Abstract
This chapter covers the subject of botulinum toxins and the different classes of fillers—collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, permanent fillers, liquid injectable silicone—available for facial rejuvenation. The authors sequentially describe each step of the presurgical, surgical, and postsurgical phases. Indications that respond best to Botox are cited, and techniques for filler injection are covered. The chapter concludes with guidelines on postsurgical care and, importantly, potential complications and their treatments.
23 Facial Rejuvenation with Injectables
23.1 Botox: Goals and Objectives
Know the indications for use of botulinum toxins including FDA-approved indications and off-label uses.
Know the different types of botulinum toxins and the advantages and disadvantages of each.
Know the contraindications and cautions for use of botulinum toxin.
Know the potential complications and side effects of treatment with Botox and what should be discussed in order to obtain informed consent.
Know proper reconstitution, storage, preparation, and the techniques for injection of Botox for different areas of the face.
23.2 Fillers: Goals and Objectives
Know the different classes of fillers and the pros and cons of each and their use in appropriate areas.
Know what the G prime value is and how it guides the use of the filler.
Know the complications that can be associated with injection of fillers.
Know the injection techniques for the upper, mid, and lower face.
Know aftercare procedures and cautions after injection with fillers.
23.3 Botox
Botulinum neurotoxin (BoNT) is an exotoxin that is secreted by the Clostridium botulinum bacteria. There are seven serotypes of botulinum toxin labeled A to G, but only types A and B are produced for clinical use. Although serotypes A and B have the same clinical effect of chemodenervation, they have different mechanisms of action. Type A botulinum toxin (BoNT-A) inhibits SNAP-24 which inhibits vesicular release of acetylcholine at the neuromuscular endplate. Type B botulinum toxin (BoNT-B) binds to and cleaves synaptobrevin which blocks presynaptic vesical fusion and neurotransmitter release. The first reported use of BoNT in humans was in 1980 for the treatment of strabismus by Dr. Alan Scott. Dr. Carruthers pioneered the use of BoNT for cosmetic treatment of glabellar rhytids in 1987. 1
BoNT is a safe drug when used appropriately. Most cosmetic patients receive 25 to 75 units per treatment. The most commonly used BoNT used for cosmetic applications is Type A. The different formulations of injectable BoNT-A available are OnabotulinumA (Botox Cosmetic, Allergan), AbobotulinumtoxinA (Dysport, Medicis), and IncobotulinumtoxinA (Xeomin, Merz). There are minor differences between the different formulations. Compared to Botox, Dysport is thought to have a faster onset, but its spread is wider which may be a factor when deciding if to use it around the ocular muscles. It requires 2.5 to 4 times the dose of Botox for the same effectiveness. Xeomin has the advantage of not needing refrigeration and its dosage is 1:1 with Botox. It does not have hemagglutinin proteins which give it a lower theoretic hypersensitivity profile than Botox or Dysport. Botox is approved by the Food and Drug Administration (FDA) for the treatment of glabellar rhytids and crow’s feet. Dysport and Xeomin are FDA approved for glabellar rhytids. RimabotulinumtoxinB (Myobloc, US WorldMeds LLC) is a BoNT-B, but it is not commonly used for cosmetic applications as it has a lower pH which makes injection more painful and it has a shorter duration of action. 1
23.4 Fillers
There are a wide variety of fillers that are FDA approved for use in facial rejuvenation. Fillers can be first divided into two main categories: biodegradable fillers which are impermanent and non-biodegradable fillers which are permanent. The biodegradable fillers can be further subdivided into collagen, hyaluronic acid (HA), calcium hydroxylapatite (CaHA), and polylactic acid fillers and within these different categories, the fillers can vary in their concentration of material, size of the particles, the viscosity, and side-effect profile. Common uses for fillers are for correction of moderate-to-severe facial wrinkles, lip augmentation, cheek augmentation to address midface age-related volume deflation, and tear trough deformities.
The fillers have different properties which guide which ones are used for different areas. The size of the particles, the amount of cross-linking, the G-prime, the viscosity, and the concentration of the product determine its properties. G-prime refers to the elastic modulus, or stiffness of the filler. The more cross-linking of the polymer, the higher the viscosity and G-prime of the product and the more volume and lift it will give and the longer it will last. 2
23.4.1 Collagen
The first facial fillers were made of bovine collagen, but they fell out of favor due to the need for skin testing before injection to detect possible hypersensitivity reactions and a short duration of action. Zyderm I and II and Zyplast (Inamed Aesthetics, Santa Barbara, CA) were derived from bovine collagen and to address the concerns of hypersensitivity, CosmoDerm I and II and CosmoPlast (Inamed Aesthetics, Santa Barbara, CA) were developed from highly purified human collagen. 3
23.4.2 Hyaluronic Acid
HA is a naturally occurring glycosaminoglycan in the skin. It is synthesized using non-animal sources and consists of cross-linked polysaccharide chains which combine with the skin’s HA and water to increase the volume and lift in the area. It is also thought to contribute to neocollagenesis. HA lasts between 4 and 12 months and is cleared by the body via hepatic metabolism. HA fillers can be used for correction of moderate-to-severe rhytides and folds and is usually injected into the mid-to-deep dermis. The side-effect and complication profile of HA includes mild pain, edema, and erythema which can be related to injection technique. Hyaluronidase can be used to dissolve HA which makes HA the only currently reversible filler available today. Currently available HA fillers include Restylane (Galderma), Juvederm (Allergan), Prevelle (Mentor), and Belotero (Merz). 4
23.4.3 Calcium Hydroxylapatite
Calcium hydroxylapatite microspheres are made of material that is similar to bone. The microspheres measure 25 to 45 µg and are suspended in an aqueous sodium carboxymethylcellulose carrier gel. The microspheres stimulate the neocollagenesis which lasts for 15 months or longer due to the slower degradation of the particles. CaHA is degraded into calcium and phosphate which is resorbed. It is currently FDA approved for HIV-associated lipoatrophy and moderate-to-severe rhytids. It should not be used for superficial injections or in the lips, tear trough, or glabella due to granuloma formation. There is no reversal agent for CaHA. Radiesse is a CaHA filler currently available in the United States. 4
23.4.4 Poly-L-Lactic Acid
Poly-L-lactic acid (PLLA) is a synthetic polymer that is broken down in the body via hydroxylation. Sculptra is a PLLA-based filler that is available in the United States. The microspheres usually measure 40 to 63 µg and after injection, it causes a subclinical inflammation which stimulates collagenesis. It is FDA approved for the treatment of HIV-related lipoatrophy and correction of moderate-to-deep rhytides. It has also been used to treat acne scars and for larger volume correction of facial asymmetry. The product requires reconstitution with sterile water hours prior to injection and may take several treatments to achieve the needed volume correction, but the results can last as long as 2 years. Complications associated with PLLA include nodule formation and clumping of the filler in dynamic areas. 4
23.4.5 Permanent Fillers
Permanent fillers are polymethylmethacrylate (PMMA) microspheres and liquid injectable silicone (LIS). PMMA microspheres measure 30 to 50 µg and are suspended in a gel that contains a small amount of bovine collagen. The first generation of PMMA fillers used smaller and less uniform microspheres which resulted in granuloma formation 6 months to 6 years after implantation. The newest generation of PMMA fillers, such as Bellafill (Suneva Medical, San Diego, CA), utilizes larger, more uniform beads and have a better side-effect profile. PMMA microspheres stimulate neocollagenesis which occurs months to more than a year after initial injection. This filler is good for the treatment for contour deformities, facial wasting, acne scars, and deep rhytids. It is meant for deep plane injection and should not be used for superficial injection and in thin skin areas due to the risk of nodules, beading, and scarring, especially with first generation of PMMA fillers. 4 The permanence of the filler is both an advantage and disadvantage of the filler. If injected correctly, the results are permanent and do not require reinjections, but if injected incorrectly, there is no way to remove the filler except surgically.
23.4.6 Liquid Injectable Silicone
Silikon 1000 (Alcon, Fort Worth, TX) and ADATO SIL-OL 5000 (Bausch & Lomb, San Dimas, CA) are two FDA-approved LIS. The FDA approval is for intraocular use only and LIS is not commonly used for facial rejuvenation due to the risk of granuloma formation and migration of the silicone. The use of a microdroplet injection technique has been shown to decrease incidence of complications. 5
23.5 Patient Presentation
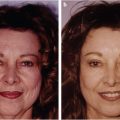
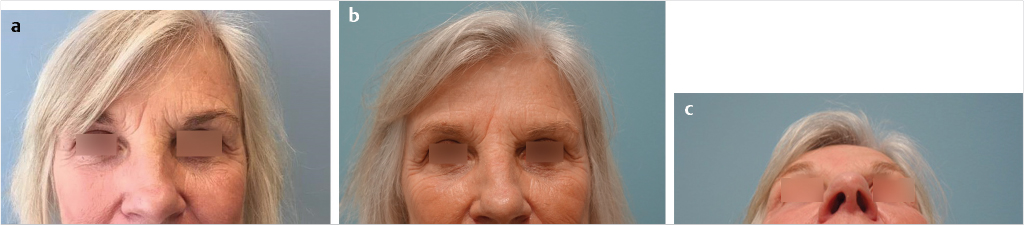
The chief concern that patients may present with in the office can vary from general requests to look younger or “less tired” to specific concerns about particular areas of the face and a request for “Botox.” Patients may complain of looking angry or upset due to rhytids that are present when their face is at rest. Others will complain of feeling like their eyes are tired at the end of the day, especially after being outside for an extended period of time. Others may present with specific concerns such as dark circles under the eyes (tear trough), deep marionette lines, or nasolabial folds (Fig. 23‑1).
The initial patient assessment includes obtaining a full history and information about prior cosmetic procedures and injections. When asking if the patient has had prior BoNT injections, the question should be directed as queries about any facial injections, not just Botox injections as some patients are not aware that Dysport and Xeomin and Myobloc are in the same class of medications as Botox.
Past medical history should be screened for neuromuscular diseases such as amyotrophic lateral sclerosis, myasthenia gravis, and Eaton–Lambert’s syndrome, which are a relative contraindication to BoNT injection. 6 Review of systems should include inquiries about problems with swallowing, bleeding problems, and eye difficulties such as ptosis and corneal exposure. It is not recommended to perform BoNT injections on patients who have recently had LASIK surgery and blepharoplasty due to a higher risk for dry eyes and corneal exposure.
Medications to watch for are any blood thinning medications including warfarin, clopidogrel, and aspirin and recent use of nonsteroidal anti-inflammatory drugs or any of the many blood thinners that are now available. The use of over-the-counter medicine such as fish oil and vitamin E should also be investigated as these may also increase the risk of bleeding and bruising. The patient will need to be off any blood-thinning medications for 2 weeks prior to injection. Aminoglycoside medications and other medications that affect neuromuscular transmission are contraindicated as they can potentiate the effect of BoNT.
Other contraindications to treatment with BoNT are allergies to botulinum toxin or human albumin. If the patient is allergic to milk protein, Dysport cannot be used as it contains milk protein.
Although BoNT and fillers have not been found to be a teratogen, it is a category C drug and injections are not recommended for women who are pregnant, nursing, or are attempting to become pregnant.
If the patient is concerned about perioral rhytids and is requesting injection around the mouth, obtaining information about his or her jobs and activities such as playing a musical instrument, any job that requires a lot of talking, and other job/activities that require a lot of movement of the mouth is important, as it may be compromised by BoNT injections around the mouth. 7
23.6 Evaluation of the Patient
The patient’s face is first examined at rest and existing rhytids and resting facial expression are noted. Common patterns of rhytids on the upper face are transverse rhytids across the forehead, vertical rhytids overlying the glabella, transverse rhytids over the nasion, and transverse and radial rhytids lateral to the lateral canthus, also known as “crow’s feet.” A snap test can be performed on the lower eyelid to test for risk for ectropion. In the lower face, the common patterns of rhytids are along the nasolabial folds, marionette lines, and vertical perioral rhytids. The resting position of the oral commissures may be downturned due to overactive depressor anguli oris (DAO) muscles. There may be a mental crease along the chin and platysmal banding on the neck.
Next, have the patient animate his or her face. The amount of movement and deepening of the rhytids should be noted. Forehead rhytids will deepen with contraction of the frontalis. The position of the brow as the frontalis contracts should be observed. Hyperactivity of the lateral brow in contrast with the medial brow may indicate a lateralized frontalis which could lead to excessive elevation of the lateral brow if not enough Botox is placed laterally. Next, have the patient frown to identify the position of the corrugator muscles. Having the patient sniff will activate the procerus and the transverse nasion rhytids can be examined. When the patient smiles, activation of the zygomaticus major (ZM)/minor will result in deepening of the nasolabial folds and the radially oriented folds at the lateral canthus. Having the patient strain his or her neck will make any platysmal banding more prominent.
BoNT injection for the treatment of a “gummy smile” has been reported. This appearance can be due to hyperactive lip elevator muscles. For patients with this complaint, there should be special consideration on the type of smile the patient has and which muscles are mainly responsible for the smile. Rubin described three basic forms of smiles. The “Mona Lisa” smile is the most common smile and is where the corners of the mouth are pulled up and out due to a dominant ZM muscle and is followed by contraction of the levator labii superioris (LLS). The “canine” smile occurs when the LLS contracts first, exposing the canine teeth followed by contraction of the ZM. The third type of smile is the “full denture” smile where the ZM and LLS contract at the same time showing both the upper and lower teeth. 8
If the patient has deep forehead rhytids at rest and there is evidence of dermatochalasis, the patient should also be evaluated to see if he or she is using his or her frontalis muscle to keep the eyelids in a position that allows for adequate peripheral vision. This exam can be done by relaxing the forehead and having the patient look down and then straight ahead while keeping a hand on the forehead. The amount of dermatochalasis should be evaluated and the patient’s peripheral vision evaluated. The patient should be counseled that by relaxing the brow with BoNT, worsening of peripheral vision may occur.
Existing asymmetries of the face should be discussed with the patient during the preinjection evaluation and there may be differences in the way the muscles respond to the injection. Since there are differences between patients in response to BoNT, an incomplete block may occur with dosages that produce a complete block in another patient.
23.6.1 Managing Expectations
It is a misperception that Botox removes all wrinkles in the area injected. In a young patient with minimal rhytids, paralysis of the muscle can result in a smooth brow due to decreased activation of muscles in the area. In a patient with deep rhytids, they need to understand that the wrinkles will remain even if the muscle is inactivated and further treatments such as skin care, resurfacing treatments, or filler will be needed to lessen the appearance of the rhytids.
The onset of Botox occurs within 3 days and the maximal effect will not be seen until 7 to 14 days after injection. If the patient has any significant events within in a week, as a wedding or public appearance, injection may want to be delayed until after the event to give time for any bruising or swelling to resolve.
The paralysis of the muscle is temporary and on average, the patient can expect it to last for 3 to 4 months with full reversal of the effects taking 3 to 6 months. 9
After injection with fillers, the patient may have bruising for 7 to 10 days after injection. Nonpermanent filler injections can last 6 to 12 months based on their properties and placement. Fillers placed in dynamic areas will not last long and more frequent injections may be needed.
23.7 Preparation for Procedure
23.7.1 Consent
Prior to injection, the patient needs to be fully informed of the risks, benefits, and alternatives to the treatment.
FDA approval for cosmetic uses currently for Botox, Dysport, Xeomin, and Myobloc is only for treatment of glabellar rhytids. Botox has the additional approval for treatment of crow’s feet. The use of BoNT in any other areas of the face is off-label. 10
The approval for fillers is generally for injection into the mid to deep dermis for treatment of moderate-to-severe facial wrinkles and folds. Restylane silk has approval for treatment of perioral rhytids and lip augmentation. Juvederm Voluma has been approved for supraperiosteal injection for mid-face volume restoration. Sculptra and Radiesse have been approved for HIV-related lipoatrophy. 11
Since injections with BoNT and fillers are often cosmetic procedures, insurance does not typically cover the treatment and the patient will be financially responsible for the treatment, follow-up visits, and for the costs for treatment of any complications that arise from the injections.
The American Society of Plastic Surgeons (ASPS) has designed a general consent for BoNT and filler injections that can be utilized by ASPS members.
23.8 Treatment
23.8.1 Prep
The skin is cleaned with a Betadine and/or alcohol prep in the area to be injected. Prior to this, the patient can place ice packs over the skin to decrease sensation over the area. EMLA or LET cream can be placed on the skin to induce some anesthesia. Some practitioners will perform regional blocks prior to injections of filler if multiple injections are needed. 12
23.8.2 Reconstitution
Botox comes lyophilized and needs to be reconstituted with saline for injection. Recommended reconstitution procedures are given by the manufacturer. For Botox, it is recommended that 2.5 mL of preservative-free 0.9% sodium chloride is added to the vial which results in 4 units per 0.1 mL. The liquid is then drawn up into a 1-cc syringe. A short 30- to 33-gauge needle is used for the injection. Large reconstitution volumes afford greater control over the amount dosed, but more concentrated formulas are less painful on injection due to the smaller volume. It is recommended that after reconstitution, it is used within 24 hours and stored in a refrigerator at 2 to 8°C. 13
Fillers come ready to use and do not require reconstitution, but some practitioners will dilute some of the fillers to facilitate the injection process and prevent clumping. 14
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree