Abstract
This chapter addresses the considerations involved in remediating bulges and hernias. The challenge in these procedures is to create a lasting connection between two divided tissues under tension. Bulges result from lessened strength and elasticity of the abdominal wall and generally pose no risk; hernias come with a higher risk that can result in serious and long-term symptoms. Presurgical steps (e.g., advising the patient to lose weight and/or stop smoking) are outlined, and the procedures for hernia repair are detailed. Unsupported repairs rely solely on sutures to hold the abdominal wall; supported repairs add a reinforcing mesh to the repair site; bridged repairs suture spanning material to the abdominal wall. The authors cover the factors, strengths, and contraindications that should be weighed in deciding which repair best suits a patient. Extended discussion is given the critical areas of skin vascularity, force distribution, and components release with perforator preservation and force distribution with mesh. Images from actual cases supplement the discussion in the text. Postoperative care guidelines and information regarding outcomes and complications are also included.
41 Abdominal Wall Reconstruction
41.1 Goals and Objectives
Understand the anatomy and forces on the abdominal wall.
Appreciate the blood supply to the skin and the abdominal wall musculature and how to preserve it during abdominal wall closures.
Establish an algorithm for the treatment of the open abdominal wound.
Develop a working knowledge of prosthetic and bioprosthetic meshes for elective hernia repair.
Develop the concept of force distribution and suture pull-through as it relates to abdominal wall closure.
41.2 Patient Presentation
The central issue to abdominal wall reconstruction (AWR) is the optimal means to create a lasting connection between two divided tissues under tension. Understanding the forces and physiology at the suture/tissue interface (STI) is critical to successful closure of the abdominal wall.
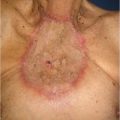
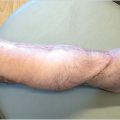
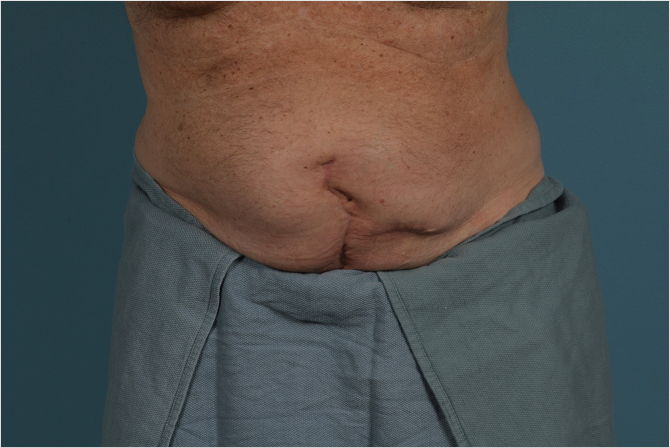
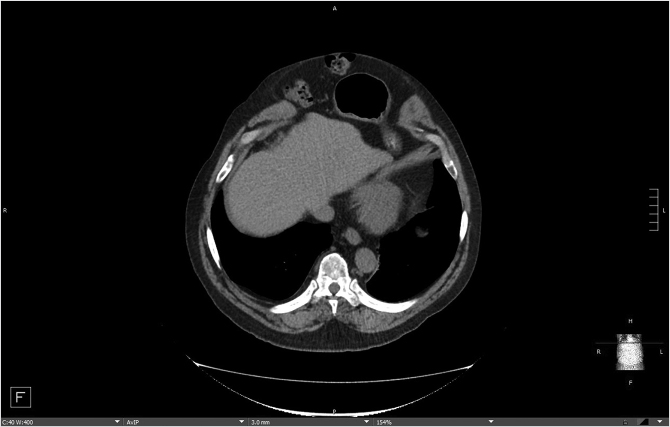
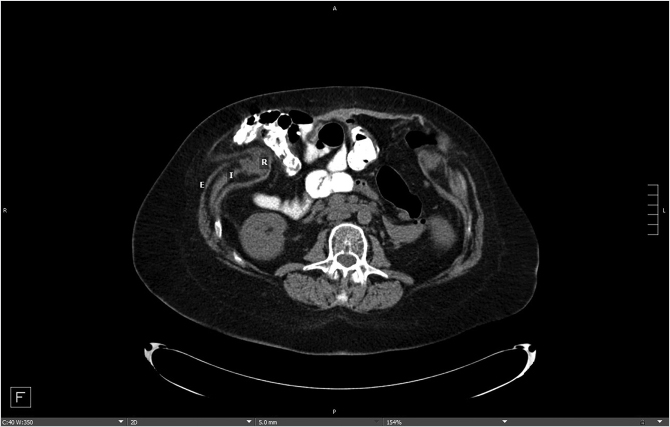
Patient presentation is straightforward, and the majority of diagnoses can be established with a history and physical examination that will distinguish between two conditions—bulges and hernias. Bulges occur when the strength and elasticity of the abdominal wall is not enough to contain the viscera uniformly from episodes of increased intra-abdominal pressure. The classic appearances of postpartum women and the epigastrium of heavier older males (the so-called “beer belly”) are consistent with rectus diastasis and bulges. In general, there is no risk to abdominal wall bulges, because the intestines face a smooth peritoneal lining without abrupt transitions and will not strangulate. In certain areas of the body such as the groin, umbilicus, and the epigastrium, a failure of the local tissue to contain the viscera creates a true hernia–distinctive from abdominal wall bulges due to a small defined ring that can permit entry of a bowel loop and possibly lead to strangulation. In contradistinction to the smooth peritoneal contour of bulges, these “ventral” hernias (as they are located on the front surface of the abdomen) have rings in the shape of the letter omega with a lip that can catch the preperitoneal fat and bowel. A third category of abdominal wall defects is incisional hernias, notable for the scar demonstrating a prior entry into the abdomen (Fig. 41‑1). The scar at the STI has not withstood the abdominal wall tensions and pressures and has failed over time. Mechanistically, the total strength of the physical construct of repair and the biologic healing is less than the loads applied at some point in the postoperative period, leading to deformation of scar and failure. There is some minor overlap between groups; for instance, a bulge from rectus diastasis can be associated with the development of a true umbilical or epigastric hernia. History and physical examination is typically adequate to differentiate these clinical entities, though an abdominal CT scan can add confirmatory information for heavier patients (Fig. 41‑2, Fig. 41‑3).
Patients will present with complaints of pain, bowel disorders, and/or change in abdominal wall contour. Large hernias and bulges associated with a loss of intra-abdominal pressure will often be associated with back pain due to a loss of “core strength” due to decreased intraabdominal pressure. Hernia size and location may not be overly correlated with symptoms, as some small hernias will be symptomatic and large hernias can be unnoticed by the patient. The indication for repair of these hernias and bulges is for the decrease of local pain and the avoidance of bowel incarceration at the hernia site. Quality-of-life improvements from repaired contour and core strength are harder to quantitate but are also present.
The majority of patients will present, as above, with a large ventral hernia, often as a recurrent incisional hernia. Less common are patients who have recovered from an abdominal catastrophe such as necrotizing fasciitis, multiple laparotomies with subsequent skin grafting over bowel, stomas, or large wounds with enterocutaneous fistulae. These patients’ complaints are similar to those with large hernias, but have the added issues of potentially larger defects of the abdominal wall with possible absence of multiple anatomic components. They often have a level of debilitation due to long illness and convalescence.
41.3 Preparation for Surgery
All patients are encouraged to lose weight prior to surgery, but this is seldom achieved. Actively smoking patients similarly are urged to stop for 1 to 2 months prior to the procedure, though smokers have not had worse outcomes for the procedures described in the following section. Immunosuppressants are managed by the transplantation surgery teams, steroids should be at stable (and low) doses, and sirolimus is to be avoided due to its profound wound healing effects. Standard medical evaluation is done if needed, including cardiac clearance by the preoperative clinic or the patient’s medical doctor. Specific assessment of nutritional status, including pre-albumin serum levels, may be required dependent on the patients underlying condition. The lead author (G.A.D.) prescribes a gentle bowel preparation the night before surgery, consisting of clear liquids, a half bottle of magnesium citrate, and two bisacodyl tablets to clear the bowel of particulate matter. This is done to decrease intra-abdominal volume and to minimize the early forces at the STI. Many surgeons do not prescribe a bowel prep if the hernia is easily reducible and no loss of domain is present.
Patient who have recovered from a significant intra-abdominal illness with a resultant large hernia should be fully stable and recovered from the initial health crisis that led to the abdominal wall defect. Additionally, patients with enterocutaneous fistula require maximal nutritional support and maturation of their fistula. If a defect is present with skin-grafted small bowel, a degree of scar maturation is needed to be able to remove the skin graft. Often times, gently pinching the skin graft provides an assessment of the degree of adherence to the underlying bowel. These patients with complex defects most often require a combined surgical effort with the primary surgical service. Confirmation of normal lower extremity anatomy and blood supply is required if flaps from the thigh are considered.
41.4 Treatment
41.4.1 Concepts of Pressure, Tension, and Type of Repair
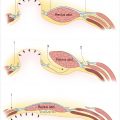
The goal of a hernia repair is to return uniformity to abdominal wall counter pressure against the viscera. “Unsupported” direct repairs rely solely on sutures to hold the abdominal wall. “Supported” direct repairs attempt to distribute the forces on the repair over a larger area by adding a reinforcing mesh to the suture repair site. In bridged repairs, a spanning material is sutured to the abdominal wall to prevent bowel from entering the hernia sac, but the abdominal wall is not approximated. These three methods of abdominal wall repair have radically different conceptualizations of the long-term efficacy of approximating the divided abdominal wall.
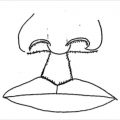
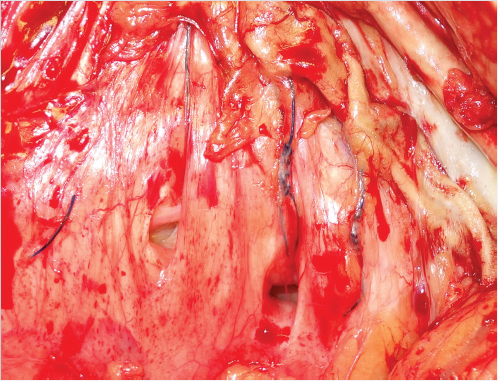
A suture acts to appose two divided tissues by applying pressure on the inner aspect of a thin flexible loop. A perfectly placed suture would apply just enough force to bring the tissues together to overcome early distraction forces. In an ideal world when forces required to appose tissue are low, the tissue inside the loop of suture remain viable, and inflammation caused by the tissue division and reapproximation incites the normal cascade of wound healing. Unimpeded biologic healing should be expected to regain 70% of the native tissue strength. 1 The long-term strength of the closure in these situations is the physical strength of the suture construct and the strength of the healed biologic tissues. The tensile strength of the physical construct is directly related to the microencapsulation of suture and mesh filaments as a foreign body response. Failure of the physical construct is due to tearing of the filaments away from its encapsulation scar. The ultimate tensile strength of the biologic healing is from scar tissue that joins and unifies the gap between the divided tissues. Therefore, the total strength of any repair is a complex mixture of scar between living tissues and around foreign bodies. High tension closures (such as in abdominal wall surgery and orthopedics) may be different from other situations in surgery, because the forces required for tissue approximation create tissue pressure that in all likelihood can exceed capillary perfusion pressure, causing necrosis and creating a micropressure sore within suture loops. This area of necrosis turns into scar 2 and has the potential to remodel, stretch, and thin over time in response to physical stresses. Visually, this effect is the commonly recognized phenomenon of suture pull-through, as the sutures can be seen to cheese wire through the intact abdominal wall (Fig. 41‑4).
The three types of abdominal wall closures (suture unsupported repair, direct supported repair, and spanning mesh repair) have markedly different concepts regarding the healing of high tension approximations. A suture repair of an abdominal wall defect regards the biologic healing and physical strength of sutures to be acceptable to resist the forces at the STI. It is well established that suture repairs trade a higher rate of long-term failure of these high tension closures for decreased local wound issues of mesh implantation. 3 Direct supported repairs attempt to reduce the forces at the STI by increasing the surface area of foreign material and encouraging microencapsulation scar. Proponents of direct supported repairs find the benefits of a stronger physical repair worth the downsides of mesh use and more tension at the closure site due to the reapproximation of divided tissue. Finally, spanning meshes used for abdominal wall defects rely solely on the physical strength of the repair, as there is no attempt to join the abdominal wall at the defect. The abdominal wall muscles are not manipulated and moved, thereby keeping low the tension at the closure (the so-called tension-free repair). The abdominal wall muscles are shortened and not at their optimal lengths, causing them to be at a weaker, less contractile point on their Starling curves. A muscle intentionally left with sarcomere overlap at the microscopic level would not generate as much force against the spanning mesh, with the thought that this lower tension state would lead to a longer-lasting hernia repair. Having presented these three viewpoints, there is general consensus that suture repairs should only be attempted for defects 2 to 3 cm in size. For the two remaining options, it has been shown that direct supported repairs have lower recurrence rates than bridged repairs in open surgery, 4 and therefore will be the focus of this chapter. The efficacy of laparoscopic-bridged repairs is touted by several authors. Such repair is most often done by general surgeons without the input of plastic surgeons. Therefore, this is outside the scope of this review.
41.4.2 Surgical Procedure for Incisional Hernias Based on Skin Vascularity and Force Distribution
Abdominal Skin Vascularity
The dominant blood supply to the abdominal wall comes from perforating blood vessels that emerge through the rectus abdominis muscle and supply the central abdominal skin. In rough terms, below the umbilicus the source of these perforators is the deep inferior epigastric artery and above the umbilicus is the superior epigastric artery. These perforators exist in medial and lateral rows and are located in the periumbilical area. In the inferolateral abdominal skin, the vascular supply is from the superficial inferior epigastric system, and over the external oblique in the upper and mid-abdomen the sources of blood are lateral intercostal perforators. Abdominal skin flap elevation is a commonly performed surgical maneuver, but is not without risk of necrotic tissue loss, as it requires adjacent angiosomes to supply the tissue whose primary perforating vessel was divided.
A working knowledge of abdominal skin blood flow is important in the performance of hernia surgery, as skin flap elevation is a necessary requirement for exposure and manipulation of the abdominal musculature. Old skin incisions interrupt the dermal circulation, and so one should assume that blood does not cross a scar. With new skin flap elevation and loss of a primary perforating vessel, the blood flow becomes less pulsatile and more laminar. The choke vessels dampen pulsatility and there is overall lower arteriolar pressure. Tissue perfusion, based on the difference between arterial and venous pressures, is also decreased because outflow is reduced due to choke vessels on the venous side. Decreased total blood flow that lacks pulsatility has been shown experimentally and clinically to have more unreliable healing than when tissue is oxygenated with pulsatile blood flow. 5 , 6 , 7 Preservation of perforators will maintain skin vascularity and be a significant aid in healing. Scarred soft-tissue vascular beds only have laminar flow, and therefore should be removed whenever possible.
Force Distribution
If the forces on the closure are greater than the sum of the strength of the physical construct of closure and the strength of biologic healing, then the scar will remodel and widen. The early separation of laparotomy closures as a predictor of eventual incisional hernia formation was demonstrated by Pollock and Evans and later confirmed by Burger et al. 8 , 9 , 10 Force distribution is a concept that focuses on lowering the forces experienced at each STI. The lower the STI at each suture and the more evenly distributed the forces for closure, the higher the success of closure. Alternatively, the greater the episodic forces experienced at the STI, the more the sutures will pull through the tissue—the central cause of hernia recurrence. 11 , 12 , 13 While the total force of closure in direct supported repairs will be greater than the forces experienced by bridging meshes, the forces at the STI are lowered in two separate ways. First, the total force of closure is divided by a greater number of sutures, thereby lowering the force at each individual STI. Second, the long-term strength of the repair is achieved both with the physical construct of mesh and the biologic healing of the overlying abdominal wall.
A compliant abdominal wall absorbs energy with coughs, the Valsalva maneuver, and forceful movements, rather than sending energy waves to the STI. Patients with weight loss, treated ascites, and a history of multiple large gestations are all known to have compliant abdominal walls and relative success with closure. Patients with noncompliant abdomens include those patients with prior intra-abdominal sepsis and scarring, a history of radiation therapy, lateral scarring from prior incisions, and patients at their maximal weight. The state of a hernia itself causes an abdominal wall to become less compliant. 14 While many view a components release as a means to bring vascularized tissue to the midline for closure, an alternative conceptualization is to regard the release of the external oblique muscle as a means to improve lateral abdominal wall compliance and thereby protect the midline sutures.
It is often remarked that hernias tend to occur at the end of the repair, and this too can be explained as a function of the STI. The best hernia repairs have smooth transitions between repaired abdominal wall and intact fascia, with a “matching” of compliances. Mismatched compliances will cause high tension at the STI and suture pull-through. Anchoring mesh to noncompliant rib or symphysis pubis in particular leads to areas of imbalance of STI forces and early failure. Another situation where compliance mismatches (and increased chance for failure) exist is when a direct supported repair for a midline incisional hernia ends adjacent to significant rectus diastasis. It is for this reason that recurrences often occur at the superior and inferior borders of mesh. In general, a longer repair with properly balanced forces at each STI (working out the “dog-ears” of the abdominal wall closure) is optimal for long-term success and is preferable to a short repair. This is opposite to the common belief that one should leave alone stretched but otherwise intact abdominal wall.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree