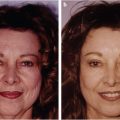
Abstract
This chapter explores in detail the range of considerations, historical perspectives, assessments, and surgical and nonsurgical options involved in offsetting facial and neck aging. Changes that occur due to facial aging, skin laxity, sun damage, lipoatrophy, soft-tissue descent, and potential skeletal changes are covered, along with effective remedies for restoring balance and harmony to the facial parts. Treatment options for every region of the face and neck are reviewed, including skin resurfacing, soft-tissue augmentation, chemodenervation, and surgery. Operative techniques—incisions, facelifts, skin management, etc.—are listed, discussed, and illustrated. Particular emphasis is paid to the risks involving the facial nerve branches. Seasoned advice on postoperative care and complications conclude the chapter.
22 Face and Neck Rejuvenation
22.1 Goals and Objectives
Recite the landmark contributions which define the historic evolution of face and neck rejuvenation surgery.
Describe the three-dimensional surgical anatomy of the SMAS, retaining ligaments of the face, facial nerve branches, safe dissection planes, and danger zones.
Outline the basics of clinical evaluation of face and neck rejuvenation patients.
Differentiate short-scar-limited SMAS interventions from traditional surgical techniques.
Explain the principles and steps of the MACS lift, the lateral SMASectomy, the extended SMAS, and composite rhytidectomy.
Summarize the potential complications and their management.
22.2 Patient Presentation
22.2.1 Clinical Evaluation
Given the plethora of surgical and nonsurgical options, a comprehensive clinical assessment is crucial to guide treatment strategies. Particular attention is given to the various aspects of facial aging including skin laxity, sun damage, lipoatrophy, soft-tissue descent, and potential skeletal changes.
22.2.2 Aesthetic Facial Evaluation with a Cephalometric Perspective
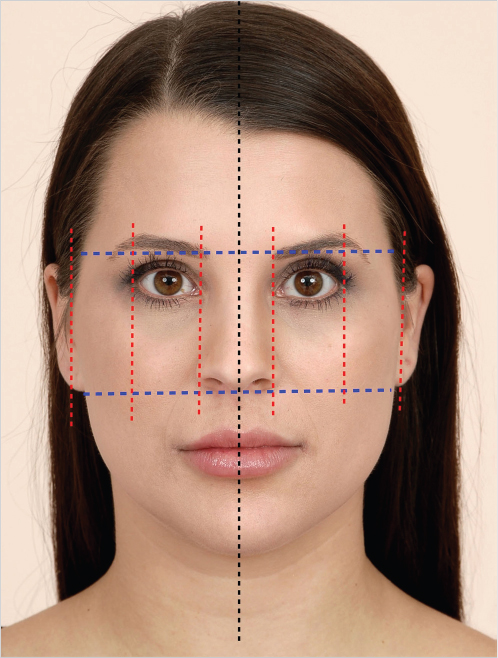
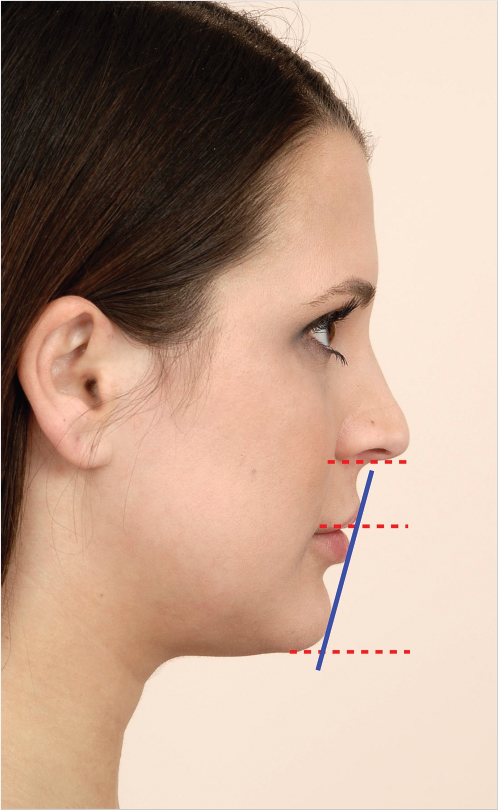
This assessment should analyze age-related deformities in the context of soft tissue and skeletal cephalometrics. The goal is to determine balance and proportions (harmony) of different facial parts, establish limitations of soft-tissue procedures, and identify potential adjunctive skeletal interventions (e.g., genioplasty). 1 , 2 The patient should be assessed with the head in “natural” position (straight forward gaze; the Frankfort horizontal is generally parallel to the ground in that posture) and with the eyebrows and lips relaxed. 1 Any deviation results in soft-tissue distortion that can lead to inaccurate assessment. 1 Although assessment should be three dimensional, frontal aesthetics hold prime significance since this is how patients picture themselves. 1
Upper Third
Aging is associated with (1) hairline recession, (2) transverse forehead rhytids, (3) vertical glabellar lines, and (4) eyebrow ptosis.
Middle Third
Aging is associated with (1) blepharochalasis, (2) ptosis, (3) lower lid laxity ± ectropion, (4) lower lid fat bulge, (5) Crow’s feet, (6) tear trough deformity, (7) palpebromalar groove, and (8) malar mound.
The Lower Third and Neck
Aging is associated with (1) nasolabial fold bulge, (2) nasolabial crease deepening, (3) marionette lines, (4) exaggeration of the labiomental fold, (5) perioral wrinkles, (6) loss of the cervicomental angle, and (7) platysmal bands.
22.3 Preparation for Surgery
Like many other aesthetic procedures, rigorous patient selection is crucial. Medically unfit patients may benefit from alternatives such as botulinum toxin and fillers. A thorough screening for hemorrhagic diathesis is essential to avoid bleeding complications. 3 Antiplatelets and vitamin K suppressors should be stopped 1 to 2 weeks prior to surgery. 3 A medical consult is occasionally necessary to document fitness for surgery, the feasibility of drug cessation, and to suggest alternatives. 3 Smoking should be stopped for at least 4 weeks in advance. 4 , 5 Finally, identifying patients with unrealistic expectations, psychiatric disease, psychosocial impairment, and body dysmorphic disorder is also critical. 6 Chronological age does not seem to be an independent risk factor for higher complications in rhytidectomy surgery. In fact, a study demonstrated that elderly patients (older than 65 years) were not at a higher risk for complications, compared with a younger control group (younger than 65 years), provided proper preoperative screening is undertaken. 3
22.4 Treatment
Face and neck rejuvenation involve multifaceted clinical treatment of cervicofacial aging using skin resurfacing, soft-tissue augmentation, chemodenervation, and surgery. The objective is to positively modify skin, cervicofacial contour, and restore volume. Safe and effective treatment requires precise knowledge of anatomy and technique. Understanding anatomical planes, the SMAS’s (Superficial Muscular and Aponeurotic System) relations to the facial nerves, and retaining ligaments is critical to safe sub-SMAS surgery. No versatile surgical technique can fit all needs. Rather surgeons should modify surgery to meet the specific requirements of their patients. While the MACS (minimal access cranial suspension) lift and the lateral SMASectomy are examples of widely used short-scar-limited SMAS interventions, the extended SMAS and the composite rhytidectomy represent prototypes of conventional sub-SMAS procedures that suit patients with significant laxity and advanced signs of aging. Lipo-filling is assuming growing significance as a regenerative and volume restoring adjunct. Nonsurgical interventions, especially botulinum toxin and soft-tissue fillers, are rising as standalone and ancillary interventions. Finally, skin-resurfacing techniques, like peeling and LASER, further optimize the surgical outcome. This chapter focuses on surgical treatment.
22.5 Historical Perspectives
Prior to Skoog’s contributions, 7 facial rejuvenation was addressed with conservative skin resection, and limited subcutaneous undermining. Skoog reported the elevation of a cervicofacial flap deep to the superficial fascia, lifting the platysma in the neck, and the “superficial fascia” in the face, opening a new era for facelift surgery. 7 This paved the way for defining the applied anatomy of the SMAS by Mitz and Peyronie in 1976. 8 Furnas then outlined the retaining ligaments of the cheek in 1989. 9 Stuzin et al further highlighted the importance of the masseteric ligaments, and the relationship between superficial and deep fascia, defining safe planes and danger zones of sub-SMAS dissections. 10 These anatomical studies stimulated different “SMAS-platysma modifications” and deeper tissue repositioning techniques in an attempt to identify procedures that are more durable.
Owsley described the SMAS-platysma facelift, which formed the foundation of SMAS flap techniques. 11 , 12 , 13 The subperiosteal approach was presented by Psillakis et al and Ramirez et al who explored deep tissue repositioning in this anatomical plane. 11 , 12 , 13 , 14 , 15 Hamra took Skoog’s operation a step further, describing first the deep plane facelift, 16 followed by the composite facelift, 17 , 18 , 19 as well as different modifications of the lower eyelid myofascial complex. 20 , 21 , 22 , 23 , 24 , 25 Barton reported his modification of Skoog’s procedure which became known as the “high SMAS technique.” 26 , 27 , 28 , 29 While the refinements by Hamra and Barton adopted a composite dissection, where skin and SMAS are elevated as one unit, other SMAS-based techniques employed a bilamellar dissection, where the SMAS is undermined and redraped independently in a vertical direction, while the skin vector is more horizontally positioned. 18 , 28 These latter procedures described variable SMAS flap designs, for example, the extended SMAS. 12 , 18 , 28 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37
In the neck, Connell combined comprehensive lipectomy with full platysma transection and mobilization. 38 Various other platysma-modifying techniques were also proposed. The most well-known of these being the “corset platysmaplasty” described by Feldman in 1990. 39 Knize, Zins, and Feldman further highlighted the utility of the isolated anterior approach to improve the neck in selected patients. 40 , 41 , 42 , 43 A trend toward less invasive techniques in facial rejuvenation was initiated by the endoscopic brow lift, intended to address drawbacks of conventional open brow lift surgery. 44 “Short-scar-limited SMAS” procedures, including the lateral SMASectomy and the MACS lift, then followed. 45 , 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 , 55 These “less invasive” procedures were designed to serve patients with minimal laxity seeking shorter recovery and less surgical risk. 48 , 49 , 50 , 53 , 55 Recently, more emphasis has been placed on ancillary facelift techniques including volume restoration with lipofilling based on Coleman’s pioneering work. 56 , 57 Finally, the evolution of botulinum toxin further allowed myogenic manipulation of wrinkles, and brow position, and soft-tissue fillers enabled wrinkle and volume adjustment in less surgery-committed patients. 58 , 59
22.6 Anatomical Considerations
“No amount of improvement in facial contouring is worth an injury to the facial nerve.” 35
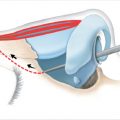
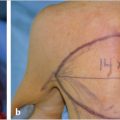
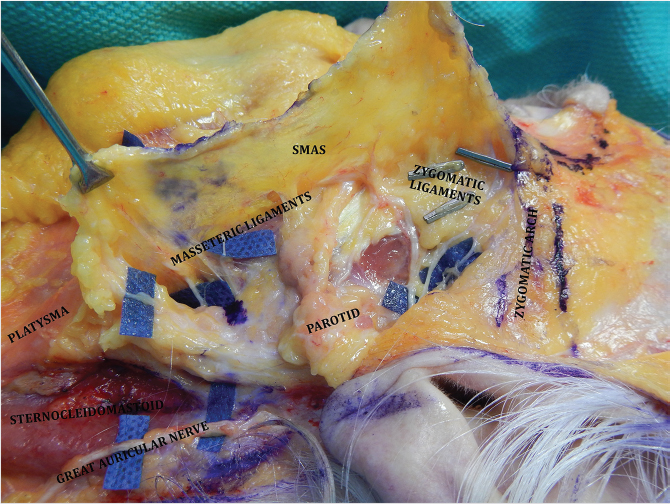
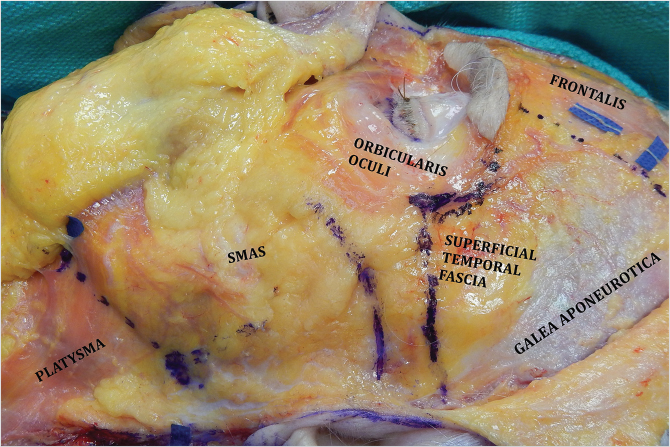
The SMAS-platysma-superficial temporal fascia plane (Fig. 22‑1).
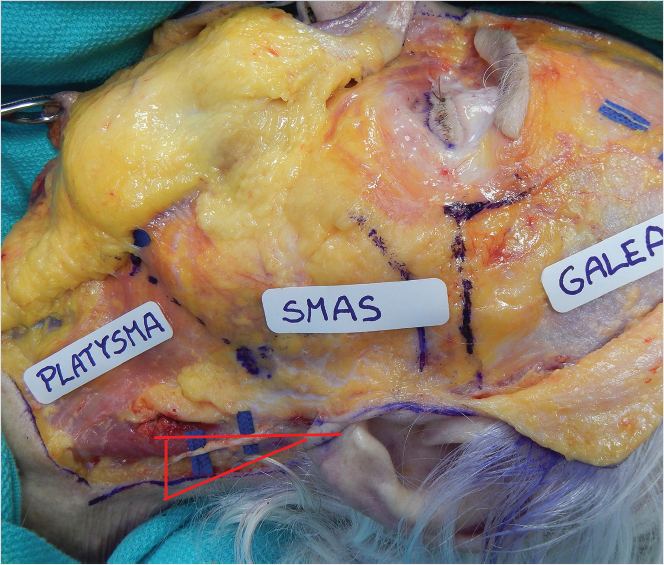
The SMAS is a subcutaneous fibromuscular structure continuous with the platysma in the neck and the superficial temporal fascia in the temporal region. 8 Further cephalad it lays in the same plane as the frontalis and galea aponeurotica. 8 More anteriorly it invests the muscles of facial expression. 10 , 27 Posteriorly it is fixed to the thick parotid fascia in addition to the thick sternocleidomastoid fascia. 8 , 10 The facial nerve branches are invariably located deep to this layer (Fig. 22‑2). 8 , 10 The SMAS is muscular caudally, where it merges with platysma, but becomes progressively aponeurotic (fibrous) more cephalad (Fig. 22‑1). 8 Posteriorly, it blends with the parotid fascia, forming a thick layer. 8 More anteriorly, it thins out and becomes more tenuous. 8 , 10
22.6.1 The Support System of the Face (Retaining Ligaments of the Face)
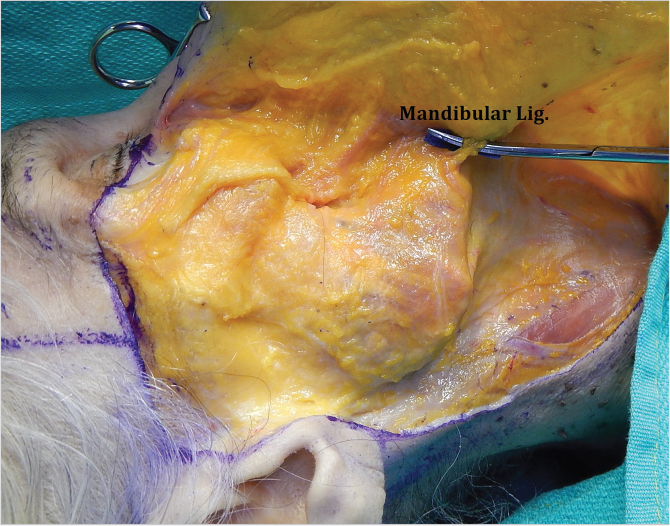
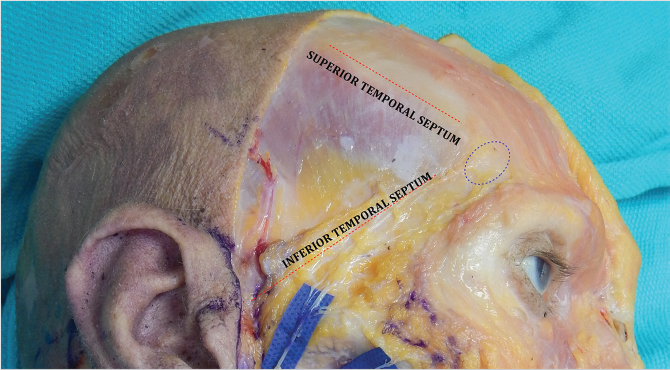
A series of retaining ligaments or more recently termed “SMAS fusion zones” supports the facial soft tissues against gravitational forces. 60 , 61 , 62 Their constant location and predictive nature with relation to facial nerve branches renders them surgically significant (Fig. 22‑2). 40 , 41 , 42 , 63 , 64 , 65 , 66 The classic plastic surgery literature has described this support system as osteocutaneous or “true” ligaments such as the zygomatic and mandibular ligaments, or false ligaments. 9 , 10 , 63 , 67 True ligaments extend from bone to dermis passing through the SMAS and arborizing to attach to dermis, similar to the trunk of a tree passing from bone and arborizing after passing through the SMAS to insert into dermis. 33 Arborization allows an enhanced effect on skin superficially. 33 Myofascial or false ligaments, such as the masseteric and parotid cutaneous ligaments, are fibrous structures fixing deeper fascial layers to overlying superficial soft tissue. 10 Moss et al, for greater precision, advocated a distinction between (1) True ligaments which are cylindrical fibrous structures holding the superficial layer (SMAS and extensions) to deeper periosteum or deep fascia, and are mostly located in the mid-, and lower face. 66 The zygomatic, masseteric, and mandibular ligaments (Fig. 22‑2, Fig. 22‑3) constitute the best-known examples of these. 66 (2) Septa, on the contrary, are fibrous walls extending from deep fascia or periosteum to the superficial layer and are basically found in the temporal and periorbital region. The superior and inferior temporal septa exemplify the prototypes of these (Fig. 22‑4). 66 (3) Finally, adhesions represent areas of low-density fibrous or fibrofatty adhesions between the superficial layer and deeper tissues. The temporal and supraorbital ligamentous adhesions are models of this latter configuration (Fig. 22‑4). 66 The mobility of the superficial layer is generally dictated by the type of underlying support, with true ligaments providing greatest mobility, septa allowing only motion perpendicular to their line of attachment, and adhesions restricting movement in all directions. 66 Furthermore, septa and adhesions represent areas of extended bonding between superficial and deep fascia generally requiring wider dissection to obtain satisfactory repositioning. 66
Recently, Pessa described this anatomy as SMAS fusion zones rather than ligaments. 60 In his analysis, these structures exist as bilaminar membranes extending between superficial and deep fascia, and constitute the boundaries of the fat compartments and the anatomical spaces of the face (e.g., the buccal space). 57 , 60 Blood vessels, nerves, and lymphatics are stabilized by traveling within these fusion zones. 60 Between superficial fascia (SMAS) and skin, these membranes further continue as unilaminar structures limiting the superficial fat compartments. 60
The disparity in nomenclature and variation in reporting have added significant confusion to the understanding of these anatomical structures. 60 , 61 , 62
The Temporal Region
The Superior Temporal Septum
This coincides with the superior temporal fusion line where the galea and periosteum adhere to bone just superior and medial to the deep temporal fascia (Fig. 22‑4). 66 It starts superior to the temporalis muscle and arches downward toward the outer corner of the superior orbital rim, defining the transition of the superficial layer from temporoparietal fascia to galea and frontalis. 66 Its lower end is expanded and forms the temporal ligamentous adhesion. 66 The septum forms the upper border of the superior temporal compartment, whose inferior boundary is formed by the inferior temporal septum. Dissection can proceed rapidly in this anatomical space since no vital structure is encountered. 66
The Temporal Ligamentous Adhesion
This is a triangular fibrous condensation 1 cm above the craniolateral corner of the orbital rim at the juncture of frontal, temporal, and periorbital areas (Fig. 22‑4). 66 Three retaining ligaments radiate from it: the supraorbital ligamentous adhesion medially, the superior temporal septum superolaterally, and the inferior temporal septum inferolaterally. 66
The Inferior Temporal Septum (Orbicularis-Temporal Ligament)
This is an area of fusion between the superficial temporal fascia (temporoparietal fascia) and the superficial layer of deep temporal fascia extending from the craniolateral corner of the orbicularis oculi to the external acoustic meatus which constitutes this structure (Fig. 22‑4). 66 It divides the temporal compartment into superior (see earlier) and inferior temporal compartments; the latter is further bordered caudally by the zygomatic ligaments. 66 This space is surgically significant for housing the frontal branches of the facial nerve, the zygomaticotemporal nerve, and the sentinel veins (Fig. 22‑5). 66 The frontal branches of the facial nerve travel medial and parallel to the inferior temporal septum. The septum thus acts as a warning sign during craniocaudal dissection in the temporal region, alerting surgeons to stay strictly on top of the superficial layer of the deep temporal fascia (or deep to it) to avoid nerve injury (Fig. 22‑4). 63 , 66
The Periorbital Area
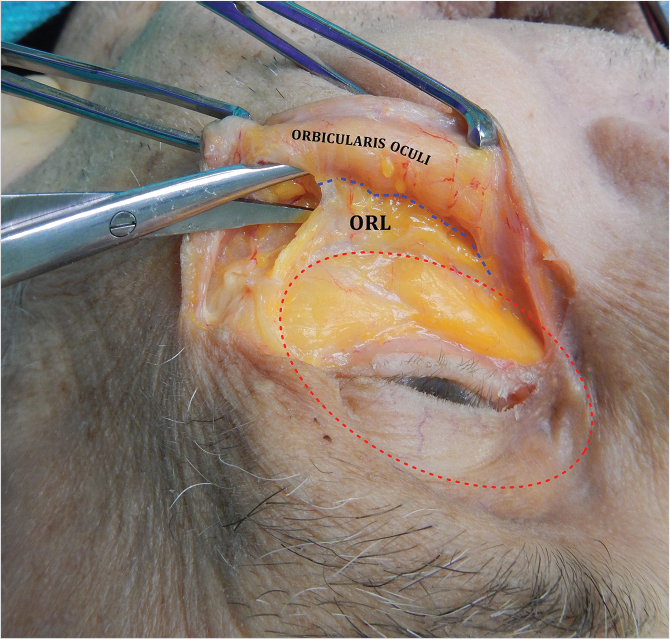
The Orbicularis-Retaining Ligament (Orbitomalar Ligament)
The anatomical delineation of this structure stems from the clinical observation of constant strong attachments between the orbicularis oculi and the inferolateral orbital rim that often need to be released for adequate periorbital mobilization. 66 , 67 , 68 , 69 The orbitomalar ligament (ORL) is an osteocutaneous ligament arising from the periosteum of the orbital rim and traversing the orbicularis oculi to insert into the dermis of the lid–cheek junction (Fig. 22‑6). 68 , 70 It is not well defined medially where the orbicularis oculi is tightly adherent to the inferior orbital rim (Fig. 22‑7). 68 The ligament extends circumferentially around the orbital rim, and thus forms the roof of the preseptal space in the upper eyelid, the floor of the preseptal space in the lower eyelid, and the roof of the prezygomatic space in the midface, which is bordered caudally by the medial extent of the zygomatic ligaments. 67 , 68 , 69 Morphologically, the ligament is a bilaminar structure where the cranial leaf is a reflection of the orbital septum, and the caudal leaf a continuation of the fascia covering the preperiosteal fat of the prezygomatic space. 68 , 69 The zygomaticofacial nerve and foramen always lie peripheral to the ORL. 68 , 69 The ORL can be thought of as the leash of a dog running from bone to dermis. The leash is tight medially, loose centrally, and again tight laterally; where it fuses with the lateral orbital thickening or superficial canthal tendon, the laxity of the ligament thus creates a V-shaped structure (Fig. 22‑7). 68 Lysis of the medial orbicularis oculi and varying portions of the ORL have become an essential part of a variety of procedures used to treat the tear trough, palpebromalar groove, and malar mound. 24 , 69 , 71 , 72 , 73 , 74 , 75 , 76
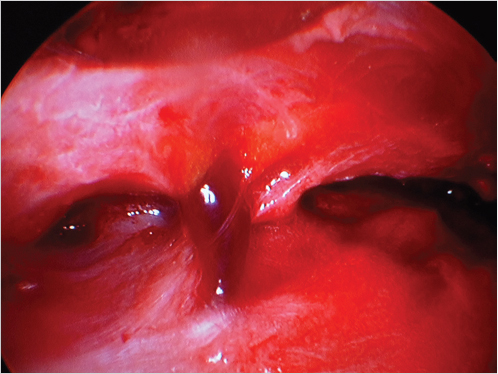
The Cheek Area
The zygomatic ligaments arise from the lower border of the zygomatic arch and from the body of the zygoma. 9 , 10 , 64 They attach directly to the dermis. 9 , 10 , 64 The medial extent of these ligaments forms the floor of the prezygomatic space, while the lateral extent constitutes the lower border of the inferior temporal compartment. 66 , 67 , 69 Furthermore, these ligaments act as the roof of the lateral cheek compartment. 63 The major zygomatic cutaneous ligament lies at the junction of the arch and body of the zygoma. 63 , 64 This ligament is surgically important because when released, the zygomaticus major (ZM) muscle comes into view. 9 , 10 , 35 This marks the end of the sub-SMAS dissection. All sub-SMAS procedures invariably pass superficial or subcutaneous at this point, in order to avoid injury to facial nerve branches (Fig. 22‑8). 9 , 17 , 35 , 37
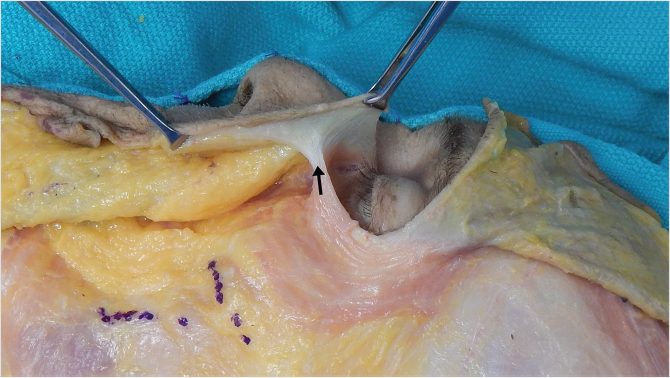
The masseteric ligaments have their origin from the fascia overlying the masseter muscle. Together with the zygomatic ligaments they form a T-shaped configuration (the confluence of the T is at the origin of the ZM muscle). 10 , 33 , 63 , 64 One centimeter inferior to the ZM in the vicinity of the stout upper masseteric ligaments is the sub-SMAS “danger zone,” where care needs to be taken when lysing these ligaments to avoid injury to zygomatic branches of the facial nerve (Fig. 22‑9). 9 , 10 , 63 , 64 The lower masseteric ligaments or zones of fusion separate the premasseteric space from the masticatory space that houses the buccal fat pad. 10 , 60 , 63
The platysma auricular ligament/fascia is the first ligament severed during a facelift. It is formed of fibrous bands connecting the platysma to the parotid fascia, securing it to the preauricular skin. 9 , 10 , 63
The Mandibular Area
The mandibular ligament is an osteocutaneous ligament arising from the anterior third of the mandible, 1 cm above the inferior border. 9 Its presence marks the anterior boundary of the jowl (Fig. 22‑3). 9 , 10 The marginal mandibular branches of the facial nerve are located posterior and approximately 1 cm cranial to it (Fig. 22‑10). 65
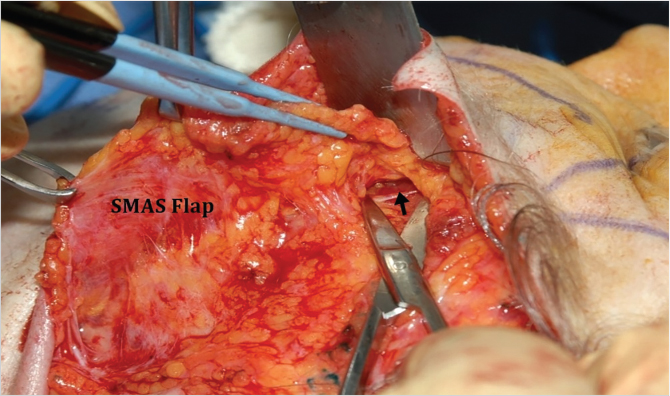
22.7 The Facial Nerve
22.7.1 Beyond the Parotid Gland, Five Groups of Branches Can Be Identified
Frontal (Temporal) Branches
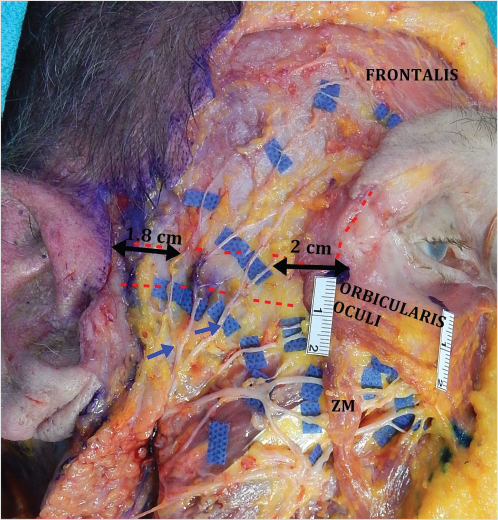
The limited number of communications with other facial nerve branches is thought to account for the higher risk of permanent injury. 77 The classic surface anatomy is the Pitanguy line which extends from 0.5 cm below the tragus to 1.5 cm above the lateral eyebrow. 78 The nerve exists in multiple (2–5) rami that intercommunicate, which may explain the return of function after injury. The branches were found to reside 2 cm posterior to the lateral orbital rim at the level of the lateral canthus. 79 , 80 At the level of the zygomatic arch, the branches run over the middle third approximately 1.8 cm anterior to the helical crus, and 2 cm posterior to the lateral orbital rim (Fig. 22‑11). 79 , 81 In terms of depth, these branches are closely applied to the periosteum of the zygomatic arch, deep to both the SMAS and the parotid masseteric fascia (parotid temporal fascia). 82 Superior to the arch, they travel on top of the superficial layer of the deep temporal fascia. 82 , 83 , 84 Trussler et al and Agarwal et al in separate studies showed the presence of two superficial fascial layers covering the zygomatic arch and the deep temporal fascia. 82 , 84 The first layer is the superficial temporal fascia, which is in continuity with the SMAS. 82 , 84 Deeper to it they showed the existence of another distinct layer (the parotid temporal fascia per Trussler et al, and innominate fascia per Agarwal et al). 82 , 84 The frontal branches travel either deep or within this deeper layer. 82 , 84 This relationship continues for at least 1.5 to 3 cm above the arch from where the nerves start to transition through the superficial temporal fascia to merge with the anterior branch of the superficial temporal artery as they approach orbicularis oculi and frontalis. 82 , 84 Appreciation of this anatomy is important during craniocaudal dissections in the temporal region, and in elevating a high SMAS flap. 82 Surgeons should stay strictly on top of the superficial layer of the deep temporal fascia (or deep to it) elevating the frontal branches with the flap. Two centimeters cephalad to the zygomatic arch, dissecting deeper to the superficial layer of the deep temporal fascia has been advocated to avoid nerve injury. 77 , 82 , 84 In endoscopic brow lift, the sentinel vein (located approximately 0.5 cm lateral to the frontozygomatic suture) defines a danger zone about 10 mm cephalad, where the frontal branch is liable to injury (Fig. 22‑5). 81 A second vein 1.5 cm lateral to the first is occasionally identified. 81
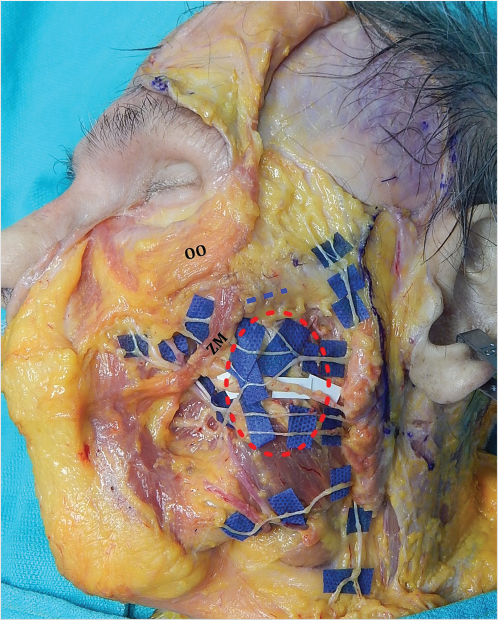
Zygomatic and Buccal Branches
These branches display higher number of rami and interconnections which may account for spontaneous recovery following injury (Fig. 22‑2 and Fig. 22‑9). 77 The buccal ramus is thought to be the most frequently injured facial nerve branch in rhytidectomy. 36 , 77 Due to extensive arborization, clinical effect is usually short-lived and rarely discernible. 36 , 77 Injury to the branches that supply the elevators of the upper lip (e.g., ZM), however, can potentially affect smile. The area inferior to the zygomatic ligaments and anterior to the upper masseteric ligaments is a danger zone 9 , 10 , 63 , 77 . Thus, sub-SMAS dissection in this region should proceed with extreme caution, always hugging the undersurface of the SMAS and keeping all fat down.
Marginal Mandibular Branch
The limited number of rami and communications with other facial nerve branches is thought to account for higher risk of permanent injury (Fig. 22‑9 and Fig. 22‑10). 77 Anatomical studies have shown the nerve to be composed of at least two rami. Posterior to the fascial vessels they travel within 1 cm (rarely 2 cm or more) of the mandibular border (more frequently cephalad than caudad). 65 , 77 , 83 , 85 In contrast, anterior to the facial vessels the marginal mandibular is always cranial to the mandibular border traveling toward the depressor anguli oris. 77 , 83 , 85 Here, it lies posterior and approximately 1 cm cranial to the mandibular ligament 65 (Fig. 22‑8). In terms of depth, the nerve emerges from the lower end of the parotid gland and travels deep to the parotid-masseteric fascia, and under cover of platysma. 65 , 77 , 83 , 85 Inferior to the mandible it courses on the surface of the posterior belly of digastric and submandibular gland remaining deep to platysma. 77 , 83 , 85 At the level of the facial vessels, the nerve crosses the mandibular border superficial to these vessels. It is at this point that the nerve is believed to be at higher risk of injury as it becomes more superficial. 77 The nerve remains deep to the platysma throughout its course, and thus keeping dissections above the platysma is a safe dissection plane. In sub-SMAS dissections, the lower masseteric ligaments are a sign of caution to the proximity of the marginal mandibular branches.
Cervical Branch
Injury to this branch is believed to result in marginal mandibular pseudo-paralysis secondary to dysfunction of the platysma which is likewise a depressor of the lower lip (Fig. 22‑9). 86 , 87 Clinical differentiation is possible by demonstrating retained ability to pucker the lower lip, which signifies residual orbicularis oris and mentalis function. 86 , 87 Return of function is almost always complete. 86 , 87 This nerve (composed of one to three rami with occasional communication to the marginal mandibular) travels close to the gonial angle and branches approximately 1.74 mm inferior to the mandibular border. 88 , 89 Platysmal incisions should be designed at least 15 mm posterior to the gonial angle, and 45 mm caudal to the mandibular border to avoid cervical nerve injury. 88 , 89 It emerges from the lower border of the parotid gland and remains deep but close to the undersurface of the platysma. 77 , 88 , 89 Thus, subplatysmal dissection in proximity to the gonial angle should proceed with caution staying superficial to the underlying fat. Blunt dissection has been advocated in this plane inferior to the tail of the parotid to minimize injuries of the marginal mandibular and cervical branches. 77 , 89
Sensory Nerves
The great auricular nerve (C2 and C3) is thought to be the most commonly injured nerve in rhytidectomy. 90 Arising from the cervical plexus, it winds around the posterior border of the sternocleidomastoid, and passes vertically upward toward the middle of the lobule of the ear. 91 The classic surface landmark is McKinney’s point which lies 6.5 cm caudal to the external acoustic meatus approximately 0.5 to 1 cm posterior to the external jugular vein. 90 Ozturk et al have identified a triangle, where the nerve can be found 100% of the time. 92 The triangle is defined by a line perpendicular to the Frankfort horizontal bisecting the lobule of the ear and another diverging 30 degrees posterior to it (Fig. 22‑12). 92 Maintaining the dissection superficial to the sternocleidomastoid fascia in this region prevents nerve injury and consequent neuroma formation. 92
22.8 Operative Techniques
22.8.1 Incisions
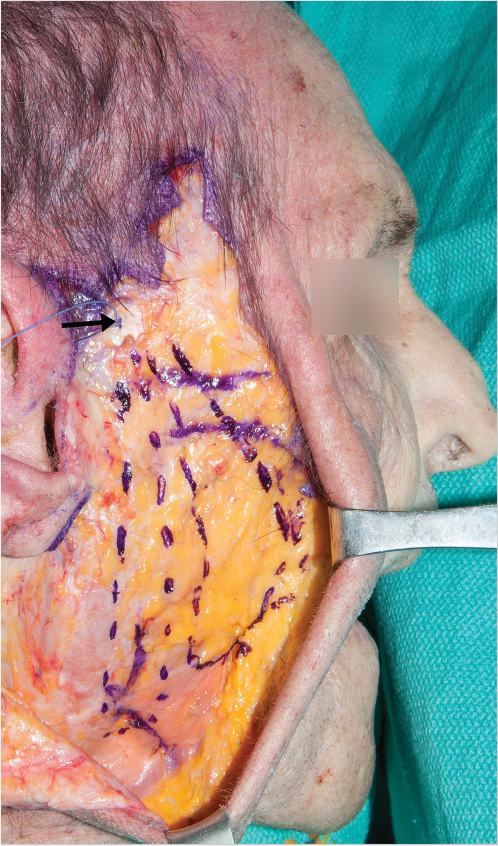
The Preauricular Part
A preauricular retrotragal incision results in less discernible postoperative scarring. 32 If properly executed, the aesthetic outcome is excellent. The potential for tragal deformity is readily preventable if care is taken to avoid cartilage injury and ensure tensionless closure (Fig. 22‑13; Fig. 22‑14). 32
The Temporal Extension
An outer canthus to temple distance less than 5 cm permits a temporal scalp extension. A distance greater than 5 cm mandates a temporal hairline incision to avoid aggravation of temporal recession (Fig. 22‑15). 32 , 47 , 48 , 93
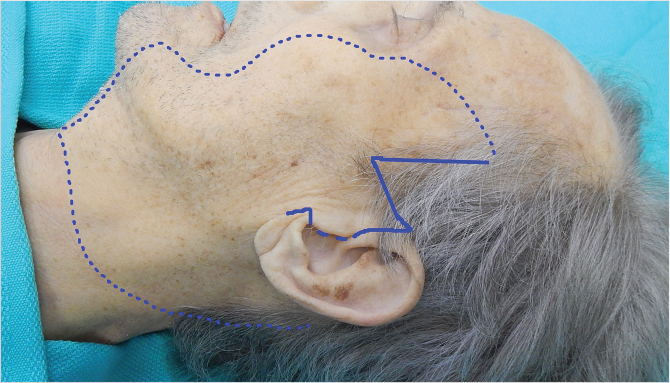
The Postauricular Extension
The incision follows the postauricular sulcus and crosses the postauricular skin where the helix and postauricular hairline touches (Feldman’s “touch point”). 94 It either continues horizontally into the occipital scalp or follows within the occipital hairline. 32 , 55 , 94 The latter prevents posterior hairline recession especially if neck skin laxity is profound (Fig. 22‑16). 32 , 55 , 94
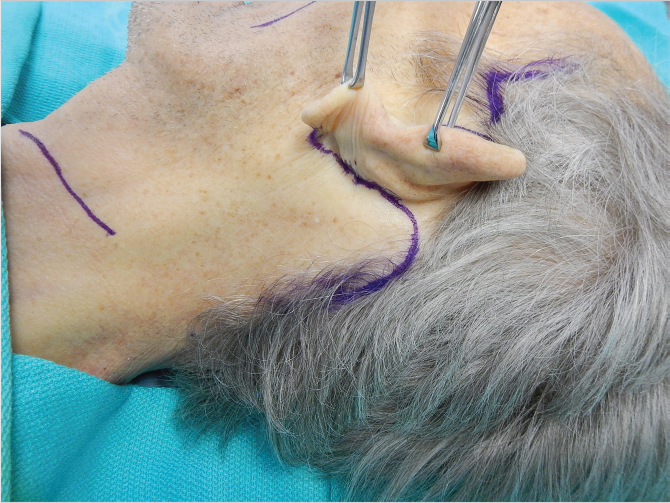
22.8.2 The Extended SMAS Facelift (Senior Author’s [JZ] Preferred Technique)
Principle
The extended SMAS technique delaminates the face by raising separate skin and SMAS flaps. 35 The independent mobility of SMAS and skin permits greater versatility in vector selection and direction of tissue repositioning. 34 , 36 , 37 Precise control of dissection plane and skin flap thickness is critical, however, to avoid tearing of the SMAS flap. 34 , 37 The SMAS thins considerably as dissection extends medially and SMAS tearing can complicate flap fixation. 8 , 34 , 37 , 46
Dissection
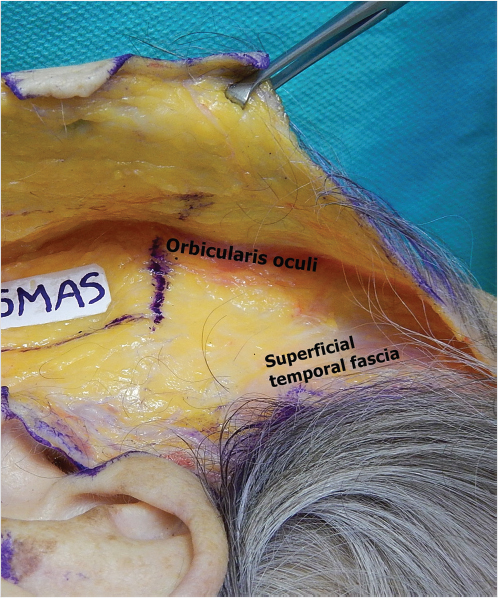
The Temporal Region
It is prudent to stay on top of the superficial temporal fascia. This is the layer immediately deep to the hair follicles. Proceeding anteriorly, the orbicularis oculi muscle is identified, and more caudal dissection should continue superficial to the plane of this muscle (Fig. 22‑17). 32
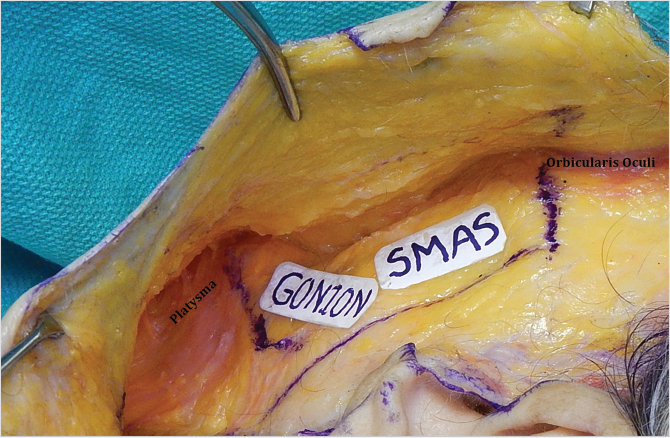
The Preauricular and Cheek Part
Immediately anterior to the helical crus, dissection should be very superficial (subdermal), to avoid injury to the superficial temporal vessels; this part of the skin is usually discarded anyway except in secondary cases. At the tragus, caution is necessary to prevent cartilage injury. In the cheek area (from the zygomatic arch to the mandible), dissection should be more superficial to preserve fat on top of the SMAS, thus providing a thicker SMAS flap. 32 , 35 Moving across the mandible to the neck, the plane transitions smoothly to a slightly deeper level. Dissection anterior to the sternocleidomastoid muscle should be hugging the superficial surface of the platysma (Fig. 22‑18).
The Postauricular Part
Skin should be elevated immediately superficial to the sternocleidomastoid fascia. The great auricular nerve is protected by superficial dissection over Ozturk’s triangle (see above; Fig. 22‑12). 92
The SMAS Flap and Sub-SMAS Dissection
Outline of the SMAS Flap
The Upper Border
Different variations in designing the upper border have been described. While Stuzin et al place it 1 cm caudal to the zygomatic arch, others incise at or above the level of the arch. 32 , 35 , 36 , 37 It is our preference to draw this line flush with the lower border of the zygomatic arch in order to capture most of the malar fat pad in the SMAS flap. The proximity of the frontal branches, their surface anatomy, and their depth (see section 22.6 Anatomical Considerations) should be appreciated.
The Lateral Border
This line is designed approximately 1 to 2 cm in front of the ear and continues downward for 4 to 5 cm into the neck (at least 1.5 cm lateral to the gonial angle to avoid the marginal mandibular nerve). This line generally coincides with the posterior border of the platysma. 32 , 35 , 36 , 37
SMAS Flap Elevation
Starting sub-SMAS dissection over the parotid gland mitigates potential nerve injury, since parotid tissue serves as an extra layer protecting the nerves. The SMAS is also thicker, muscular, and more distinct in this region (Fig. 22‑8). Anterior to the parotid, it is critical to hug the deep surface of the SMAS flap, maintaining all fat over the masseteric fascia protecting the facial nerve branches lying deep (Fig. 22‑8). Once the proper plane is identified, more cephalad dissection can be undertaken. Malar SMAS is mobilized severing zygomatic and upper masseteric ligaments with extreme care (see section 22.6 Anatomical Considerations). 32 , 35 , 36 , 37 Amidst this dissection, ZM muscle is identified (Fig. 22‑8). 32 , 35 Dissecting medial to ZM disconnects any remaining zygomatic ligaments, while caudal to it, the masseteric ligaments are split. 35 Maintaining the dissection superficial to the plane of this muscle protects the nerves. Using blunt spreading motion, dissection continues to the nasolabial crease. 35 This maneuver ameliorates the depth of the crease and simultaneously lifts the malar fat pad. 35 Further, malar fat mobility can be achieved using finger dissection in the same plane: “FAME” (finger-assisted malar elevation). 95 An effective SMAS flap should improve the jowl and the neck and reposition the malar fat pad. 32 , 35 The SMAS elevation has a decidedly positive and powerful effect on platysma tightening and gonial angle improvement because the platysma is continuous with the SMAS and has no bony attachments.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree