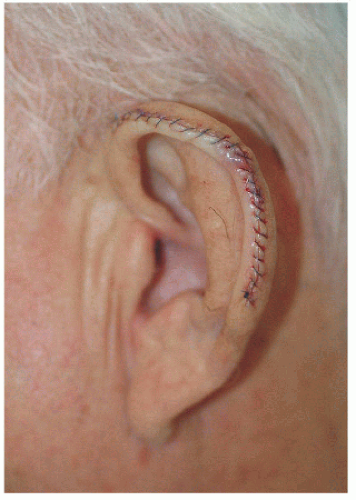
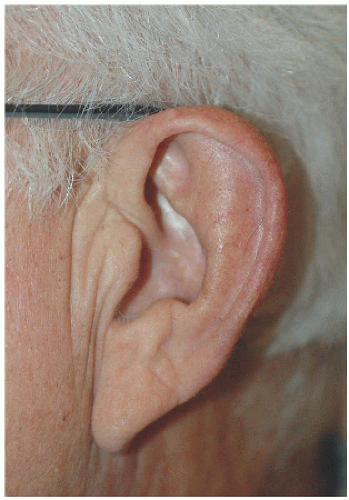
7.1 HELICAL RIM: SIDE-TO-SIDE REPAIR
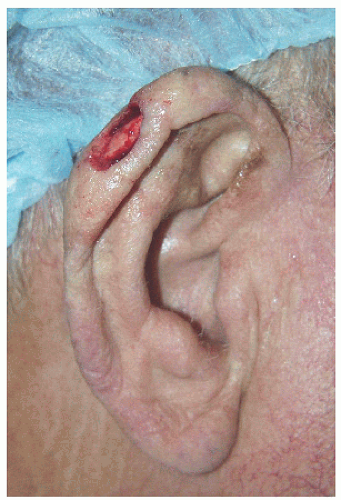
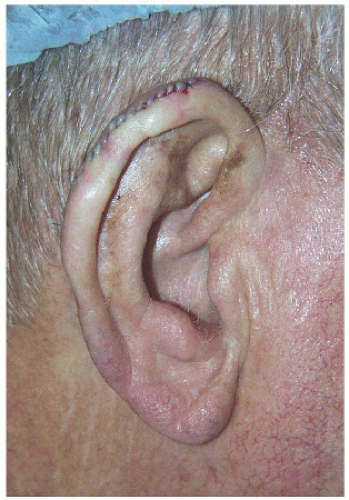
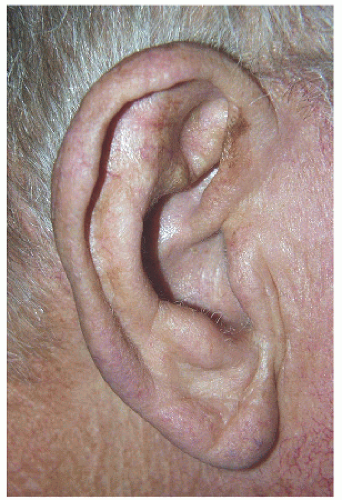
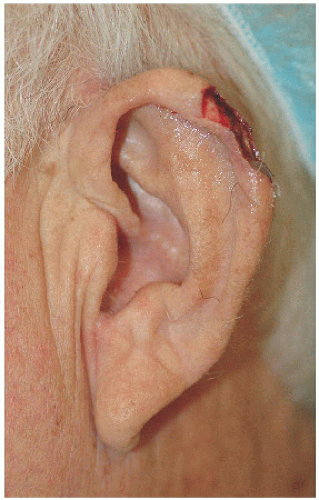
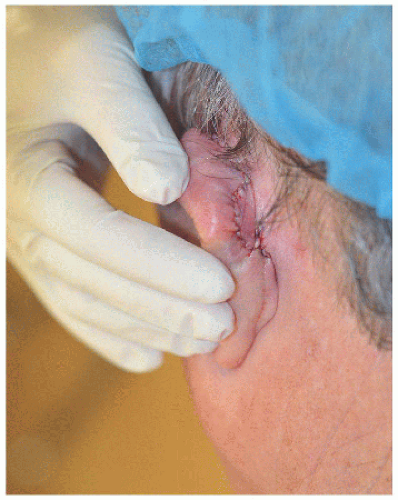
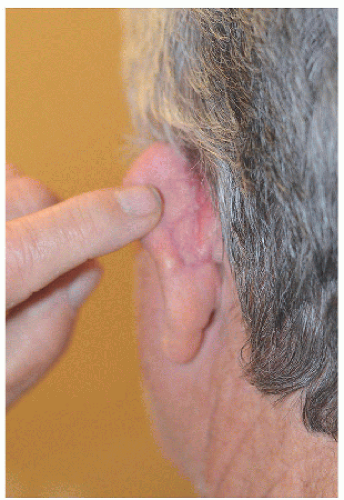
Smaller defects often require simpler repairs. This small defect on the helical rim (Fig. 7.1A) is easily closed with a side-to-side or complex linear repair (Figs. 7.1B and C). Especially in older patients, side-to-side repair may be an excellent alternative in cases where flap reconstruction might have to be considered. Areas with significant laxity and redundant tissue combined with adequate undermining (e.g., in this case on the posterior pinna) can facilitate closure in a side-to side fashion (Figs. 7.1D, E, F).
Key Points
Side-to-side or complex linear closure may work well for small- to medium-sized defects of helical rim, lobule, or postauricular sulcus defects, especially in older patients with redundant tissue.
Over a curved surface such as the helical rim, the usual 3:1 length-to-width ratio for closure of a side-to-side wound may have to be increased to 4:1 or more to avoid a persistent standing cone or tricone at the pole(s) of the incision.
7.2 MEDIAL PINNA: TRANSPOSITION FLAPS (RHOMBIC AND BILOBED)
Defects on the medial pinna too large to close in a side-to-side fashion can be repaired with a transposition flap, ultimately recruiting loose tissue from the postauricular sulcus. For defects adjacent to the sulcus, a rhombic transposition flap can be designed to reconstruct the defect, transposing tissue over the uninvolved skin of the medial pinna (Figs. 7.2A, B, C, D, E, F). For defects distant from the postauricular sulcus, a bilobed transposition flap can effectively “walk” tissue from the postauricular sulcus across the medial pinna to the surgical defect (Figs. 7.2G, H, I, J, K). Use of a transposition flap helps to minimize secondary tension vectors that could distort the delicate cartilaginous framework of the pinna.
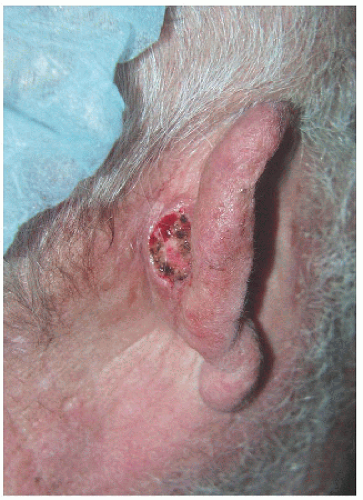 Figure 7.2A. Surgical defect on the right medial (or posterior) pinna adjacent to the helical sulcus. |
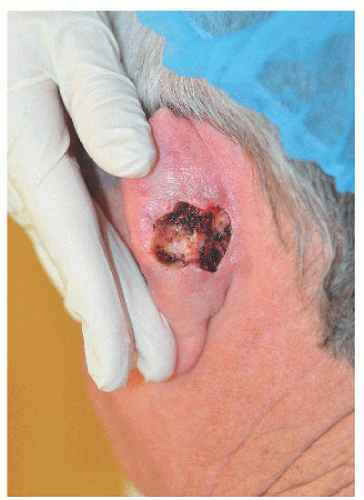 Figure 7.2D. Larger surgical defect on the posterior or medial pinna and adjacent to the postauricular sulcus. |
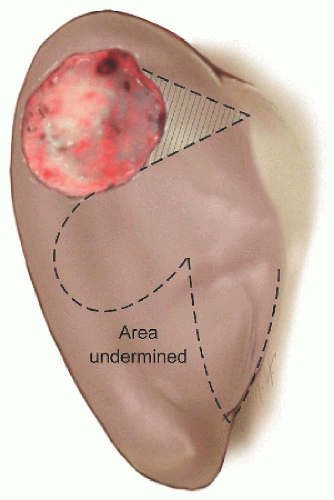 Figure 7.2H. Design of bilobed transposition flap moves tissue from the postauricular sulcus to surgical defect. (For details on bilobed transposition flaps design, see Section 5.4.) The lobes are approximately the same size as the defect, and the angle between the defect and the first lobe or the first and second lobes is approximately 45 to 50 degrees. The second lobe is within the postauricular sulcus, effectively “walking” or transposing tissue across the medial pinna to the surgical defect. |
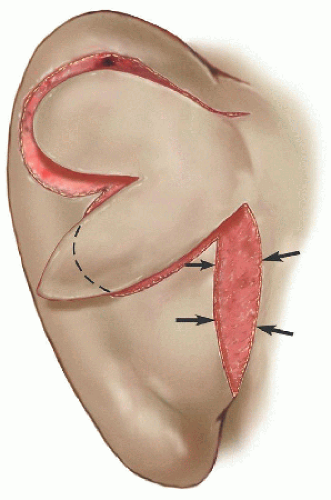 Figure 7.2I. The secondary defect in the postauricular sulcus is closed first, causing the two lobes to transpose into the defect of the first lobe and the surgical defect. |
Key Points
Defects on the medial (or posterior) pinna too large for side-to-side repair can be repaired with a transposition flap.
If a defect is adjacent to the postauricular sulcus, a rhombic transposition flap can be designed. For defects away from the postauricular sulcus, a bilobed transposition flap can effectively walk tissue across the pinna.
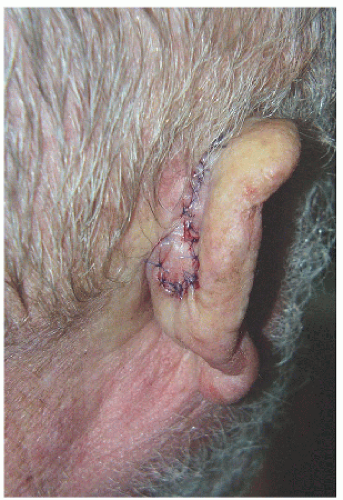
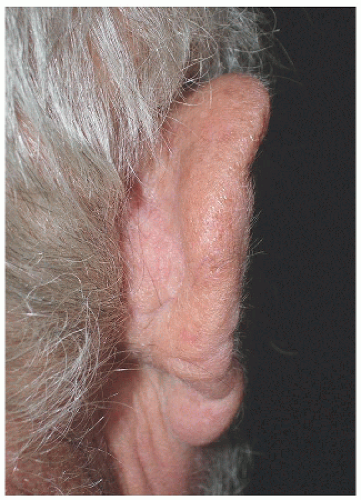
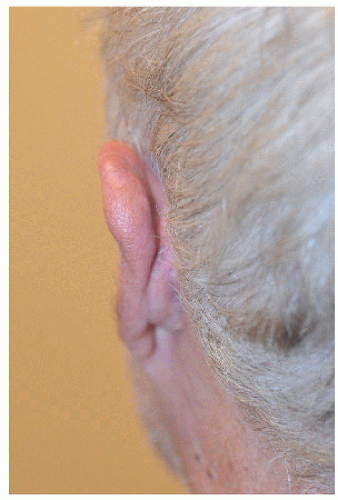
7.3 TRAGUS (AND CONCHAL BOWL): PREAURICULAR TRANSPOSITION FLAP
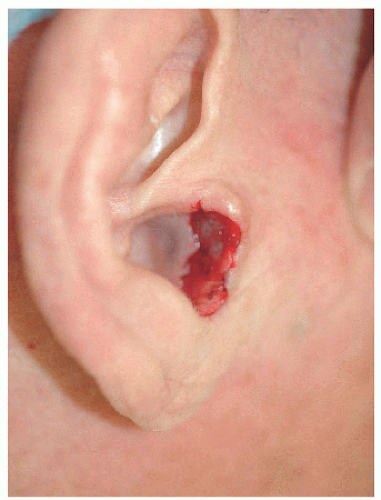
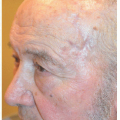
This postoperative defect on the posterior or medial aspect of the tragus could be repaired in a number of ways (Fig. 7.3A). A full-thickness skin graft would reconstruct the defect but would require neovascularization or “take” of the skin graft on exposed cartilage. A flap provides its own blood supply, and therefore the chance of a successful repair on exposed cartilage is greater.
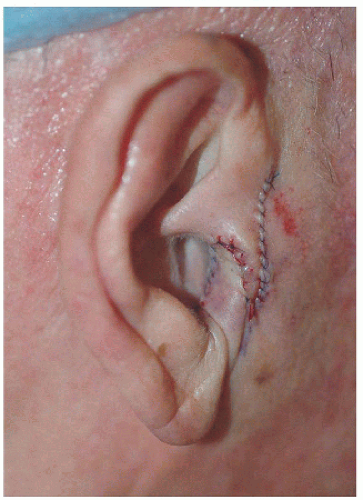
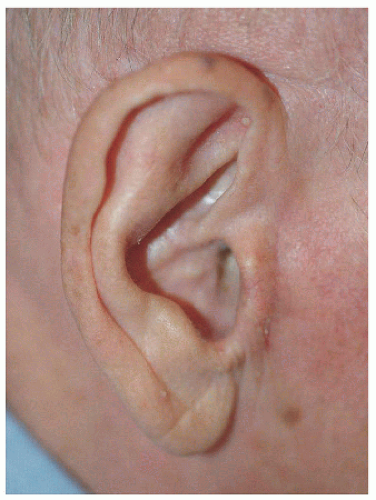
The preauricular cheek holds a reservoir of loose tissue for most people, especially in older patients. This 76-year-old man had enough loose tissue in the preauricular cheek to allow creation of a transposition flap, which would transpose around the tragus and repair the defect on the medial aspect of this structure (Figs. 7.3B and C). As with other transposition flap repair, proper planning is key to ensure that an adequate amount of tissue is transposed and that it reaches all of the edges of the surgical defect. Like other transposition flaps, closure of the secondary defect first allows the flap to more easily transpose into place, essentially “falling” into the surgical defect.
The same type of repair may be useful for a defect on the inferior aspect of the conchal bowl, the main requirement being that the available tissue is adequate to complete the repair and that the defect is adjacent to the donor site (pretragal cheek). In this second example, the defect is located on the inferior aspect of the conchal bowl extending toward the external auditory meatus (Fig. 7.3D). The transposition flap allows for faster healing of the conchal bowl and decreases scar contracture that might constrict the EAM or tragus (Figs. 7.3E and F).
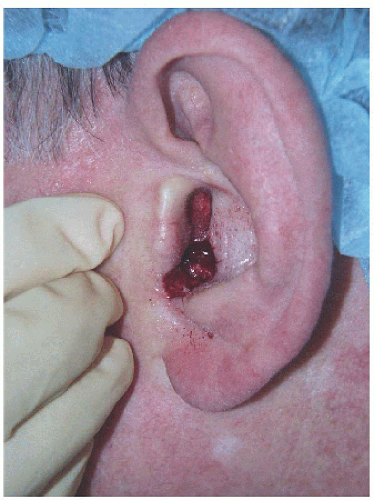 Figure 7.3D. Surgical defect on the inferior aspect of the conchal bowl, extending to the external auditory meatus (blood-soaked cotton ball in the external auditory canal). |
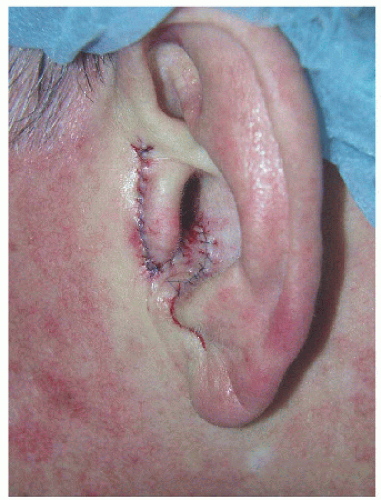 Figure 7.3E. Preauricular transposition flap utilized to reconstruct surgical defect pivoting around the tragus and placed in the intertragal notch. |
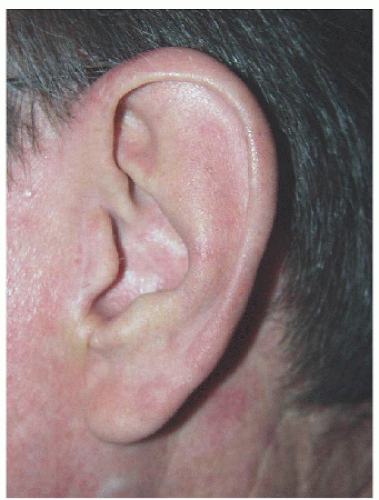 Figure 7.3F. Healed result. Flap repair helps minimize potential scar contracture around the external auditory meatus. |
Key Points
Preauricular skin may be a good donor site for flaps (in addition to its traditional role as donor site for full-thickness skin grafts).
For defects in the conchal bowl, second intention healing should be considered (e.g., Section 7.4) unless the defect involves the EAM or unless scar contracture may cause distortion of the ear. Donor sites for conchal bowl defects include preauricular or postauricular tissue for grafts or flaps (e.g., Sections 7.3 and 7.5).
The preauricular transposition flap should be designed so that the flap transposes around the tragus with the pedicle crossing the intertragal notch. Closure of the secondary defect (i.e., site of the flap) should fall within the pretragal crease.
7.4 CONCHAL BOWL (AND TRIANGULAR FOSSA): SECOND INTENTION HEALING
Second intention healing may be an excellent alternative for defects in concave areas, including the conchal bowl. One caveat is that if the defect approximates the EAM, especially encircling at least 50% of the EAM, graft or flap repair should be considered to decrease the possibility of constriction of the EAM and potential impact on hearing. Likewise, large defects or defects near free margins might be better off with flap or graft repair to decrease the chance that scar contracture might distort the free margin.
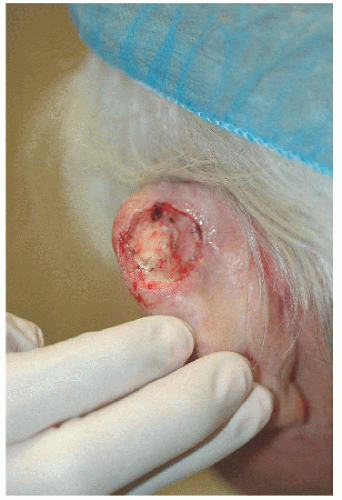
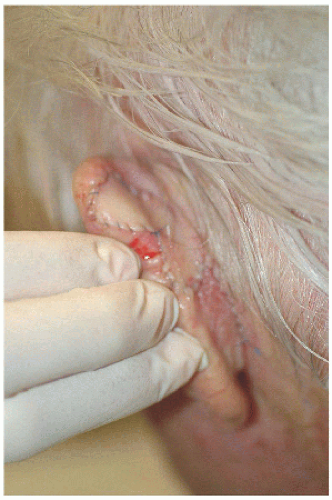
In the first example, the surgical defect involves the entirety of the cavum of the conchal bowl, avoiding the area of the EAM (Figs. 7.4A and B). The second example is a larger defect involving the triangular fossa but far enough away from free margins (e.g., helical rim) to entertain the option of second intention healing (Fig. 7.4C). In this second example, small, 3-mm circular pieces of cartilage were removed in the wound bed to facilitate granulation of the wound through the transcartilaginous migration of granulation tissue from the subcutaneous tissue on the medial pinna (Figs. 7.4D and E). Both examples demonstrate that second intention healing may be an excellent alternative for specific surgical defects on concave surfaces. A third and final example involving the area between the scaphoid and triangular fossae show that even over a convex surface second intention healing may be a good consideration for repair as long as there is no impact upon structure or function (Figs. 7.4F and B).
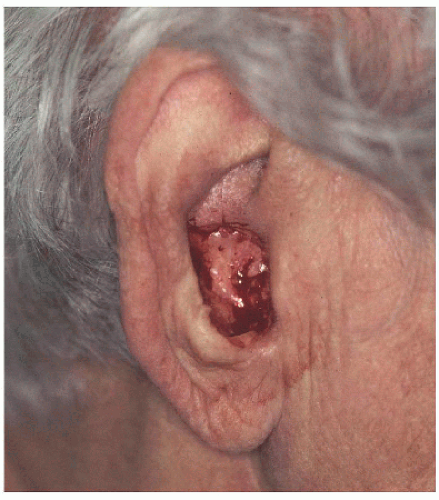 Figure 7.4A. This 88-year-old female was left with a 2.5 × 1.5 cm defect involving the conchal bowl following Mohs surgery of a basal cell carcinoma. |
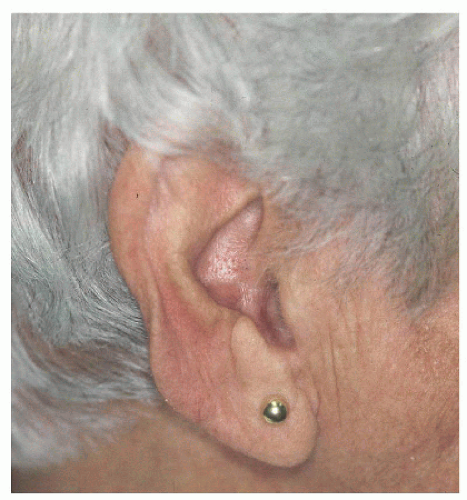 Figure 7.4B. Final healed result by second intention healing.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|