Abstract
This chapter is a careful discussion of the function of the palate in normal speech and of the problems caused by cleft palate. After surveying the history of cleft palate repair, the author delves into the steps involved in the double opposing z-plasty repair. Hard palate repairs are also discussed and illustrated. Postoperative care of the airway and bleeding is covered, and the steps in long-term follow-up are outlined.
13 Cleft Palate Repair by Double Opposing Z-Plasty
13.1 Goals and Objectives
Function of palate in speech, both velar and hard palate portions.
Brief history of cleft palate repair and its importance in the development of palate repair methods.
The purposes and design of the double opposing Z-plasty palate repair.
Why velopharyngeal function is critical to normal speech.
How to determine velopharyngeal function related to cleft palate.
13.2 Patient Presentation
Cleft palate repair carries a large responsibility, because repair does not guarantee normal speech, and because abnormal speech is a serious social disadvantage.
In the United States, the standard of care for those with cleft lip and palate is by a team, which usually includes one or more of the following: plastic surgeons, pediatricians who are familiar with the syndromes associated with patients with cleft palate only (CPO) and cleft lip and palate (CLP), otolaryngologists, orthodontists, oral surgeons, speech–language pathologists (SLP), and a supporting team including a nurse and an administrator. Plastic surgeons not associated with a team should consider referring patients to a team.
It is important to consider the concerns and anxieties of the parents and to have the appropriate team members address them at every visit, especially the first. On initial examination, the plastic surgeon should note the child’s age, weight, hemoglobin and hematocrit, and a family history of cleft and any congenital abnormality that might be cleft-related; record the characteristics of the cleft; unilateral CLP (UCLP), bilateral CLP (BCLP), CPO; complete/incomplete, width of the cleft, and the slope of the hard palate shelves; evidence of previous surgery (review the chart if the lip has been repaired); and evidence of a syndrome (particularly such as Pierre Robin, which might impact the surgical plans by delaying repair and avoiding a pharyngeal flap, or velocardiofacial syndrome, which is a difficult surgical problem carrying a lower expectation of success). The parents should be asked to try to have the child feeding from a cup before palatal surgery. In older patients, speech characteristics should be noted and velopharyngeal (VP) function tested.
13.3 Preparation for Surgery
After examination of the child and discussion of the patient by the team, the plastic surgeon should explain why the function of the palate is important in speech, what palate repair must accomplish, and that the goals of repair are not always reached at the first operation. The planned operation should be described for the parents with a discussion of postoperative complications; let them know what to expect. The sequence and timing of procedures and operations should be discussed, with the reasons that subsequent operations might be necessary, and an estimate of how frequently they are usually needed. The possibility of operations for velopharyngeal insufficiency (VPI), fistula repair, alveolar bone grafting, or correction of maxillary recession should be emphasized in the discussion. These points should be reiterated at the preoperative visit.
13.3.1 Palatal Function
Proper treatment of the patient with cleft palate requires an understanding of palatal function.
During speech, for various consonants, the lips and tongue permit (“really”), restrict (“fish”), or block (“Dad”) airflow through the oral cavity. The normal hard palate separates the oral and nasal cavities. It is important for the growth of the midface. The normal soft palate (velum) is a muscular valve that opens and closes to permit, restrict, or prevent airflow and sound into the nasal cavity. This function is required for normal speech.
For most of our English words, the velopharyngeal valve is closed, to direct air and sound through the mouth only (“brother, papa, supercalifragilisticexpialidocious”). The velum provides quick, precisely timed velopharyngeal valving to permit airflow and sound through the nose when the lips, tongue, and teeth block oral airflow (“noon, rain”), and coordinates with the lips and tongue to control airflow and sound through nose and mouth for many words (“mop, king, ban”), or with a mid-word switch (“sentinel, bumper, single, maintain”). Listen to how the oral and nasal vowel sounds relate to the associated consonants. 1 , 2 , 3 , 4
The palatal cleft mars speech by failing to block airflow into the nasal cavity on consonants, and sound on vowel production. The prime purpose of cleft palate repair is to make normal speech possible by converting the cleft hard and soft palate into a competent velopharyngeal valve mechanism.
A successful cleft palate repair (1) produces a velum that permits precise, instantaneous, essentially complete velar closure, and velar opening sufficient for nasal breathing; (2) constructs an intact hard palate that partitions the nasal cavity from the oral cavity and permits adequate midface growth; (3) protects middle ear function; (4) achieves all, preferably by the age of 1 year, before the child learns speech.
Palate repair is by no means always successful. If velopharyngeal closure is inadequate or a hard palate fistula connects the oral and nasal cavities, compensatory articulations, substitute sounds learned when the attempt to make a desired sound by closing the velum does not work, may mar speech after a second operation has provided VP competence. 5 , 6 , 7
13.4 Treatment
13.4.1 History
The history of cleft palate repair is interesting, particularly because improvements have come so slowly and because over its first century and a half, the history has defined the requirements for the best assurance of a successful surgical outcome.
The first successful surgical repair was in 1819 in Paris by Dr. Roux on the Canadian John Stephenson, a medical student in Edinburgh. A thesis was required for graduation, and Stephenson’s thesis is a very interesting, revealing personal account: by a person with a cleft palate, and as the patient receiving this seminal surgery. 8 Stephenson returned to Montreal to practice medicine, where he was the key person in the founding of McGill Medical School as the first College of McGill University.
For many years, cleft palate repair was clearly difficult for both patient and surgeon. It was not until 13 years after Stephenson’s repair, in 1832 in Jefferson, Georgia, that Dr. Crawford W. Long introduced ether anesthesia, subsequently popularized in Boston. However, general anesthesia was not used for palate repair until about 1867, also the year that Joseph Lister, in Scotland, introduced the concept of asepsis in surgery. 9 , 10 Surgery was usually performed using outside sunlight until the 1880s, when Tesla’s alternating current illuminated the 1883 Chicago World’s Fair with Edison’s light bulbs. In 1920, Magill introduced the endotracheal tube. Other aids such as epinephrine, intravenous fluids, lidocaine, suction, and the Dingman mouth gag did not become available until sometime in the 20th century.
In 1861, von Langenbeck described tension-relieving mucoperiosteal flap elevation through lateral relaxing incisions, which made hard palate closure easier, and must have reduced fistula rates. 11 It is an operation frequently used today, the repair this author learned and used before the double-opposing Z-plasty (DOZ-pl). 12 Variations include Bardach’s two-flap repair, which detached von Langenbeck’s flaps anteriorly for easier flap elevation and closure. 13
In the 1920s, Veau, Dorrance, Wardill, and Kilner’s pushback procedures lengthened the velum with hard palate mucoperiosteum. 14 , 15 , 16 , 17 However, without velar muscle lift, the added velar length did not significantly improve speech results, but palatal fistulas and midface retrusion increased. 18 , 19 The procedure is widely used today. The palatal island flap provided more hard palate mucoperiosteum for velar length, 20 resulting in greater retrusion. 21
In the 1940s, an increase in midface retrusion, likely from the pushback’s increased hard palate scarring, led to Schweckendiek’s two-stage repair. 22 , 23 Its intent was to engage the repaired velum in speech while leaving the unrepaired hard palate mucoperiosteum unscarred for growth.
Maxillary growth was much improved, but the intentional hard palate fistula led to speech seriously marred by compensatory articulations. 24 , 25 Variations on the two-stage operation have generally moved the hard palate closure into early childhood or later in infancy.
In the 1960s, attention was finally turned to the velar muscles by Ruding, Braithwaite, and Kriens. 26 , 27 , 28 By most reports, the end-to-end, anterior intravelar veloplasty (IVV) led to some improvement in speech outcomes, but much room for improvement in speech outcomes remained. Today, IVV is usually a part of palate repair, regardless of design. Sommerlad’s recent results with his careful IVV using the operating microscope has produced very good outcomes, even though velar lengthening is not part of the operative plan. 29
From these history lessons of palate repair and an awareness of the aberrant anatomy, we may conclude that the best characteristics of a repair should
Put the levator muscles at functional tension by retroposing and transversely overlapping them.
Permanently lengthen the velum.
Provide complete oral and nasal mucosal coverage.
Leave the hard palate mucoperiosteum where it belongs.
Scar the hard palate as little as possible.
Complete the cleft repair by or close to age 1 year, in one operation if possible.
13.4.2 Double Opposing Z-Plasty Repair
The design of the double opposing Z-plasty repair achieves the six attributes noted above; patients’ VP competency rate of the authors’ went from 48% using a von Langenbeck repair either without or with IVV to 91% with double opposing P-plasty (Table 13‑1). 12 , 30 , 31
The experience of others has confirmed its effectiveness. 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39 , 40
The operation has been much modified by others, usually by reducing the size of the Z-plasties and by freely using lateral relaxing incisions in the hard and soft palate, more recently by treating the muscles and their Z-plasty flaps as separate units. 40 , 41
What follows briefly describes the Furlow version for the hard palate and the velum as the author finally came to perform it after his experience performing some 350 cases: 37 in private practice, and the rest later on volunteer surgery trips.
The DOZ-pl repair was developed in the author’s private practice in which a consecutive series of 33 infants and 4 older patients (11 with various syndromes) were treated. Some 300 cases done on volunteer surgery trips added greatly to his operative experience. Much of what is described below was learned on these trips: from other surgeons, from the increasing experience, and the opportunity to do one case after another, remembering problems and solutions from case to case. Unfortunately, the lack of follow-up was very disappointing.
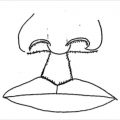
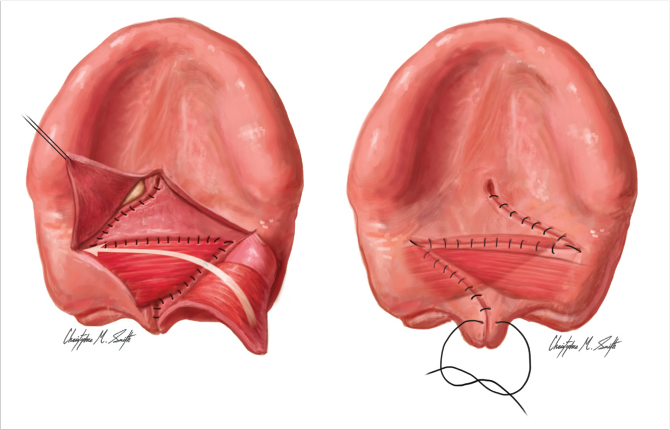
The soft palate cleft is closed with two large mirror-image Z-plasties; the cleft is the central limb of each Z-plasty. The palatal muscles are elevated as part of each posteriorly based flap; the anteriorly based flaps are mucosa only. As the lateral limb incision of the oral posteriorly-based flap is deepened to nasal mucosa, the palatal aponeurosis is automatically divided, making velar relaxing incisions unnecessary (this author has never used them). The incision is carried to, but not into the superior constrictor laterally (Fig. 13‑1). 12 , 42
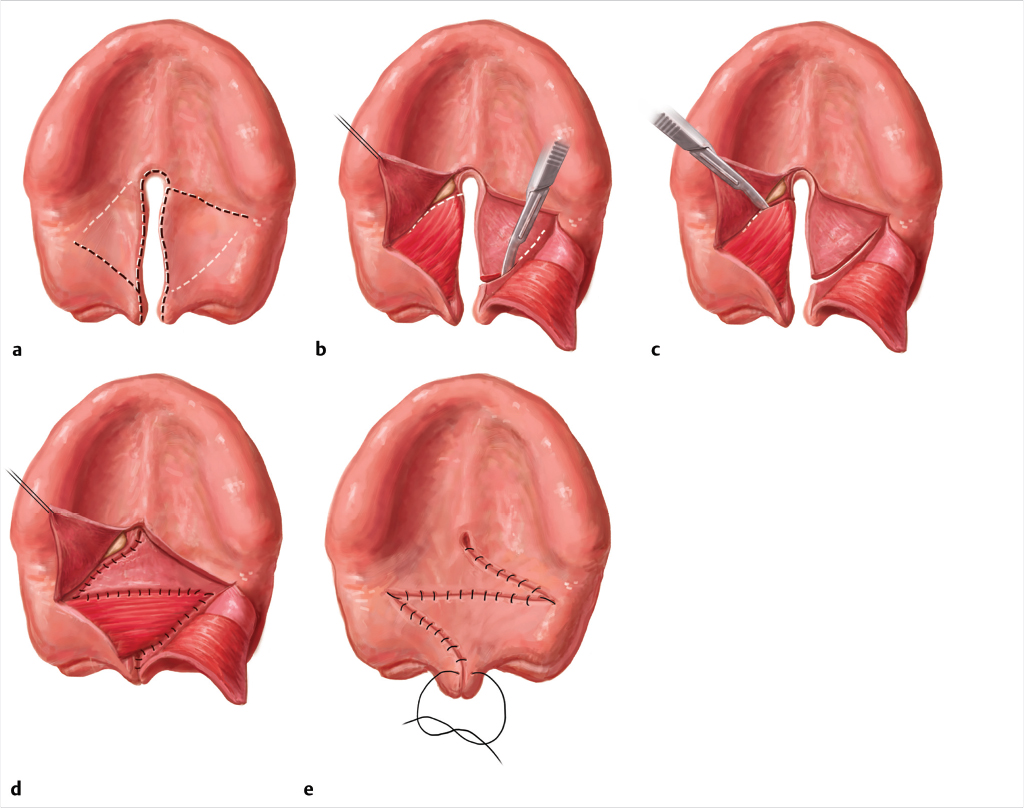
The flap with its palatal muscle is carefully elevated from the nasal mucosa. A Freer elevator is used to push the levator posteriorly along the medial surface of the superior constrictor, and the flap with its levator is rotated transversely. 12 , 42
Transposition of each z-plasty carries the levator in its posterior flap, overlapping the muscles. The anterior flaps close the oral and nasal mucosa. The large z-plasty design gives wide access for precise dissection and positioning of the flaps and their muscles.
In short, the velar Z-plasties repair the cleft, create a retroposed levator sling by rotating the levators transversely and overlapping them, lengthen the velum, leave the hard palate mucoperiosteum where it belongs, and close the oral and nasal mucosa anterior to the muscle sling, all at the same time.
Several technical details:
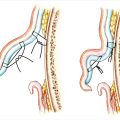
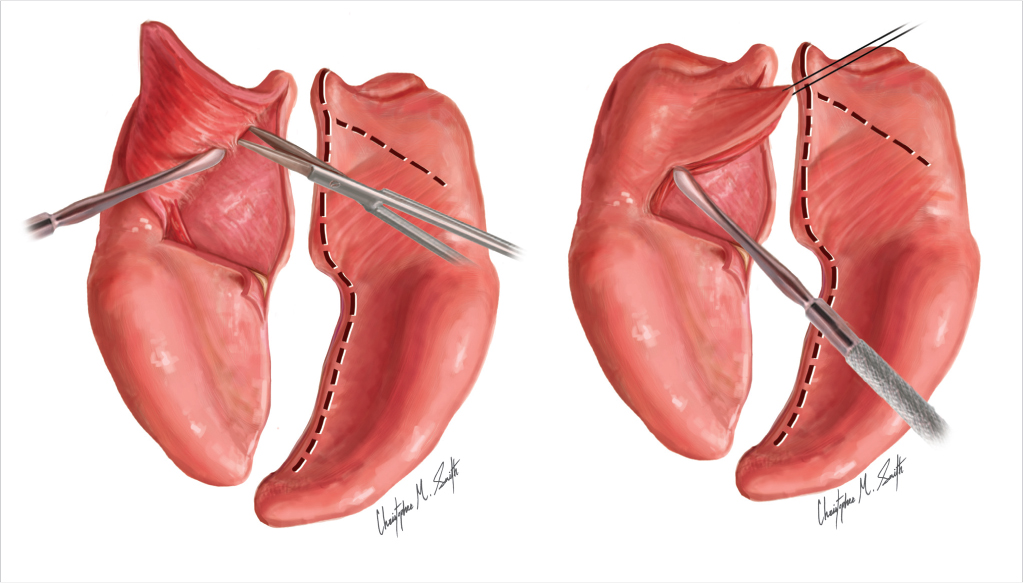
The end of the lateral limb incisions of the anteriorly-based flaps position the tip of the myomucosal flaps, which sets the A-P direction, dorsal position, and overlap of the velar muscles. As the nasal lateral limb incision is carried laterally its end also curves posteriorly toward the posterior pharyngeal wall, so that a wide z-plasty constructs the levator sling nearer the posterior pharyngeal wall than a small one, under more functional tension, making VP closure easier for the patient.
The left nasal mucosal Z-plasty flap ends precisely under the levator muscle base to position the right levator tip directly under the left levator base, creating a transverse levator sling, its levators under functional tension. 30
The oral myomucosal flap tip with its levator muscle is inset directly over the opposite levator base, not farther anteriorly over the hamulus Fig. 13‑2. 30
An intraoperative view of widely overlapped exactly aligned levators is what one should see before the final oral mucosal z-plasty flap is placed. 31
The anteriorly-based flaps provide complete mucosal closure anterior to the velar muscles, so the levator sling is not pulled forward over time by scar contracture, shortening the velum Fig. 13‑3. 12
Guneren and Uysal found a mean increased velar length of 12.45 mm (55%) 4.5 years after DOZ-pl. 44
Other helpful variations are described in Furlow. 30
13.5 Hard Palate
Lateral relaxing incisions and pushback procedures are to be avoided because they add hard palate mucosal scars and bare areas. The greater the hard palate mucoperiosteal deficit the greater the midface growth deficit. 18 , 21
When the mucoperiosteal flaps are brought from the vault to horizontal, lateral relaxing incisions are infrequently needed. 42
The relationship of palatal vault to the distance from alveolus to alveolus, not cleft width, is critical for closure without lateral relaxing incisions. To avoid lateral relaxing incisions the mucoperiosteal flaps must reach from the base of each alveolus to each other at the cleft.
When the cleft is wide, if the vault of the hard palate is high, mucoperiosteal flaps may reach each other when they are brought out of the vault to horizontal, whereas if the cleft is narrower but the vault is flatter, mucoperiosteal flaps may not reach. 31
To determine if the flaps will reach, plan ahead. Before beginning surgery, at the hard-soft palate junction measure the distance on each side from the cleft margin to the base of the alveolus, which is the breadth of each mucoperiosteal flap that can be brought out of the vault to horizontal to close the cleft. If the two measurements will bridge the gap from alveolus to alveolus, lateral relaxing incisions will not be necessary. If they will not reach, an alternate plan can be made before beginning the operation.
Two q-tips can be used to measure the available mucoperiosteal flap width by placing the end of each q-tip stick (the handle) on the cleft margin of each side at the hard-soft palate junction and marking the base of that side’s alveolus with a pen or methylene blue, then adding one handle marking to the other q-tip handle to mark the sum of the mucoperiosteal flap breadths. Bending the q-tip at the mark permits the segment to be put across the gap from alveolus to alveolus. If it reaches, proceed without lateral relaxing incisions. 31
If it won’t reach, after doing velar DOZ-pl, do lateral relaxing incisions, 2-flap elevation and closure, or another plan for hard palate closure. 45 , 46
When closing the hard palate, to avoid fistulas, always use mattress sutures. The raw surfaces of each mucoperiosteal flap must be in apposition; a raw edge is unlikely to heal to a mucosal surface.
13.6 Postoperative Care
The main immediate concerns are with airway and bleeding; a pulse-oximeter is indicated. Traction on a tongue stitch placed at the end of the palatoplasty can be very helpful in preventing airway obstruction, and sedation and elevation of the head of the infant’s bed will stop most postoperative oozing. As soon as the infant is awake, the moistening from drinking from a cup or syringe may be calming and safe, and may reduce the need for pain medicine. If analgesics are needed, acetaminophen or ibuprofen is nearly always effective. Elbow restraints are probably not necessary.
When the infant is drinking clear liquids, the intravenous fluids can be discontinued and the infant sent home on a liquid diet if the parents are reliable and comfortable with providing care.
Initial follow-up should be in about 2 weeks after hospital discharge, and thereafter in craniofacial clinic by its schedule.
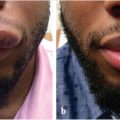
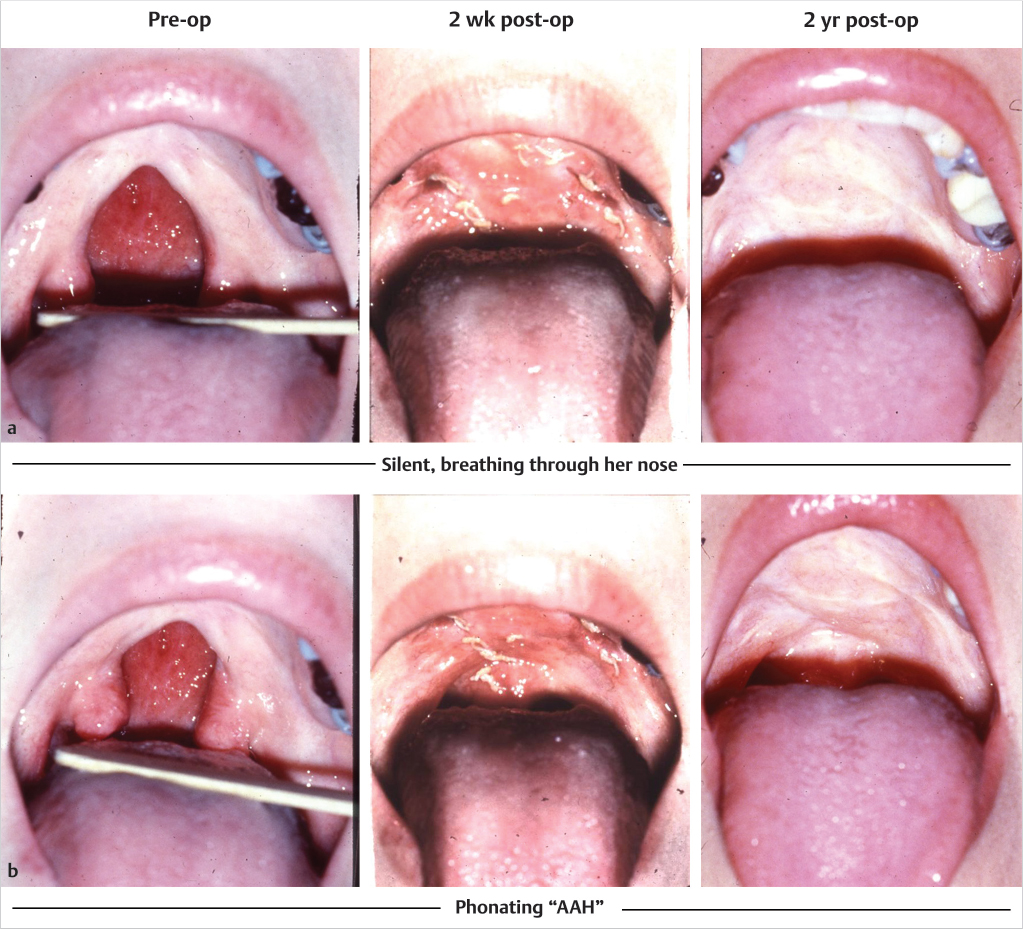
This 34 year-old patient with a Veau II cleft had never been told it could be repaired (Fig. 13‑4). Velum elevates well at two weeks and at two years. She developed velopharyngeal competence, her increased velar length persists, and her “dimple of elevation” remains well posterior. Her articulation was good before her repair and her speech became normal.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree