Synopsis
- ▪
Normal speech is the primary goal of cleft palate repair; minimizing effects of maxillary growth is also important but ultimately secondary.
- ▪
Cleft palate repair prior to 1 year of age (ideally 9–10 months) results in better speech outcomes than later repairs.
- ▪
The levator veli palatini muscle is longitudinally oriented in the cleft palate patient. Realignment of the muscle to a transverse and posterior position in the soft palate is the key to a successful functional result.
- ▪
Eustachian tube function is abnormal in cleft patients due to abnormal position of the tensor veli palatini muscle; this must be addressed in every cleft palate patient, usually with ventilating tubes.
Keywords
cleft, palate, congenital, speech, feeding, repair
Synopsis
- ▪
Normal speech is the primary goal of cleft palate repair; minimizing effects of maxillary growth is also important but ultimately secondary.
- ▪
Cleft palate repair prior to 1 year of age (ideally 9–10 months) results in better speech outcomes than later repairs.
- ▪
The levator veli palatini muscle is longitudinally oriented in the cleft palate patient. Realignment of the muscle to a transverse and posterior position in the soft palate is the key to a successful functional result.
- ▪
Eustachian tube function is abnormal in cleft patients due to abnormal position of the tensor veli palatini muscle; this must be addressed in every cleft palate patient, usually with ventilating tubes.
Brief introduction
- ▪
The failure of fusion of the frontonasal and maxillary processes gives rise to the cleft of the primary palate, which includes the lip, alveolar process, and the hard palate anterior to the incisive foramen.
- ▪
This results in a cleft in the typical location between the premaxilla and the lateral maxilla, on either one or both sides.
- ▪
The lateral palatal shelves fuse later than the primary palate, around 7–8 weeks’ gestation, as they rotate from vertical to horizontal orientation.
- ▪
This fusion proceeds from anterior to posterior, which helps to understand the spectrum of clefts of the secondary palate.
- ▪
The levator palatini and other pharyngeal muscles are derived from the fourth branchial arch and are innervated by cranial nerve X (vagus). The sole exception to this is the tensor palatini muscle, which arises from the first branchial arch and is innervated by cranial nerve V (trigeminal).
- ▪
The incidence of otitis media effusion has been found to be 96–100% in cleft palate patients.
- ▪
It is estimated that there is a 20–30% incidence of pure-tone hearing loss in cleft palate patients by audiography.
- ▪
It has long been suggested that closure of the palate reduces risk of permanent hearing loss.
Preoperative considerations
Submucous cleft palate
- ▪
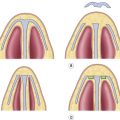
Submucous cleft palate occurs when the palate has mucosal continuity but the underlying levator palatini muscle is discontinuous across the midline and longitudinally oriented, similar to the muscle anatomy in overt clefts of the palate ( Fig. 12.1 ) .
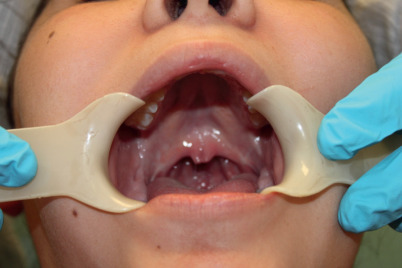
Figure 12.1
Submucous cleft palate – note bifid uvula and thinning of central palate. On palpation, there is a notch in the posterior hard palate rather than a posterior nasal spine.
- ▪
Calnan’s classic triad is diagnostic of this condition:
- •
A midline clear zone (zona pellucida).
- •
A bifid uvula.
- •
A palpable notch in the posterior hard palate.
- •
With contraction of velar musculature, a distinct midline muscle diastasis may be seen (see Fig. 12.1 ).
- •
- ▪
The significance of a submucous cleft may be difficult to assess clinically; the child with submucous cleft palate is often undiagnosed in infancy.
- ▪
It has been reported that 45–55% of patients with isolated submucous cleft palate are symptomatic with regard to speech, serous otitis media, or hearing loss.
- ▪
However, an infant identified with submucous cleft palate need not routinely undergo repair because a significant number of individuals with submucous cleft palate will not develop velopharyngeal insufficiency.
- ▪
Rather, these patients should be closely monitored with serial speech evaluations and audiometric surveillance.
- ▪
Patients who present with velopharyngeal insufficiency and submucous cleft palate on examination require full evaluation, including speech evaluation and endoscopy.
- ▪
Even in the absence of obvious findings on clinical examination, anatomic abnormalities are found in most patients (>90%) at the time of surgery.
- ▪
Corrective surgical technique for submucous cleft palate is focused on anatomic correction of the velar muscle diastasis.
- ▪
Although pharyngeal flaps and sphincter pharyngoplasty have been proposed as primary means of treatment, most surgeons focus on repair of the abnormal levator muscle position.
- ▪
The Furlow double-opposing Z-plasty (see below) is an ideal procedure for these patients because there is no width discrepancy to overcome.
Pierre Robin sequence
- ▪
Pierre Robin described the triad of micrognathia, glossoptosis, and respiratory distress.
- ▪
60–90% of patients diagnosed with Pierre Robin sequence have cleft palate.
- ▪
Infants with Pierre Robin sequence also have increased incidence of associated anomalies, particularly cardiac and renal problems.
- ▪
Newborns with Pierre Robin sequence may have severe respiratory and feeding difficulty because of the posterior displacement of the tongue.
- ▪
Initial treatment consists of placing the child prone and use of gastric lavage feeding tubes to push the tongue forward.
- ▪
Nasal airways have been used for the same purpose with reported success rates of 80–90%.
- ▪
If these conservative measures fail, surgical management of the airway may be required.
- ▪
A tongue–lip adhesion has been used as an alternative to tracheostomy and is generally effective.
- ▪
More recently, mandibular distraction osteogenesis has been used in neonates with success in averting tracheostomy.
- ▪
Palatoplasty in children with Pierre Robin sequence must be carefully timed with growth of the child, particularly the mandible, as closure of the palate narrows the effective area for respiration and can lead to respiratory distress.
- •
If the mandible attains reasonable size in the first year of life, palate repair can still be performed safely before 1 year of age.
- •
In the rare patient who has previously undergone tracheostomy, the palate should be repaired before decannulation.
- •
The risk of airway compromise after palatoplasty reaches 25%, with an emergent tracheostomy or reintubation rate of 11% at one institution.
- •
Syndromes
- ▪
Cleft palate without associated cleft lip has been reported to be associated with a syndrome in as many as 50% of cases, while cleft lip and palate together have an incidence of syndromes of about 30%.
- ▪
Van der Woude syndrome is associated with a mutation in the interferon regulatory factor 6 ( IRF6 ) gene; this is an autosomal dominant syndrome associated with lower lip sinus tracts (“lip pits”), and has variable penetrance including the full range of cleft lips as well as palates.
- ▪
Velocardiofacial syndrome is associated with a 22q chromosomal deletion, detected by fluorescent immunohybridization. These children have a characteristic “birdlike” facial appearance, soft palate dysfunction, developmental delay, and various cardiac conditions.
- ▪
The same deletion gives rise to DiGeorge syndrome with associated B-cell and immune dysfunction.
- ▪
Infants with profound developmental delay and severely shortened life span projection should have surgical intervention delayed or should undergo palatoplasty under special circumstances only.
- ▪
Palate repair in severely disabled children can lead to altered airway status and obstructed upper airway in those with neuromuscular delay.
Growth
- ▪
At birth, the average weight is the same for cleft and unaffected newborns. However, cleft infants have been shown to exhibit poor weight gain in early infancy.
- ▪
After repair of the palate, average growth returns to normal compared with unaffected children by the age of 4 years.
- ▪
Children with orofacial clefting stabilize and continue normal growth to at least 6 years of age, with no statistically significant differences in height and weight when compared to unaffected children.
- ▪
In later childhood, however, the average weight and height of children with cleft palate appears to diminish compared with those of control subjects.
Feeding and swallowing
- ▪
Oral intake is divided into two separate activities: generation of suction force (negative intraoral pressure) and swallowing.
- ▪
For negative intraoral pressure to be produced, the velum seals off the pharynx posteriorly; the lips close anteriorly, and negative pressure is produced by moving the tongue away from the palate and by opening the mandible, which effectively increases the intraoral volume within a closed system.
- ▪
If the individual is unable to close the nasopharynx or to generate a seal of the lips, or if the palate is not intact at the point of contact with the tongue, negative pressure cannot be produced.
- ▪
In the cleft palate infant, the communication between the oropharynx and nasopharynx prevents a seal of the tongue against the palate, and negative pressure cannot be generated. Suckling is therefore not productive, and breast-feeding is ineffective.
- ▪
Swallowing involves a coordinated, complex interaction of the tongue and pharynx and is dependent on neuromuscular control and rhythmic coordinated contraction of the tongue and pharynx.
- ▪
Children with clefts generally do not have difficulty with swallowing and aspiration unless intrinsic neuromuscular abnormality of the tongue or pharynx is present.
- ▪
When the palate has an open cleft, food may reflux into the nasal passage.
- ▪
Nasal reflux is irritating to the nasal mucosa and can predispose to sinusitis and ulceration.
- ▪
In the older child, a persistent communication through a fistula in the palate may result in regurgitation of food through the nose, which is socially unacceptable.
- ▪
Most infants with clefts are unable to breast-feed. Infants who are unable to breast-feed because of cleft palate have a number of options for feeding, including nipples; crosscutting of standard nipples; long, soft nipples that place the liquid at the posterior tongue; and special flow bottles.
- ▪
All of these techniques are effective, and selection is generally by personal preference and the baby’s acceptance of the method.
- ▪
Other key considerations are elevated head positioning during feeding and careful observation of feeding time and volume ingested.
- ▪
Weight gain and skeletal growth confirm success of the feeding regimen. Once the palate is successfully closed surgically, special feeding methods are generally unnecessary.
Speech
- ▪
The primary goal of palatoplasty is normal speech.
- ▪
Patients can grow and even thrive, despite feeding difficulties, but speech cannot be normalized if the palate is not repaired.
- ▪
The ability to partition the oropharynx and nasopharynx is crucial for normal speech production.
- ▪
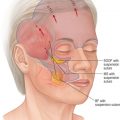
The palate elevates during production of any sounds requiring positive pressure in the oropharynx; the levator palatini is primarily responsible for this movement ( Fig. 12.2 ) .
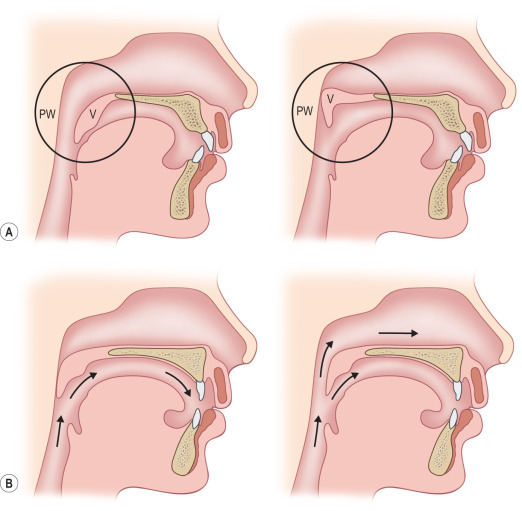
Figure 12.2
(A) In the lateral cephalogram, the soft palate or velum (V) is shown at rest (above left) and during speech, making contact with the posterior pharyngeal wall (PW) (above right). (B) This line drawing shows the air flow during normal speech (left), with the velum making contact with the posterior pharynx to direct air out the mouth. If the velum is too short or movement is inadequate (right), air can escape through the nose during speech, creating velopharyngeal insufficiency (VPI).
- ▪
Speech is a complex issue, and many factors may influence speech development in a child with a cleft palate.
- ▪
In addition to the importance of the palate itself, speech development may be influenced by motor or neurologic developmental delay (often seen in syndromes), by hearing, and by environmental stimuli.
- ▪
If palate function is not corrected, velopharyngeal insufficiency results, with speech that is hypernasal, often with hoarse quality due to difficulty in directing airflow through the mouth.
- ▪
When complete closure cannot be anatomically or functionally obtained, compensatory mechanisms for sound production are learned.
- ▪
These are maladaptive patterns that interfere with global intelligibility and include glottal stops and pharyngeal fricatives.
- ▪
Two crucial aspects of palatoplasty are important in optimal speech outcome: (1) surgical technique and (2) timing of palate repair.
- ▪
Most would agree that the best speech results are correlated with closure of the palate near the time of the infant beginning language acquisition, which, for the normal-developing child, is before 12 months of age.
- ▪
Despite the absence of hard evidence supporting earlier palate repairs, a growing body of opinion seems to support palate repair around 9–10 months of age for children with apparently normal development.
- ▪
Very early repair of the palate (6 months or younger) has been proposed by some surgeons, primarily as a means of improving feeding; however, long-term results are lacking for any large cohort of these patients.
Maxillary growth
- ▪
Palatoplasty has been shown to detrimentally affect maxillary growth.
- ▪
Many children with repaired cleft palate display typical findings of transverse maxillary deficiency requiring orthodontic widening of the maxilla once permanent teeth have erupted.
- ▪
There may be a sagittal growth deficiency as well, whereas 35–40% of children will develop an anterior crossbite; as many as 15–20% of children with cleft palate go on to require a Le Fort I maxillary advancement in some series.
- ▪
Although it might seem preferable to wait until a more advanced age for palate repair, given the growth effects on the maxilla, it is far more difficult to establish normal speech in older children after cleft repair than to correct occlusion with a combination of orthodontic treatment and orthognathic surgery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree