Abstract
Sternal reconstruction, as the name implies, is a sternal/chest wall reconstruction made necessary due to infection, oncologic resection, and trauma. A presurgical examination drawing on imaging (CT, nuclear), nutritional markers, and knowledge of any prior reconstruction the patient has had to the chest wall is necessary to determining the proper treatment. The authors guide the reader through each stage of treatment, from debridement of infected and necrotic tissue, to soft tissue cover options (pectoralis major, rectus abdominus, latissimus dorsi, and omental flaps), to adequate drainage. Pictures from actual cases illustrate the discussion in the text. Guidelines for postoperative precautions are followed by closing paragraphs on outcomes and potential complications.
40 Sternal Reconstruction
40.1 Goals and Objectives
Understand the etiology and risk factors for sternal wound breakdown.
Define the goals of reconstruction and various reconstructive options.
Have an understanding of the indications for use of sternal plating systems and vacuum-assisted closure devices.
40.2 Patient Presentation
The most common indications for sternal wound reconstruction are infection, oncologic resection (+/– subsequent radiation affects), and trauma. 1 The chest wall, composed of 12 paired ribs and a midline sternum, serves to protect the heart, lungs, and great vessels. Numerous antagonistic muscle groups rely on the rigid thoracic framework to produce functional breathing. When the overall structure of the chest is affected, it can lead to significant physiologic changes and associated morbidity. Thus, it is imperative to address any disruption or wound early to limit the effect on the patient’s long-term functional status.
Infection most commonly occurs in the setting of cardiac surgery and can manifest in a number of ways. Patients who undergo sternotomy for coronary artery bypass, in which the internal mammary artery (IMA) is harvested, are at an increased risk for sternal nonunion and subsequent wound infections. Studies have shown sternal blood flow is decreased as much as 90% when the IMA is harvested. 2 Infections that occur between the skin and pectoralis fascia are termed superficial sternal wound infections (SSWI) and carry a mortality/morbidity rate between 0.5 and 9%. However, infections that are deep to the pectoralis musculature, deep sternal wound infections (DSWI) or mediastinitis, are more serious with a mortality rate between 10 and 47%. Risk factors for development of DSWI include diabetes, chronic obstructive pulmonary disease (COPD), obesity, tobacco, osteoporosis, prolonged operative times, and reoperation. 1 In addition, patients who develop sternal wound complications after chest surgery are prone to an average of 20 more days in the hospital and a nearly threefold increase in hospital costs. 1 In 1986, Pairolero and Arnold classified infected sternal wounds based on interval between surgery and presentation of symptoms (Table 40‑1). 3
Most commonly patients present with an open wound or draining sinus tract. Patients may also present with a sternal click on exam or the complaint of “bony pain” with deep inspiration or coughing. Intermittent low-grade fevers and overall malaise can be subtle signs of an indolent chronic infection.
40.3 Preparation for Surgery
Reconstruction can be generalized into soft-tissue coverage and rigid skeletal support. Restoration of skeletal support and reestablishing the thoracic framework prevent paradoxical chest wall motion and facilitate physiologic respiration. Preoperative imaging, including CT and nuclear medicine scans, can assist in planned bony resection and assess for the potential need for rigid fixation. However, the decision to use a sternal plating system must take into account any potential infection remaining in the wound bed. Finally, preoperative antibiotic therapy should be guided by available wound biopsies. A treatment duration of 6 to 8 weeks is usually required after adequate bone and soft-tissue debridement.
Poor nutritional markers (prealbumin/albumin) along with suboptimal glucose control can reduce the likelihood of a successful reconstruction. An effort should be made to maximize nutrition and maintain normal glucose levels perioperatively.
Finally, while there are a number of flaps that can be used to reconstruct thoracic defects, the previous use of the internal mammary vessels in coronary bypass, location of the sternal wound, and prior surgeries may limit the options. If there is any question about the availability or patency of a certain flap, preoperative imaging may help reveal potential flap inadequacies.
40.4 Treatment
Wound Preparation and Rigid Skeletal Reconstruction
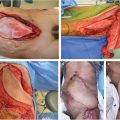
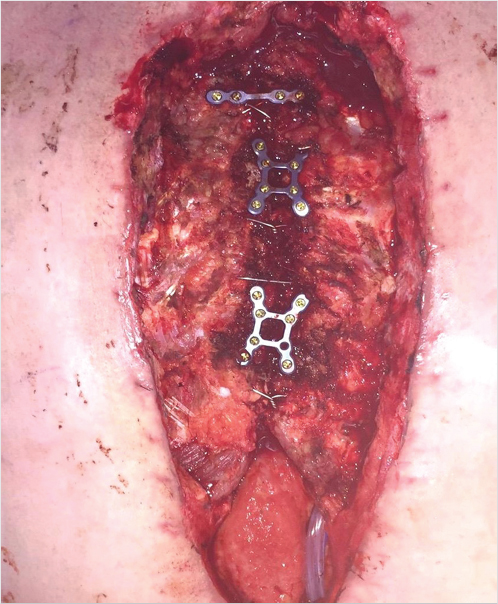
The thorough and complete debridement of all infected and necrotic tissue followed by vascularized soft-tissue coverage with adequate drainage is key. Tissue cultures showing >105 organisms/cm3 are considered positive and should be treated with debridement, antibiotics, and possibly vacuum-assisted wound dressing followed by delayed coverage. 4 The immediate use of rigid plating systems should be avoided when possible in the face of positive wound cultures. Debridement should include all devitalized tissue and removal of previously placed implants including sternal wires, plates, and pacing wires. Bone debridement should be performed until bleeding is seen from edges and the cortex is no longer soft and friable (Fig. 40‑1, Fig. 40‑2). Radical sternectomy is not indicated and sternal salvage should be attempted if the bone is viable. 4
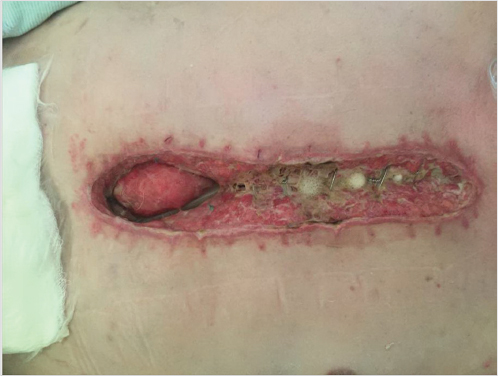
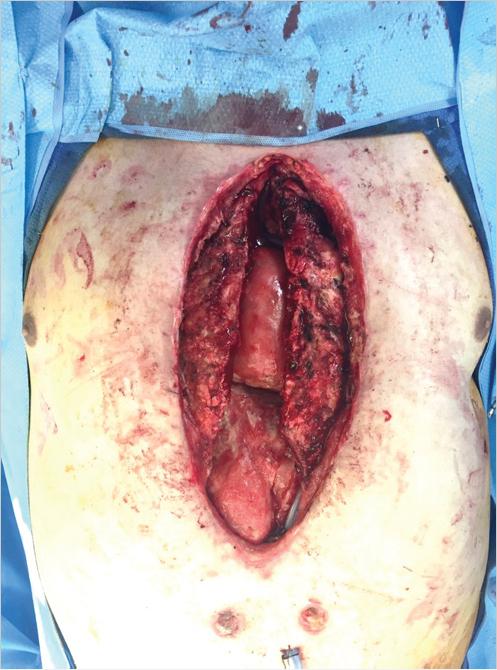
Subatmospheric pressure wound therapy may be utilized to increase peristernal blood flow, even in the face of internal mammary harvest. 5 It has also been shown to decrease bacterial counts and decrease dead space by increasing granulation tissue formation. 6 , 7 Through these mechanisms, negative pressure wound therapy has been shown to decrease the number of days between wound debridement and definitive closure (8.5–6.2 days) and the number of flaps required per patient (1.5–0.9). 8 Finally, by only having to change the negative pressure dressing three times per week instead of the traditional twice per day gauze changes, there is less exposure of the wound and fewer procedures on potentially debilitated patients. The use of vacuum-assisted wound therapy is now the standard of care in many situations. These include in patients who are unstable, whose chest cannot be closed due to cardiopulmonary compromise, or situations in which pathologic disease-free margins must be confirmed. 9
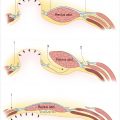
Rigid fixation is ideal and paramount for bony union and stabilization of the chest wall. It has been shown to prevent paradoxical chest wall motion, improve patient comfort, accelerate healing, and decrease mediastinal hernias compared to flap coverage alone (Fig. 40‑3). 10 Studies have shown plating promotes earlier bony union from accelerated osseous healing. 11 The increased stabilization and re-establishment of normal chest function has been shown to decrease ventilator dependence and reduce overall hospital length of stay. 11 Recurrent plate infections can be minimized with thorough debridement, culture-directed antibiotic therapy, and adequate soft-tissue coverage. Cicilioni et al performed rigid fixation on 50 consecutive cases in which 37 cases were culture positive for osteomyelitis and report only one plate infection. 12
40.4.1 Soft Tissue Coverage
Since 1980, when Maurice J. Jurkiewicz and his Emory colleagues published their 20-year experience, flap coverage has been the gold standard for sternal wounds. 13 Recruitment of adjacent tissue is necessary, as there is typically a very limited amount of soft tissue immediately available over the sternum and even less after wide debridement. The muscle flaps most commonly utilized include the pectoralis major, rectus abdominus, and the latissimus dorsi flaps. The omentum is often used for reconstruction of large, deep wounds and can be used alone or in conjunction with other flaps. It is essential to understand the indications and limitations of each flap, as the resultant wound can become greater than anticipated after debridement and one must be able to improvise.
Pectoralis Major Flap
The pectoralis major muscle serves to internally rotate and adduct the arm. It originates along the sternum, medial clavicle, and superior six costal cartilages and inserts on the bicipital groove of the humerus. The flap is a type V muscle flap, with the thoracoacromial artery being the major supply and additional segmental flow arising from intercostal perforators derived from the IMA. The thoracoacromial artery is a branch of the second portion of the axillary artery. It emerges from under the clavicle between the middle and lateral third of the clavicle. It passes on the undersurface of the muscle between the pectoralis major and minor. The flap can be raised in a fairly avascular plane as a muscle-only or myocutaneous flap. The dissection for flaps based on the thoracoacromial trunk should start medially at the sternal attachment. The superior attachment to the clavicle should be taken down as well. Lateral dissection should be carried out until adequate medialization into the defect is accomplished. The muscle may also be released from its insertion on the humerus to gain more medial advancement and a greater arc of rotation. This is usually performed through a counter incision near the axillae. Conversely, the flap can be used as a turnover flap based on the internal mammary perforators. This should only be performed once the patency of the ipsilateral internal mammary has been confirmed. This dissection proceeds in a suprafascial dissection above the muscle and is carried out until it can be divided from its humoral insertion. Again, this may require a counter incision to fully visualize the insertion. The thoracoacromial artery is divided as the muscle is flipped into the defect. The majority of sternal wounds can be closed with a single pectoralis flap; however, more extensive defects may require bilateral muscle flaps. Inferior sternal defects are difficult to obtain consistent coverage with pectoralis flaps. In such cases, studies have shown that extending the pectoralis dissection to include anterior rectus fascia or splitting the pectoralis turnover flap can help provide coverage inferiorly.
Rectus Abdominus Flap
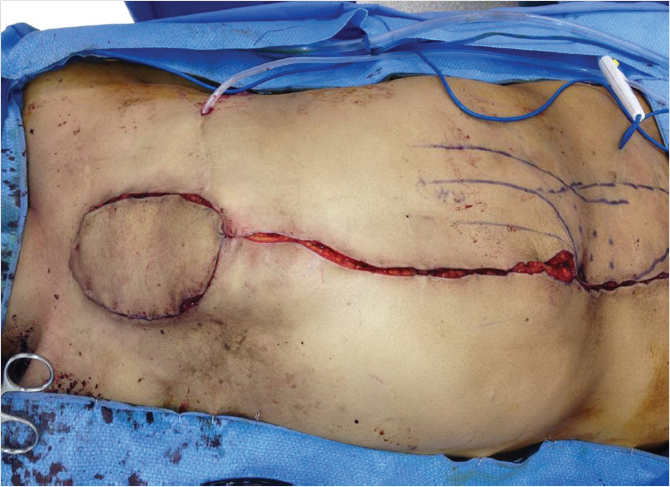
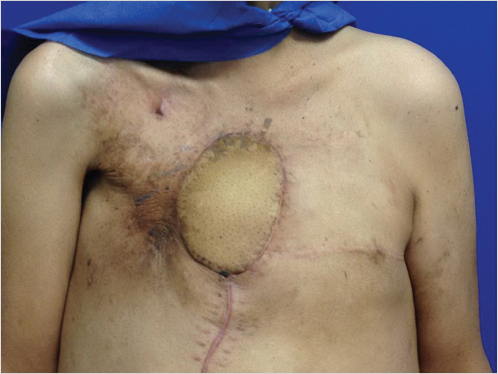
Each rectus abdominus muscle serves to flex the trunk. It originates on the pubis and inserts on the costal margin. It is a type III muscle flap with the superior and inferior deep epigastric arteries being the two major pedicles. The deep superior epigastric vessel is the continuation of the internal mammary vessel onto the abdomen. It pierces the posterior rectus sheath superiorly just after the final costal cartilage and travels on the posterior surface of the muscle. It anastomoses with the deep inferior epigastric, an ascending branch of the external iliac, at the level of the umbilicus. The flap can be raised as a muscle-only or myocutaneous flap depending on the reconstruction required. In general, the rectus flap is not a good option if the IMA on the same side has been used for bypass. However, in dire settings, when no other flaps are available, some have reported that the rectus flap can be pedicled superiorly even in the absence of the internal mammary system, based on accessary blood supply from the eighth subcostal artery. 14 , 15 , 16 If a muscle-only flap is to be harvested, a variety of incisions can be used. The most common are the midline and the paramedian incision oriented over the rectus muscle. The dissection is taken down to the anterior rectus fascia. This is divided longitudinally exposing the entire length of the muscle. The anterior sheath is dissected off the muscle without disrupting the inscriptions and violating the integrity of the muscle. Localization and division of the deep inferior epigastric vessels on the lateral side of the muscle near its origin allows for elevation of the flap in an avascular plane from inferior to superior. Care must be taken to avoid injury to the pedicle as it runs along the underside of the muscle. The muscle can be transposed into the defect and covered with local soft tissue and skin, or grafted skin. The divided anterior rectus fascia is approximated and the abdomen is closed. If a myocutaneous flap is desired, a template can be designed from the defect and oriented either vertically, obliquely, or horizontally over the rectus muscle. After division of the skin and soft tissue, the anterior rectus fascia under the skin paddle is excised and is taken with the flap (Fig. 40‑4, Fig. 40‑5). The resultant anterior fascial defect may be too large for primary closure, requiring that mesh repair be performed. It is important to explain to the patient preoperatively that this may ultimately result in abdominal wall laxity (i.e., bulging) or hernia. It is critical to assess the patient before surgery for prior abdominal operations that may have compromised cutaneous perforators or violated the intramuscular blood supply. 4
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree