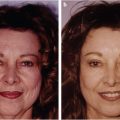
Abstract
This chapter thoroughly discusses the anatomy and various defects of the scalp and forehead that require reconstruction and introduces an algorithmic approach to the matter. Emphasis is given the etiology, diagnosis, and treatment of the three usual sources of scalp defects: congenital, posttraumatic, and oncologic. Substantial differences between scalp and forehead defects are identified, and treatment proper to the forehead is examined. Numerous photographs illustrate the irregularities and treatments discussed.
17 Reconstruction of the Scalp and Forehead
17.1 Goals and Objectives
Understand the relevant anatomy of the scalp and forehead in regard to reconstruction.
Review the various etiologies of scalp and forehead defects requiring reconstruction.
Gain an appreciation and algorithmic approach to reconstruction of defects of the scalp and forehead.
17.2 Scalp
17.2.1 Patient Presentation
Anatomy
The pertinent anatomy of the scalp that has clinical relevance can be divided into the specific regions as well as the cross-sectional components. The cross-section anatomy is summed in the acronym SCALP (Skin, Connective Tissue, Aponeurosis, Loose Areolar, Periosteum)—the skin containing hair follicles, sebaceous and sweat glands, a thin layer of subcutaneous fat, the galeal aponeurosis, a zone of subgaleal loose areolar tissue, and lastly, periosteum or pericranium. Skin, in contrast to the subgaleal space, is tightly adherent to the underlying galea. The galea has an interesting teleological history since the aponeurosis may represent a vestige of the panniculus carnosus, a layer of immediate subdermal striated muscle seen in nonprimate mammals that enables twitching of isolated areas of skin. In that vein, the galea is a fusion sheet between the frontalis of the forehead and the occipitalis posteriorly, two other structures that represent vestigial remnants of the panniculus carnosus. Laterally, the galea is contiguous with the temporoparietal fascia.
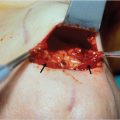
The tightly adhered skin to the underlying galea and the lack of laxity of the skin envelope to the supporting skeleton, the skull, implies that primary closure of soft-tissue defects can be challenging. The looseness of the subgaleal areolar layer has clinical implications: undermining is most easily accomplished in this plane for the creation of scalp flaps or to facilitate closure, and avulsions occur within this plane. Also, any hematoma or abscess in this plane is easily extended to more remote areas of the scalp (Fig. 17‑1).
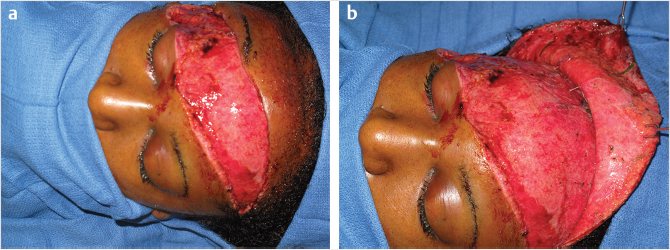
Regionally, the scalp can be divided into the same areas of the skull: temporal, parietal, and occipital. The modified anatomy of the temporal scalp region is of interest because of the surgical corollaries. The additional presence of the temporalis muscle and the investing fascia, the deep and superficial layers adds a level of complexity. Approaches to the zygomatic arch from the bicoronal route dictates a knowledge of this anatomy to avoid injury to the temporal branch of the frontal nerve. Division of the superficial layer of the deep temporal fascia at a point approximately 2 cm superior to the arch and a traverse through the superficial temporal fat pad will provide access to the arch, deep to the plane of the temporal branch that innervates the frontalis muscle.
The scalp is richly vascularized, setting the table for design of soft-tissue flaps of generous length and width proportions if oriented properly. The principal arterial supply is the supraorbital arteries anteriorly, the superficial temporal arteries laterally, the occipital, and to a lesser degree, the posterior auricular arteries posterior. The paired supraorbital nerves of the ophthalmic division of cranial nerve V provides sensation to the forehead and anterior one-half of the scalp. The greater occipital nerves, branches of C2, are the sensory innervation of the posterior scalp. The temporal region is innervated by the auriculotemporal nerve, a branch of the mandibular division of V.
17.2.2 Etiology of Scalp Defects
The causation of scalp defects falls into one of three of the usual broad group: congenital, posttraumatic, and oncologic.
17.2.3 Congenital Defects
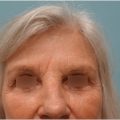
The principal presentation of scalp defects of congenital etiology is aplasia cutis congenital. 1 , 2 Other causes such as giant nevus and sebaceous nevus are discussed under the oncologic section. Aplasia cutis congenita, a rare condition (1 in 10,000 births), is absence of skin, subcutaneous tissue, and in approximately 25% of the cases, skull and dura as well. An autosomal dominant with variable penetrance inheritance pattern has been described, although many, if not most, cases appear sporadic in nature. Treatment and the urgency of same hinges on size and severity (see “Treatment” section). 3
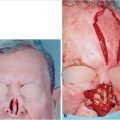
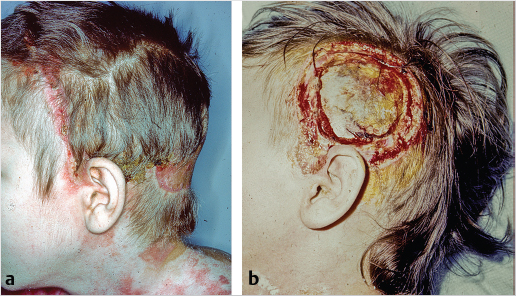
Another set of congenital soft-tissue entities is melanocytic and sebaceous nevi. Both are of a treatment concern because of the potential of malignant conversion. Estimations vary but melanoma in giant congenital melanocytic nevi, GCMN, and basal cell carcinoma within sebaceous nevi has been described (Fig. 17‑2). The nature of the pathology will dictate the reconstructive approach to oncologic defects. Certainly, in instances in GCMN, the parents will have both aesthetic and cancer conversion concerns.
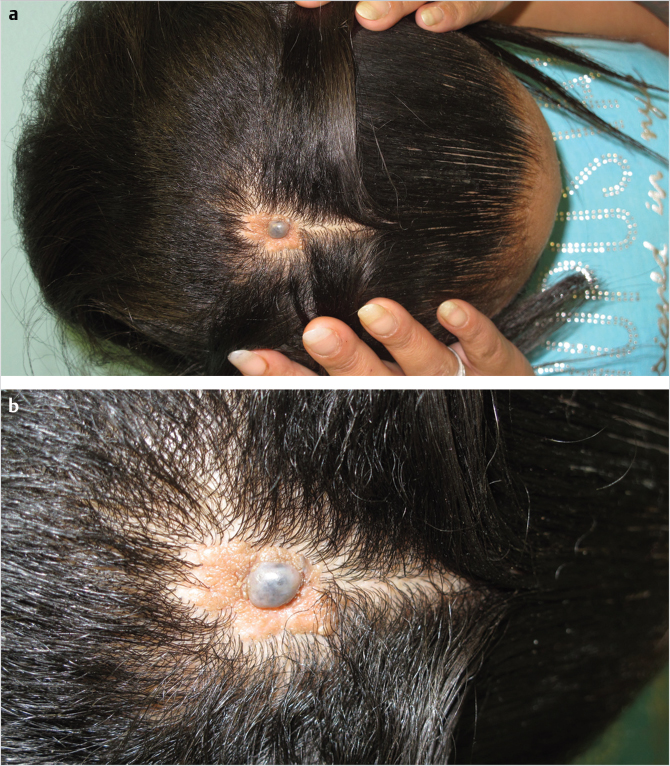
17.2.4 Traumatic
Although the possible causes of posttraumatic scalp defects are protean, the vast majority of cases are secondary to avulsive loss, direct contusion and loss, or burn injury, of which a significant portion are from electrical burns. Avulsive scalp injuries, particularly when total or subtotal, occur either at the level of pericranium for the reasons cited in the “Anatomy” subsection, but frequently and unfortunately result in loss of pericranium or periosteum as well with significant treatment implications (Fig. 17‑3).
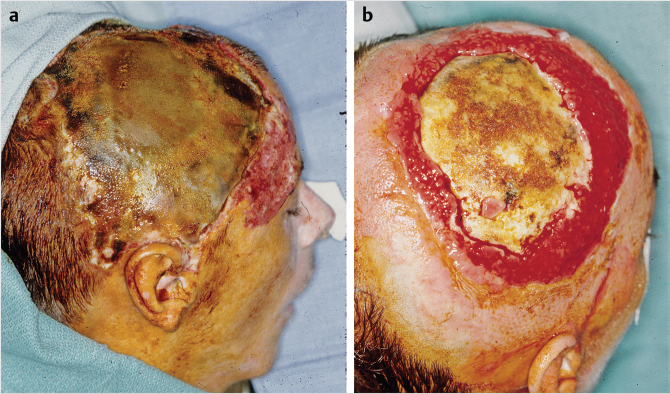
Similarly, although flame burns may be full thickness and yet spare subcutaneous tissue, electrical burns or injury, particularly high voltage, may destroy not only all layers of the scalp but perhaps also may involve cranial bone, again with a different set of treatment implications.
17.2.5 Oncologic
The most frequent scalp reconstructive setting for most surgeons will be oncologic. Mentioned earlier were the benign conditions of congenital melanocytic nevus and sebaceous nevus. Any skin tumor, benign or malignant, drawn from the lengthy list of benign and malignant skin and soft-tissue neoplasms, can occur on the scalp. In addition, the treatment of intracranial neoplasms may have scalp reconstructive connotations. The occurrence of postcraniotomy scalp necrosis and/or bone flap infection may require intervention by the reconstructive surgeon. In any oncologic setting, reconstructive needs may be rendered more complex by the presence of radiation damage from prior radiotherapy, prior incisions and scars, and, of course, the extent of the tumor.
17.3 Treatment
The assessment in preparation for reconstruction of any defect, present or planned, requires adherence to a logic framework to ensure as much as feasible a successful result. One such framework is the Here–There concept 4 that elicits analysis of the defect, anatomically and functionally (“Here”), the forces responsible and the desired objective (“There”), and finally the treatment plan to arrive at the objective. Implicit in each of the treatment scenarios described below is the use of this framework.
17.3.1 Here–There Operative Planning
Where is Here (What are the components, functional and structural, of the defect?)
How did we get There? (What were the forces, traumatic, oncologic, congenital responsible for the defect?)
Where is There? (What are the reconstructive needs?)
So do we get There? (Treatment or operative plan including steps and sequences.)
How do we want to get There? (A mutual decision-making process of patient and surgeon.)
17.3.2 Congenital
As outlined earlier, management of aplasia cutis congenita pivots on the magnitude and severity of the defect from simple to complex. The simplest of presentations, a defect of 2 to 4 cm2 skin can only be managed nonoperatively with dressings. Larger defects (Fig. 17‑4) require a more aggressive approach, particularly in the absence of bone and the presence of exposed dura since the described complications of hemorrhage from exposed intradural sinuses as the sagittal or large dural veins can result in a fatal outcome. In the absence of coverage, the thin membrane becomes desiccated and an eschar forms, the debridement of which can result in massive bleeding. Contamination of the cerebrospinal fluid can result in meningitis and intracerebral infection. Coverage with split-thickness autografts, with or without a preliminary allograft, does protect the underlying dura and brain. The neonate has the osteogenic capability to generate new bone to heal the osseous defect. The negative aspects of the use of skin grafts include the difficulty and hazards of dissection and excision in preparation for eventual cranioplasty if needed. In addition, some concerns exist that the contractile nature of the grafts does not permit normal dural and brain growth. 3
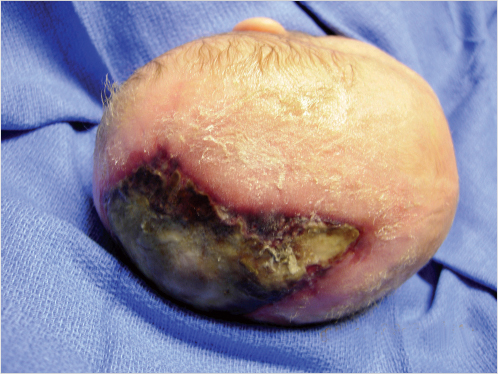
Although described earlier, the use of local flaps would seem fraught with hazard. In addition to the concerns about inevitable blood loss and operating time on a neonate without good temperature regulation and physiologic homeostasis of an older infant, an abnormal vascularity of the intact scalp may exist, the result of an in utero epithelialization of perhaps an even larger defect.
An intriguing solution to this treatment dilemma is the use of a dermal substitute. The placement of Integra once incorporated in 2 to 3 weeks, followed by removal of the silicone layer and placement of a thin split-thickness skin graft has been described in a case report and has the potential to obviate the shortcomings of skin graft alone. 5 Perhaps, a full-thickness autograft could accomplish the same objective with a single stage but with a considerable size donor site.
17.3.3 Traumatic
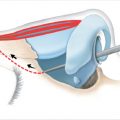
Again, the analysis begins with the nature of the defect. Intact periosteum permits simple wound closure with split-thickness skin graft (STSG) to set the stage perhaps for definitive reconstruction with tissue expansion. The absence of periosteum implies the usual necessity of provision of full-thickness coverage with vascularized tissue, either pedicled or by microvascular free flaps. An exception is the creation of a vascularized bed by decortication of the skull and exposure of the cancellous layer with delayed skin grafting after stimulation of a granulating surface, hastened by the use of negative wound pressure therapy, negative would pressure therapy (NWPT) (Fig. 17‑5). 6
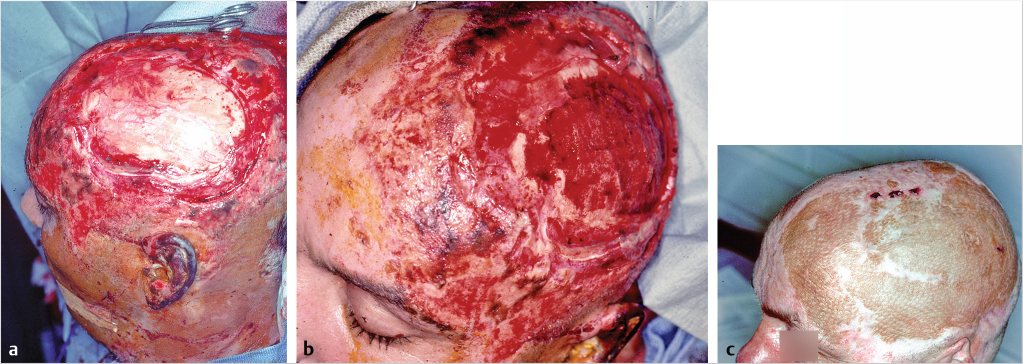
The long-term durability of split-thickness skin grafts placed on decorticated bone is of concern, since late ulceration and wound breakdown is a frequent sequela, perhaps because of the thin layer of dermis.
The employment of a dermal substitute in the reconstructive plan may, as in the management of the aplasia cutis congenita defect, provide a course between the two horns of the dilemma. 7 Both Integra, a dermal substitute composed of bovine collagen and ground substance derived from shark, and Alloderm, an acellular human dermal matrix, are the two most widely used dermal substitutes (Fig. 17‑6).
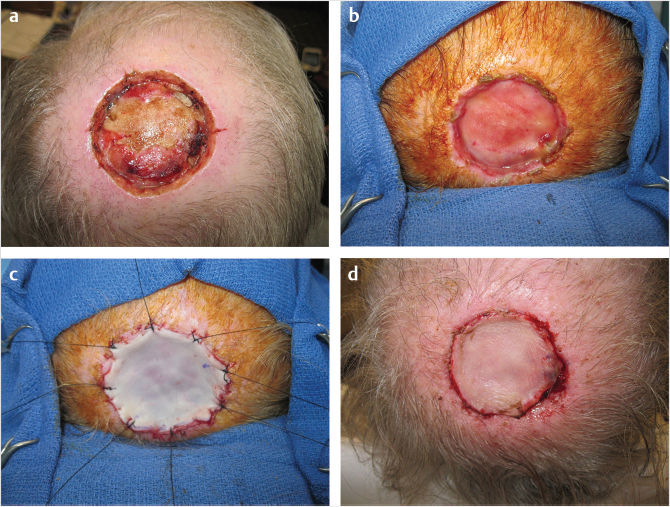
The incorporation of both requires vascularization and the use of NWPT appears to be of benefit in this process. The final wound closure still requires a split-thickness skin graft, albeit considerably thinner than without the use of a dermal substitute. In addition, infection beneath the dermal template can occur, an enhanced probability in the presence of contamination and certainly with potential dire consequences.
The one set of circumstances that prescribes a discreetly different plan is an electrical injury of scalp and skull. In this instance, injury of scalp and skull, none of the above options of acellular dermis plus skin graft are available because of the full-thickness burn of the bony skull. In these circumstances, vascularized coverage, pedicled or free flaps, will be necessary. If performed in the early postinjury period before colonization of the bone, the evidence suggests that the devitalized bone can be regarded as an in situ graft with a perfect fit that will be reconstituted by the process of bone graft take or “creeping substitution” (Fig. 17‑7). 8
The next rung on the reconstructive ladder is the use of local and regional flaps, discussed in the following section. The principles would still be applied in posttraumatic defects, if so indicated.
17.3.4 Oncologic
Extensive excisions of lesions as congenital melanocytic nevi are not amenable to direct primary closure and may be managed by serial tissue expansion (Fig. 17‑8). Patients most often present in early childhood, attended by parents with concerns about malignant transformation as well as the inherent aesthetic problem. The elective setting allows a cyclic approach of expansion, excision, and repeat expansion and excision.
As mentioned earlier, virtually any skin-soft tissue tumor that occurs elsewhere can present on the scalp. An additional dimension is the reconstructive deficit that involves the treatment of intracranial neoplasms, complicated by skin flap necrosis and/or infected bone flap. Both scenarios, resective defects created by excision of skin-soft tissue malignancies and the complications of treatment of intracerebral tumors, may be rendered more complex by the presence of previously irradiated tissue.
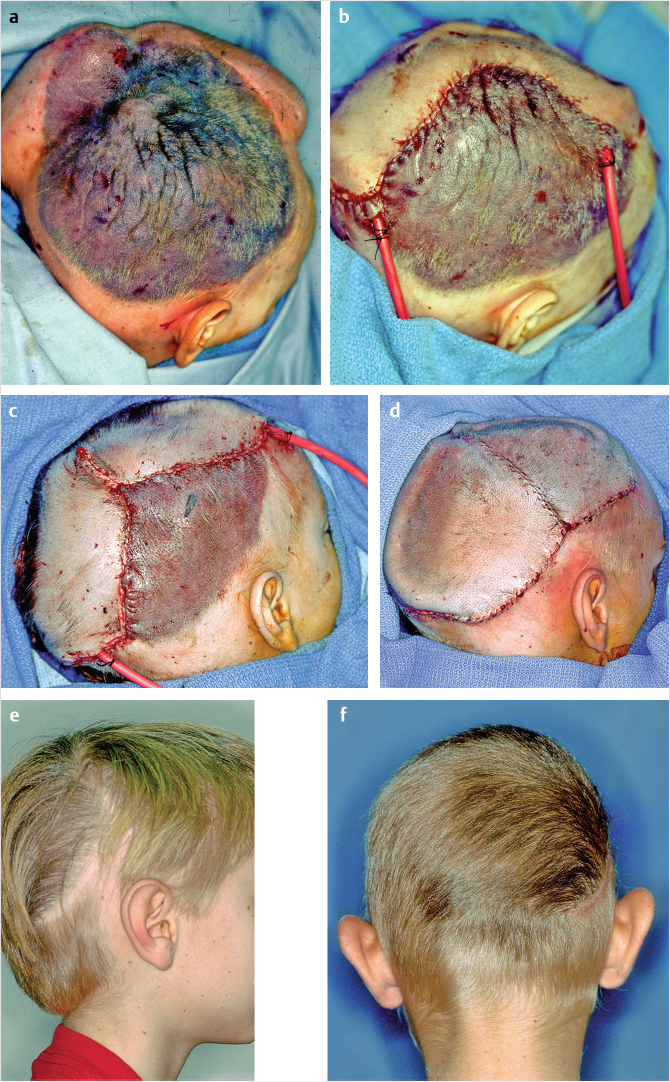
Defects of the scalp larger than 2.5 to 3.0 cm may be difficult to close without extensive undermining and the excessive tension can produce a widened, alopecic scar. 9 Local flaps such as a yin and yang design still permit primary closure of the donor site but larger, rotation-style flaps do not and a commitment to a skin-grafted donor site is unavoidable. Advancement, transposition, or rhomboid flaps perform poorly on the scalp because of the lack of skin laxity. An option for vertex defects, even of a considerable size, is the Orticochea flap tissue rearrangement approach, mobilization of the entire scalp by division into three flaps, similar to peeling a banana (Fig. 17‑9). 10
Two paired flaps are each based on their respective superficial temporal artery, the posterior flap on the occipital arteries. Descriptions of the Orticochea flaps universally include scoring of the galea. Galeal scoring can be problematic from two aspects: one, the additional length obtained is modest and because the vasculature lies in the immediate superficial plane to the galeal aponeurosis, the blood supply can be damaged, creating a tradeoff of length versus ischemia. 9 If performed, the design is one of cross hatching to create the maximal relaxation (Fig. 17‑10).
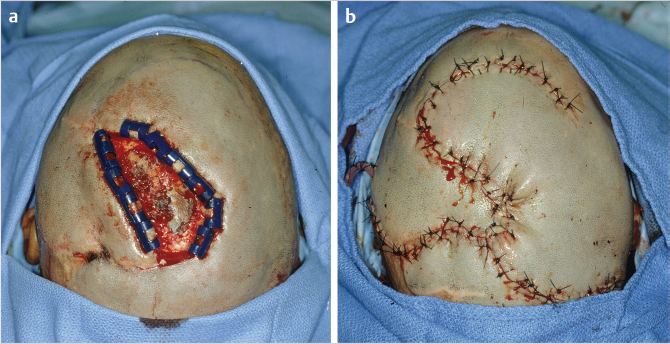
In the author’s experience, galeal scoring is bloody and of little advantage. If a scalp rotation flap is selected, a compulsive and carefully considered plan is mandatory. The elements include incorporation of a flap shrinkage factor (25%) into the defect dimensions, determination of the length and pivot point by simulation with gauze or suture, and orientation of the flap base to include a named blood supply (Fig. 17‑11).
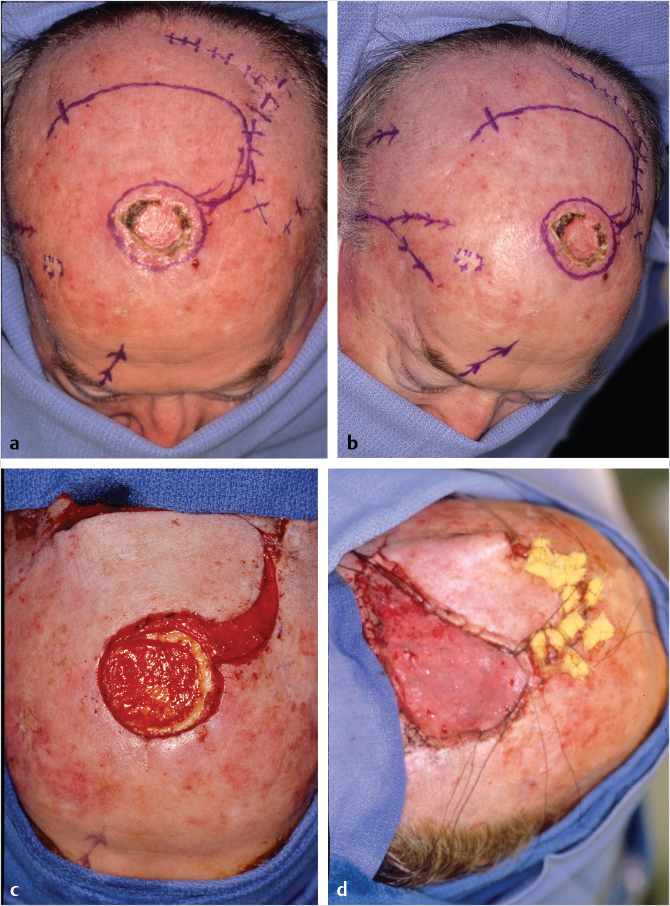
The scalp, because of the convexity of the skull and absence of laxity, dictates the design of larger flaps than what the novice may anticipate. A large, unsightly dog ear will occur at the site of the “standing cone” that begs immediate release/excision. To do so will compromise the vascular supply and with time the dog ear will shrink remarkably.
The above scenarios assume the absence of prior irradiation. Prior irradiation to skin-soft tissue, particularly with larger radiotherapy portals, will usually imply regional pedicled or free flap coverage if the dimensions of the reconstructive defect, present or proposed, is of substantial size.
The presentation of a previously irradiated brain tumor patient to the reconstructive surgeon is most commonly as wound dehiscence/infection with or without infection of the underlying bone flap. Some clinical judgment is necessary in the estimation of the degree of radiation injury to the scalp. Review of the radiotherapy plan can be helpful, but the essence of the judgment is the vascularity or lack of same or proposed wound closure. The tautness of the scalp skin, the magnitude of radiation-induced alopecia, and the size and location of the defect all play a role. Wound dehiscence only without evidence of infection can be managed with wide undermining and advancement to close relatively small defects of 2 to 3 cm. More extensive defects that include debridement of necrotic tissue will require a rotational flap plus STSG. Management of the underlying cranial bone is a separate topic, but the treatment will be determined by the extent of infection. Localized osteomyelitis secondary to hardware alone can be managed with removal of the plates and screws and limited debridement, while more extensive infection requires removal of the bone flap and late secondary reconstruction.
Larger postoncologic defects, greater than 50% of the scalp, often cannot be satisfactorily addressed with the above methods (Fig. 17‑12).
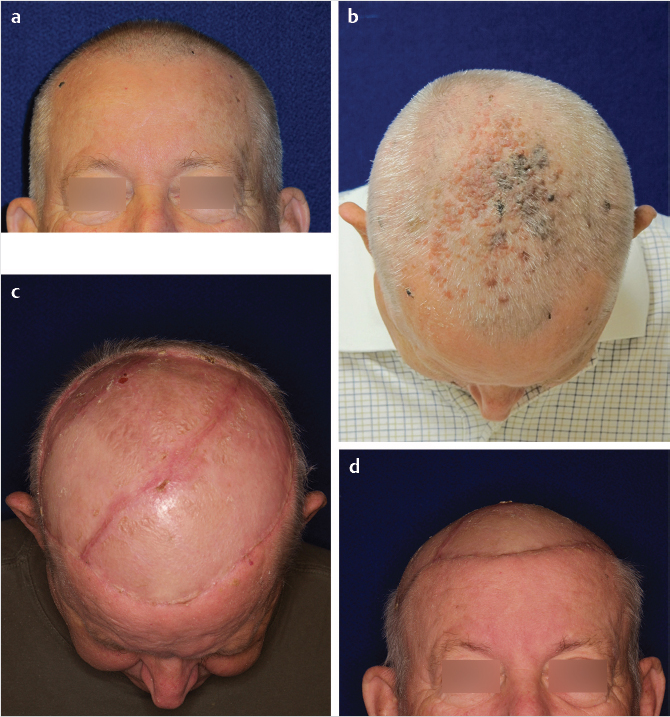
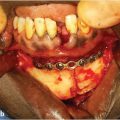
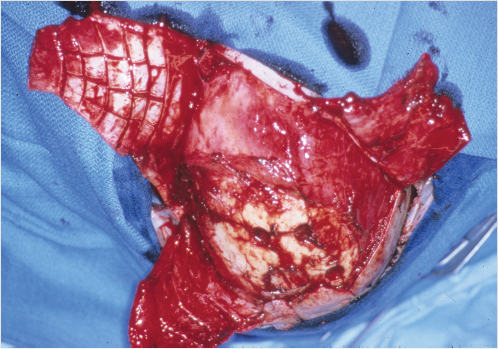
If the scalp has been previously irradiated for skin-soft tissue malignancy, even considerably smaller defects will compel the use of regional or distant flap coverage, although the use of Integra and delayed skin graft has been described for smaller irradiated scalp defects. 11 The use of Integra has been described for the coverage of a total scalp loss in the pediatric patient. 12 The only scalp location for which the choice of a pedicled regional flap can be entertained is the temporal region, for example, after a temporal bone resection, reconstructed with an extended pectoralis major flap (Fig. 17‑13).
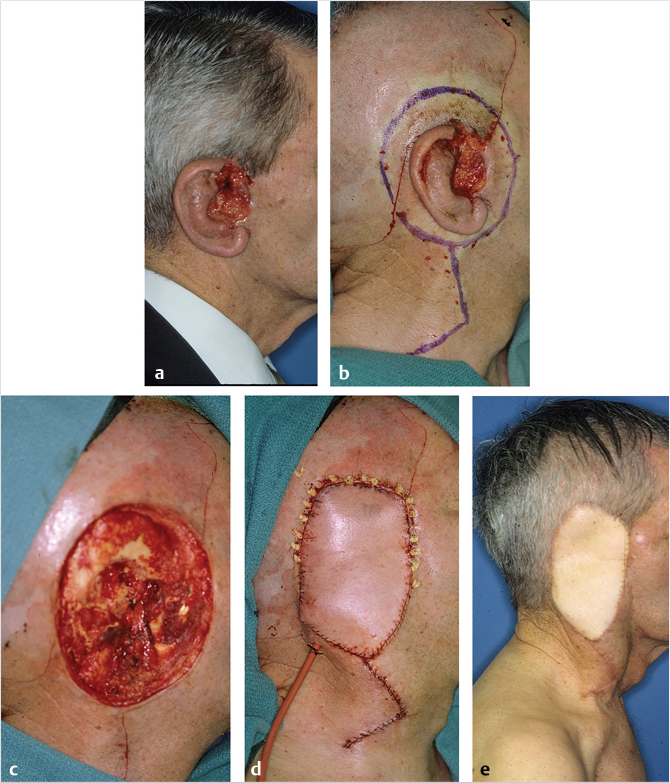
The inferior occipital scalp is amenable to a posterior trapezius perforator flap, perfused by the dorsal scapular artery. More often, the challenge is one of previously treated, often inadequately, because of positive margins and/or bone involvement with the employment of postoperative radiation in an adjunct setting. 13 The hostile environment created by irradiation, recurrence-persistence of the neoplasm, and possibly contamination, if not of limited size and within the inferior or occipital region, all point to the indication for microvascular flap coverage.
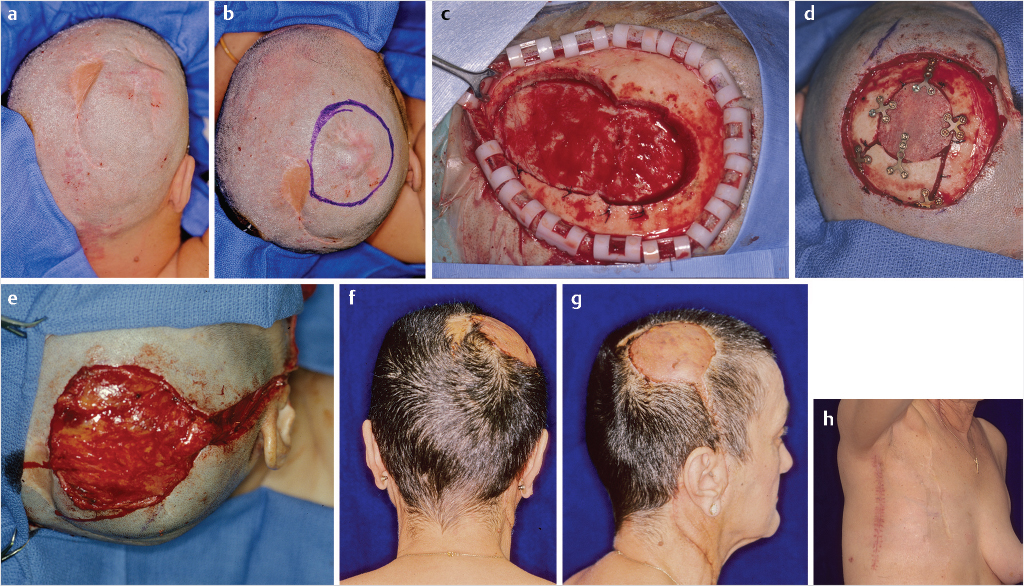
Although an array of free flaps are available, essentially three (and possibly a fourth) have been most frequently selected and described. Large defects, for example, a subtotal scalp excision, covered with a latissimus muscle and skin graft, may be ideal from several perspectives: size, ability to tailor the muscle, and the value of well-vascularized muscle in a contaminated field. A skin island may be included if the size of the defect permits primary closure of the back and patient is not obese. The radial forearm as a fasciocutaneous flap provides, as does the latissimus, a pedicle of adequate length and is suitable for smaller, medium-size defects. The anterolateral thigh flap does provide a skin island, has a long and sizable pedicle, and offers, as does the radial forearm, the opportunity for a two-team approach, a harvest simultaneously with independent preparation of the recipient site. The fourth flap possibility is the scapular or parascapular, both fasciocutaneous flaps in nature. Similar to the latissimus, a patient position change may be necessary.
The most readily accessible recipient vessels are the superficial temporal artery and vein. At times, the vessels, particularly the vein, may be friable and difficult to use. The next echelon is within the neck, access to the superior thyroid or facial vessels. More extreme situations may require the use of vein grafts to access undamaged, nonradiated recipient vessels (Fig. 17‑14).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree