Abstract
Over the last few decades, treatment of cancer has evolved to include therapies that may be broadly categorized as traditional cytotoxic chemotherapies, hormonal therapies, targeted therapies, and immunotherapies. From the dermatologic perspective, cytotoxic chemotherapies have long generated much interest because of the potential to cause alopecia, their hallmark adverse event (AE). The introduction of various targeted therapies and immunotherapies in recent years, however, has led to the recognition of a broad spectrum of dermatologic AEs. In this chapter, we provide an overview of the dermatologic AEs associated with major classes of cancer therapies, as well as the management options for common entities. Besides the physical and emotional impact, impairments in patients’ quality of life, and financial burden, these AEs may also disrupt cancer treatments. Therefore, their timely recognition and prompt management are crucial for optimal clinical outcomes.
Keywords
Antineoplastic agents, adverse events, Drug eruption, drug therapy, Epidermal growth factor–receptor inhibitors, Molecular molecularly targeted therapies
- •
Dermatologic adverse events (AEs) of cancer therapies can cause patient discomfort and impair their quality of life, increase the cost of medical care, and even result in dose modifications, all of which can negatively impact clinical outcomes.
- •
The traditional cytotoxic chemotherapies are commonly associated with dermatologic AEs such as alopecia, hypersensitivity reactions, xerosis, skin hyperpigmentation, nail changes, and hand-foot syndrome (HFS).
- •
Targeted therapies encompass drugs that inhibit selective molecules and pathways critical for carcinogenesis, tumor growth, and survival. The ensuing dermatologic AEs tend to vary depending on the entity targeted (e.g., epidermal growth factor receptor [EGFR], vascular endothelial growth factor/vascular endothelial growth factor receptor, immune checkpoints, mammalian target of rapamycin), although there is overlap.
- •
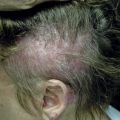
Acneiform rash, xerosis, skin hyperpigmentation, alopecia, hair disorders (e.g. dyspigmentation, hypertrichosis, trichomegaly, textural changes), paronychia, and mucosal inflammation are characteristic dermatologic AEs of the EGFR inhibitors. Treatment with inhibitors of angiogenesis often leads to HFS, exanthems, hair disorders (alopecia, dyspigmentation, and textural changes), xerosis, mucositis, and in the case of bevacizumab/aflibercept, impairment of wound healing. The immune checkpoint inhibitors often result in pruritus and maculopapular cutaneous eruptions, although their AE profile, as with other newly developed drugs, is emerging and pending further characterization.
- •
Timely recognition, prompt management of dermatologic AEs related to cancer treatment, and appropriate counseling are crucial for optimal clinical outcomes, and require a multidisciplinary approach involving oncologists, dermatologists, and nurses.
Introduction
Over the last few decades, treatment of cancer has evolved to include therapies that may be broadly categorized as traditional cytotoxic chemotherapies, hormonal therapies, targeted therapies, and immunotherapies ( Table 18-1 ). From the dermatologic perspective, cytotoxic chemotherapies have long generated much interest because of the potential to cause alopecia, their hallmark adverse event (AE). The introduction of various targeted therapies and immunotherapies in recent years, however, has led to the recognition of a broad spectrum of dermatologic AEs ( Table 18-2 ). Besides the physical and emotional impact, impairments in patients’ quality of life, and financial burden, these AEs may also disrupt cancer treatments. Therefore, their timely recognition and prompt management are crucial for optimal clinical outcomes.
| Cytotoxic Chemotherapeutic Agents | ||||||
| Alkylating agents | Nitrogen mustards | Aziridines and epoxides | Alkyl sulfonates | Nitrosoureas | Hydrazines and triazine derivatives | |
| Mechlorethamine Cyclophosphamide Ifosfamide Melphalan Chlorambucil | ThioTEPA Mitomycin-C | Busulfan | Carmustine Streptozocin | Procarbazine, Dacarbazine Temozolomide | Hydroxyurea | |
| Antimetabolite agents | Folate Antagonists Methotrexate Pemetrexed | Pyrimidine Analogs 5-Fluorouracil Capecitabine Cytarabine Gemcitabine | Purine Analogs Mercaptopurine Thioguanine Fludarabine Cladribine |
| Topoisomerase-interacting agents | Topoisomerase I inhibitors Irinotecan, Topotecan | Topoisomerase II inhibitors Anthracyclines Etoposide, Tenoposide Mitoxantrone |
| Antimicrotubule agents | Taxanes Paclitaxel Docetaxel Nab-paclitaxel | Vinca alkaloids Vinblastine Vincristine Vinorelbine Vindesine Vinflunine | Estramustine phosphate sodium |
| Epigenetic modulators | Histone deacetylase inhibitors Vorinostat Romidepsin | Proteasome inhibitors Bortezomib Carfilzomib | Demethylating agents 5-Azacitidine Decitabine |
| Retinoids | Bexarotene All-trans retinoic acid | ||
| Arsenicals | Arsenic trioxide | ||
| Targeted Anticancer Agents | |||
| EGFR inhibitors | EGFR inhibitors Erlotinib Cetuximab Panitumumab Gefitinib | EGFR/HER2 inhibitors Afatinib Lapatinib | EGFR/VEGFR inhibitors Vandetanib |
| Angiogenesis inhibitors | VEGF inhibitors Bevacizumab Aflibercept | VEGFR inhibitors Sorafenib Sunitinib Pazopanib Axitinib Regorafenib Cabozantinib |
| BRAF inhibitors | Vemurafenib, Dabrafenib | |
| BCR-ABL inhibitors | Imatinib, Nilotinib, Dasatinib, Ponatinib, Bosutinib | |
| mTOR inhibitors | Everolimus, Temsirolimus | |
| MEK inhibitors | Trametinib | |
| SMO inhibitors | Vismodegib | |
| JAK inhibitors | Ruxolitinib | |
| PI3K inhibitors | Idelalisib | |
| BTK inhibitors | Ibrutinib | |
| ALK inhibitors | Crizotinib | |
| Immune checkpoint inhibitors | Ipilimumab, Nivolumab, Pembrolizumab | |
| Other monoclonal antibodies | HER-2 Trastuzumab Ado-trastuzumab emtansine Pertuzumab | CD20 Rituximab Ofatumumab Obinutuzumab | CD30 Brentuximab | CD-52 Alemtuzumab |
| Other Anticancer Agents | ||||||||
| Endocrine agents | SERMs | ERDs | Aromatase inhibitors | LHRH agonists | Androgens | Antiandrogens | Somatostatin analogs | |
| Tamoxifen Toremifene Raloxifene | Fulvestrant | Exemestane Anastrozole Letrozole | Leuprolide | Fluoxy-mesterone | Flutamide Bicalutamide | Megestrol acetate | Octreotide acetate | |
| Miscellaneous | |||
l -Asparaginase | Bleomycin | Thalidomides Thalidomide Lenalidomide Pomalidomide | |
| Primary Molecular Target | EGFR | Multikinase | VEGF | VEGFR/PDGFR | BRAF | mTOR/PI3K | CD20 | HER-2 | CTLA-4 | PD-1 |
|---|---|---|---|---|---|---|---|---|---|---|
| Anticancer agents | Cetuximab, Panitumumab, Erlotinib, Afatinib, Lapatinib | Imatinib (I), Nilotinib (N), Dasatinib (D) | Bevacizumab (B), Aflibercept (A) | Sorafenib (So), Sunitinib (Su), Pazopanib (P), Axitinib (Ax), Regorafenib (R), Cabozantinib (C) | Vemurafenib, Dabrafenib | Everolimus (E), Temsirolimus (T), Idelalisib (I) | Rituximab, Ibritumomab tiuxetan | Trastuzumab (Tr), Trastuzumab emtansine (T-DM1) | Ipilimumab | Nivolumab (Nv), Pembrolizumab (P) |
| Dermatologic adverse event | ||||||||||
| Skin | ||||||||||
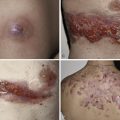
| Cutaneous eruptions | +++ | +++ (B) | ++ | ++ | +++ (E, T) ++ (I) | + (Tr) | +++ | +++ | ||
| Maculopapular | ||||||||||
| Papulopustular (“acneiform”) | +++ ∗ | +++ (E, T) | + (Nv) | |||||||
| Keratosis pilaris-like | + | ++ | ||||||||
| Xerosis | ++ | ++ | ++ | ++ | ++ (E, T) | |||||
| Fissures (fingertips, toes) | ++ | |||||||||
| Skin infections (Bacterial, viral, fungal) | + | + | ||||||||
| Pruritus | ++ | ++ | ++ (B) | ++ | + | +++ (E, T) | +++ | +++ | ||
| Hand-foot skin reaction | + | +++ | + | +++ (T-DM1) | ||||||
| Photosensitivity | +++ | + | + | +++ | + (Nv) | |||||
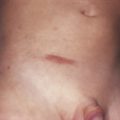
| Pigmentary changes | + | + (A) | ||||||||
| Hyperpigmentation | ||||||||||
| Hypopigmentation | + (I) | + (Su) | ||||||||
| Depigmentation (vitiligo) | ++ (I) | ++ | + | |||||||
| Impaired wound healing | + | +++ | ||||||||
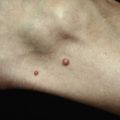
| Skin neoplasms (KA/cuSCC) † | + (So, R) | +++ | ||||||||
| Psoriasis exacerbation or psoriasiform eruptions | ++ (I) | |||||||||
| Lichenoid eruptions | + (I) | |||||||||
| Eruptive nevi | + (So) | |||||||||
| Facial edema | +++ | ++ (Su) | ||||||||
| Appendages | ||||||||||
| Hair | ++ | + | + | ++ (So, Su); + (P) | ++ | ++ (Tr) | + | |||
| Alopecia | ||||||||||
| Hypertrichosis, trichomegaly | +++ | |||||||||
| Curling | ++ | ++ | ++ | |||||||
| Hyperpigmentation | + (P) | |||||||||
| Hypopigmentation | + | + (Su); ++ (P) | + (P) | |||||||
| Nail | ++ | ++ (E, T) | ||||||||
| Paronychia | ||||||||||
| Nail abnormalities | ++ | ++ | ||||||||
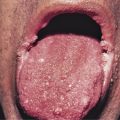
| MUCOSAE | ++ | ++ | + | + | +++ (E, T) | |||||
| Mucositis, stomatitis | ||||||||||
| Bleeding, hemorrhages | + (B) | |||||||||
| Cutaneous and mucosal telangiectasia | + | +++ (T-DM1) | ||||||||
| Geographic tongue | + (B) | |||||||||
| Mucocutaneous reactions ‡ | + | |||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree