Summary
As with all cosmetic procedures, autologous fat grafting (AFG) or transfer (AFT) carries risks along with benefits. The benefits are well established and well documented; these include correcting facial imperfections and asymmetries, adding volume with the patient’s own nonallergenic, autologous material and the benefits of adipose-derived stem cells. The ideal implant material is nonantigenic, durable, nontoxic, and resistant to infection. In terms of an injectable filler, fat comes closest to this ideal. It is important to consider the risks, benefits, potential complications, alternative treatments, and imponderables of fat transfer before electing to perform this procedure. As the benefits have been mentioned above and widely elucidated elsewhere, this chapter will focus on the description of possible risks and complications of facial fat transfer.
6 Fat Transfers
6.1 Introduction
Fat injection as a cosmetic treatment is natural, versatile, long-lasting, and safe. Fat transfer has become a preferred method of facial volume enhancement for patients who are allergic to traditional dermal fillers such as bovine-derived collagen, or simply prefer autologous material. One of the biggest benefits of volume enhancement via fat injection is that there is absolutely no chance of an adverse allergic reaction to the injected material. Another popular reason for choosing fat transfer/injection is the duration of benefit. Even though up to 50% of injected fat will be reabsorbed into the body within a few months of the fat injection, the remaining 50% will usually last for years. For longer lasting results, many patients choose to have several fat transfer sessions over the course of a few months. This approach helps with precision and seems to improve durability.
6.2 Risks and Complications
Fat transfer risks are few, rare, and usually minimal. Soreness and edema are the norms, but not experienced by every patient. The most significant downside of the procedure is that your face may simply reabsorb all or most of the injected fat. This may be technique dependent, but can happen in the most experienced of hands. The chances of this happening can’t be predicted for a given individual, but it happens in up to 35% of all patients who receive fat injection treatments to the face.
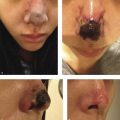
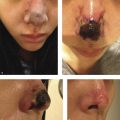
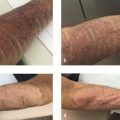
Complications of fat transfer are uncommon but can include reaction to anesthesia, permanent or temporary discoloration caused by blood vessel injury at the treatment site, Tyndall effect, calcification, a distorted look if overcorrection is permanent, perioperative bleeding, a blood clot at the treatment or donor site, a blood-borne infection, venous thromboembolism, scar tissue, fat necrosis, and a fat embolism due to fat injection into a blood vessel which can cause necrosis, blindness, central nervous system (CNS) complications or even death.
Being disappointed with the results of a fat transfer procedure is also considered a risk; therefore, setting realistic expectations is a must. Discussion of all patient concerns and desires before undergoing a fat transfer procedure is mandatory and should be documented. The placement of the fat transfer may not be adequate, the patient may reject the transfer, or the duration of the effect may not be to the patient’s expectation. It is possible, the patient’s face or lips may not look exactly as they had envisioned. Lips can be challenging both due to potential trauma during the procedure and the constant motion of the orbicularis oris.
6.3 Vascular Occlusion
Visual loss is one of the most severe complications reported in patients undergoing facial filler procedures. In the majority of reported cases, visual deterioration was severe and irreversible 1 , 2 , 3 (Fig. 6.1). Visual loss can be caused by ophthalmic artery occlusion (OAO), central retinal artery occlusion (CRAO), either localized or generalized posterior ciliary artery occlusion (PCAO) with sparing of the central retinal artery, branch retinal artery occlusion (BRAO) or posterior ischemic optic neuropathy (PION). Autologous fat injections are associated with the most severe visual impairment (CRAO or OAO). 1 , 2 , 4 , 5 Due to differences in particle size among the different types of facial fillers, patients with ocular symptoms following other types of fillers, such as hyaluronic acid injections, are more likely to suffer from localized ocular occlusion with milder clinical manifestations and better visual prognosis than patients who received autologous fat injections. 2 , 4 , 5 , 6 Preventative strategies proposed in the literature to reduce the risk of vascular complications following fat injections include: using small-diameter nontraumatic blunt-tip cannulas instead of sharp cannulas and needles, limiting syringe size to 1 mL, aspirating before injection, injecting the filler slowly with minimal pressure, limiting the amount of filler to less than 0.1 mL with each pass, mixing the filler with vasoconstrictor, using a topical vasoconstrictor before injection, and moving the needle tip while injecting. 2 , 3 , 7 , 8 Avoiding injection at the sites of previous trauma, or at sites with chronic inflammation or scarring, is also advised. Facial filler injections should be performed with extra caution in patients who have previously undergone facial or plastic surgeries. 2 , 3 , 9
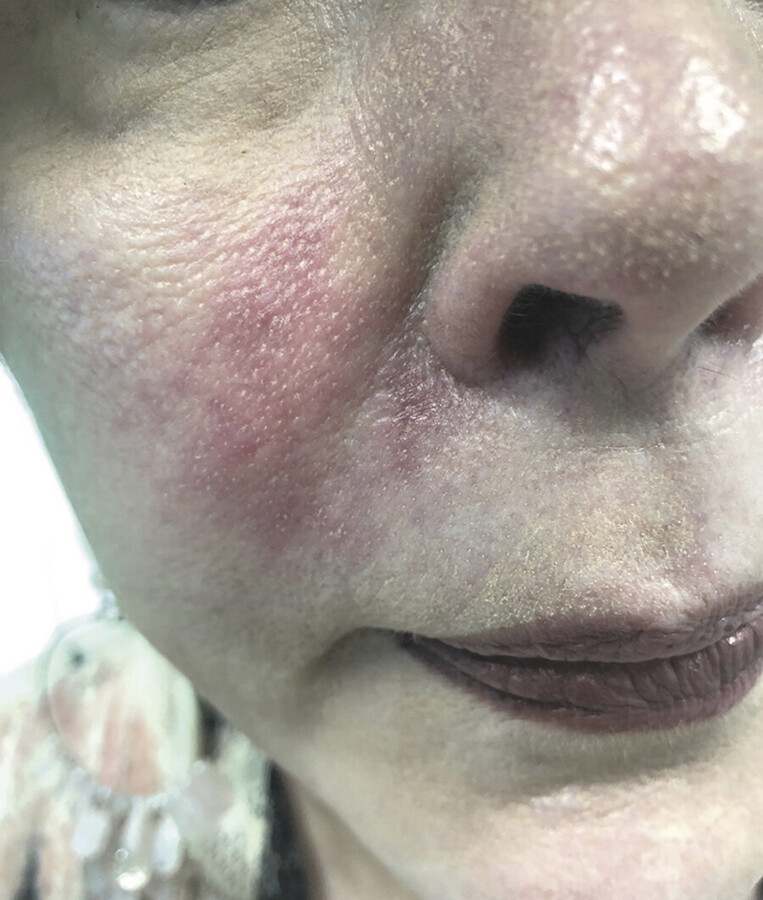
According to the literature, no fully effective treatment for CRAO or OAO is available. Nevertheless, there are several papers presenting various treatment modalities that recover some degree of visual function for the patient. The aim of the treatment after facial fat or filler embolism is to restore the perfusion of the retina as soon as possible. The retina is very sensitive to hypoxia, and after 90 minutes damage due to retinal ischemia becomes permanent. 2 , 3 , 10 , 11 , 12 Most previously reported cases of vision loss secondary to fat embolism have remained without any improvement. Therefore, no evidence-based treatment strategies are available. Slightly better outcomes have been reported in cases of vision loss following injection of fillers other than autologous fat. 2 , 3 From 23 cases of ocular complications following injections of hyaluronic acid into the facial area, permanent vision loss was observed in 9 cases, and in 6 cases some level of improvement in visual acuity was reported. 2 Early, immediate identification of arterial occlusion is key with treatment aimed at lowering intraocular pressure to boost retinal perfusion, dilating arteries to eliminate embolus or dislodging embolus to a more peripheral location, and reducing the inflammatory component of the injury. To decrease intraocular pressure, Diamox (PO, IV, or OS) or mannitol (IV) is immediately given if there are no contraindications, eye drops are administered (e.g., alpha agonists, B-blockers, carbonic anhydrase inhibitors), and ocular massage and anterior chamber paracentesis are performed. Carbon dioxide and oxygen inhalation or vasodilatory agents such as prostaglandin E1 provoke arterial dilation. Hyperbaric oxygen therapy and systemic corticosteroids decrease the inflammatory response. 1 , 2 , 9 , 10 , 11 , 12 Table 6.1 presents the currently recommended treatments and their underlying mechanism of action.
The crucial factor in the management strategy is the immediate assessment and referral to the ophthalmologist or retina specialist, to start the proper treatment within the 90-minute time span after which the damage to the retina is irreversible. 6 , 12 Loh et al 12 have proposed a treatment algorithm for managing vision loss following filler injections. When a patient presents the first symptoms of vascular compromise in the retina, filler injection should be instantly stopped and the patient should be laid in a supine position. Immediate treatment (including topical instillation of timolol, and/or oral acetazolamide and ocular massage) should be administered by a nonophthalmologist injector before arranging and transferring the patient to a specialist for a definitive therapy, which consists of further medical administration and—when indicated—anterior chamber paracentesis. In cases of hyaluronic acid–induced vision loss, hyaluronidase may be used to dissolve hyaluronic acid emboli. 1 , 2 , 3 , 4 , 5 , 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 Unfortunately, there is no effect on fat. After these acute measures have been taken, additional supportive therapy (corticosteroids, hyperbaric oxygen therapy, anticoagulants) should be introduced to protect retinal cells.
In previously reported cases, the treatment provided has usually been incomplete, since the majority of patients suffering from ocular complications received at most a 3-step therapy. 3 , 9 As noted by Chen et al, 3 combination therapy may contribute to the recovery of visual symptoms. In addition to commonly used treatment options such as ocular massage, acetazolamide, mannitol, and corticosteroids, alprostadil and vinpocetine have been successfully used in the treatment regimen. Alprostadil is a synthetic variant of prostaglandin E1. It causes vasodilatation through directly affecting the vascular smooth muscles and increases blood flow in the retina. Alprostadil also decreases thrombocyte activation, improves cell metabolism by increasing oxygen supply to the tissues, and decreases neutrophil activation and the release of their toxic metabolites, helping to reduce tissue damage caused by inflammation and possibly by hypoxia. The use of prostaglandin E1 as a part of comprehensive therapy for visual loss following hyaluronic acid injection was also reported by Chen et al. 3 This resulted in improvement in visual acuity, extraocular movement, and visual field defects. Vinpocetine, chemically known as ethyl apovincamine, is a vinca alkaloid that increases cerebral blood flow and has neuroprotective effects. 15 This drug is used in the treatment of ischemic cerebrovascular diseases and vascular dementia. Vinpocetine may help to increase retinal perfusion (Table 6.1).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree