Summary
Cryolipolysis and deoxycholic acid injections are effective and generally safe and well-tolerated non-invasive fat removal modalities. Cryolipolysis involves the use of controlled cooling to noninvasively target and destroy subcutaneous fat. ATX-101 refers to synthetic deoxycholic acid (a bile acid), which is used to disrupt adipocyte cell membranes. Common postprocedural side effects from both may include temporary erythema, bruising, swelling, numbness, and/or tenderness/pain. However, it is important for health practitioners to be aware of rare concerning complications associated with both procedures.
15 Cryolipolysis
15.1 Introduction
Body contouring and noninvasive fat removal procedures have grown strongly in popularity within the past decade. The American Society for Dermatologic Surgery (ASDS) 2019 consumer survey on dermatologic procedures shows that 58% of consumers are interested in body sculpting procedures. 1 More and more patients seek noninvasive body contouring and fat removal procedures that lack the risks associated with surgery and liposuction, which include infection, anesthesia complications, and death. 2 , 3 Two effective noninvasive techniques for the removal of unwanted subcutaneous fat are cryolipolysis and deoxycholic acid injections. While these are generally considered safe and effective treatment modalities, 4 , 5 it is important for health practitioners to be aware of associated adverse events and potential complications.
15.2 Cryolipolysis Mechanism of Action
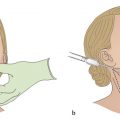
Cryolipolysis refers to the localized application of controlled cooling to noninvasively target and destroy subcutaneous adipose tissue. The controlled cooling promotes crystallization of lipids within adipocytes, which triggers a localized panniculitis. This inflammatory response leads to selective loss of adipocytes through apoptosis without injuring surrounding tissue. The apoptotic cells are eliminated by macrophages, a process that peaks at about 2 weeks and resolves at about 3 months. 6 , 7 More than 7 million procedures have been performed globally on different body sites as of 2019, 8 and cryolipolysis (CoolSculpting; Allergan, Inc. Irvine, CA) has been cleared by the US Food and Drug Administration (FDA) for reduction of fat in the flank area (2010), abdomen (2012), thighs (2014), submental area (2015), and arms/back/bra fat/area beneath buttocks (2016). 7 , 9 The treatment cycle involves positioning a negative pressure applicator on a region of skin overlying excess adiposity; the negative pressure helps bring the treatment area into complete contact with two cooling plates. The original treatment cycles were 60 minutes, but newer applicators such as the CoolAdvantage family of applicators have helped reduce treatment times to as low as 35 minutes. 9
15.3 Safety Profile of Cryolipolysis
Cryolipolysis is generally considered a safe and well-tolerated body contouring procedure with minimal treatment-associated discomfort. 10 , 11 Studies have demonstrated no significant changes in serum total cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), and triglycerides after treatment. 11 Postprocedural side effects include temporary numbness, dysesthesia, erythema, edema, induration, bruising, and/or tenderness that typically self-resolve within 14 days. 10 , 11 , 12 , 13 About two-thirds of patients may experience numbness in the treatment area for about 2 months after the procedure. 14 , 15
A 2013 review by Stevens et al of 528 patients who underwent 2,729 cryolipolysis treatment cycles at 1,785 anatomic sites resulted in no serious adverse events; there were only three cases of mild or moderate pain. 15 , 16 A 2015 systematic review by Ingargiola et al highlighted the following complications after cryolipolysis—temporary erythema, bruising, swelling, dysesthesia/numbness, and pain, which resolved within a few weeks. No cases of scarring, ulceration, blistering, bleeding, infection, or dyspigmentation were noted. However, the authors noted a case of paradoxical adipose hyperplasia (PAH), a rare but significant adverse event that will be discussed in detail later in this chapter. 3
A 2015 review of the FDA Manufacturer and User Facility Device Experience (MAUDE) database lists 62 adverse events from cryolipolysis reported from 2011 to 2013. The MAUDE database is a record of all adverse events that patients voluntarily report after using various medical devices. Adverse events within the MAUDE database included fat hypertrophy, firmness, pain/dysesthesia, hernia, indentation, laxity of the skin, laxity of the fascia, hyperpigmentation, and edema. 17 It is unclear if the reported hernias were present before or after cryolipolysis treatment. 15 , 17 One limitation of the MAUDE database is that it is based upon the voluntary anecdotal reporting of physicians, other health care providers and patients. Thus, the actual incidence of adverse side effects is not elucidated within the MAUDE database. By contrast, industry reports are mandated.
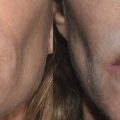
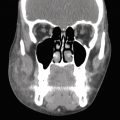
Side effects from submental cryolipolysis are mild and generally self-limited. Side effects, although uncommon, include swelling, bruising, tenderness, and paresthesias. 18 Kilmer et al highlight two cases of prolonged erythema lasting 2 to 3 weeks, one incident of hyperpigmentation resolving at 4 weeks, and one patient with the sensation of fullness at the back of the throat lasting 1 to 2 months from swelling after submental cryolipolysis. 5 Lee et al discuss a rare case of marginal mandibular nerve (MMN) palsy resolving 2 months after submental cryolipolysis using CoolMini applicators (Cool Sculpting; Allergan, Inc., Irvine, CA). 18 Gregory et al highlight two rare cases of submental neuropathic pain associated with mandibular nerve injury due to submental cryolipolysis. 19 One case demonstrated sudden severe posttreatment pain of the left submental region extending toward the left ear. The second case highlighted a subacute onset of significant discomfort extending from the treatment area into the oral cavity and manifesting as dental pain.
15.4 Paradoxical Adipose Hyperplasia in Cryolipolysis
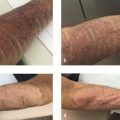
While most side effects associated with cryolipolysis are mild, well-tolerated, and occur within hours to days of the procedure, there are several rare and more notable delayed-onset symptoms. PAH is the most common, significant complication of cryolipolysis. PAH refers to large well-demarcated, painless, firm masses that develop in areas treated with cryolipolysis 2 to 6 months after treatment. 20 , 21 Reported cases have been noted on the abdomen, chest, back, flanks, and thighs. 9 Initially described in two patients by Jalian et al in 2014, the estimated incidence of PAH was previously thought to be 1 in 20,000 treatment cycles; 20 , 22 however, with more recent data, the incidence rate is estimated as 0.029% or 1 in 3,500 treatment cycles. 23 Some still believe that this incidence rate may even be underrepresented, and a study by Singh et al showed that the incidence of PAH may be as high as 2%. 24
Histological studies on PAH are inconsistent, showing either tissue hypovascularity and adipocyte hypocellularity, or hypervascularity and adipocyte hypercellularity. 20 Jalian et al reported biopsied tissue specimens showing septal fat thickening, decreased adipocyte organization, and hypervascularity. 10 , 22 However, Seaman et al reported adipose tissue demonstrating hypocellularity and hypovascularity compared to controls. 10 Septal thickening and hypervascularity may represent a reactive fibrosis and angiogenesis resulting from hypoxic injury to adipocytes that have been cooled. 9
The pathophysiology of PAH is poorly understood but appears to involve both genetic and hormonal factors. 25 A systematic review by Ho and Jagdeo showed that PAH was more common in men and in patients with Hispanic and Latino backgrounds, especially when large applicators were utilized. 9 In addition, there are several cases of PAH developing in men after pseudogynecomastia treatments, suggesting that caution must be taken when treating the male chest. 20 The negative suction from the cryolipolysis applicator has been implicated in adipocyte stimulation and proliferation. 26 Specifically, inadequate suction may lead to suboptimal cooling of the targeted tissue, thereby preventing a cold panniculitis from developing. Therefore, poor suction without the necessary panniculitis may stimulate the adipose tissue to grow larger. This idea has been demonstrated by a breast augmentation device (BRAVA, Biomecanica, Inc, Miami, FL) that uses low suction to increase fibroglandular tissue and adipose tissue within breasts. 20 , 27
The treatment of choice for PAH is liposuction. No evidence of spontaneous resolution of PAH has been described to date, and therefore treatment with liposuction should be considered. 25 Liposuction is recommended between 6 and 9 months after cryolipolysis treatment to allow the newly expanded tissue to soften after a firm inflammatory phase. 25 Additional treatment with cryolipolysis is not recommended, as it may exacerbate the PAH. 9 In a case series of 11 patients, Kelley et al describe one patient with recurrence of PAH at 2 months following ultrasound-assisted liposuction, but no recurrence was noted with power-assisted liposuction. 25 Ward et al describe the use of ATX-101 (deoxycholic acid), another noninvasive fat removal modality, in the treatment of PAH. 28
15.5 Delayed Post-treatment Pain in Cryolipolysis
While PAH is more common in men, women are at increased risk of delayed posttreatment pain with cryolipolysis. Delayed posttreatment pain refers to neuropathic symptoms, increased pain disrupting sleep at night, and/or discomfort unalleviated by analgesic medications. 11 The pathophysiology is unknown, but it is thought that there may be sensory nerve variation in women versus men, and that sensory nerves may be affected by the strong inflammation within the treated area. 11 Keaney et al conducted a retrospective study on 125 patients (27 men and 98 women) who received a total of 554 cryolipolysis treatment cycles (CoolSculpting; Allergan, Inc., Irvine, CA) for the lower and upper abdomen, flanks, back, thighs, and/or chest using one of four small applicators, a large applicator, or flat applicator. 11 A total of 19 patients (15.2%) developed delayed posttreatment pain, and all these patients were female. The pain developed on average 3 days after cryolipolysis and resolved with an average duration of 11 days. Pain was most commonly localized to the abdomen, which was also the most commonly treated area. The delayed posttreatment pain was managed with compression garments, lidocaine transdermal patches, low-dose gabapentin, and/or acetaminophen with codeine.
15.6 Miscellaneous Complications in Cryolipolysis
There are additional isolated complications associated with cryolipolysis. The device being used appears to be a major factor in these cases. Skin necrosis has been reported with the improper use of cryolipolysis. Nseir et al illustrate a case of skin necrosis on the left thigh following cryolipolysis performed without the required interface gel pad. 13 Motor nerve palsy has been reported with cryolipolysis of the arms. Lee et al reports a case of a young woman who underwent cryolipolysis to the distal upper arms with the MiCool cryolipolysis device (MiCool, Hironic Co., Seongnam, Korea). 12 Days later, the patient developed radial nerve injury characterized by left hand extensor weakness and difficulty in lifting heavy objects. Electromyography was consistent with axonal damage to the distal branch of the radial nerve, which bifurcates at the elbow. The left hand motor neuropathy fully recovered 6 months later. Khan reports a case of cryolipolysis to the lower anterior thighs resulting in unsatisfactory contour irregularities and mild hyperpigmentation, which was later corrected with fat grafting and nonablative fractional laser, respectively. 8
15.7 Introduction to ATX-101
In addition to cryolipolysis, the injectable compound known as ATX-101 is an effective treatment for the noninvasive removal of unwanted fat. ATX-101 (Kybella; Allergan, Inc., Irvine, CA) was approved by the FDA in 2015 for the treatment of moderate-to-severe submental fat. 15 , 29 However, it has been used off-label for the jowls, brassiere line fat, and lipomas. 30 The compound contains synthetic deoxycholic acid (a bile acid), which causes lipolysis by disrupting adipocyte cell membranes. 7 , 15 , 31 Submental injections are performed in recommended dosages of 0.1-mL to 0.2-mL aliquots spaced 1-cm apart from one another, using a 30-gauge 0.5-inch needle. 31 In clinical practice, other dosages are often used.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree