13 Breast Implant–Associated Anaplastic Large Cell Lymphoma
Summary
Breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) is a rare peripheral T-cell lymphoma that develops after placement of breast implants for cosmetic or reconstructive purposes. BIA-ALCL most commonly follows a fairly indolent course, with excellent prognosis in early-stage disease but disseminated cancer and deaths from BIA-ALCL have been reported. BIA-ALCL typically presents approximately one decade after implant placement with capsule-confined disease, seroma, or less frequently, as mass adjacent to the implant. Over 500 confirmed unique cases have been identified worldwide. The Food and Drug Administration has published multiple safety communications cautioning women with breast implants about the risk of BIA-ALCL. The National Comprehensive Cancer Network consensus guidelines have been established and widely adopted for the diagnosis and management of breast implant ALCL. Treatment with complete surgical excision has shown better overall survival and event-free survival when compared to patients treated with partial capsulectomy, systemic chemotherapy, or radiation. In this chapter, we will review the clinical features, diagnosis, treatment, and prognosis of BIA-ALCL.
Key Teaching Points
Breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) is a rare CD30-positive peripheral T-cell lymphoma arising around textured breast implants placed for reconstructive or cosmetic purposes.
BIA-ALCL most commonly presents approximately one decade after implant placement with a seroma or less commonly, a mass adjacent to the implant. The tumor involves the fibrous capsule surrounding the implant with varied degrees of infiltration.
Tumor cells are CD30 positive, lack expression of anaplastic lymphoma kinase (ALK) and lack gene rearrangements involving the ALK gene on chromosome 2p23.
Evaluation for BIA-ALCL includes bilateral breast examination, ultrasound, seroma fluid aspiration, and capsule biopsy. Diagnosis is made on the basis of clinical features in conjunction with morphologic and immunohistochemical patterns found on biopsy.
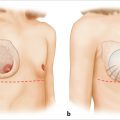
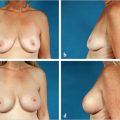
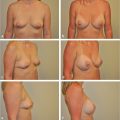
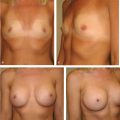
NCCN BIA-ALCL consensus guidelines guide diagnosis and treatment, and recommend implant removal, capsulectomy, and resection of any associated masses for all patients with BIA-ALCL.
Patients who fail surgical therapy alone or have disseminated disease should be treated with an anthracycline-based regimen (CHOP: cyclophosphamide, doxorubicin, vincristine, and prednisone) or brentuximab vedotin, based on NCCN guidelines.
Breast implant associated anaplastic large cell lymphoma (BIA-ALCL) is a rare peripheral T-cell lymphoma that develops after placement of breast implants for cosmetic or reconstructive purposes. BIA-ALCL most commonly follows a fairly indolent course, with excellent prognosis in early-stage disease, but disseminated cancer and deaths from BIA-ALCL have been reported. BIA-ALCL typically presents approximately one decade after implant placement with capsule-confined disease, seroma, or less frequently, as mass adjacent to the implant. Over 500 confirmed unique cases have been identified worldwide. The Food and Drug Administration (FDA) has published multiple safety communications cautioning women with breast implants about the risk of BIA-ALCL. The National Comprehensive Cancer Network (NCCN) consensus guidelines have been established and widely adopted for the diagnosis and management of breast implant ALCL. Treatment with complete surgical excision has shown better overall survival and event-free survival when compared to patients treated with partial capsulectomy, systemic chemotherapy, or radiation. In this chapter, we will review the clinical features, diagnosis, treatment, and prognosis of BIA-ALCL.
13.1 Classification
Lymphoma is a cancer of the immune system, which develops from B or T lymphocytes, natural killer cells, or plasma cells and includes Hodgkin’s lymphoma (10%), non-Hodgkin’s lymphoma (NHL), multiple myeloma, and immunoproliferative diseases. Lymphoma is the most common malignancy of the blood affecting up to 1 in 50 in the general population. ALCL is a type of NHL characterized by large anaplastic lymphoid cells that express the cell surface protein CD30, which is a marker for activated T cells.
Anaplastic large cell lymphoma (ALCL), a subset of peripheral T-cell lymphoma, comprises less than 2% of all adult lymphomas. The World Health Organization (WHO) 2016 classification of lymphoid neoplasms recognizes three distinct lymphoma forms of ALCL: anaplastic lymphoma kinase (ALK)-positive systemic ALCL, ALK-negative systemic ALCL, and now provisionally classifies BIA-ALCL. Primary cutaneous ALCL (PC-ALCL) is recognized as a lymphoproliferative disorder compared to systemic ALCL and BIA-ALCL, which are classified as invasive lymphoma. ALK expression is a result of the t(2;5) translocation involving chromosomes 2p23 and 5q35 creating an oncogenic fusion protein of the ALK gene and nucleophosmin gene. ALCL subtypes exhibit variable degrees of clinical aggressiveness. Systemic ALCL classically follows a very aggressive course with rapid evolution when left untreated. Most patients with systemic ALCL present with disseminated stage III or IV disease, systemic symptoms (B symptoms), and extranodal disease (40–60%). ALK positivity indicates a more favorable prognosis. BIA-ALCL does not express ALK in all reported cases to date.
PC-ALCL is usually clinically indolent with an excellent prognosis and disease-specific survival rates of 85% at 5 to 10 years. Systemic spread is uncommon (approximately 5%), however, when present, it does not change the prognosis of PC-ALCL, which remains favorable. The WHO provisionally classifies BIA-ALCL as a lymphoma. In early-stage disease, BIA-ALCL is most commonly confined to an effusion, becoming infiltrative in later stages.
13.2 Pathogenesis
13.2.1 Theories
While no clear etiology of chronic immune stimulation has been identified in BIA-ALCL, proposed theories describing the pathogenesis include chronic inflammatory response to particulate matter, presence of subclinical biofilm, induction after mechanical abrasion, and genetic predisposition. Deva, Prince, and colleagues postulate that four primary factors provide impetus for inflammation, contributing to the malignant degeneration of ALCL including (1) bacterial contamination, (2) high surface area texturing, (3) long exposure time, and (4) host susceptibility by genetic predisposition.
Bacteria Biofilms and Inflammatory Response
The biofilm hypothesis states that textured implants harbor significantly higher levels of gram-negative bacteria that correlate with lymphocyte activation. Bacteria have been isolated from normal capsules, capsular contracture, and some BIA-ALCL patient specimens. Chronic activation of local and systemic immune systems in BIA-ALCL may also cause neoplastic transformation. Host genetic factors influence the likelihood of malignant transformation in the context of chronic inflammation. In genetically at-risk hosts, polyclonal activation of T cells may result in monoclonal T-cell expansion, ultimately leading to lymphoma.
Bacteria are significantly more likely to attach to textured implants, therefore, textured implants develop a higher load of bacterial biofilm. Once a threshold biofilm is reached, on both smooth and textured implants, predilection toward capsular contracture increases. Bacterial biofilm of patients with BIA-ALCL shows a microbiome with predominance of gram-negative bacteria, while the microbiome in nontumor capsular contracture capsules is gram positive and Staphylococcus spp. predominant.
Chronic biofilm infection of mammary prosthesis is associated with a T-cell predominant lymphocytic infiltration and increased bacterial load around implants. In porcine models, increased T-cell predominant lymphocytes have been demonstrated within textured implant capsules compared to smooth implants. Preliminary studies suggest that BIA-ALCL cells may be derived from Th1 or Th17 cells in capsular tissues and seromas. Th17 are antigen-driven memory T cells. BIA-ALCL cells display diffuse CD30 expression on their cell surface. Investigation regarding whether select biomarkers (JunB, SATB1) will identify nonmalignant precursors of BIA-ALCL are in process. BIA-ALCL appears to result in the setting of a background of allergic inflammation with expression of interleukin 10 and immunoglobulin E, allergic mediators.
Texturing
To date, all reported cases of BIA-ALCL with a known clinical history exclusively involve a textured device. According to the 2017 FDA update, 359 adverse event reports of BIA-ALCL have been reported. Of the reported cases, 231 had information regarding surface characteristics available, 92.3% were reported as textured implants. The 28 cases of BIA-ALCL following smooth implant reported to the FDA were unreliable with minimal or no clinical history available. All patients with BIA-ALCL and known implant characteristics have a history of textured implants.
Lista and colleagues reviewed adverse sequelae associated with textured implants including capsular contracture, malrotation, and late seroma over 9 years and identified no cases of BIA-ALCL in 440 patients reviewed. However, with a proposed incidence of BIA-ALCL ranging from 1 in 1,000 to 1 in 30,000 women with breast implants (see Epidemiology section), a larger sample population is necessary for adequate evaluation of BIA-ALCL. While not intended to be an epidemiologic study, the prospective CA/CARE Trial reported by McGuire et al represents the largest prospective series on textured implants to date. This study revealed six cases of BIA-ALCL in 17,656 women, an incidence of one in 2,943 women with textured Biocell breast implants (95% confidence interval: 1,350–8,000).
Tribology is the observed effect of friction between two objects and a hypothesized driver of carcinogenicity in orthopedic implants. Brody suggests that the etiology of BIA-ALCL is multifactorial and that texturing alone may be sufficient to stimulate BIA-ALCL mutation and lead to disease development.
Genetic Predisposition
Genetic predisposition may help explain the rarity of BIA-ALCL. Blombery et al identified acquired activating mutations in JAK1 and STAT3 in two cases of BIA-ALCL. A germline JAK3 variant may be a contributor to disease development. The aberrancy in the JAK/STAT3 pathway supports the inflammatory model of pathogenesis suggesting that similar fundamental driving genetic lesions exist between BIA-ALCL and systemic ALK-negative ALCL, despite clinical differences. Di Napoli confirmed that mutations in JAK/STAT pathway genes occur in BIA-ALCL. Additionally, Di Napoli identified a SOCS1 mutation in this disease and TP53 and DNMT3A mutations as additional somatic events. Pathogenesis is possible given a sufficient amount of exposure time to a textured implant in the setting of allergic inflammation and genetic predisposition.
13.3 Epidemiology
BIA-ALCL is a rare disease, the exact incidence is unknown. It most commonly presents one decade after implant placement with a median age of onset of 50-year-olds. The absolute risk of developing BIA-ALCL is very low; however, the relative risk of developing BIA-ALCL in women with textured breast implants, when compared with the general population is high. In the setting of breast implants, an 18-fold higher rate of developing BIA-ALCL has been reported. Epidemiologic studies that fail to show an association between breast implants and lymphoma are limited by length of follow-up and sample size.
There was a significant increase in reporting in 2011 reflecting increased physician awareness of the disease after the FDA safety communication. The MAUDE database contains 359 adverse event reports of BIA-ALCL through February 1, 2017. Worldwide, 464 adverse event reports of BIA-ALCL have been reported to government authorities; however, these values may include case duplication and unconfirmed cases.
BIA-ALCL risk appears to be equivalent between cosmetic and reconstructive implant recipients. Risk has not been linked to implant type (saline vs. silicone), but BIA-ALCL has only been reported with textured implants. A lifetime prevalence of 1 in 30,000 is estimated when comparing the number of textured implants sold to cases of BIA-ALCL, a prevalence 67.6 times higher than primary ALCL in the general population. The Therapeutic Goods Administration, in Australia, estimated a BIA-ALCL disease risk of 1 in 1,000 to 1 in 10,000 women with textured breast implants. The risk discrepancy between the United States and Australia requires further investigation but may be due to discrepancy in physician reporting or geographic predisposition. Interestingly, the United States CA/CARE Trial, the largest published prospective textured implant study to date, reports a BIA-ALCL prevalence within the Australian estimated range (1 in 2,943 women with Biocell implants).
13.4 Disease Characteristics
Onset of BIA-ALCL has been reported between 2 and 28 years after breast implantation, most commonly occurring between 7 and 10 years. Unilateral disease is much more common than bilateral disease, which occurs rarely in approximately 4.9% of patients. BIA-ALCL most commonly arises as an effusion adjacent to an implant and then infiltrates into the luminal surface of the implant scar capsule. Approximately 60 to 80% of patients present with a seroma surrounding the implant and 17 to 40% present with a mass. BIA-ALCL occurs predominantly but not exclusively in women—three transsexual patients with textured implants have been confirmed.
13.5 Diagnosis
13.5.1 NCCN Consensus Guidelines for Diagnosis and Disease Management
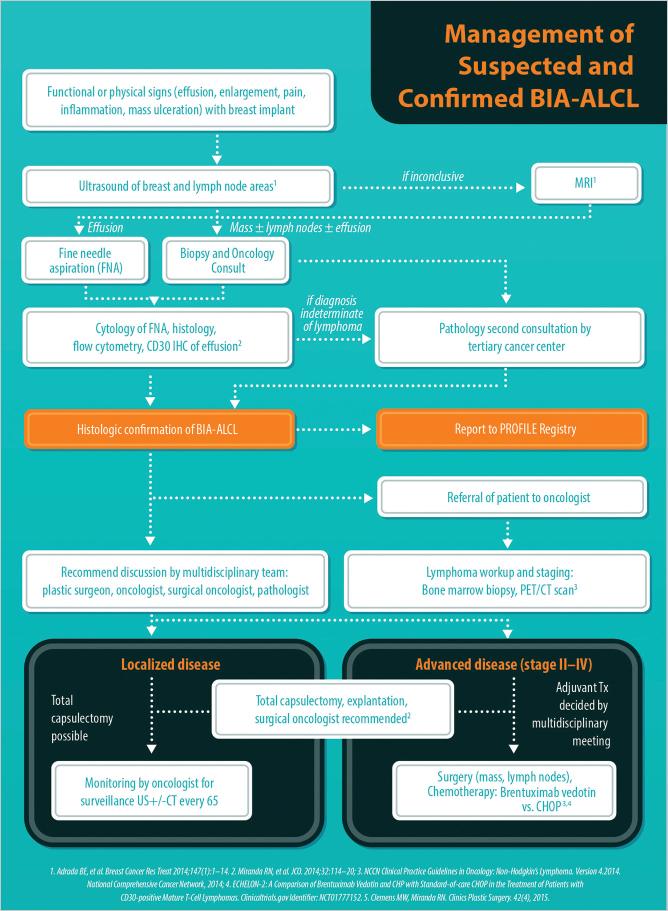
Standardized diagnosis and management guidelines for BIA-ALCL were developed in 2016 by NCCN. These guidelines have since been adopted by the American Society of Plastic Surgeons (ASPS) and the American Society for Aesthetic Plastic Surgery (ASAPS). NCCN guidelines represent the authoritative oncology standards utilized worldwide and are available for free from www.nccn.org (▶Fig. 13.1). The guidelines are important for justification of insurance coverage. NCCN guidelines represent the most up-to-date evidence-based approach to this disease and should be used as part of a multidisciplinary approach to formulate individual treatment plans.
13.5.2 Signs and Symptoms
Pearl
The finding of seroma presenting more than 1 year after placement of breast implants should raise concern and prompt investigation for BIA-ALCL.
BIA-ALCL has been estimated to occur in 9 to 15% of delayed seroma presentations. Physical examination should include inspection and palpation of bilateral breast, axillae, neck, and chest wall. The most common presentation of BIA-ALCL is a periprosthetic fluid collection or mass. Other symptoms include breast enlargement, skin rash, capsular contracture, and lymphadenopathy. Capsular contracture in isolation has not been described and therefore may be coincidental in relation to BIA-ALCL.
13.5.3 Imaging and Diagnostic Tests
Pearl
Ultrasound, the preferred initial imaging modality to investigate BIA-ALCL, can be used to define the extent of a fluid collection (sensitivity 84%, specificity 75%), identify masses (sensitivity 46%, specificity 100%), and evaluate enlarged regional lymph nodes. Magnetic resonance imaging or positron emission tomography scan can be used if ultrasound is indeterminate.
The sensitivity of mammography is inferior for detection of both effusion and mass and therefore is not an acceptable imaging modality for BIA-ALCL. Typically, ultrasound evaluation is used as a screening tool, while positron emission tomography or computed tomography (PET or CT) scan is utilized after diagnosis has been established, for further diagnostic oncologic perioperative work-up.
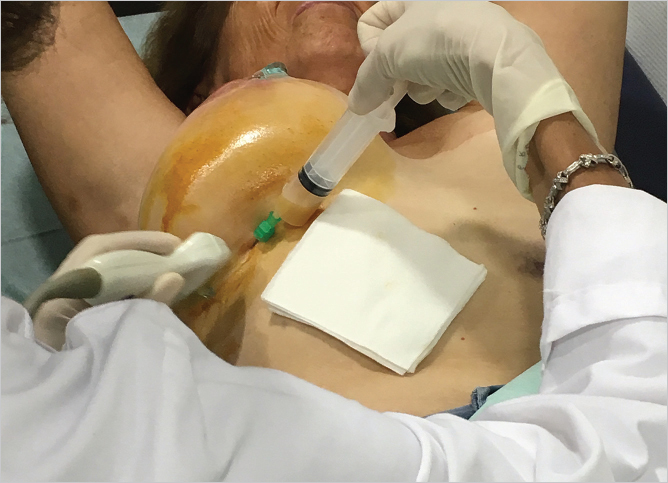
While a small volume (10–15 mL) of periprosthetic fluid may be normal, clinically significant seromas should be investigated with fine-needle aspiration (FNA). FNA can be performed under ultrasound guidance in a clinic setting or in interventional radiology (▶Fig. 13.2). Seroma fluid should be aspirated for analysis along with samples of the capsule and any suspicious masses and should be sent for cellblock cytology. Surgeons investigating a suspicious seroma must supply the pathologist with at least of 50 mL of fluid for analysis, and ideally 100 mL for thorough evaluation. Cell cytology, CD30 immunohistochemistry, and flow cytometry for T-cell clones are necessary to establish the diagnosis of BIA-ALCL.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree