Summary
The use of cosmetic injectable fillers has become ever more prevalent. The physician’s ability to safely produce consistent results relies on prevention and appropriate management of complications, should they arise. Knowledge of patient-, product-, and technique-related factors are all integral to being a complete injector. When the unfortunate complication does arise, having a systematic method for diagnosis of management of early and late complications can help facilitate recovery. Complications can occur for injectors of all skill level and experience and treatment protocols are constantly evolving. Constant review, understanding, and implementation of current treatment are paramount to high-quality patient care with fillers.
5 Fillers
5.1 Introduction
Cosmetic, nonsurgical procedures such as injectable dermal fillers have the ability to produce consistent, effective, and safe results. In 2018, 810,240 dermal filler procedures were performed in the United States alone. 1 With increasing popularity and patient-driven demand, the clinician should be well versed in the prevention, identification, and management of complications. A recent global consensus statement from ASAPS declared that “optimal complication management is the largest unmet need with fillers.” The safety profile of the numerous dermal filler agents has been widely established. However, there is constant evolution of injection paradigms and protocols. This includes the volume utilized, injection technique, development of new product classes, and indefinite number of repeated injections. These all mandate that the treating clinician remain vigilant to complications that may arise and practice in a manner capable of preventing and managing complications in a timely manner. 2 The focus of this chapter is to identify techniques for minimizing and managing complications secondary to dermal fillers and incorporating them into an injectable practice.
5.2 General Principles
A key tenet of complication management is prevention. The treating clinician for this diverse population must be aware of patient-, product-, and technique-related factors that can impact adverse reactions and the treatment of such complications.
5.2.1 Patient-Related Factors
Patient selection is key. Identifying both appropriate and inappropriate candidates for filler is the first step for avoidance and management of complications. Patient demographics, history and physical examination, and patient goals and desires are all considered. In addition to aesthetic patients, these may also include patients with prior facial trauma who desire improvement of facial disharmony. This population should heed caution as injecting over solid surgical implants could theoretically be a nidus of infection. Also of mention, while there has been no association established between use of fillers and autoimmune conditions, patients with uncontrolled immune deficiencies or patients with prior organ transplant may be affected by the filler injection or the condition may affect the behavior of the filler. 3 Immune suppression or depression is not an absolute contraindication but the presence of these medications or steroid use should yield a more discerning look into one’s medical history.
A discussion of patient-related factors includes not only a relevant cosmetic surgical history and disclosure of previous complications but also assessing skin-related conditions and factors (quality, atrophy, etc.). 4 A history of prior surgeries and any complications with wound healing is helpful to note, but also a history of prior dental, facial surgery, or trauma as noted above requires caution.
This is important as there is potential for unusual vascular distribution due to neovascularization and the local soft tissue changes that can occur after trauma and healing. 5 Eliciting prior filler complications, significant allergies, and medical conditions is also imperative. Medical conditions that may heed caution include careful attention to any ongoing skin infections: particularly HSV, perioral HPV, Mollusca contagiosum, impetigo, or excessive amounts of Propionibacterium acnes. All of the aforementioned can make the patient an unsuitable candidate. In addition, active inflammatory dermatitis including atopic, allergic contact, or seborrheic dermatitis mandates caution but is at the discretion of the clinician. Patients should be counseled regarding the possibility of a rosacea flare secondary to the normal inflammatory effect on local tissue and mechanics of injection associated with filler placement. Active HSV infection should preclude treatment and prophylaxis should be prescribed (acyclovir, famciclovir, valacyclovir) for future injections in the perioral area specifically. There is also caution in performing dermal filler injections in patients with active sinusitis, periodontal disease, or other similar infections as these infections may subsequently involve areas where filler has been placed and induce a biofilm reaction. 6 A recent consensus guideline published in the Journal of Clinical, Cosmetic and Investigative Dermatology outlines a comprehensive list of preexisting conditions that may contraindicate or warrant caution in the use of dermal fillers and is worthy of review.
Medication review should focus on the use of immune modulators, anticoagulants, nonsteroidal anti-inflammatory medications, as well as vitamins and herbal supplements. As therapeutic anticoagulation is only a relative contraindication, injection technique can be modified by the clinician to still provide a safe, effective injection in this patient population. Technical modifications include the use of small needles, microcannulas, and immediate prolonged pressure. 2 Vitamin and herbal medications associated with anticoagulation include but are not limited to vitamin E, fish oil, krill oil, gingko, garlic, ginger, ginseng, and should ideally be discontinued 7 to 10 days prior to the procedure. 3 Also, worth mention is screening occupation/occupation factors that may increase the potential for facial skin microbial colonization, i.e., methicillin-resistant Staphylococcus aureus (MRSA) prevalence in health care workers and community-acquired staphylococcal microbes. While these numerous factors elicited above are not contraindications, front-end awareness allows for a timely diagnosis and treatment should a complication arise.
In a multifaceted approach to facial rejuvenation, adjunctive noninvasive or minimally invasive procedures are often combined with dermal filler. It is generally recommended that microdermabrasion, chemical peels, laser treatments, and intense pulsed light (IPL) should be carried out 1 to 2 weeks pre- or posttreatment in the same area to allow time for any erythema and edema to subside and the skin barrier to reestablish. 4 From a more routine and practical standpoint as well, dental procedures should be performed at least 2 weeks pre- or posttreatment to minimize risk of hematogenous spread or biofilm formation, and makeup application is often avoided for the first 24 hours after injection. 6
5.2.2 Product-Related Factors
Pertinent product-related factors include: concentration and flow properties of the filler, as well as the manufacturing and purification process. For hyaluronic acid (HA) fillers, having a working knowledge of these properties will aid in avoiding undesirable outcomes; an example being a higher resistance to deformation is desirable in volumizing in the supraperiosteal or subcutaneous planes, but not superficially where it is more visible. 3
Factors such as G′ have been studied with HA fillers. G′ refers to the elasticity of the product or its ability to retain shape when pressure is applied. Lower G′ fillers spread more easily and are therefore better suited for more superficial placement. Of the non-HA dermal fillers, calcium hydroxylapatite (Radiesse, Merz Aesthetics) is a particulate suspension in methylcellulose gel and poly-L-lactic acid (Sculptra Aesthetic, Galderma) is a synthetic peptide polymer, both of which may stimulate collagen neogenesis and are considered by some to be semipermanent fillers. 7 A comprehensive review of the individual properties of the known dermal fillers is beyond the scope of this chapter, but an in-depth understanding of filler properties and filler selection will undoubtedly aid in avoiding and minimizing complications.
5.2.3 Technique-Related Factors
Multiple accepted injection techniques exist in the literature and are mostly dependent on the area being treated and the filler being utilized. However, there are several agreed upon principles that should be adhered to from a technique standpoint. While HA filler and non-HA fillers can be injected safely and effectively by microcannula or needle, the use of needles must be used with caution in areas prone to vascular complications. These distinct regions, such as the temple and glabella, may be better suited for blunt-tip microcannulas. 2 In addition, aspiration via needle or cannula is often heralded as imperative in these high-risk vascular areas but a negative aspiration should not be relied on in terms of avoiding a vascular complication. General technique principles include a slow injection with a low flow rate in small quantities at various points. Fast injections, aggressive fanning, and high-volume deposition should be avoided, to decrease the possibility of vasculature compression.
5.2.4 Pretreatment Prophylaxis
There is yet to be specific universal consensus guidelines outlining skin preparation prior to injectable soft tissue fillers. Given more recent awareness of risk of infection and possibility of a biofilm, the data for infection prevention for facial procedures is extrapolated from other surgical experiences of patients undergoing clean-contaminated surgery, which is similar to the wound classification during facial procedures. Selection of skin preparation should consider the risks of an adverse reaction vs. a possible small further decrease in the risk of infection. Skin cleansing with chlorhexidine-alcohol is more protective than povidone-iodine against superficial and deep incisional infections. 8 However, there is the greater possibility of significant allergic reaction and the risk of corneal ulceration. Some injectors favor the use of 2% chlorhexidine gluconate in 70% isopropyl alcohol on the facial skin with the exception of the periocular region, given the risk of keratitis and ocular injury. 2 , 9 Others believe that considering the increased associated risks with chlorhexidine skin prep it should not be used and only betadine should be used. Most physicians consider fillers similar to an injectable implant and therefore favor skin cleansing and prep prior to injection. There are currently prep solutions under review that may prove to be better than the current commonly used prep solutions.
5.3 Adverse Reactions and Complications
Complications have been categorized throughout the dermal filler literature in several different structural formats. A recent review in Facial Plastic Surgery grouped complications by type into three categories including injector-dependent adverse events (AEs), lumps and bumps, and ischemia and necrosis. 3 Secondarily, one of the most commonly used classification paradigms is based on timing of the complication for both identification and management, such as early and late, which is the format that is commonly used and one that our discussion will follow. Early complications, often within the first 14 days to 4 weeks, are often attributed to acute inflammation, acute infection, or ischemia. Late (14 d to 1 y) and delayed (up to 1 y) are generally secondary to granuloma formation and possibility of biofilm development. 3
5.4 Early Adverse Reactions and Management of Complications
5.4.1 Bruising
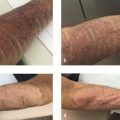
Bruising may be seen as a “normal” complication if performing filler treatments on the face, but to the patient this can be quite concerning on a social level. Bruising, bleeding, and edema may be prevented or decreased by using smaller gauge instruments and/or cannulas (discussed below), slow injection, smaller aliquot deposition, exercise avoidance, and manual or cold compression. Arnica by mouth (started 2 d prior to injectable procedure), arnica cream or gel, and topical vitamin K are adjuncts as well to managing these early limited AEs. For those who can tolerate it, eating pineapple for the anti-inflammatory benefits of the bromelain may help to reduce related inflammation. Some favor using an AccuVein device to identify veins not readily visible on the skin surface; however, most physicians do not routinely utilize this device. 10 Some physicians favor the option of trying to treat a bruise with a vascular laser to boost recovery. Fitzgerald et al reported that resolution can be rapid if the bruise is at a level reached by the laser. Issue is that frequently the bruise is deep in the dermis or even the soft tissue in which case lasering the skin surface is unlikely to affect the bruise.
5.4.2 Edema
Edema and swelling are expected components of all injectable fillers but may vary in severity and time of onset depending on the particular filler used and the location treated. Swelling is most common to the lips and periorbital regions. Onset is typically within the first few hours after injection and may subside within a few days with the use of compresses, antihistamines, or corticosteroids. Periorbital edema can be idiosyncratic, due to too much volume used in the tear trough or incorrect placement. However, patients with preexisting malar edema and malar festoons should be counseled regarding an increased risk for persistent edema following injection. This is due to obstruction at the level of the microlymphatic channels responsible for draining this area.
Avoiding use of certain more hydrophilic products in this area should be considered, such as some clinicians’ preference to avoid Juvederm Ultra and Ultraplusperiorbitals. 3
5.4.3 Placement Related: Inappropriate Placement and Depth, Overcorrection
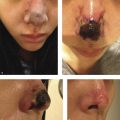
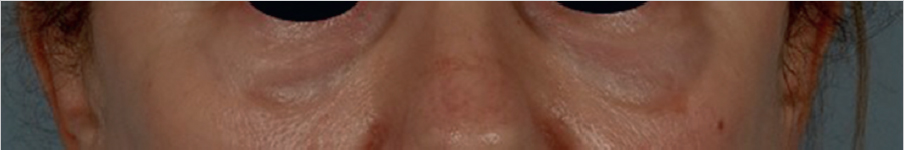
Contour deformities, lumps, and asymmetries may be seen immediately or early following injection. The Tyndall effect (Fig. 5.1), a bluish discoloration due to filler acting as an optical chamber, is a known occurrence with augmentation and too superficial placement within the thin skin of the infraorbital region and the nasolabial folds. This can be treated with hyaluronidase (HYAL) and gentle massage as superficial filler may last for long periods of time in these tissues. Incorrect placement in the retroseptal region or poor filler selection may yield a characteristic “sausage” appearance as it settles along the orbital rim adjacent to the orbital-retaining ligament. 3 Filler migration may occur due to injection technique (excessive volume or pressure) or placement in a plane that is subject to muscular contracture that mechanically pushes the filler to an area other than the initial injection site, which is commonly seen in perioral and periorbital areas due to the shape and function of the orbicularis in each location.
Lumps and contour irregularities as described above following treatment with an HA filler can be altered or adjusted with the use of HYAL. Its use is FDA-approved for drug dispersion, i.e., local anesthetics into soft tissues as it is a mucolytic enzyme that hydrolyzes natural and cross-linked HA fillers. The majority of its use in the aesthetic practice is an off-label use for dissolution of HA filler product that has been incorrectly placed or has generated a suboptimal cosmetic result as well as following the occurrence of any number of AEs. In light of the risk of possible allergic reactions, limited use is recommended and the Global Aesthetics Consensus panel set forward a specific schema for judicious use of HYAL: 10 to 20 U single injection for an area less than 2.5 mm, and for an area 2.5 to 1 cm, two to four injection points with 10 to 20 U per injection point. Considering the risk of allergic reactions to HYAL, when possible, some physicians favor an intradermal skin test prior to injection. Clinical judgment is recommended in order to titrate the dosage to the desired effect as this depends on the volume placed and product itself as not all HA fillers respond similarly to HYAL. 2 The ability of HYAL to dissolve filler is dependent on multiple factors that are discussed later in the vascular compromises section of the chapter. In short, heavier products with higher cross-linking require increased amounts of HYAL to obtain the desired effect.
Paresthesias and hypesthesias are reported after filler injection. These complications can be related to filler placement and depth of injection, a localized inflammatory response, compression or direct trauma to the nerves. Symptoms include transient numbness or pain and may include facial nerve branches dependent on location and depth of injection. Other placement-related complications include parotid duct obstruction or injury and exacerbation of temporomandibular joint pain. Parotid-related issues may arise secondary to chemical dermatitis from saliva tracking into the facial soft tissues or direct obstruction/duct injury with symptoms consisting of edema, erythema, pain, and trismus. Treatment options consist of compresses to the affected side, antibiotics, oral steroids, and HYAL after 24 hours of antibiotic therapy.
5.4.4 A Tool for Management of Early Adverse Events: Use of Blunt-Tip Cannula
Some studies have advocated for the use of blunt-tip cannulas to reduce bruising. This push toward cannula injection comes from a safety perspective as it relates to vascular events, particularly intravascular injection. 2 , 3 , 7 , 11 , 12 In a recent study by Pavicic et al investigating precision and avoiding filler diffusion, “if precision in filler injection is defined as the filler material remaining in the plane of intended implantation, then using cannulas resulted in a more precise injection of material as compared to needles. Others disagree with this approach and favor precise injection with a needle. Applications with needles resulted in the distribution of material into more superficial layers, which was not noted for cannulas.” More importantly, this study also noted the similar ease of vessel wall penetration between 27 g needles and 27 g cannulas, thus suggesting the use of cannulas 25 g and larger when placing dermal filler. 13
Hexel et al completed a double-blinded, randomized controlled trial to compare safety and efficacy between the use of metallic cannula vs. standard needle to augment the nasolabial folds. A total of 25 participants were included.This trial reported fewer side effects on the side injected with cannula for all parameters: pain, edema, erythema, and hematoma formation. 14
5.5 Inflammatory Reactions
5.5.1 Allergic Reactions and HA Hypersensitivity
HA filler hypersensitivity treatment is a function of severity. With more purified and synthetic derivatives the rate of hypersensitivity reactions has decreased to about 0.02%. 7 Immediate hypersensitivity is often less severe and is more common in products with an anesthetic component. In the majority of cases it is self-limited and resolves with supportive measures within a few hours or days. If the patient’s history suggests a more mast cell–mediated allergic profile the reaction may respond to antihistamines. Immediate substantial swelling and angioedema may occur, and rare anaphylactic reactions have been documented in the literature. 15 , 16 The authors recommend having an EpiPen on hand in the office to begin immediate treatment due to the possibility of impending airway compromise. Delayed hypersensitivity usually resolve without untoward consequences but may be treated with oral steroids depending on the severity of the reaction and the allergen should be removed if possible.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree